Abstract
Objective:
Viral hepatitis, particularly hepatitis B virus and hepatitis C virus, is the leading cause of global liver-related morbidity and mortality. Concomitant infections of hepatitis B virus, hepatitis C virus, and tuberculosis are risks of hepatotoxicity and death due to antituberculosis therapy. Hepatitis and human immunodeficiency virus coinfection poses challenges in treating hepatotoxic patients and leads to mortality during antituberculosis treatment. Thus, this study aimed to determine the prevalence of hepatitis B virus and hepatitis C virus infections, and associated factors among human immunodeficiency virus-positive and human immunodeficiency virus-negative tuberculosis patients attending public health facilities, Northeast Ethiopia.
Methods:
A comparative cross-sectional study was conducted among 229 tuberculosis patients from January 1 to April 30, 2021 in public health facilities’ tuberculosis treatment centers. Study participants were selected using a consecutive sampling technique. Data on sociodemographic and other risk factors were collected using an interviewer-based pretested questionnaire by trained data collectors. Anti-hepatitis C virus and hepatitis B surface antigen were determined in serum using enzyme-linked immunosorbent assay. Data were entered and analyzed using SPSS version 22. Logistic regression analysis was computed, and then variables with a p value <0.05 were considered as statistically significant.
Result:
The overall hepatitis virus infection among human immunodeficiency virus-positive and human immunodeficiency virus-negative tuberculosis patients was 14.03% and 8.14%, respectively. The prevalence of hepatitis B virus infection in human immunodeficiency virus positives and human immunodeficiency virus negatives was 10.5% and 6.4% and hepatitis C virus infection in human immunodeficiency virus positives and human immunodeficiency virus negatives was 3.5% and 1.75%, respectively. Hepatitis B virus and hepatitis C virus coinfections were not observed. Older age, history of problematic alcohol use, history of blood transfusion, ear–noise piercing, and history of multiple heterosexual partners were predictors for the hepatitis virus infection.
Conclusion:
Hepatitis virus infection increases morbidity and mortality of tuberculosis patients. Therefore, screening tuberculosis patients for hepatitis virus infection is necessary to reduce the risk of antituberculosis complications.
Introduction
Hepatitis B virus (HBV) and hepatitis C virus (HCV) are oncogenic viruses that cause acute and chronic hepatitis, affecting the liver adversely. 1 If untreated, chronic hepatitis may contribute to life-threatening conditions such as cirrhosis, hepatocellular carcinoma, liver failure,2,3 extrahepatic hepato-renal syndrome, and bacterial peritonitis.4,5 Chronic viral hepatitis infection affects 550 million people globally, compared to 33 million with human immunodeficiency virus (HIV). Overall, 296 million people are infected with HBV, while 58 million live with chronic HCV.6,7 Hepatitis virus infections are a significant public health problem causing a significant rate of mortality worldwide, accounting for over 1 million deaths per year worldwide.8,9
HBV and tuberculosis (TB) may coexist in patients from endemic areas and HIV is significantly associated with TB infections. Thus, HIV and/or HBV coinfections increase the risk of anti-TB drug-induced hepatotoxicity, which may necessitate the discontinuation of treatment. 10 The risk of anti-TB hepatotoxicity has been reported to be 3–5 times more than that in TB patients who do not have a viral infection as well as 14-fold increases in HIV and HCV coinfected patients. 11 Both HBV and HCV are clinically identical in both acute and chronic infection stages, but HCV is more likely to become a chronic disease than HBV. 12
The HBV epidemic mostly affects the WHO African Region and the Western Pacific Region, but the HCV epidemic affects all regions, with large variations between and within countries. 13 The burden of HBV and/or HCV in the Sub-Saharan population is high.8,14 About 6.1% of the African population has HBV infection and around 18 million people have chronic HCV infection. 8 Hepatitis B is endemic in Ethiopia. 2 Owing to the shared use of infected injection devices and blood, multiple sexual partners, 15 history of dental and surgical procedures, 16 those who inject drugs, HIV infection,17 –19 as well as the history of blood transfusion and tattooing 20 were the significant risk factors for acquiring HBV and HCV infection.
A revival of TB has been caused by the HIV/AIDS pandemic, resulting in increased morbidity and mortality worldwide. HIV and TB connect synergistically; each accentuates the other’s progression. In people with HIV, hepatitis coinfection is one of the leading causes of morbidity and mortality globally. Due to similar routes of transmission, coinfection of HIV with hepatitis (B and C) had substantial clinical effects due to the complicated clinical path, case management, and HIV therapy and vice versa.21 –23
TB/HIV coinfected patients are more prone to hepatotoxicity than HIV-negative TB patients who are infected with HBV.23,24 Thus, coinfections with HIV and/or viral hepatitis (HBV and HCV) raise the risk of anti-TB drug-induced hepatotoxicity and/or drug-induced liver injury and pose particular public health challenges and complications such as progressive liver fibrosis and cirrhosis, liver failure, and liver cancer.10,25 –27 HBV and HCV are risk factors for the development of liver dysfunction characterized by abnormal liver function tests and mortality during anti-TB therapy among TB/HIV coinfected patients. 28 Liver enzyme elevation in serum is common in these patients due to isoniazid and rifampicin chemotherapy.28 –30
Despite no further attempts made to escape these problems and/or risks, the WHO Global Health Sector Plan 2016–2021 has launched an elimination strategy to prevent and monitor HBV and HCV infection. 4 Thus, further study is very important to get epidemiological data which are crucial to control and monitor HBV and HCV infection during TB treatment. 15 This is also important to improve the quality of life of TB patients, reduce treatment failure or interruption, and promote effective management. 15 Furthermore, studies on HBV and HCV infection do not fully reflect a unique situation in TB patients with or without HIV. Thus, HBV and HCV infection among HIV-positive and HIV-negative TB patients has not been thoroughly investigated globally, especially in our country Ethiopia. 15 Moreover, very little is known about the rates of coinfection of HBV and HCV among TB patients.10,15,25 Therefore, this study aimed to determine the prevalence of HBV and HCV infections and their associated factors among HIV-positive and HIV-negative TB patients attending public health facilities’ TB treatment centers, Northeast, Ethiopia.
