Abstract
Massive ovarian edema is a rare, non-neoplastic solid tumor-like lesion. It results from compromised venous and lymphatic drainage due to partial or intermittent torsion of ovarian pedicle. Pain, distension or abdominal mass, menstrual irregularities, infertility and hormone-related symptoms can be the clinical presentation. We report a case of massive ovarian edema in a 28-year-old female who presented with acute pain abdomen. She was diagnosed clinico-radiologically with solid ovarian tumor and massive ovarian edema on histology. Massive ovarian edema should be suspected in female of reproductive age group presenting with abdominal pain, solid enlargement of ovary on radiology and normal serum tumor markers.
Introduction
Massive ovarian edema is a rare, benign, non-neoplastic solid tumor-like gross enlargement of one or both ovaries due accumulation of edema fluid within the stroma.1–3 It results from partial or intermittent torsion of an otherwise normal ovary.1–3 To the best our knowledge, less than 200 cases are reported in the literature. 3 Venous and lymphatic drainage are compromised due to incomplete and intermittent ovarian torsion which can leads to the enlargement of the ovary which can present as a solid, adnexal mass without ischemic necrosis.1,2 The young females in the child-bearing period (second to third decades) are most commonly affected age group although rarely seen in prepubertal girl.1,4 The patient may present as acute abdomen when torsion is acute and has to be differentiated from appendicitis if the right-sided ovary is affected.2,3 Pain, distension or abdominal mass, precocious puberty, menstrual irregularities, infertility and virilization can be the clinical presentation if the torsion is gradual.2,3 We present a case of massive ovarian edema in a 28-year-old female who presented with acute pain abdomen and diagnosed radiologically and clinically with solid ovarian tumor, for which she underwent oophorectomy.
Case reports
A 28-year-old married female was admitted to the gynecology department with acute pain abdomen. There was no history of any drug intake. On physical examination, abdomen was soft, tenderness on right lower abdominal quadrant and no mass was felt. The patient’s routine blood analysis, liver function test and renal function test were within normal limits: hemoglobin, 11.5 g/dL (11.5–15.5 g/dL); total leukocytes count, 8.5 × 103/mm3, with 65% neutrophils, 29% lymphocytes, 3% monocytes and 3% eosinophils in the differential count; and platelet count, 230,000/mm3. The total bilirubin, 0.5 mg/dL (0.1–1.2 mg/dL); alanine transaminase, 24 U/L (4–36 U/L); aspartate aminotransferase, 22 U/L (8–33 U/L); urea, 20 mg/dL (15–40 mg/dL); and creatinine, 0.65 mg/dL (0.6–1.2 mg/dL). On ultrasonography (USG), right adnexa showed a solid, heterogeneous, hypoechoic mass of 8.5 cm × 3 cm. There was mild free fluid in the pouch of Douglas. Uterus and other side adnexa were unremarkable. A diagnosis of solid ovarian tumor suggestive of fibroma was made. The preoperative serum level of cancer antigen 125 (CA-125), 13.2 U/mL (0–35 U/mL); β human chorionic gonadotropin (β-HCG), 1.9 IU/L (0–5 IU/L); and α-fetoprotein, 3.2 µg/L (<8.5 µg/L) were within normal values. The patient underwent for exploratory laparotomy, and right oophorectomy was done. The tissue was sent for histopathological examination. We received an oophorectomy specimen weighing 154 grams and measuring 9.5 cm × 6.5 cm × 3.5 cm in size. External surface is grayish white and lobulated. Capsule was intact and smooth. Cut surface was solid, grayish brown, edematous to gelatinous with multiple tiny cysts of varying sizes, largest one measured 0.5 cm in diameter. Most of the cysts were located at the periphery (Figure 1). Histological examination of the right ovary showed stromal edema with separation of stromal cells by abundant pale staining fluid, multiple follicle cysts, occasional corpus luteum, areas of hemorrhage and few dilated lymphovascular channels. The superficial cortex was spared (Figure 2). No evidence of secondary pathology seen after extensive sampling. Based on histopathological examination, final diagnosis of ovarian edema of right ovary was made. Postoperative period was uneventful and patient did well on 4 months of regular follow-up.
