Abstract
Background:
Fibromyalgia is a chronic pain disorder characterized by widespread musculoskeletal pain, fatigue, and psychological distress. Despite its high prevalence, the neurobiological mechanisms underlying fibromyalgia remain poorly understood. This study aims to investigate the structural and functional correlates of the left thalamus in fibromyalgia patients using a combined Granger causality and voxel-based morphometry approach.
Methods:
We analyzed functional and structural magnetic resonance imaging data from 20 female fibromyalgia patients and 20 female age-matched healthy controls. Voxel-based morphometry was employed to evaluate grey matter volume differences, while Granger causality analysis explored effective connectivity between the left thalamus and other brain regions. Psychological metrics, including pain intensity, pain self-perception, anxiety, depression, and pain catastrophizing, were assessed to determine correlations with neuroimaging findings.
Results:
Our analysis revealed significantly increased grey matter volume in the left thalamus among fibromyalgia patients compared to controls, alongside a decrease in the right amygdala. Correlation analyses indicated strong associations between increased thalamic volume and pain intensity, pain catastrophizing, and anxiety. Granger causality analysis demonstrated decreased outflow from the left thalamus to key regions involved in pain processing, including bilateral caudate, right fusiform, and right hippocampus, as well as increased inflow from areas associated with visual and cognitive processing, including right inferior occipital cortex, right middle frontal cortex, and right inferior parietal cortex, suggesting a complex interplay between structural changes and psychological factors in pain processing.
Conclusions:
This study provides novel insights into the neurobiological underpinnings of fibromyalgia, highlighting the role of the left thalamus in both pain perception and emotional regulation. Our findings underscore the necessity for a multi-faceted understanding of fibromyalgia that integrates neuroimaging, psychological assessments, and their implications for future therapeutic interventions. Further research is warranted to explore the longitudinal effects of treatment on structural and functional brain changes in fibromyalgia.
Keywords
Background
Fibromyalgia (FM) is a complex, chronic pain disorder characterized by widespread musculoskeletal pain, fatigue, sleep disturbances, and cognitive dysfunction. It affects approximately 2%–4% of the population, predominantly women, and is often associated with a range of comorbid conditions, such as depression, anxiety, and irritable bowel syndrome.1,2 The pathophysiology of FM is not fully understood, but it is thought to involve central sensitization, where the central nervous system becomes hyper-responsive to sensory stimuli. 3 This heightened pain sensitivity is believed to result from abnormal processing in both the spinal cord and brain. 4
In recent times, voxel-based morphometry (VBM) has been used to study deficits of grey matter volume (GMV) in several disorders either on a voxel-by-voxel or seed-to-voxel scale.5,6 VBM is a technique that allows for the assessment of GMV in the brain, which reflects the density of neuronal cell bodies and synapses in a given region. 7 Changes in GMV have been linked to a variety of neurological and psychiatric conditions, including chronic pain disorders like FM. Alterations in GMV in FM patients have been reported in key brain regions related to pain processing, such as the postcentral gyri, amygdalae, hippocampi, superior frontal gyri, anterior cingulate cortex (ACC), and prefrontal cortex.2,8 Mosch et al. 6 also reported decreased GMV in FM patients in the bilateral middle temporal gyrus, parahippocampal gyrus, left dorsal anterior cingulate cortex, right putamen, right caudate nucleus, and left dorsolateral prefrontal cortex, and increased GMV in the bilateral cerebellum and left thalamus. 6 These regions are integral to the perception and modulation of pain, as well as emotional and cognitive processing, suggesting that structural changes in these areas may underlie the chronic pain experience in FM.
In addition to structural changes, understanding the effective connectivity between brain regions is essential for elucidating the neural correlates of FM. Granger causality (GC) analysis has been widely employed in neuroimaging studies.5,9–11 This method is a statistical technique used to depict information flow by determining whether one time series precedes and allows the prediction of another time series. Thus, the neural activity in one brain region precedes and enables the prediction of the neural activity in another. Even though VBM and GCA have been used in several FM studies, no study has explored the effective connectivity of the left thalamus in FM. This study aims to investigate the neural correlates of FM through a combined GC and VBM approach.
The thalamus plays key roles in pain processing. Research has consistently demonstrated that the thalamus processes not only the sensory aspects of pain but also integrates emotional and cognitive components, making it a pivotal structure in understanding chronic pain conditions like FM.6,12 We hypothesized that structural differences between FM and HC subjects will have a causal effect on brain functioning in pain perception and processing. By examining both structural and functional aspects of brain connectivity, we hope to uncover the underlying neurobiological mechanisms that contribute to the chronic pain experience in FM, ultimately paving the way for more effective therapeutic interventions.
