Abstract
Objective:
This scoping review aims to examine studies led or participated in by clinical pharmacists or nurses, investigating the work performed and roles played by clinical pharmacists and nurses in managing patients’ adverse reactions such as oral mucositis, summarizing the existing research foundation and evidence.
Methods:
This scoping review followed the methodology defined by the Joanna Briggs Institute, and adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews checklist. Literature search was conducted in February 2024 across PubMed, Scopus, Cochrane, and Embase. Two independent researchers initially screened titles and abstracts in Covidence, followed by a full-text review. The extracted information and summaries were organized using Excel.
Results:
Twelve publications were included (randomized controls n = 8; retrospective descriptive studies n = 2; prospective cohort studies n = 2). Three publications studied clinical pharmacists providing pharmaceutical care to patients, while nine publications focused on nurses monitoring adverse reactions.
Conclusion:
In the chemoradiotherapy of head and neck cancers, the effective prevention and management of oral mucositis are crucial for improving patient prognosis. Clinical pharmacists and nurses, with their expertise and skills, play vital roles in the multidisciplinary treatment team, providing optimized treatment plans and comprehensive care support to patients. The direct care model implemented by pharmacists and nurses effectively alleviates symptoms and enhances the quality of life, thus fostering better treatment outcomes and patient satisfaction.
Introduction
Head and neck cancer (HNC) is the seventh most prevalent cancer worldwide. A significant proportion of HNC patients undergo radiation therapy (RT) as part of their treatment regimen. However, the oral and throat regions of these patients are often exposed to radiation, resulting in a high incidence of oral mucositis (OM). Research by Trotti et al. 1 reveals that approximately 80% of HNC patients receiving RT experience OM to varying degrees, with 34% of those undergoing conventional RT developing severe OM (grades 3–4). 1
The development of OM significantly increases pain levels in HNC patients and profoundly affects their quality of life (QoL). 2 Severe oral mucositis (SOM) is also closely associated with compromised nutritional status and weight loss. 3 A decline in overall health can reduce patients’ tolerance to RT doses and may even necessitate the suspension of treatment, thereby affecting therapeutic outcomes.
Additionally, OM can lead to mouth ulcers and open wounds, increasing the risk of infection. SOM may cause bacterial, fungal, or viral infections, 4 further exacerbating the patient’s condition.
Despite growing awareness, effective prevention and management of OM remain a persistent challenge in cancer care. Adequate prevention and treatment by healthcare professionals can potentially reduce the severity of radiation-induced OM, alleviate patients’ pain and discomfort, prevent malnutrition and infections, enhance treatment compliance, and thus improve patients’ QoL and treatment outcomes.
Guideline for the management of treatment-induced OM published by Multinational Association of Supportive Care in Cancer and International Society of Oral Oncology (MASCC/ISOO) recommend the use of mouthwashes or photobiomodulation to prevent OM in patients with HNC, with topical 0.2% morphine mouthwash to alleviate pain associated with OM. 5 The 2020 guideline from the European Oral Care in Cancer Group emphasizes oral care as central to preventing and minimizing oral complications during and after treatment. The oral care team may vary by medical setting but typically includes dental professionals, dietitians, nurses, physicians, and pharmacists. 6 The support provided by the team, along with good communication with patients, forms the core of all care plans, crucial for maintaining patients’ oral health. While the involvement of pharmacists and nurses is essential in managing treatment-related adverse reactions in oncology patients, research describing their specific roles in OM management remains limited. Studies as early as 2009 highlighted the significant contributions of clinical pharmacists in supportive care for oncology patients. 7 Subsequent research has shown the effectiveness of pharmacists in managing OM caused by chemoradiotherapy (CRT) in HNC patients,8–11 but there is a lack of specific measures and standardized workflows for pharmacist and nurse-led care for OM induced by RT and chemotherapy (CT) in HNC in real-world settings.