Method and materials
Study design, period settings, and population
A comparative cross-sectional study was conducted from January 1 to April 30, 2021. All public health facilities that delivered TB care and treatment at the TB Directly Observed Therapies (DOTs) clinic were included in this study. A total of 11 health facilities in Dessie town (Dessie Comprehensive Specialized Hospital, Boru-Meda General Hospital, Selam General Hospital, Dessie, Buanbua Wuha, Segno Gebaya, Metero, Kurkur, Boru Silasie, Gerado, Tita Health Center) and 4 health facilities in Kombolcha town (Kombolcha 02, 03, 05, and 07 Health Center) were included. Dessie and Kombolcha towns are located at a distance of 401 and 376 km, respectively, away from the capital city of the country, Addis Ababa. Based on the 2007 census conducted by the Central Statistical Agency of Ethiopia, Dessie town has a total population of 151,174 of whom 72,932 were men and 78,242 were women. Likewise, Kombolcha towns’ total population was 485,337 of whom 41,947 were men and 43,390 were women. In all health facilities, a total of 331 (74 HIV positives and 257 HIV negatives) TB patients whose age were 18 years and above were on anti-TB treatment at the TB DOTs clinic. All adult (18 years and above) HIV-positive and HIV-negative TB patients who were at TB DOTs clinic for anti-TB treatment during data collection period were included. On the other hand, TB patients who were not able to give written informed consent due to physical, mental, and critically ill conditions were excluded.
Sample size determination and sampling techniques
The sample size was determined using a double population proportion formula by considering the following statistical assumptions: 95% confidence interval (CI), power 95%, the ratio of HIV negatives to HIV positives = 3, taking the prevalence of HBV, p1 = 35.27% in HIV-positive TB patients and p2 = 15.1% in HIV-negative TB patients from the previous study done in Amhara region Ethiopia 31 based on the rules of sample size determination suggested by Geoff Fosgate for comparison of two proportions of unequal group sizes using the following formula. 32 The sample size for HIV positives and HIV negatives was 52 and 156, respectively. Finally, after adding a 10% nonresponse rate, the final sample size was 229 (57 HIV positives and 172 HIV negatives). Then, a consecutive sampling technique was used to recruit the study participants (Figure 1).

Enrollment procedure of HIV-positive and HIV-negative TB patients in the study.
Definitions of terms
Problematic alcohol use (PAU) was defined using the following four simple
Hepatitis infection was defined as the presence of HBV and/or HCV infections.
Chronic disease was defined as the presence of at least one of the diseases such as diabetes mellitus (DM), hypertension (HTN), cardiac disease, and renal disease.
HBV infection grade: The prevalence of positive hepatitis B surface antigen (HBsAg) was graded as high, intermediate, and low if it is ⩾8%, 2–7%, and <2%, respectively. 15
HCV infection grade was graded as high, moderate, or low endemic when the prevalence is >3.5%, 1.5–3.5%, and <1.5%, respectively. 15
Data collection and laboratory procedures
A pretested structured questionnaire adopted from literatures10,16,18,21,31 (Supplemental File 1) was used to collect sociodemographic, clinical, behavioral, and/or risky behaviors characteristics of the study participants. Data collectors such as nurses and laboratory technologists were trained on how to collect data, specimen collection, and manage the overall activities. Moreover, retrospective data such as type of TB, type of anti-TB medication, duration of anti-TB medication, and HIV status were collected from TB registration logbook for each study participant.
Five milliliters of venous blood was collected using a plain tube from each study participant by a laboratory technologist. The tube was labeled with a unique identification number at the time of collection. The blood sample was allowed to clot at room temperature and serum was separated by centrifugation at 5000 rpm for 15 min in each health facility. Then, serum samples were transported to the Ethiopian Red Cross Society Blood Bank, Dessie branch using a cold box and stored at −20°C until tested. Serum specimens were tested for HBV and HCV using AiD™ HBsAg enzyme-linked immunosorbent assay (ELISA; Wantai Hepatitis B Virus Diagnostics, Beijing, China) with a sensitivity of 100% and a specificity of 99.92% and AiD™ anti-HCV ELISAPlus (Wantai Hepatitis C Virus Diagnostics) with a sensitivity of 100% and a specificity of 99.55%.