Methods
Participants
The dataset used in our study (Rest and T1 scans taken before listening to music and pink noise) is a publicly available neuroimaging dataset collected from a cross-sectional study data of 20 female FM patients and 20 healthy controls (HCs), which can be accessed at https://openneuro.org/datasets/ds001928/versions/1.1.0. 13 This dataset originates from a larger, previously published studies13–15 examining neurobiological correlates of music induced analgesia in FM, including structural and functional magnetic resonance imaging (MRI) data (taken before and after listening to music and pink noise) alongside psychological assessments. We selected this dataset because it provides high-quality, well-characterized neuroimaging data with comprehensive clinical and psychological measures, enabling us to investigate the structural and functional brain alterations associated with FM within a robust and standardized framework. Additionally, its accessibility ensures transparency and reproducibility of our analyses. FM patients were recruited from the Hospital General of the Secretaria de Salud and an FM help group, both in the city of Queretaro, Mexico. The inclusion criteria for FM individuals were: (1) age 18 years or older, (2) female, (3) meeting the American College of Rheumatology (ACR) 1990 classification criteria 1 and the ACR 2010 diagnostic criteria 16 for FM by a trained rheumatologist, (4) right-handed, and (5) spontaneous and continuous pain in daily life (Visual Rating Scale > 5, average of a month). 17 The exclusion criteria were: (1) inability to move or walk, (2) uncontrolled endocrine problems, (3) neurological diseases (e.g., stroke, epilepsy, traumatic brain injury), (4) auditory problems, (5) pregnancy and/or breastfeeding, and (6) MRI contraindications. HC subjects were enrolled and age-matched to the recruited FM participants. The inclusion criteria were: (1) healthy adult females, (2) right-handed. The exclusion criteria were: (1) acute or chronic pain (e.g., osteoarthritis), (2) pregnancy and/or lactating women, and (3) MRI contraindications. Written informed consent was obtained from each participant before the study, which was conducted following the Declaration of Helsinki. Patients received no compensation for taking part in the study. Ethical permission was obtained from the Bioethics Committee of the Institute of Neurobiology, UNAM Juriquilla, Queretaro, Mexico (IRB number: 007.H-RM). FM patients were asked not to use painkillers on the day of testing. HC subjects were screened to ensure none of them experienced any type of pain on the day of testing.
To evaluate pain before MRI scanning, pain intensity (PI) was measured only in FM patients, using a verbal rating scale (0 = no pain, 10 = worst pain possible). Both FM and HC subjects answered the following questionnaires: the Spanish version of the Pain Catastrophizing Scale (PCS) to measure thoughts and feelings when experiencing pain, 18 State-Trait Anxiety Inventory (STAI) to measure both immediate (state) and broad (trait) emotional, cognitive, and behavioral elements of anxiety, 19 and Center for Epidemiologic Studies Depression Scale (CESD), a questionnaire for depression and depressive disorder. 20 Helplessness, pain magnification, rumination, pain self-perception (PSP), and FM years were also recorded for symptom assessment. English versions of PCS, STAI, and CESD are shown in Supplemental Figures 1–3 respectively. FM patients’ data were taken in 2010 and the HCs subjects’ data were taken in 2016.
MRI acquisition
The image acquisition was performed with a 3-T GE Discovery MR750 scanner (HD, GE Healthcare, Waukesha, WI, USA) and a commercial 32-channel head coil array. High-resolution T1-weighted anatomical images (15 min scan) were obtained using the FSPGR BRAVO pulse sequence: plane orientation = sagittal, TR = 7.7 ms, TE = 3.2 ms, flip angle = 12°, matrix = 256 × 256, FOV = 256 × 256 mm2, slice thickness = 1.1 mm, number of slices = 168, gap = 0 mm, slice order = interleaved, view order = bottom-up. A gradient echo sequence was used to collect rs-fMRI data (5 min scan) using the following parameters: plane orientation = axial, TR = 3000 ms, TE = 40 ms, flip angle = 90°, matrix = 128 × 128, FOV = 256 mm2, slice thickness = 3 mm, voxel size = 2 × 2 mm, number of slices = 43, gap = 0 mm, slice order = interleaved, view order = bottom-up.
Data preprocessing
Functional data preprocessing
The first five time points were discarded to remove the T1 saturation effects, followed by slice time correction, realignment, co-registration of T1 images to corresponding functional images, segmentation, normalization by Diffeomorphic Anatomical Registration using Exponentiated Lie Algebra, 21 and resampling to 3 mm3 × 3 mm3 × 3 mm3 voxels, nuisance covariates regression using Friston 24, 22 spatial smoothing with an 8-mm full width at half maximum (FWHM) Gaussian kernel, linear detrending to remove slow, non-stationary trends in the signals, and filtering using a bandpass filter of 0.01–0.08 Hz to capture the relevant resting-state oscillations and further stabilize the data by reducing low-frequency drifts and high-frequency noise. Subjects with a maximum translation >2 mm or rotation >2° were excluded from further analysis.