This scoping review aims to investigate the roles of clinical pharmacists and nurses in managing adverse reactions among HNC patients, particularly OM. It seeks to summarize existing research, highlight evidence-based practices, and explore opportunities for optimizing pharmacist- and nurse-led care for OM in clinical settings.
Methods
Study design
This review adheres to the JBI scoping review methodology updated in 2020 12 and Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) Checklist, 13 shown in Table 1.
PRISMA-ScR checklist of this scoping review.

Search strategy
From February to March 2024, two independent researchers conducted searches of Scopus database, the Cochrane Library database, Embase database, and PubMed database. Use the following terms: (((((((Head and Neck Neoplasms[MeSH Major Topic]) OR (Upper Aerodigestive Tract NCancer[Title/Abstract])) OR (UADT Neoplasm[Title/Abstract])) OR (Otorhinolaryngeal Cancer[Title/Abstract])) OR (Tongue Neoplasm[Title/Abstract])) OR (Oral Cancer[Title/Abstract])) AND (((Stomatitis[MeSH Major Topic]) OR (Stomatitides[Title/Abstract])) OR (Oromucositi*[Title/Abstract]) OR (Oral Mucositi*[Title/Abstract])) OR (Aphthous[Title/Abstract])) AND ((((((((((((((Pharmacists[MeSH Major Topic]) OR (Nurses[MeSH Major Topic])) OR (Nursing[MeSH Major Topic])) OR (Pharmaceutical services[MeSH Major Topic])) OR (Practice patterns, Nurses’[MeSH Major Topic])) OR (Practice patterns, pharmacists’[MeSH Major Topic])) OR (pharmaceutical care[Title/Abstract])) OR (pharmaceutical service*[Title/Abstract])) OR (Nursing*[Title/Abstract])) OR (Nursing care[Title/Abstract])) OR (Oncology Nursing[Title/Abstract])) OR (clinical pharmacist*[Title/Abstract])) OR (Pharmacist*[Title/Abstract])) OR (Nurse*[Title/Abstract])). These search criteria were selected to capture the management by all study clinical pharmacists and nurses of patients with HNC who developed OM following CRT.
Inclusion and exclusion criteria
Covidence system was used for conducting this scoping review. The criteria for exclusion and inclusion adopted in this scoping review are delineated as follows:
Exclusion criteria:
(1) Reviews.
(2) Non-English publications.
(3) Publications without interventions on OM.
(4) Publications prior to 2009.
(5) Publications with inaccessible full texts.
(6) Studies involving patients under the age of 18.
(7) Patients without head and neck tumors.
Inclusion criteria:
(1) Studies on interventions for OM.
(2) Nurse or pharmacist-led clinical studies.
(3) Studies over the past 15 years.
(4) Studies with interventions targeting adults.
Upon importing the searched publications from the four databases into Covidence, the exclusion criteria were utilized for the screening process within the Covidence system. Table 2 shows the research findings obtained from the four databases.
Searched database outcomes and Covidence importation results.

Data extraction
Two independent researchers extracted data from the 12 included publications. Following extraction, the researchers cross-verified the data to ensure consistency and accuracy. Data were extracted using Microsoft Excel in a tabular format, including publication year, authors, country, study type, patient disease type, treatment methods, primary intervenors, main intervention contents, intervention content for the intervention group, numbers of participants in the intervention group, intervention content for the control group, numbers of participants in the control group, methods and content of intervention effect assessment, intervention outcomes, study results, and study limitations.
Quality assessment
The quality of the publications was assessed, since there is no unified assessment method or tool for scoping reviews, we referred to the evaluation methods from two review papers. The assessment methodology proposed by Hölbl et al. 14 in their 2018 study involves assigning scores to the intrinsic quality of the included studies and their relevance to the research. González-García et al. 15 in 2021 subsequently applied this evaluative approach within the context of scoping review research. The quality assessment method utilized in this scoping review is predicated upon these two aforementioned studies. These studies received scores based on the criteria listed in the table, scored as follows: No or almost none = 0; Moderate, with relevant content = 1; Adequately described the issue = 2. The criteria are illustrated in Table 3.