Quality control
All quality control checks were done before, during, and after data collection to generate quality and reliable data. The structured questionnaire was pretested on 5% of the study participants in Haik Health Center. Data collectors were trained for an hour at each site before data collection began. Moreover, the quality of laboratory test results was assured using internal quality control of the ELISA and by using known negative and positive samples. Pre-analytical, analytical, and post-analytical stages of quality assurance were strictly followed. All laboratory analyses were carried out using standard operating procedures. Moreover, negative control and positive control samples were run in parallel with the patients’ sample and the result of the patients’ sample was validated if and only if the controls were passed.
Statistical analysis
Data were coded, cleaned, entered, and analyzed using SPSS version 22.0 (IBM, USA). The prevalence of HBV and HCV infections was computed. Fisher’s exact test was used to assess the differences in the proportions of hepatitis virus infections between HIV-positive and HIV-negative groups. Bi-variable logistic regression was computed and variables with a p value < 0.25 were entered into the multivariable logistic regression analysis to control confounding variables. Finally, variables with an adjusted odds ratio (AOR) and a p value
Ethical considerations
Ethical approval was obtained from the College of Medicine and Health Sciences Research and Ethics Review Committee of Wollo University. Written informed consent was obtained from each study participant. Finally, participants’ who were positive for HBsAg and/or anti-HCV antibody were linked to the nearby health facility for further management, consultation, and care.
Results
Sociodemographic and clinical characteristics of study participants
A total of 229 (57 HIV positives and 172 HIV negatives) TB patients who were on anti-TB medications were recruited (Figure 1). Out of the total study participants, 120 (52.4%) were females, of which 31 (25.8%) and 89 (74.2%) were HIV positive and HIV negative, respectively. The mean age of the participants was 34.1 years (ranging from 15 to 83 years) with a SD of ± 13.0 years. A majority of the study participants were less than 30 years of age (98; 42.8%); of these, 83 (84.7%) were HIV negatives. In addition, 150 (65.5%) participants were patients with pulmonary TB type of which 34 (22.6%) and 116 (77.4%) were HIV positives and HIV negatives, respectively. A majority of study participants (214, 93.4%) had no previous history of anti-TB medication. More than half (60.3%) of the study participants were on the intensive phase of treatment during data collection. Moreover, 15.7% of participants had chronic diseases; of these DM and HTN accounted for 7.0% and 3.9%, respectively (Table 1).
Sociodemographic and clinical characteristics of TB patients (N = 229) attending public health facilities, Northeast Ethiopia from January 1 to April 30, 2021.
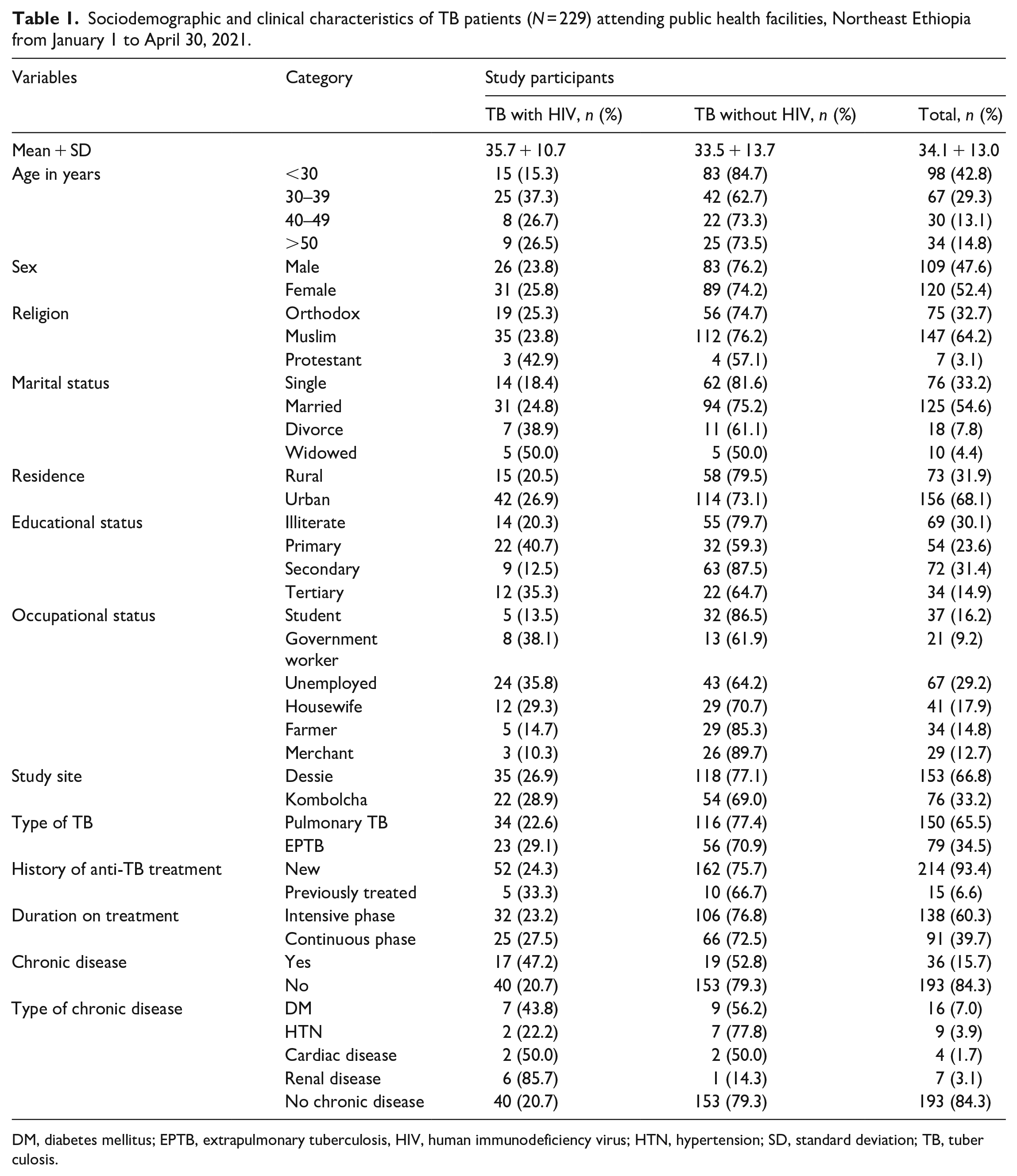
DM, diabetes mellitus; EPTB, extrapulmonary tuberculosis, HIV, human immunodeficiency virus; HTN, hypertension; SD, standard deviation; TB, tuberculosis.