Structural data preprocessing
High-spatial-resolution T1-weighted MRI data were processed using the Computational Anatomy Toolbox in statistical parametric mapping software (SPM12). All T1 images were first checked for artifacts and reoriented to adjust image origins at the anterior commissure. Segmentation was then done to separate the T1 images into grey matter (GM), white matter, and cerebrospinal fluid and resampled to a volume image resolution of 3 mm3 × 3 mm3 × 3 mm3. After the data quality and sample homogeneity check, the segmented GM images were smoothed using an 8-mm FWHM Gaussian kernel. Total intracranial volume (TIV) was estimated for each subject. The smoothed GM images were used for subsequent analyses.
VBM analysis
To determine the GM alterations in FM, a two-sample t-test as implemented in SPM12 was performed on the smoothed GM images of the two groups. Age and TIV were used as covariates for statistical analyses. Family-wise error rate (FWE) correction was employed for multiple comparison corrections.
Partial correlation analysis
To determine the relationship between abnormal GM regions and FM symptom metrics, we extracted regions that showed significant GM differences between FM patients and HC subjects and performed a partial correlation analysis. FM metrics used include; FM years, rumination, pain magnification, STAI scores, CESD scores, PI, PCS scores, PSP scores, and helplessness. Age effects were regressed out as a covariate.
Granger causality analysis
We used the signed path coefficient-based GCA implemented in the REST toolkit (http://www.restfmri.net). GCA assesses effective connectivity by examining directed influences between brain regions, which reflects a statistical association rather than definitive causality. While longer scan durations can improve the reliability of effective connectivity estimates, several previous studies have demonstrated that resting-state fMRI data of this length are sufficient for detecting consistent and significant directed connectivity patterns using GC analysis.9,23 To mitigate the potential limitations of a relatively short time series, we employed preprocessing steps such as temporal filtering and nuisance regression, and we used robust statistical methods to control for multiple comparisons. The GCA was performed on preprocessed BOLD time series sampled at a TR of 2 s, establishing a discrete, regular sampling interval. This sampling rate determined the temporal resolution of the analysis, with each time point representing 2 s of brain activity. The causal lag in GCA is modelled concerning this sampling interval, with the effective lag corresponding to one or a few TRs. Although the hemodynamic delay inherent to the BOLD signal limits temporal precision, prior research has demonstrated that resting-state GCA using TRs around 2 s can reliably infer directed interactions within brain networks,5,24 supporting the validity of our approach in investigating directional influences in the context of FM. GCA was performed on a voxel-wise basis for all the voxels. The left thalamus region with Montreal Neurologic Institute coordinates (−14, −21, −2 with radius = 7 mm) was selected from a two-sample t-test of a whole-brain voxel-based morphometric comparison between FM patients and HC subjects. The thalamus generally plays a key role in pain processing 25 and have been reported to show structural changes in FM. 6 This region also correlated with STAI, rumination, PI, and PC and was, therefore, selected for the GCA analysis. The order was set to 1, and GCA was Z-transformed. GCA from the seed region (left thalamus) to the whole brain (Outflow) and from the whole brain to the seed region (Inflow) were measured. Age and mean framewise displacement (mean FD) were regressed out as covariates during the analyses. FWE (p < 0.05) was used for multiple comparison corrections.
Post hoc power analysis
To determine whether the study had enough power to detect a significant effect. Mean and standard deviation values of FM patients (0.156404 ± 0.024185) and HC (0.116407 ± 0.018393) subjects in the left thalamus (key primary finding) was used for post-hoc power analysis. We first calculated the standardized effect size (Cohen’s d), followed by determining the power of a two-sample t-test based on this effect size, sample size, and a conventional significance level (α = 0.05, two-sided).
Results
Demographic and clinical characteristics
Our study conducted a post hoc analysis of previously collected data consisting of 20 female FM patients and age-matched 20 female HC subjects. These participants were originally recruited and characterized in an earlier study, 13 and we did not actively recruit or perform original data collection for this research. The demographic and clinical information of the participants are shown in Table 1. The two groups did not differ significantly in age. FM patients had a mean disease duration of 5.2 ± 5.0 years and a mean PI of 7.2 ± 1.6 indicating they were in severe pain. The interquartile range of the disease duration is 5 years. Compared to HCs, FM patients demonstrated higher scores in PC, helplessness, pain magnification, rumination, PSP, anxiety, and depressive symptoms.
Data demographics and clinical characteristics.