Quality assessment questions and scoring criteria for included studies.

Result
Search results
All identified records were imported into the Covidence. 16 Of the 226 publications, duplicates were automatically identified and removed by the platform, leaving 168 publications. These records were independently screened by two researchers. During the initial screening, titles and abstracts were reviewed, and publications that did not meet the inclusion criteria were excluded, resulting in 117 exclusions. The full texts of the remaining 63 publications were then evaluated, and a second screening was conducted based on predefined full-text exclusion criteria. Ultimately, 12 publications met the inclusion criteria and were included in this scoping review. The detailed process of the literature search is illustrated in Figure 1.
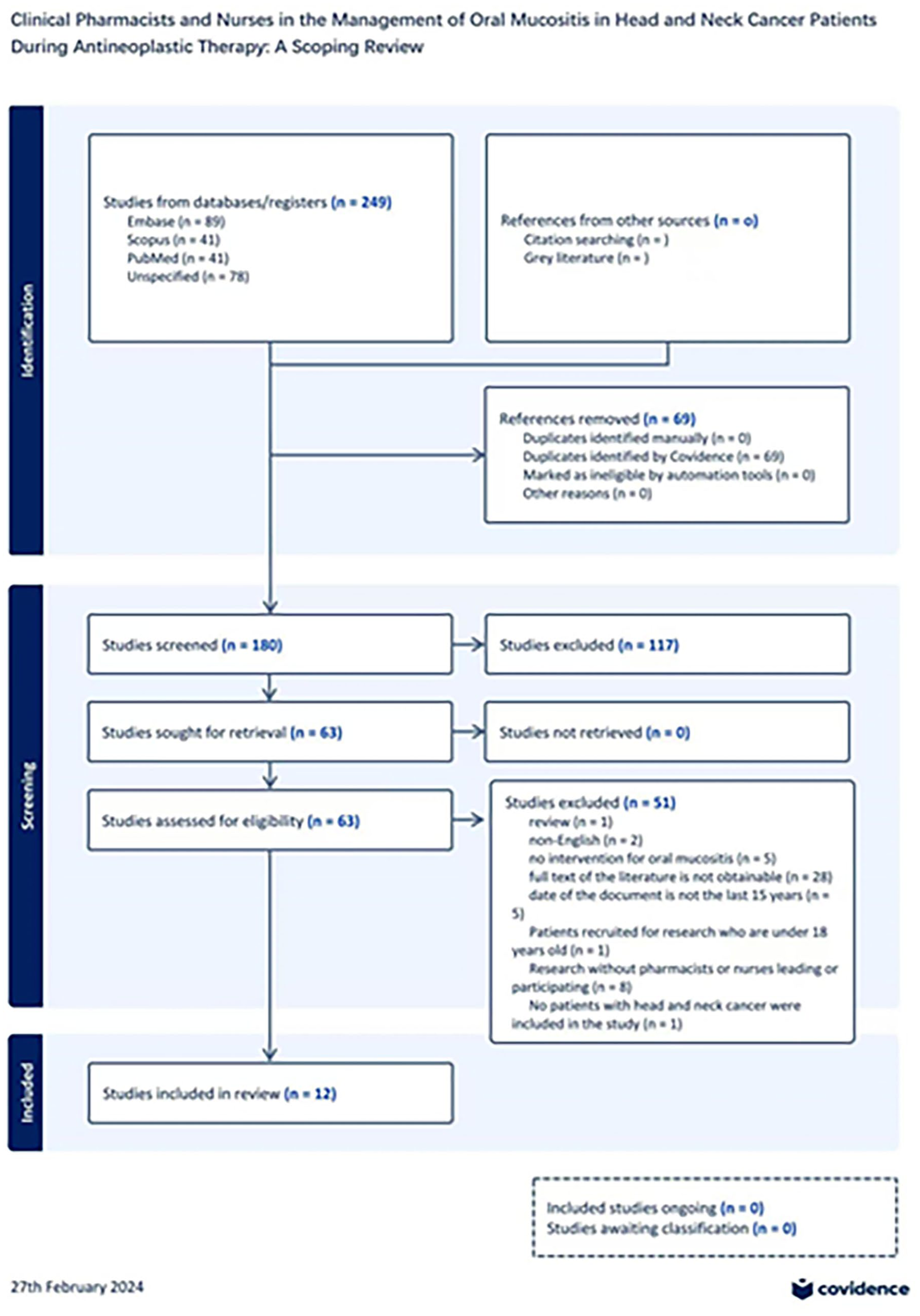
Literature search flowchart.