Prevalence of HBV and HCV infections
Overall, 9.6% (95% CI = 6.1–14.0) were found positive for hepatitis infections; positive for at least one virus infection; that is, HBV or HCV. Moreover, the prevalence of hepatitis among HIV-positive TB patients was 14.0% (8/57; 95% CI = 5.3–22.8). Likewise, the prevalence of hepatitis among HIV-negative TB patients was 8.14% (14/172; 95% CI = 4.1–12.3). The prevalence of HBV infection among HIV-positive and HIV-negative TB participants was 10.5% (6/57; 95% CI = 3.5–19.3) and 6.4% (11/172; 95% CI = 2.9–9.9), respectively. Similarly, the prevalence of anti-HCV antibodies among HIV-positive and HIV-negative TB patients was 3.5% (2/57; 95% CI = 0.8–8.8) and 1.7% (3/172; 95% CI = 0.1–4.1), respectively (Table 2). HBV and HCV coinfections were not observed in both HIV-positive or HIV-negative TB patients. The proportion of TB-HIV-HBV, TB-HIV-HCV, TB-HBV, and TB-HCV was 2.6% (6/229), 0.9% (2/229), 4.8% (11/229), and 1.3% (3/229), respectively.
Statistical association test for HBV, HCV, and hepatitis virus infection among HIV-positive and HIV-negative TB patients attending public health facilities, Northeast Ethiopia from January 1 to April 30, 2021.

%: Percent, since assumption of chi-square (χ2) test was violated, Fisher’s exact test was used.
HBV, hepatitis B virus; HBC, hepatitis B virus; HIV, human immunodeficiency virus; TB, tuberculosis.
A statistically significant difference was not observed (p > 0.05) between HIV-positive and HIV-negative TB patients with HBV infection (p = 0.303), HCV infection (p = 0.600), and overall hepatitis virus infection (p = 0.200) (Table 2).
Factors associated with HBV and HCV infections
Factors associated with HBV infections
This study showed that tattooing (p = 0.031) was a significant predictor of HBV among HIV-positive patients and history of multiple heterosexual practices (p = 0.041) was also found to be independently associated with HBV infection among HIV-negative patients. Moreover, PAU was a significant predictor of HBV infection for both HIV-positive (p = 0.020) and HIV-negative patients (p = 0.048) (Supplemental File 2). Regardless of HIV status, multivariable logistic regression analysis showed that PAU (AOR = 7.102, 95% CI = 1.956–25.79, p = 0.003), previous history of multiple heterosexual practices (AOR = 2.437, 95% CI = 1.12–7.328, p = 0.036), and ear and noise piercing practices (AOR = 4.12, 95% CI = 1.357–12.55, p = 0.012) were found to be independently associated with HBV infection (Table 3).
Bi-variable and multivariable analysis of factors associated with HBV infection among TB patients (N = 229) attending public health facilities, Northeast Ethiopia from January 1 to April 30, 2021.

AOR, adjusted odds ratio; CI, confidence interval; COR, crude odds ratio; PAU, problematic alcohol use; STI, sexually transmitted infection.
Statistically significant.
Factors associated with HCV infections
There was no significant predicting factor (p > 0.05) seen in this study for both HIV-positive and HIV-negative TB patients (Supplemental File 3). Moreover, multivariable analysis of factors showed that only a history of blood transfusion (AOR = 14.5, 95% CI = 4.68–112.36, p ⩽ 0.0001) was independently associated with HCV infection among TB patients (Table 4).
Bi-variable and multivariable analysis of factors associated with HCV infection among TB patients (N = 229) attending public health facilities, Northeast Ethiopia from January 1 to April 30, 2021.