FM: fibromyalgia; HC: healthy controls; PI: pain intensity; PCS: pain catastrophizing scale; PSP: pain self-perception scale; STAI: state-trait anxiety inventory; CESD: center for epidemiologic studies depression scale.
VBM results
A two-sample t-test was used to determine the significant GMV differences between FM and HC subjects. From Figure 1 and Supplemental Table 1, our results revealed increased GMV in the left and right thalamus of FM patients while decreased GMV in the right amygdala was observed in FM patients.
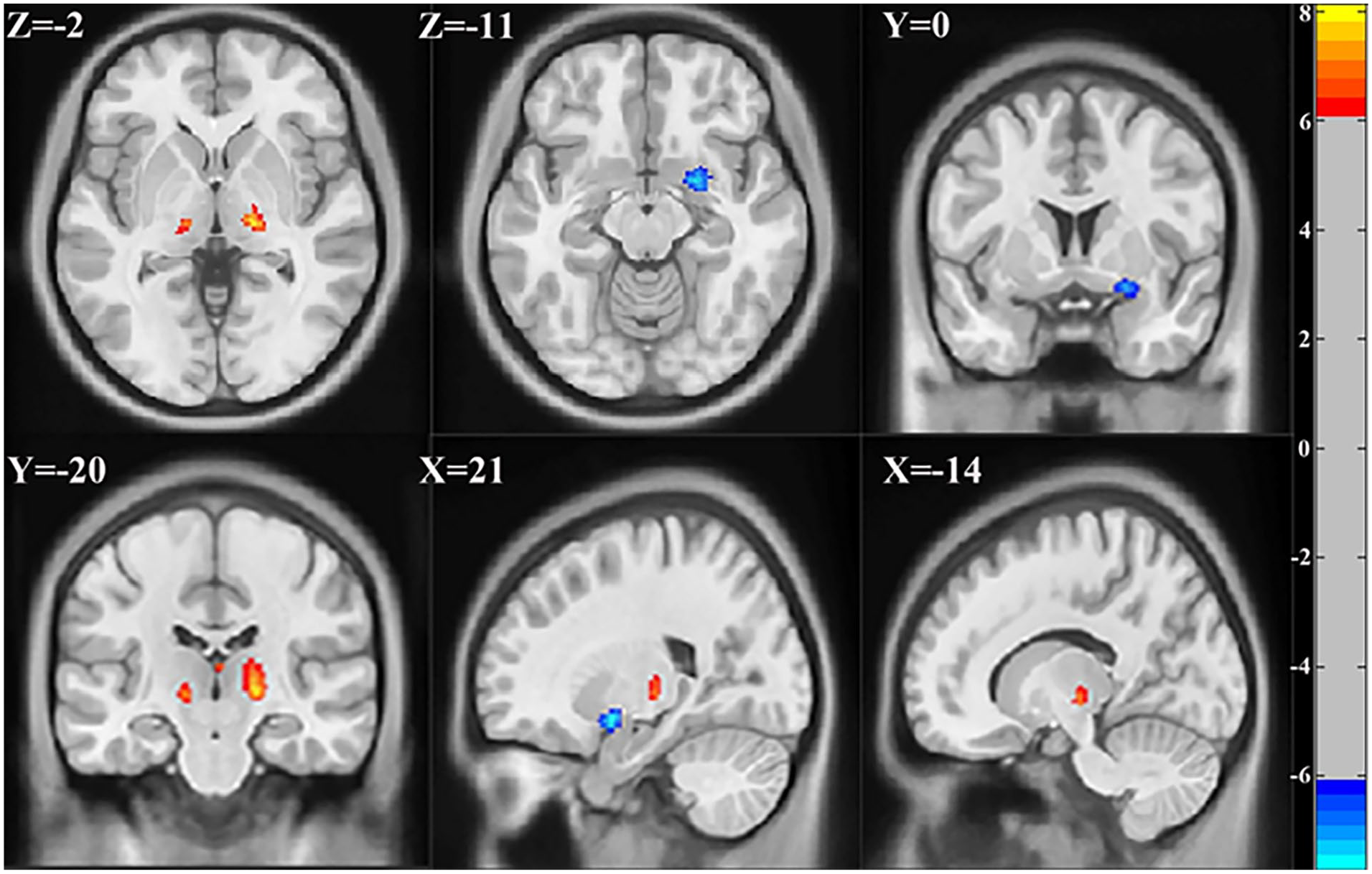
Regions showing significant GMV differences between FM and HC subjects. Hot (red) color represents increased GMV in FM and blue color represents decreased GMV in FM. FWE corrected p < 0.05.