Quality assessment
The quality assessment was independently conducted by two researchers simultaneously, with the final results being consolidated and any discrepancies resolved. The quality scores of the included studies ranged from 5 to 8. Specifically, four studies scored 5, one scored 6, five scored 7, and two scored 8. The average scores for the assessment questions were as follows: Question 1 had an average score of 2 ± 0; Question 2 had an average score of 1.33 ± 0.49; Question 3 had an average score of 1.42 ± 0.79; and Question 4 had an average score of 1.67 ± 0.49. The overall average quality score of the 12 studies was 6.42 ± 1.16, indicating generally high methodological quality.
Data extraction results
The national backgrounds of the 12 studies were extracted, representing five countries: China, Turkey, Japan, Sweden, and the USA. The 12 included publications focused on the prevention or treatment of OM in patients with HNC. Of these, eight publications were randomized controlled trials, two were retrospective studies, and two were cohort studies. The basic information of the 12 extracted publications, as filtered and shown in Excel, is presented in appendix document, the document summarizes the study methods, participants, interventions, indicators, and outcomes.
Summary of research indicators
The extracted indicators included QoL (n = 6), Pain Index (n = 7), Incidence of OM (n = 7), OM Score (n = 2), Xerostomia Score (n = 1), Nutritional Status Score (n = 2), and Incidence of Esophagitis (n = 1). Studies consistently showed that interventions aimed at improving QoL and reducing pain index scores were effective. All six studies indicated that clinical interventions by pharmacists and nurses had a positive impact on the patients’ QoL. This underscores the indispensable roles of pharmacists and nurses in the clinical management of OM in patients undergoing radiotherapy and CT.
Seven evaluation metrics utilized 12 different scoring tools: five categories of tools were used to assess QoL, two were used for the Pain Index, two for OM scoring, one for assessing Xerostomia, two for evaluating Nutritional Status, and additionally, two studies employed the “Oral Assessment Guide” developed by Eilers et al. to evaluate the overall oral health of patients, it includes eight categories: voice, swallowing, lips, tongue, saliva, mucosa, gums, and teeth or dentures. The results are shown in Table 4.
Statistics of scoring tools used in included studies.

Nursing interventions for OM in included studies
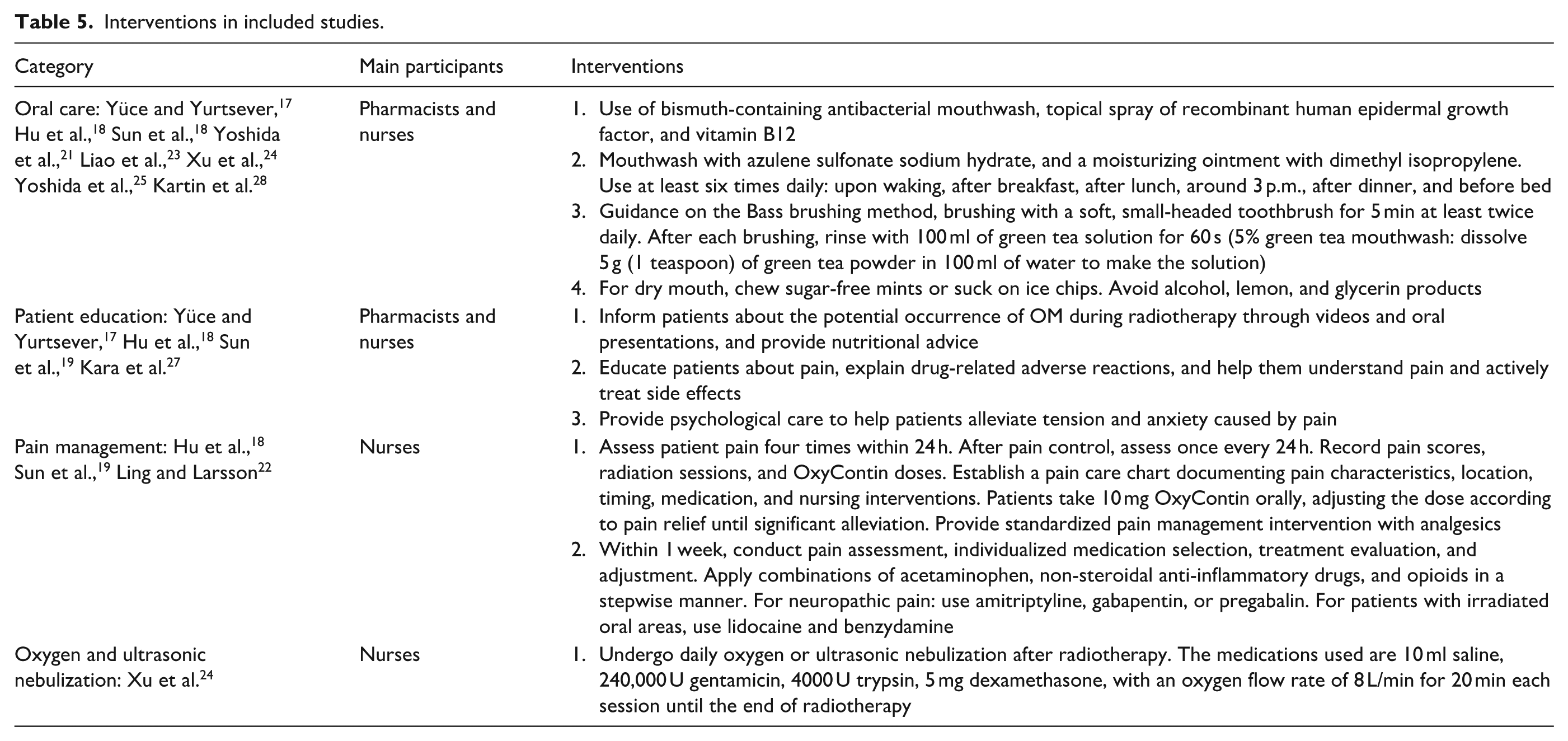