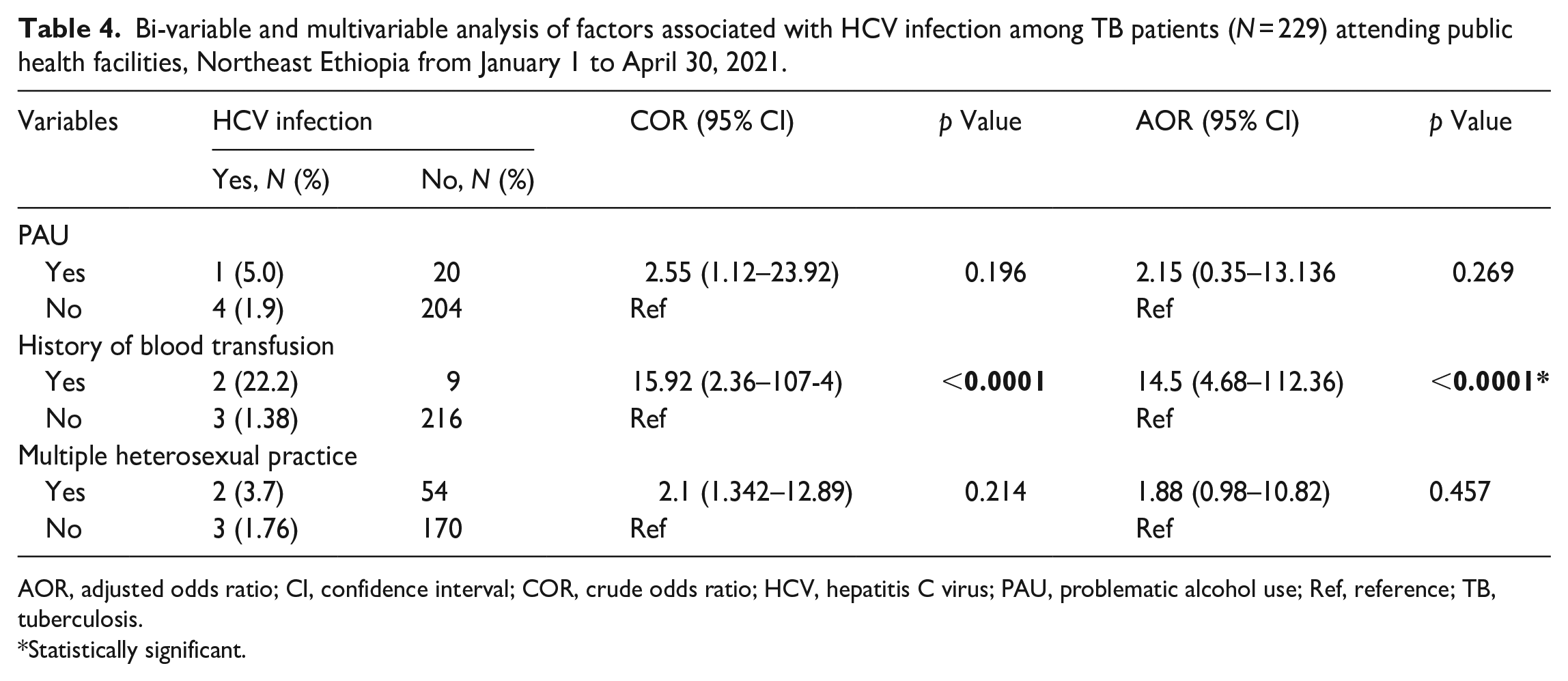
AOR, adjusted odds ratio; CI, confidence interval; COR, crude odds ratio; HCV, hepatitis C virus; PAU, problematic alcohol use; Ref, reference; TB, tuberculosis.
Statistically significant.
Factors associated with overall hepatitis virus infections
Hepatitis virus infection was independently associated with PAU (p = 0.005) and chronic diseases (p = 0.018) in HIV-positive TB patients. Similarly, multiple heterosexual partners (p = 0.013) were independently associated with hepatitis virus infection in HIV-negative groups. There was no common predicting factor for both groups (HIV positive and HIV negative) (Supplemental File 4). Furthermore, after adjusting the dependent variable all variables with a p value < 0.25; hepatitis virus infection was independently associated with a history of PAU (AOR = 5.219; 95% CI = 1.71–15.87; p = 0.004), history of blood transfusion (AOR = 4.73; 95% CI= 1.63–23.5; p = 0.020), ear–noise piercing (AOR = 2.93; 95%CI = 123–7.41; p = 0.048), and history of multiple heterosexual partners (AOR = 4.88; 95%CI = 1.57–15.14; p = 0.006) (Table 5).
Analysis of factors associated with overall hepatitis virus infection among TB patients (N = 229) attending public health facilities, Northeast Ethiopia from January 1 to April 30, 2021.

AOR, adjusted odds ratio; CI, confidence interval; COR, crude odds ratio; HIV, human immunodeficiency; PAU, problematic alcohol use; Ref, reference; TB, tuberculosis.
Statistically significant.
Discussion
TB and viral hepatitis are global health problems, and common in developing countries. 35 HBV and HVC infections are major public health problems leading to significant morbidity. 8 HBV and HCV infections are also responsible for drug-induced hepatotoxicity and/or drug-induced liver injury, liver dysfunction, and mortality during anti-TB therapy among TB/HIV patients. 28 Moreover, these viruses also pose particular public health challenges and complications like progressive liver fibrosis and cirrhosis, liver failure, and liver cancer.10,25 –27 Therefore, this comparative cross-sectional study was conducted to address the prevalence of HBV and HCV among HIV-positive and HIV-negative TB patients and their associated risk factors of the diseases at public health facilities of Northeast Ethiopia.