Partial correlation results
To determine the relationship between GM anomalies and FM symptom metrics, we used regions that exhibited significant differences between FM and HC subjects for correlation analysis. Only significant correlations were reported. The partial correlation analysis revealed that increased GMV of the left thalamus in FM patients showed significant positive relationships with PC (r = 0.489, p = 0.034; Figure 2(a)), PI (r = 0.455, p = 0.050; Figure 2(b)), rumination (r = 0.522, p = 0.022; Figure 2(c)), and STAI (r = 0.507, p = 0.027; Figure 2(d)). This suggests that an increase in GMV of the left thalamus may result in increases in these FM symptoms.

Scatter plots of the partial correlation analysis. (a) Positive correlation between the left thalamus volume and pain catastrophizing. (b) Positive correlation between the left thalamus volume and pain intensity. (c) Positive correlation between the left thalamus volume and rumination. (d) Positive correlation between the left thalamus volume and STAI.
GCA results
Compared to HC subjects, FM patients revealed significantly decreased outflow from the left thalamus (seed region) to the bilateral caudate, right fusiform gyrus, and right hippocampus (Figure 3(a) and Supplemental Table 2). Also, FM patients exhibited significantly increased inflow from the right inferior occipital cortex, right middle frontal gyrus, and right inferior parietal to the left thalamus (Figure 3(b) and Supplemental Table 3).
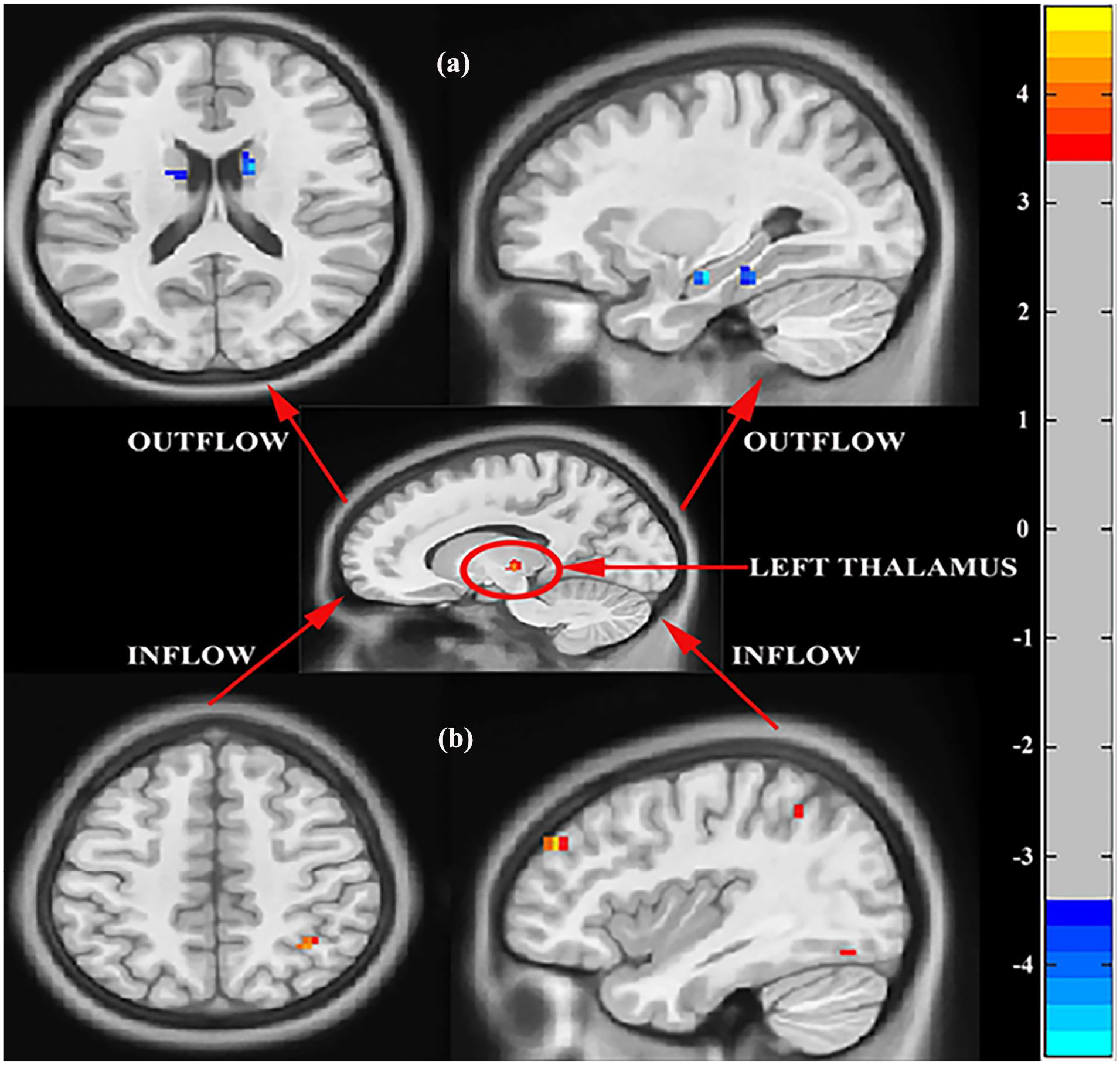
Regions showing significant causal connectivity to and from the left thalamus between FM and HC subjects (a) Causal effect from the left thalamus to whole-brain (Outflow). (b) Causal effects from whole-brain to the left thalamus (Inflow). The arrows indicate the direction of causality. p < 0.05, FWE corrected.
