Abstract
Objectives:
To report a rare case of delayed Type IIIb endoleak secondary to fabric tear following implantation of a Medtronic Talent endovascular device.
Methods:
A 83-year old gentleman underwent elective endovascular aneurysm repair for infrarenal abdominal aortic aneurysm with a Medtronic bifurcated stent graft in 2008.
Results:
Seven years after the initial repair, imaging surveillance revealed significant endoleak and brisk aneurysm sac expansion due to Type IIIb endoleak secondary to endograft limb fabric tear.
Conclusions:
This case illustrates the imperative role of imaging surveillance in detection of long-term endovascular aneurysm repair complications. Furthermore, we discuss exclusion of the graft tear with aortouniiliac stent grafting as the treatment for this complication.
Introduction
Endovascular aneurysm repair (EVAR) has revolutionized the management of abdominal aortic aneurysms (AAA). This technique offers many short-term benefits over open surgical repair, including improved peri-operative morbidity and mortality. 1 Despite these benefits, there is question surrounding the long-term durability of EVAR. 1 Late endoleaks and subsequent sac expansion may result in post-EVAR rupture and death and are thus of particular concern. Type IIIb endoleaks arise from a fabric defect and is associated with high risk of aneurysm rupture. 2 We report the case of a very late Type IIIb endoleak from a hole in the fabric of a bifurcated Medtronic Talent graft, 7 years after implantation.
Case presentation
The patient is an 83-year-old gentleman who underwent elective EVAR for an asymptomatic 5.9 cm infrarenal AAA with a Medtronic Talent bifurcated stent graft (Medtronic, Dublin, Ireland) in 2008. The operation was performed successfully and uneventfully. The main body of the graft (30 mm × 20 mm × 140 mm) was deployed under fluoroscopic guidance via cannulation of the right femoral artery. The contralateral graft limb (14 mm × 18 mm × 19 mm) was deployed, along with a contralateral extension (20 mm × 20 mm × 79 mm). Finally, an ipsilateral extension (20 mm × 20 mm × 79 mm) was placed. Balloon dilation was performed of all junctions. Bilateral renal arteries and internal iliac arteries were patent. Completion angiogram revealed no Type 1 endoleak, with late filling of the sac secondary to Type II endoleak from the lumbar arteries (Figure 1). There were no complications peri-operatively, and the Type II endoleak ceased on the 1-month follow-up computerized tomography (CT). Post-operatively, this patient was enrolled in our EVAR surveillance program where patients are followed up with CT scan at 1 month, 6 months and annually thereafter to assess for graft-related complications. CT scan at 2-year follow-up demonstrated sac regression with no endoleaks (Figure 2).

Intra-operative completion angiogram during initial EVAR demonstrating no Type 1 or Type III endoleak.

Contrast-enhanced CT scan at 2-year follow-up after initial EVAR demonstrating sac regression and no Type III endoleak.
Seven years later in 2015, ultrasound revealed sudden expansion of his aneurysm sac from 5.2 to 8.0 cm within the 1-year scan. Based on initial surveillance ultrasound and subsequent CT scan, the source of endoleak was challenging to deduce; a Type 1a leak was initially suspected though stent migration or neck expansion was excluded (Figure 3). Angiogram was then performed revealing Type IIIb endoleak arising from a tear in the distal main body or proximal left graft limb (Figure 4). Given the rate of sac expansion, subsequent repair was urgently performed. Intraoperatively, we confirmed the Type III leak and its location at the distal main body by doing selective balloon occlusions and simultaneous angiograms. We treated this endoleak with an aortouniiliac endograft and femorofemoral artery bypass in order to cover the site of Type IIIb endoleak. The main body (Medtronic Endurant II aortouniiliac (36 mm × 14 mm)) was deployed within the previous Talent graft at the same level as this previous device, landing distally in the left common iliac artery. This was further extended into the left common iliac artery using an Endurant II iliac limb (16 mm × 20 mm × 93 mm) to successfully exclude the Type IIIb endoleak. A Talent occluder (20 mm) was placed in the right common iliac artery to prevent retrograde filling (Figure 5). The patient recovered well and was discharged 2 days post-operatively.

Contrast-enhanced CT scan demonstrating Type IIIb endoleak and sac expansion 7 years after initial EVAR.