In our study, the overall prevalence of hepatitis infection (HBV and/or HCV) was 9.6%. The present finding implies that nearly one patient in every 10 TB patients might be infected by the hepatitis virus indicating that TB patients infected by the hepatitis virus might progress to hepatotoxicity and treatment failure as well as increased mortality rate.10,25 –28 Moreover, this finding indicated the need for regular screening of TB patients for viral hepatitis to reduce clinical complications. In addition, the finding of our study was higher than a study’s findings reported in Iraq, 2.33% 16 but lower than the findings reported in Cameroon, 16.16% 11 ; Sudan, 12% 36 ; and 16.3%. 37 These might be due to differences in the proportion of HIV patients involved in the study as well as differences in exposure status of participants. Moreover, 14.0% and 8.1% of HIV-positive and HIV-negative TB patients, respectively, were seropositive for at least one of the viruses. Despite the difference not being statistically significant (p > 0.05), the descriptive values of hepatitis virus infection were higher in HIV-positive TB patients compared with HIV-negative TB patients. This might be because these infections share a common mode of acquisition.21,38 In addition to the modes of transmission, HIV and HBV coinfections are common and potentiate each other 39 ; and HIV also poses an increased risk of reactivation of previously existing asymptomatic and chronic HBV and HCV infections 39 in relation to immune suppression which may exacerbate the disease progression. HIV has been a risk factor for the reactivation of HBV in patients with severe immunodeficiency, 40 because impairment of cellular immunity allows HBV to replicate more readily. 41
In our study, the prevalence of HBV among HIV-positive TB patients was 10.5%. This finding was comparable with the prevalence of HBV infection reported in Ethiopia, 8.92% 21 and Cameroon, 4.9% 11 but it was higher than a study conducted in Nigeria, 1.5%. 23 The high frequency of HBV infection observed in this study could be explained by lack of access to universal infection prevention and control measures including vaccination. 11 On the other hand, the present study was lower than a study documented in West Gojjam zone, Ethiopia, 35.27% 31 ; Nigeria, 50.7% 35 ; and Brazil, 36.9%. 10 These might be due to differences in sample size since other studies were conducted among larger sample sizes10,31 and differences in proportion of HIV-positive to HIV-negative participants where it was higher in a study done in Cameroon (1 to1.58) 11 than in this study (1 to 4). Similarly, the prevalence of HBV among HIV-negative TB patients was 6.4%, which was comparable with a finding reported in Pakistan with a prevalence of 5.5%. 42 Our finding was higher than a study finding in Iraq, 1.8% 16 but lower than the studies documented in Brazil, 20% 10 and 14% 43 and other African countries such as 12.7% in Cameroon, 11 12% 23 and 14.9% 35 in Nigeria, and 15.1% in Ethiopia. 31 These discrepancies might be due to variations in geographic regions or peculiarities of each environment. 31 Additionally, variations in study settings and participants who generally express risky behaviors such as men who have sex with men, illicit drug users, and intravenous drug users might contribute for the differences; nevertheless, none of these factors were not assessed in our study, and be considered as the limitation of this study. Moreover, the presence of occult HBV infection might be also the reason which was not possibly ruled out by the routine laboratory tests including our study supported by previous Cameroon findings. 44 However, previous studies in Brazil were quantified HBV-DNA which may rule out the presence of HBV-DNA in the absence of HBsAg.10,43
The prevalence of HCV in TB/HIV patients was 3.5%, which was comparable with a study reported a prevalence rate of 1.81% in Nigeria. 35 Based on this, the present finding was lower than a study reported 11% in Cameroon 11 and 46.09% in West Gojjam zone, Ethiopia. 31 These might be due sociodemographic differences such as lower proportion of high-risk groups in the study that is, proportion of HIV-infected patients in this study (25%) while a higher proportion of HIV-infected patients (38.6 %) was participated in a study done in Cameroon. 11 Furthermore, HIV increases the likelihood of acquiring HCV due to the shared route of transmission.
Moreover, the prevalence rate of HCV among HIV-negative TB patients was 1.75%. This was comparable with a previous findings reported in Iraq, 0.9% 16 and Sudan, 1% 37 but lower than a study reported in Cameroon, 4.23% 11 ; Nigeria, 5.4% 35 , Ethiopia; 17.3% 31 ; and Pakistan, 31%. 42 This might be due to variations in sample size, risk exposure, and government attention or concern for prevention and control of diseases. Moreover, sociodemographic variations and cultural variations such as body piercing and tattooing might contribute for the existence of this discrepancy.