Post hoc power analysis results
Using the provided summary statistics (means and standard deviations) and a sample size of 20 in each FM and HC group, the post-hoc power analysis reveals a large effect size (Cohen’s d ≈ 1.86) and very high statistical power (≈99%) for detecting differences in GMV in the left thalamus. This indicates that the study is sufficiently powered given the data, enhancing confidence in the observed findings.
Discussion
This study sought to deepen the understanding of the neurobiological foundations of FM through the use of MRI. Our findings revealed significant structural brain differences between FM and HC subjects, including increased GMV in the thalamus and decreased GMV in the amygdala. Increased GMV in the left thalamus exhibited positive relationships with PI, PC, rumination, and STAI scores. Using the left thalamus as a seed, GCA revealed causal effects from the seed region to the bilateral caudate, right fusiform, and right hippocampus. Also, the right inferior occipital cortex, right middle frontal gyrus, and right inferior parietal cortex confer causal effects on the left thalamus.
In line with the findings of Lutz et al., 8 our study observed a decrease in GMV in the amygdala of FM patients. The amygdala is crucial for processing emotions and memories related to fear and is activated in response to pain-related fear. 26 Research indicates that amygdala retraining can lead to improvements in energy, physical health, pain, fatigue, and overall symptom distress in individuals with FM and chronic pain. 27 This suggests that the amygdala plays a significant role in the pathology of FM. The observed reduction in GMV may indicate difficulties in emotional regulation, particularly concerning fear and anxiety, which are commonly heightened in FM patients. 2
Consistent with previous studies, 6 increased GMV was observed in the thalamus of FM patients. The thalamus not only serves as a relay center but also plays a key role in processing nociceptive information before transmitting the information to cortical parts of the brain. 12 Research has shown that individuals with chronic pain conditions often exhibit structural brain changes, including increased GMV in pain-related regions, which may be a result of neuroplasticity in response to ongoing pain experiences. 25
Although our findings demonstrated significant positive correlations between increased GMV in the left thalamus and variables such as PC, PI, rumination, and anxiety, the lack of significant associations with other measured metrics, including years of FM duration, pain magnification, CES-D scores, PSP, and helplessness deserves attention. These psychological factors may interact in complex, non-linear ways with pain perception and brain connectivity, leading to inconsistent empirical findings. Variability in clinical and psychological profiles across FM patients often dilutes the strength of associations, making consistent correlations difficult to detect. 28 This absence of correlation aligns with the notion that neuroplastic changes in brain structure are often more reflective of current symptom severity and psychological factors rather than the chronicity or duration of the disease.29,30
The positive correlations found between the left thalamus and PC, rumination, PI, and STAI scores highlight the thalamus's role not only in nociceptive information processing but also in the emotional and cognitive dimensions of pain. A study by Seminowicz and Davis 31 demonstrated that individuals with chronic pain exhibited increased GMV in the thalamus, which was associated with the severity of pain and emotional distress, this explains the positive relationship between the increased GMV in the left thalamus and PI in the current study, suggesting that the increased GMV in the thalamus of FM patients may be a reflection of the brain’s attempt to adapt to the persistent pain experience. PC, characterized by an exaggerated negative mental set during actual or anticipated pain experiences, has been shown to exacerbate pain perception and increase emotional distress. 32 The correlation between increased GMV in the left thalamus and PC suggests that structural changes in this region may be associated with maladaptive cognitive patterns that amplify the pain experience.