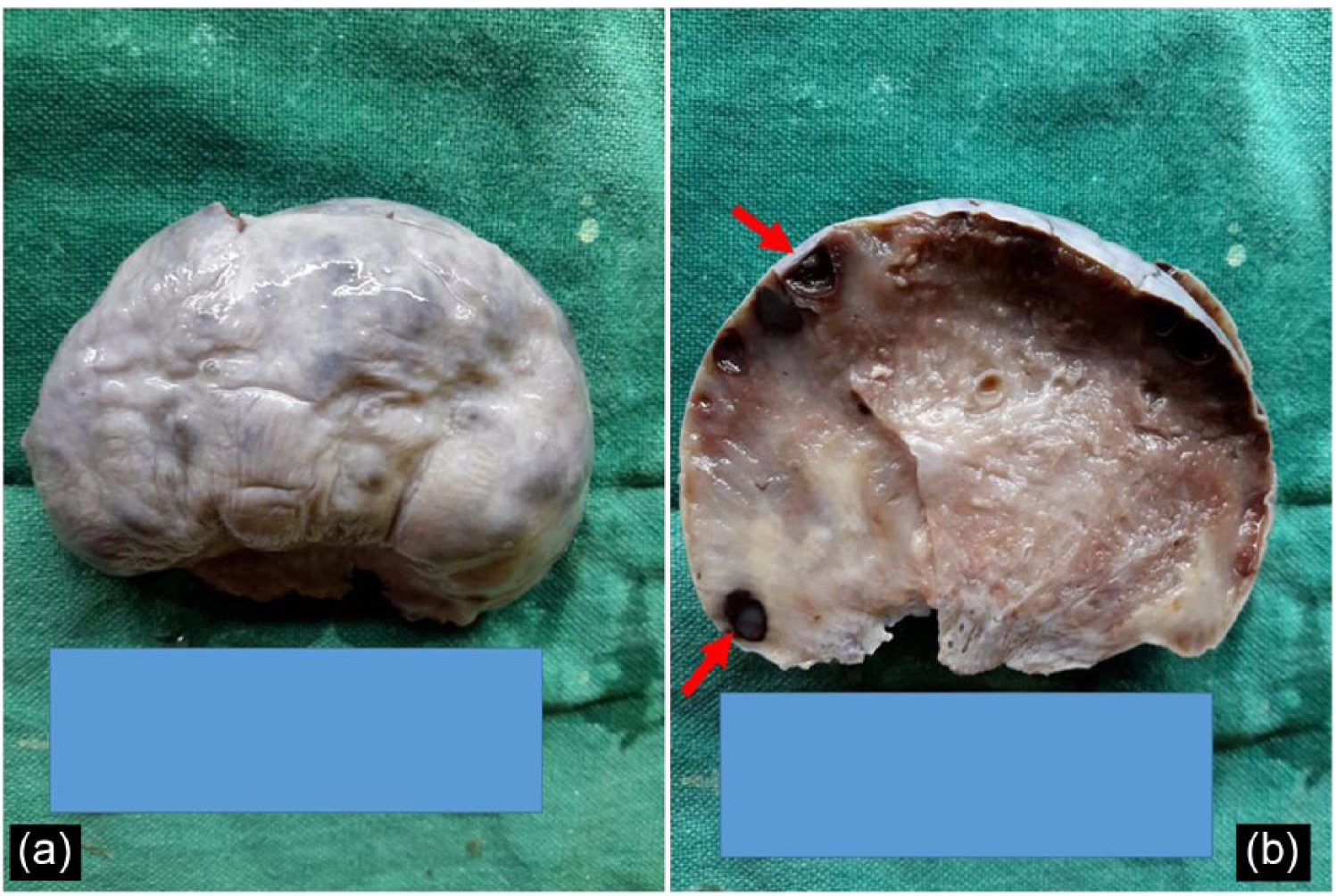
(a) Large ovarian mass with grayish white, lobulated external surface and intact capsule; and (b) solid, grayish brown, edematous to gelatinous cut surface with multiple tiny cysts (red arrow) at periphery.

(a) Stromal edema with follicular cysts and hemorrhage (red arrow), (b) stromal edema (blue star) with corpus luteum (green arrow) and areas of hemorrhage, (c) spared superficial cortex and (d) stromal cells separated by abundant pale staining fluid (blue star).
This specimen was routinely submitted in the Department of Pathology for histopathology; therefore, informed consent was taken from the patient for any academic and publication use of gross and microphotographs.
Discussion
Massive ovarian edema (MOE) was first described in 1969.3–6 MOE was defined as an accumulation of edema fluid within the ovarian stroma separating normal follicular structures forming as a primary or secondary edema.2–6 MOE divides into primary MOE, which occurs in a normal ovary constituting approximately 85% of MOE cases, and secondary MOE, which occurs when there is an ovarian mass or cyst or due to drugs used for ovulation induction.3,4 MOE mostly affects young postmenarchal female in their reproductive age with an average age of 20 years and can involve unilateral or bilateral ovaries.4,5 However, MOE also reported in prepubertal and postmenopausal females. 4 In the present case, patient was postmenarchal 28-year-old female without any history of drug intake for ovulation. There was no secondary pathology seen in the ovary even on extensive sampling.
In primary MOE, pain abdomen is the commonest clinical presentation and the nature of pain depends on the character of the torsion.2,3 An acute abdomen is the clinical presentation if torsion is acute, and profound diffuse pain occurs if torsion is incomplete and intermittent. 2 However, abdominal distension, menstrual irregularities and infertility may also be the clinical presentation.2,4 In the present case, patient was presented with acute abdomen.