In this study, HBV and HCV coinfections were not observed, which is supported by a study in Ghana. 38 However, our finding disagreed with a study reported in Nigeria, accounted for 2.9% of HBV-HCV coinfection. 35 The absence of coinfection could be due to the dominancy of HCV over the replication of HBV in the replication of coinfected cells leading to the suppression of HBV replication. The activation of cellular activities of HCV by micro-RNA contributes to the dominancy of HCV and suppression of HBV. 45 In this condition, occult hepatitis B can occur which is characterized by the presence of HBV-DNA in serum or liver in the absence of detectable HBsAg by currently available assays. 45 Therefore, the absence of diagnostic facilities that are used for the detection of occult HBV infection might be the probable reason for the absence of coinfection.44,45 Based on this, increased spontaneous clearance of HCV infection with unclear reason can reduce the possibility of HCV detection which, in turn, is the reason for the absence of coinfection. 46
HIV-positive participants who had chronic disease (p = 0.018) and a history of PAU (p = 0.005) had higher odds of hepatitis than HIV-negative participants which is consistent with the findings reported in Ethiopia.31,47 This might be due to the fact that individuals who use alcohol problematically may engage in unprotected sexual activity, which puts them at greater risk of contracting the virus. 48 Chronic diseases such as substance abuse, alcohol consumption habits, metabolic syndromes, DM, and renal disease might predispose these patients to acquire the diseases31,47,49 than HIV negatives. In addition, these factors are common risky behavior of HIV and hepatitis virus infection.
History of multiple heterosexual practice was an independent predicting risk factor of hepatitis virus infection among HIV-negative TB patients. This might not be independently explained in smaller sample size of HIV-infected patients. Furthermore, regardless of HIV infection, history of PAU, ear–nose piercing, and multiple heterosexual practice was a predictor for acquisition of hepatitis virus infection. However, history of transfusion was a significant predictor in overall analysis regardless of HIV status that could be explained as they were confounded by smaller sample size in each group (HIV positive and HIV negative). Moreover, sociodemographic characteristics and risky behavior variations between the two groups might contribute for differences in the risk groups.
In this study, the association between hepatitis infection and history of blood transfusion (AOR = 4.73; 95% CI = 1.63–23.5; p = 0.002) was statistically significant among TB patients regardless of HIV status. The possible explanation might be lack of improved laboratory screening methods for hepatitis infection from blood donors before or posttransfusion, since the virus can survive in traces of blood left on equipment. In addition, it can be related to the window period in cases of HCV where the individuals are asymptomatic but has viremia and is infectious to others. In this period, the individual antibody production status can be delayed and cannot be detected by routine anti-HCV detection method. Therefore, blood donation by asymptomatic individuals during this window period poses for hepatitis infection, particularly for HCV infection. 50 Nevertheless, HBsAg was the mainstay in the serology of HBV screening of blood donors; limited access to nucleic acid testing for HBV-DNA will be the reason for the possible transmission of HBV in HBsAg-negative donors to recipients. Therefore, occult HBV infection should be screened to reduce the risk of HBV transfusion–transmission with blood components from donors. 51 Simultaneous screening of HBV using anti-HBc and HBsAg might be useful to ensure the exclusion of infective donors and to reduce the possibility of HBV transfusion–transmission.
Participants who had a history of ear–nose piercing (p = 0.048) has been 2.93 times more likely to acquire hepatitis virus infection compared to their counterparts. This might be due to sharing and repeated use of unsterilized instruments for the tattooing procedure, not using infection protective tools that can prevent transmission of infections, and tattooing procedure being done by unskilled and/or traditional personnel.
In addition, PAU was significantly associated with hepatitis virus infection. Problematic alcohol users were about 4.219 times more likely to have hepatitis virus infection than non-problematic alcohol users. This might be due to increment of the vulnerability among participants who had a history of PAU to commit unprotected sexual intercourse31,52 which exposes them to acquiring the virus more easily than non-problematic alcohol users.
Moreover, participants who had a history of multiple heterosexual partners (more than two) regardless of HIV status had 4.88 times higher hepatitis virus infection compared to their counterparts. This was supported by findings reported in Southern Ethiopia.53,54 The high prevalence rate might be because HBV and HCV infections are sexually transmitted infections and the transmission rises with the duration of sexual activity and the number of sexual partners. It is also supported by the fact that adolescents having sex with multiple partners are at higher risk of acquiring sexually transmitted infections.54,55
Nevertheless, sociodemographic characteristics were not statistically significant (p > 0.05), descriptive findings showed that the skewed rate of hepatitis virus infection was higher among participants with age greater than or equal to 50 years of (p = 0.154) and female (p = 0.345) participants. The proxy for lifetime exposure in which the cumulative risk of hepatitis infection is linked to sexual and percutaneous exposures over time 10 might be the plausible reason for a higher proportion among ⩾50 years of age. Additionally, the coordinated effect of age and HIV such as impaired immune system in elderly individuals might contribute for the increased risk of developing hepatitis virus infection.21,40,41 This indicates the need for the coordinated effort in the healthcare system to improve the patients’ lives. Regarding gender, the positivity rate of hepatitis virus infection in our study was higher among females than males (11.7% versus 7.3%; AOR: 1.78; 95% CI = 0.78–6.89, p = 0.345). The anatomy and physiology of female’s reproductive organ favors for the acquisition of hepatitis viruses31,54 than male’s.
Limitation of the study
This study has certain limitations like risky behaviors of hepatitis virus infection such as illicit drug users and intravenous drug users were not assessed. Due to lack of resources, quantitative real-time polymerase chain reaction was not used. Thus, occult HBV infection was not ruled out which might underestimate the prevalence of hepatitis virus infection particularly HBV infection. Despite these limitations, the results of a study can validly be generalized to the population from which the sample was drawn as the samples were taken in random, which ensures that the sample is representative of the population.