Eight studies reported nursing interventions for OM, which included pharmacological treatments and patient education. Nursing interventions incorporated seven basic care medications, eight medications for managing oral pain, and three nebulized treatments. Four studies provided three types of patient education: how to prevent and manage OM, medication education, and psychological care to alleviate anxiety and other emotions, as shown in Table 5.
Interventions in included studies.
Classification of results based on study objectives
Eight studies implemented preventive measures for OM. The results of two studies demonstrated that these preventive measures significantly reduced the severity of OM in the experimental group, as well as improved medication adherence and QoL in these patients.17,25 One study found that early intervention in patients reduced the pain and swallowing difficulties associated with OM compared to a control group that received later intervention. 22 Yoshida et al. 25 in their 2021 study highlighted that early monitoring by a multidisciplinary team can significantly decrease hospital stay lengths and weight loss in patients, reduce the frequency of analgesic use, significantly enhance patients’ QoL, and increase the cost-effectiveness of disease treatment. 17
Three studies focused on symptomatic interventions. Results from one of these studies showed a notable improvement in the QoL of the experimental group patients. Research by Fujii and others demonstrated that post-intervention, the incidence of Grade 2 or higher OM significantly decreased, and there was a significant improvement in patients’ QoL. 21
OM interventions led by clinical pharmacists
Three studies were primarily led by clinical pharmacists. In a retrospective study conducted in 2022 by Fujii et al., 20 comprehensive pharmaceutical services were provided to patients undergoing CT. The involvement of clinical pharmacists reduced the incidence of adverse reactions in the experimental group and also improved the patients’ QoL Yoshida et al.’s 25 study in 2021 provided patients with regular medication guidance and education, showing that the experimental group had a significant reduction in the use of local anesthetics and opioids, a notable decrease in hospital stay durations, and improved weight loss outcomes. In a study by Yoshida et al., 21 only medication guidance was performed, which showed that the incidence of Grade 2 OM was lower in the patient group guided by clinical pharmacists compared to the control group, with a reduction in steroid usage and improved medication adherence. This demonstrates that professional pharmaceutical care by clinical pharmacists has positive outcomes on patients’ oncological treatments, not only reducing the incidence of OM but also enhancing patients’ QoL and overall benefits.