Pre-operative angiogram demonstrating Type IIIb endoleak from proximal left graft limb.
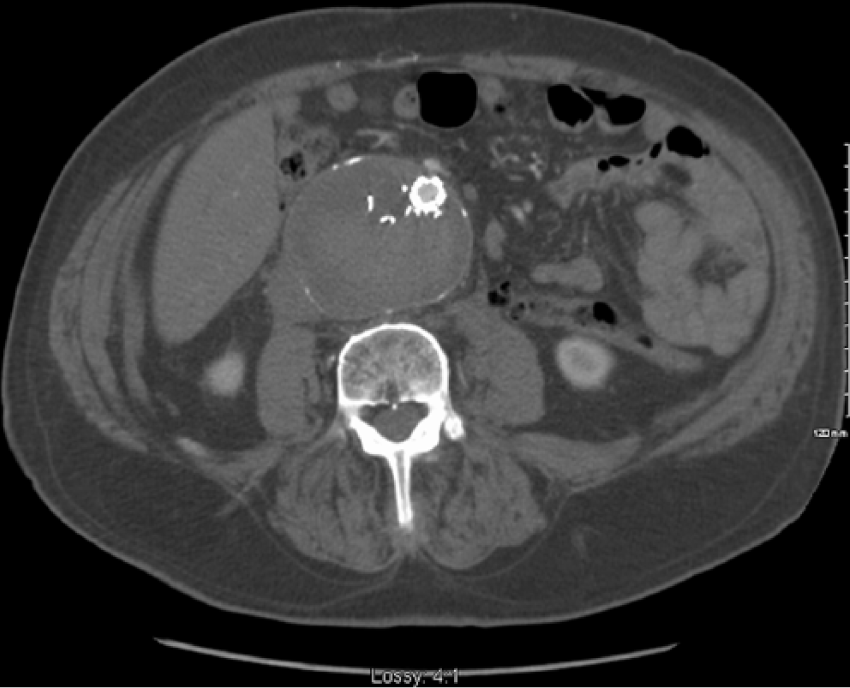
Post-operative CT scan demonstrating successful exclusion of Type IIIb endoleak with aortouniiliac Endurant II graft. No endoleak is demonstrated and aneurysm is stable.
Repair of this significantly delayed Type IIIb endoleak was carried out successfully. Post-operative contrast-enhanced CT scan revealed no filling of the aneurysm sac, suggesting the endoleak has sealed itself off. Clinical follow-up has been unremarkable. This patient will continue to undergo EVAR imaging surveillance to detect future complications. Patient consent was obtained for this case report.
Discussion
This case highlights (1) the occurrence of late fabric tears, even after several uneventful years, with a current-generation stent graft; (2) the diagnostic challenges associated with late Type IIIb endoleaks; (3) the need for ongoing surveillance of EVAR several years after initial implantation; and (4) the selective role of aortouniiliac devices in the repair of Type IIIb endoleaks.
The Medtronic Talent abdominal stent graft system has been available on the market since the mid-1990s. The graft has undergone two revisions: change of graft material to monofilament polyester in 1999 and chemical etching of the nitinol springs to improve graft durability and fatigue resistance, as well as, relocation of the connecting bar from medial to lateral side of the graft to improve kink resistance in 2002. Furthermore, the graft delivery system has undergone several revisions; the latest Xcelerant Hydro Delivery system was commercialized in 2008. Our patient underwent EVAR with the latest Talent stent graft and delivery system. 3
Late tears in Medtronic Talent endograft fabric are exceedingly rare. To our knowledge, this is the only report of 7-year-delayed Type III endoleak secondary to graft tear in a Talent stent graft. Few reports exist of this delayed complication in other stent graft systems. Specifically, Type IIIb endoleak has been reported in Cook Zenith grafts at 19 months, 4 3 years, 5 5 years 6 and 7 years; 7 in the Ancure graft at 2.5, 8 3 8 and 6 years; 9 and Gore excluder at 1 year. 10 According to Medtronic, microscopic holes and fabric wear may arise in the Talent stent graft; however, the majority of these defects should resolve spontaneously by normal hemostatic mechanisms. 3 In our case, we observed a rapid 2 cm aneurysm sac expansion in less than 1 year. This resulted from a fabric tear and represents a major device failure.
This case also illustrates the diagnostic challenge presented by delayed onset Type IIIb endoleak. Our surveillance protocol calls for annual screening in asymptomatic patients post-EVAR. However, given its sudden onset and the rapid rate of aneurysm sac expansion, this complication may be difficult to detect and prevent by means of surveillance. Furthermore, the defect is challenging to visualize on ultrasound and CT scan. In the current case, the endoleak was initially suspected to arise from around the proximal neck of the endovascular device (i.e. Type 1a endoleak), based on both the screening ultrasound and the follow-up CT scan. Angiography was necessary to make the correct diagnosis. Numerous angiograms were taken wherein individual segments of the graft were excluded using an inflatable balloon to isolate the specific location of the fabric defect. The defect was localized to the left proximal ipsilateral iliac limb or distal main body. Type IIIb endoleaks can lead to sac expansion and rupture; 11 the lifetime relative risk of rupture has been calculated at 8.95. 2 As such, urgent follow-up is recommended in cases where post-EVAR surveillance reveals sudden large-volume endoleak and rapid sac expansion, even if the patient is clinically asymptomatic.
Given the potential for devastating complications, urgent repair for Type IIIb endoleaks is indicated. Repair can be performed via open or endovascular approach; the standard treatment modality has not yet been established. 12 The open approach provides definitive repair for the failed endograft procedure; however, patients may have higher peri-operative morbidity and increased mortality. 1 Therefore, endovascular options if feasible are favored. Sometimes it is possible to deploy a new bifurcated graft within the existing graft to seal the Type IIIb endoleak; 11 however, this approach is complicated and requires custom-sized stent grafts. The technique employed in this case involved relining the Talent stent graft with an aortouniiliac device, Medtronic Endurant II, and performing a femorofemoral bypass. This approach has been used in other cases of Type IIIb endoleak.11–13 This technique is straightforward and allows for successful exclusion of the torn graft segment. Complications are minimal and comparable to those experienced by patients undergoing sole femorofemoral bypass including hematomas, graft infection and graft thrombosis. Mortality for this repair is reportedly low at 0.7%, secondary to graft infection. 14 Furthermore, this technique can be performed with standard-sized endografts, which is valuable in cases where emergent intervention is necessary.
This report describes the first known case of late Type IIIb endoleak secondary to major defect in the fabric of the Talent abdominal endograft. This is a potentially lethal complication that illustrates the necessity of long-term post-EVAR surveillance.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