Conclusion and recommendation
The overall prevalence of HBV and HCV infection was graded as intermediate and moderate, respectively. History of PAU and chronic diseases were independently associated with the overall hepatitis virus infection (HBV or HCV) among HIV-positive TB patients. Only history of multiple sexual partners was a significant predictor of hepatitis virus infection among HIV-negative TB patients. PAU, previous history of multiple heterosexual practice, and ear and noise piercing practice were independently associated with HBV infection, and blood transfusion was independently associated with HCV infection, regardless of their HIV status. The current finding implies the need for further attention in designing preventive and control strategy especially among high-risk groups such as HIV-positive patients as its medication exacerbates the hepatotoxicity among TB patients. Therefore, TB patients who are coinfected with HIV and hepatitis virus require close follow-up and a priority concern, and routine HBV and HCV screening. Furthermore, developing and implementing specific viral hepatitis (HBV and HCV) prevention and control guidelines with integrated risk reduction strategies in TB-HIV coinfected patients is highly recommended. In addition, further study should be undertaken with a higher sample proportion of HIV-positive and HIV-negative TB patients using advanced molecular methods.
Supplemental Material
sj-docx-1-smo-10.1177_20503121231166642 – Supplemental material for Hepatitis B and C virus infections and associated factors among HIV-positive and HIV-negative tuberculosis patients in public health facilities, Northeast Ethiopia: A comparative cross-sectional study
Supplemental material, sj-docx-1-smo-10.1177_20503121231166642 for Hepatitis B and C virus infections and associated factors among HIV-positive and HIV-negative tuberculosis patients in public health facilities, Northeast Ethiopia: A comparative cross-sectional study by Alemu Gedefie, Abdurahaman Seid, Genet Molla Fenta, Mihret Tilahun, Agumas Shibabaw and Abdurrahman Ali in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121231166642 – Supplemental material for Hepatitis B and C virus infections and associated factors among HIV-positive and HIV-negative tuberculosis patients in public health facilities, Northeast Ethiopia: A comparative cross-sectional study
Supplemental material, sj-docx-2-smo-10.1177_20503121231166642 for Hepatitis B and C virus infections and associated factors among HIV-positive and HIV-negative tuberculosis patients in public health facilities, Northeast Ethiopia: A comparative cross-sectional study by Alemu Gedefie, Abdurahaman Seid, Genet Molla Fenta, Mihret Tilahun, Agumas Shibabaw and Abdurrahman Ali in SAGE Open Medicine
Supplemental Material
sj-docx-3-smo-10.1177_20503121231166642 – Supplemental material for Hepatitis B and C virus infections and associated factors among HIV-positive and HIV-negative tuberculosis patients in public health facilities, Northeast Ethiopia: A comparative cross-sectional study
Supplemental material, sj-docx-3-smo-10.1177_20503121231166642 for Hepatitis B and C virus infections and associated factors among HIV-positive and HIV-negative tuberculosis patients in public health facilities, Northeast Ethiopia: A comparative cross-sectional study by Alemu Gedefie, Abdurahaman Seid, Genet Molla Fenta, Mihret Tilahun, Agumas Shibabaw and Abdurrahman Ali in SAGE Open Medicine
Supplemental Material
sj-docx-4-smo-10.1177_20503121231166642 – Supplemental material for Hepatitis B and C virus infections and associated factors among HIV-positive and HIV-negative tuberculosis patients in public health facilities, Northeast Ethiopia: A comparative cross-sectional study
Supplemental material, sj-docx-4-smo-10.1177_20503121231166642 for Hepatitis B and C virus infections and associated factors among HIV-positive and HIV-negative tuberculosis patients in public health facilities, Northeast Ethiopia: A comparative cross-sectional study by Alemu Gedefie, Abdurahaman Seid, Genet Molla Fenta, Mihret Tilahun, Agumas Shibabaw and Abdurrahman Ali in SAGE Open Medicine
Footnotes
Acknowledgements
The authors would like to thank Dessie Blood Bank for providing laboratory setup, equipment, reagents, and facilities for laboratory investigations. All health facilities, study participants, and data collectors are acknowledged for their active participation and cooperation during data collection.
Authors’ contributions
AG, AS, and GM were involved in proposal writing and designing the study and participated in analysis and interpretation of data. AG, AS, GM, MT, and AS were involved in data collection and drafting of the original article. AG, AS, AS, and AA finalized the write up of the manuscript. All authors critically revised the manuscript and read and approved the final manuscript for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The study was done from TB patients from anti-TB clinics of health facilities of the Dessie and Kombolcha towns. Supportive letter to conduct this clinical study (cross-sectional study) was obtained from College of Medicine and Health Sciences community service, research and postgraduate office, Wollo University after being assessed and reviewed by the college research review Committee (RRC) (Registration number CMHS/RRC/0380/2021). A permission letter was obtained from the Dessie and Kombolcha town health offices and then in each health institutions.
Written informed consent
After discussing the purpose and method of the study, written informed consent was obtained before data collection. Illiterate participants provided written informed consent after the consent form was read to them and they signed it with their finger. Only patient identifiable codes were used to maintain the confidentiality of individuals’ identities and the data were analyzed only for the intended purpose. The results were communicated in an aggregated manner to the district health office. All methods were carried out in accordance with the Declaration of Helsinki guidelines and regulations.
Trial registration
Not applicable.
Data availability
The data can be obtained from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