Clinical care led by nurses
In nine studies where nurses were the principal investigators, the nurses provided patient education prior to the commencement of oncologic treatments, implemented standardized pain management interventions, assessed nursing outcomes after completing supportive care, and conducted follow-ups. The results of a randomized controlled trial conducted by Hu et al. 18 in 2018 indicated that early pain intervention could effectively improve patients’ QoL. The nursing outcomes from Duzova et al.’s 26 Nurse Navigation Program demonstrated effective prevention of weight loss, reduced incidence of OM, decreased xerostomia, swallowing difficulties, patient pain, and fatigue, and improved QoL. Kartin et al.’s 28 study showed that through nurse-led oral care and nutritional programs, patients’ nutritional statuses could be improved. Yüce and Yurtsever 17 focused solely on patient education in the intervention group, and the results indicated not only a reduction in the incidence of OM but also maintained stable QoL. This illustrates that nurses play pivotal roles in the care of oncology patients, providing clear insights into patients’ living habits and accurately delivering personalized patient education. They can respond promptly to adverse reactions, playing crucial roles throughout the treatment process.
Discussion
During the treatment of HNC, the incidence rate of OM ranges from 59.4% to 100%. In addition to the damage to normal cells caused by conventional RCT, other risk factors contribute to a higher incidence rate of OM in HNC patients compared to other types of cancer. These factors include tumor location, cumulative radiation dose, radiation field, radiation technique, and concurrent CT, all of which influence the incidence of OM. 29 This scoping review aims to investigate the roles of clinical pharmacists and nurses in managing adverse reactions among HNC patients, particularly OM. Our analysis of the 12 retrieved studies was summarized across 8 key aspects. The evaluation of study indicators revealed that the majority of studies focused on QoL scores, pain scores, and the incidence of OM. However, conditions such as xerostomia, nutritional risks, and esophagitis-related infections were less frequently investigated, and the assessment tools used were not standardized. These clinical symptoms in OM patients warrant attention from both nurses and pharmacists, highlighting the need to refine patient care protocols. Regarding OM intervention strategies, most studies emphasized basic oral care and pain management. In studies involving pharmacists, patient education and psychological care were incorporated, providing long-term support that significantly improved patients’ QoL. This suggests that when pharmacists join oncology treatment teams, their role extends beyond medication guidance; they can also enhance treatment adherence through patient education. Furthermore, we summarized nurse-led OM interventions, which demonstrated a broader scope compared to pharmacist-led interventions. These studies addressed not only quality of QoL but also xerostomia, dysphagia, and nutritional status, offering comprehensive patient care. This underscores the indispensable role of nurses in managing cancer patients, as they provide precise and timely care for adverse reactions at the clinical forefront, playing a crucial role in optimizing patient outcomes.