Similarly, the tendency to focus on distressing thoughts (rumination) has been linked to increased pain sensitivity and emotional distress in chronic pain populations. 33 The positive relationship between the left thalamic GMV and rumination in FM patients indicates that alterations in thalamic structure may contribute to the cognitive processes that perpetuate the pain experience. This aligns with findings from studies showing that rumination can magnify pain perception and contribute to the maintenance of chronic pain states.34,35 Also, the positive correlation between the left thalamus and STAI scores further emphasizes the emotional component of pain in FM. Anxiety is a common comorbidity in FM, and heightened anxiety levels can exacerbate pain perception and lead to increased disability. 2 The thalamus’s involvement in emotional regulation suggests that structural changes in this region may influence the emotional responses to pain, potentially leading to a vicious cycle where increased pain sensitivity fuels anxiety, which in turn exacerbates pain. Research has shown that the thalamus interacts with various brain regions involved in the emotional and cognitive processing of pain. 8 The structural changes in the thalamus may therefore reflect a broader network of altered connectivity that influences both pain perception and emotional regulation in FM patients. However, the specificity of significant correlations to the left thalamus, rather than the right, may reflect lateralized thalamic alterations in FM. Structural and functional studies have demonstrated specific atrophy and connectivity changes in the left posterior thalamus, including the pulvinar and ventral posterior lateral nuclei, which are linked with pain processing and modulation. 36 This lateralization may relate to the functional specialization of thalamic subregions and their interactions with cortical areas involved in sensory and motor functions. Considering dexterity is important, as motor dominance and asymmetric sensorimotor integration could influence lateralized findings in thalamic connectivity and its correlations with FM metrics. 37
The GC analysis results revealed significant alterations in the effective connectivity of the left thalamus in FM patients compared to HCs. Specifically, FM patients exhibited decreased outflow from the left thalamus to several brain regions, including the bilateral caudate, right fusiform gyrus, and right hippocampus. Conversely, there was an increased inflow to the left thalamus from the right inferior occipital cortex, right middle frontal gyrus, and right inferior parietal cortex. These findings suggest that the left thalamus may play a central role in the altered neural circuitry associated with FM. The thalamus is known to be a critical relay station for sensory information and is involved in pain processing. 25 The decreased outflow from the thalamus could indicate a disruption in the normal processing and integration of sensory and emotional information, which is consistent with the heightened pain sensitivity observed in FM patients. 2 Also, the caudate, hippocampus, and fusiform alterations have been reported in FM,38–40 hence, the causal effects conferred on these regions by the left thalamus further strengthen their involvement in the pathophysiology of FM.
Also, the increased inflow to the thalamus from regions associated with visual and cognitive processing, such as the right inferior occipital cortex, right middle frontal gyrus, and right inferior parietal cortex may reflect compensatory mechanisms in response to the altered pain processing pathways. This aligns with the notion that FM is characterized by a complex interplay between sensory, emotional, and cognitive factors.3,8 These findings highlight the importance of understanding effective connectivity in FM, as it provides insights into how different brain regions interact and contribute to the chronic pain experience. Previous studies have shown that altered connectivity patterns in pain-related networks are associated with the severity of pain and other symptoms in FM. 41 The thalamus, together with the outflow and inflow regions found in the current study are part of the pain matrix, 15 therefore, disruptions in thalamic connectivity may underlie the pathophysiology of FM.
Interestingly, our study did not identify significant alterations within the default mode network (DMN), despite its reported relevance in FM. 42 The absence of significant findings related to the DMN in our study could be attributed to several methodological and clinical factors, despite the well-established relevance of DMN alterations in FM and its connectivity with the thalamus. Previous studies have shown that factors such as transient pain state variability among participants, patient heterogeneity, and differences in methodological approaches42,43 can obscure the consistent detection of the DMN alterations in FM.