Hormone-related symptoms such as precocious puberty, virilization and masculinization in MOE are due to stromal cell luteinization that occurs as a response to torsion and subsequent ischemia.2,3 Eden 5 proposed that derangement of a local paracrine factor, such as insulin-like growth factor, epidermal growth factor or cytokines, is responsible for edema and abnormal hormone production. Some authors suggest that primary MOE may occur due to lymphatic dysfunction with inhibition of vascular endothelial growth factor receptor (VEGFR) prohibiting the normal luteinization of the ovary and causes hormone-related symptoms. 2 No hormone-related symptoms were observed in the present case.
Radiological findings of MOE has been also reported in the literature using USG and magnetic resonance imaging (MRI).4,7 The most important indicator of MOE on radiology is the presence of multiple ovarian follicles situated around the periphery of the cortex of the enlarged ovary.3,4,7 However, it may be missed and over-diagnosed as ovarian neoplasm on radiology. In the present case, the patient was diagnosed to have solid ovarian tumor on USG.
MOE ranges from 5.5 to 35 cm in maximum dimensions. 6 The affected ovary is soft, fluctuant with oozing of thin edema fluid on cutting.4,6 Microscopic features are presence of edema fluid, widely separating the ovarian stromal cells, presence of atretic follicles and compressed cortical stroma at the periphery.2,4,6 The focus of luteinized cells is seen in approximately 40% cases.2,4,6 Necrosis and hemorrhage are unusual.2,4,6 The electron microscopic principal finding is the presence of both myofibroblasts and fibroblasts in the stroma, which is thought to be as a reaction to the edema.2,4 In the present case, the diagnosis was made on histology postoperatively.
Ovarian neoplasm that exhibits edematous or myxoid appearance comes in close differential diagnosis includes fibroma, ovarian fibromatosis, ovarian myxoma and sclerosing stromal tumor.1,6
Fibroma occurs commonly in patients with age more than 30 years.1,6 These are hormonally inactive and present with abdominal mass and ascites. 1 Cut surface is firm, solid, flat, gray white with whorled appearance.6,8 Histology reveals circumscribed/nodular lesion with diffusely arranged spindle-shaped cells forming collagen, follicles are present at periphery and rarely luteinized cells are seen.1,6,8 Tumor displaces the ovarian structures and adjacent ovary may be hyperplastic. 1 In MOE, age is almost less than 30 years, often hormonally active, involves the ovary uniformly and incorporates the ovarian structures. 1
Fibromatosis is characterized by proliferation of collagen-producing spindle cells enveloping normal follicular structures and thickening of the superficial cortex.1,8 The similar age range, clinical manifestations and the overlap histological features suggest that ovarian fibromatosis and MOE are closely related and may reflect differing morphologic expressions of partial or intermittent torsion leading to venous/lymphatic obstruction.1,3
Ovarian myxoma is a rare entity with ages ranging from 13 to 65 years and usually present with an asymptomatic unilateral adnexal mass which has soft and cystic consistency.1,8 Microscopy reveals spindle-shaped and stellate cells in myxoid background which are not seen in MOE.1,8
Sclerosing stromal tumor mostly present at age less than 30 years, and tumor is solid, well demarcated, whitish mass with occasional edema and cyst formation.1,8 Microscopically, there is marked pseudolobulation consisting spindle-shaped cells and round cells, prominent sclerosis around clusters and individual cells, and prominent vasculature, which are not seen in MOE1,8
Radiology along with serum tumors markers such as CA-125, β-HCG, lactic dehydrogenase and alpha-fetoprotein helps to differentiate MOE from dysgerminomatous and mixed germ cell tumors. 3
MOE had been over-treated in most of the cases reported with unilateral salpingo-oophorectomy as they are mistaken for ovarian neoplasm preoperatively.3,4,8 The suitable management of MOE is wedge resection which involves removal of 30% or more of the ovarian volume to exclude secondary MOE.2–4,8 At the time of surgery, frozen section examination is also valuable in the diagnosis and preventing unnecessary oophorectomy due to fertility issues.2–4,8 Laparoscopy can also be a therapeutic option for MOE as it combines diagnosis and therapy.2,3 Detort the twisted pedicle of ovary laparoscopically, allowing the ovary to recover its vitality. It should be followed by ovarian biopsy for diagnosis and then fixation of the ovary the posterior aspect of the uterus which prevents the further episode of torsion.2,3
MOE is a non-neoplastic disorder; therefore, conservative management must be considered where fertility preservation is needed.4,8 Use of oral contraceptives therapy for a few months after conservative management may be beneficial in MOE without the evidence of torsion. 3 There is no known medical management described for MOE. Present case was also over-treated due to preoperative diagnosis of fibroma clinically and radiologically.
Conclusion
MOE should be suspected in female of reproductive age group who presents with abdominal pain, radiologically showing solid enlargement of ovary with evidence of multiple peripheral ovarian follicles and normal serum tumor markers. In suspicion of MOE, definitive treatment should be performed only after pathological confirmation and to perform fertility-sparing procedures in young patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