An assessment of the current management status of OM as an adverse reaction in cancer patients reveals a high incidence rate, yet the management protocols remain incomplete. Existing practice guidelines are updated infrequently and contain numerous recommendations, which may complicate the standardization of care. Generally speaking, OM interventions are conducted by specialist doctors, nurses, and stomatologists. MASCC/ISOO guideline recommends the implementation of a multi-drug combined oral care program for HNC patients receiving radiotherapy to prevent OM. Patients receiving moderate-dose radiotherapy (<50 Gy) and concurrent radiotherapy with CT are advised to use Benzydamine mouthwash (a non-steroidal anti-inflammatory drug type mouthwash) to prevent OM. For patients undergoing radiotherapy alone, low-level laser therapy is recommended. Additionally, the guideline suggests using 0.2% topical morphine mouthwash for managing pain related to OM. Alongside mouthwash for pain relief, the guideline recommends the use of two natural substances to prevent OM inflammation in patients with HNC.
The MASCC/ISOO Guidelines for the Management of OM offer a comprehensive synthesis of existing research. 5 However, several clinical symptoms related to OM have not been addressed for intervention measures. Dry mouth, nutritional status, and incidence of esophagitis, which are mentioned in the 12 studies included, are clinically relevant issues associated with secondary infections and nutritional support. These aspects still require further study. Additionally, a comprehensive clinical nursing framework for managing OM in patients with HNC has not yet been established.
The guidelines recommend various treatment methods that require the involvement of professional clinical pharmacists to address patient medication issues. However, the role of pharmacists in this area is rarely emphasized. In the three pharmacist-led studies examined in this scoping review, pharmacists provided patients with nutritional counseling, delivered pain education, elucidated adverse drug reactions, facilitated patients’ understanding of pain, actively managed side effects, and alleviated tension and anxiety related to pain through the use of promotional videos and verbal presentations. However, these studies failed to address key areas such as comprehensive pharmaceutical monitoring by clinical pharmacists, personalized pharmaceutical care, and routine medication education for patients. Additionally, there appears to be a lack of clarity regarding the delineation of responsibilities between pharmacists and nurses in clinical settings, with nurses often assuming roles in medication education and personalized pharmaceutical services. Therefore, it is imperative for clinical pharmacists to clearly define and formalize their roles within the clinical process and diligently execute their clinical tasks accordingly.
Studies have shown that when pharmacists provide medication advice, even if the types of medications do not change, pharmacist-guided medication significantly benefits rational drug use and can impact health outcomes. 30 Furthermore, collaboration between nurses and pharmacists in medication coordination can prevent potential harm. 31 Optimizing pharmacist–nurse interventions for OM, clarifying their responsibilities, and highlighting their roles are crucial for ensuring long-term patient treatment and clinical benefits.
This study is the first to conduct a scoping review summarizing the prevention and targeted care of OM in patients undergoing CRT for HNC over the past 15 years. It emphasizes that supportive care provided by pharmacists and nurses in clinical practice benefits patients. Currently, OM remains an underestimated side effect of cancer treatment. The data regarding OM induced by RCT are predominantly derived from adverse event reports in cancer treatment-related studies, with a paucity of large-scale study data. The data utilized in this study are largely estimated frequencies based on the outcomes of relevant research. Furthermore, a multicenter randomized controlled trial conducted in 2019 compared the severity assessment scores of RCT-induced OM across different regions, revealing significant differences in incidence rates. 32 The subjective judgments of the assessors were found to be relevant to the trial results. Clinically, our researchers have also observed that OM is typically documented only when it reaches a more severe stage. Additionally, the absence of a standardized scale for severity assessment poses challenges in OM evaluation. 30 A limited number of interventions supported by high level evidence may not be universally applicable to all types of OM patients, leading to considerable variability in treatment protocols across different medical centers. SOM can result in premature termination or readjustment of treatment, increased hospitalization days, heightened public health costs, and a diminished QoL for patients. There is an urgent need for collaborative efforts from basic, translational, and clinical workers to improve the QoL of cancer patients and reduce management costs.