Overall, our findings align with central sensitization theories, which suggest that persistent pain induces neuroplastic changes in pain-related brain regions, such as the thalamus. Increased GMV in such regions is commonly associated with chronic pain and reflects heightened pain sensitivity. A study found positive correlation between thalamic GMV and pain severity, supporting the notion that central sensitization results in the brain becoming more responsive to nociceptive input. 31 Decreased outflow from the left thalamus to pain-processing regions suggests a disruption in pain processing pathways, a hallmark of central sensitization. A study found similar altered brain connectivity in chronic pain patients, which reflects the brain’s adaptation to continuous pain stimuli, consistent with the findings in FM. 41 Increased thalamic GMV correlates with psychological factors such as PI, anxiety, and PC, reinforcing the idea that emotional and cognitive factors amplify pain perception. This aligns with central sensitization theories, which emphasize an integrated brain response to both sensory and emotional stimuli. Previous studies have highlighted the interaction between pain and emotional distress in chronic pain populations, supporting this concept.8,25
Understanding the brain changes associated with FM is crucial for improving its clinical management and treatment. Identifying brain regions connected to FM symptoms opens up the possibility of developing targeted therapeutic approaches. For example, interventions like cognitive-behavioral therapy could be tailored to address maladaptive thought patterns that correspond with GMV changes in areas like the left thalamus. CBT has already been shown to reduce pain and improve coping mechanisms in chronic pain populations, including individuals with FM. 44 These insights could also guide pharmacological treatments. Medications that influence neurotransmitter systems involved in pain processing, such as serotonin-norepinephrine reuptake inhibitors (SNRIs), may be especially beneficial for patients with marked GMV changes in pain-related brain regions. Previous research has found that SNRIs are effective in reducing pain and improving daily functioning in people with FM. 45 Furthermore, neurofeedback techniques could be considered as a potential therapeutic option. These techniques aim to help individuals regulate their brain activity, and studies suggest that neurofeedback can enhance self-regulation of brain function, offering potential benefits for chronic pain management, including pain perception and emotional regulation. 46
Although we observed significant structural changes and causal connectivity of the left thalamus in patients with FM, several limitations must be taken into account when interpreting these findings. A primary limitation is the relatively small sample size; while our results are encouraging, a larger cohort would enhance statistical power and provide further validation of these outcomes. Also, FM has many comorbidities, however, we could not determine whether these results are solely based on FM or other comorbidities such as chronic fatigue syndrome, diabetes, and depression, among others. Notably, FM patients in the current study exhibited high depression scores, suggesting that the brain changes observed may be influenced by depression. Future studies should determine whether these brain alterations observed are as a result of FM or these comorbidities. Furthermore, the current study employed only female subjects. Even though FM is predominant in women, the results of the current study might not be a direct reflection of all FM patients. Future studies should use datasets that include both genders to confirm these results. Additionally, many FM patients are treated with various medications to alleviate their symptoms, which could potentially influence brain structure. However, controlling for the effects of these medications poses a challenge. Future studies should consider the impact of pharmacological treatments to ascertain whether the brain changes observed are attributable to FM itself or if they are confounded by medication use. Last but not least, GCA results should be interpreted with caution, as they indicate directed influence or predictive relationships rather than causation in the strict biological sense.
Conclusion
In conclusion, this study provides significant insights into the structural and functional alterations associated with FM and chronic pain as a whole, particularly highlighting the role of the left thalamus in pain perception and processing. Our findings demonstrate increased GMV in the thalamus and decreased GMV in the right amygdala of FM patients, suggesting these regions might be involved in the heightened pain experience characteristic of this condition. Positive correlations observed between increased GMV in the left thalamus and various psychological factors, such as anxiety, PI, and PC, underscore the complex interplay between emotional and sensory processing in chronic pain. The identification of specific brain regions linked to FM symptoms and the enhanced understanding of the thalamus’s role opens the door for more targeted therapeutic approaches. Interventions such as cognitive-behavioral therapy, pharmacological treatments, and neurofeedback could all be tailored to address the underlying neurobiological and psychological factors contributing to the condition. By highlighting the necessity for integrated approaches that encompass neuroimaging and psychological assessments, we advocate for a shift in how FM is studied and treated. Future research efforts should focus on longitudinal studies to track changes in brain structure and connectivity over time and in response to different therapeutic interventions. Such investigations will be critical in elucidating the dynamic relationship between brain alterations, symptomatology, and treatment outcomes in FM. By targeting altered brain areas and addressing maladaptive thought patterns, these therapies have the potential to improve pain management, emotional regulation, and overall functioning in FM patients. The findings from this study not only enhance our understanding of FM as a complex interplay of brain and behavior but also underline the urgent need for global health initiatives that address the multifaceted nature of chronic pain disorders.
Supplemental Material
sj-docx-1-smo-10.1177_20503121251352360 – Supplemental material for Investigating the neural correlates of the left thalamus in women with fibromyalgia: A Granger causality and voxel-based morphometry approach
Supplemental material, sj-docx-1-smo-10.1177_20503121251352360 for Investigating the neural correlates of the left thalamus in women with fibromyalgia: A Granger causality and voxel-based morphometry approach by Elijah Agoalikum, Hongzhou Wu, Benjamin Klugah-Brown and Michael Maes in SAGE Open Medicine
Footnotes
Acknowledgements
We would like to thank Mrs. Lydia Fusieni for reviewing the manuscript prior to publication.
Ethical considerations
Ethical permission was obtained from the Bioethics Committee of the Institute of Neurobiology, UNAM Juriquilla, Queretaro, Mexico (IRB number: 007.H-RM).
Consent to participate
Written informed consent was obtained from each participant before the study, which was conducted following the Declaration of Helsinki.
Consent for publication
All authors have consented to the publication of this manuscript.
Author contributions
EA and HW: organized and pre-processed the data. EA and HW: formal analysis. EA, HW, and BK-B: statistical analysis and results. EA: writing the first draft. BK-B and MM: revision and editing. All authors contributed to the article and approved the submitted version.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