MASCC/ISOO established a mucositis research section in 1998 to address this important issue. Clinical practice guidelines published in 2004 recommended oral care protocols, including patient education, to mitigate the severity of mucositis caused by CT or radiotherapy. 33 In the 2014 clinical practice guideline, the group recommended oral care protocols as a preventive measure for cancer treatment-induced OM. 34 Subsequent studies have demonstrated the beneficial effects of basic oral care in preventing OM. According to the 2020 clinical practice guidelines, basic oral care remains the best method for oral care in cancer patients, and educating patients about its benefits is still appropriate. 5
In the 12 studies we analyzed, several studies involved pharmacists and nurses providing basic oral care to patients in the intervention group. These interventions resulted in reduced pain, lower OM grades, improved QoL, reduced incidence of OM, lower xerostomia scores, improved nutritional status scores, and reduced incidence of esophagitis. Although this study did not include large sample trials or standardized scoring tools, it preliminarily indicates that multidisciplinary team oral basic care involving pharmacists and nurses can help cancer patients achieve optimal treatment experiences and outcomes.
From the research results, it is evident that SOM during cancer treatment significantly disrupts treatment progression. In the study by Yüce and Yurtsever, 17 nine patients in the non-education group developed grade 3 or higher OM by the third week, whereas none did in the education group. Additionally, the non-education group experienced significantly more cases of xerostomia, swallowing obstruction, and taste deterioration, with lower QoL at the end of treatment compared to the education group. Yoshida et al.’s 25 study revealed differences in albumin levels, WBC counts, and neutrophil counts among OM patients receiving pharmacist intervention, with the control group showing more severe opioid use and weight loss. Hava Cara’s 2023 study demonstrated that nurse-provided oral care training and counseling helped maintain stable stress indices during treatment. These findings indicate that providing care counseling and patient education when patients experience such adverse reactions can effectively prevent OM and improve patients’ understanding of the disease, thereby enhancing medication adherence and reducing psychological burdens. With the assistance of pharmacists, medication cost-effectiveness and patients’ QoL can be improved. This highlights the importance of the services provided by clinical pharmacists and nurses.
However, this scoping review has several limitations, such as the small sample size (551 patients in the experimental group and 395 in the control group) and regional constraints, including China, Turkey, Japan, Sweden, and the United States. Representation from these countries is insufficient, making it challenging to accurately reflect the clinical practices of pharmacists and nurses more. The norms and responsibilities of pharmacists vary significantly among these countries. Moreover, the study did not account for several confounding factors, including the impact of patients’ dietary habits, as immune responses play a crucial role in the OM development following radiation-induced cell damage. High consumption of pro-inflammatory foods (such as saturated fatty acids, fried foods, and added sugars) can induce chronic inflammation, while low intake of fruits and vegetables increases the risk of DNA damage in cells. 29 The severity of OM is also influenced by various treatment factors, such as total radiation dose, daily radiation dose, and volume of irradiated mucosa. 35 A cumulative dose of 20–30 Gy in the oral cavity has been shown to induce OM. 36 Compared with RT alone, concurrent CRT is associated with an increased risk of radiation-induced mucositis, resulting in a higher incidence and prolonged duration of OM. 37 Tobacco consumption is also associated with increased Radiation-Induced Mucositis incidence and severity.35,38,39 Pre-CRT inflammatory periodontal disease and poor dental health are additional environmental risk factors for RT and CT-induced OM. 40 This scoping review further identified that discrepancies in guidance provided by nurses and clinical pharmacists may engender patient distrust in their therapeutic process, thereby potentially compromising treatment outcomes. This underscores the presence of communication voids and an ambiguous delineation of responsibilities within clinical practice, highlighting the necessity for further research to address these critical issues.
Conclusion
In the management of HNC, effective prevention and management of OM during CRT are crucial for enhancing patient outcomes. Clinical pharmacists and nurses play indispensable roles within the multidisciplinary treatment team by leveraging their expertise and skills to provide optimized therapeutic strategies and comprehensive care support for patients. The direct care model employed by pharmacists and nurses significantly alleviates symptoms and enhances the QoL for patients, thereby fostering improved treatment efficacy and patient satisfaction.
Supplemental Material
sj-docx-1-smo-10.1177_20503121251339581 – Supplemental material for Clinical pharmacists and nurses in the management of oral mucositis in head and neck cancer patients during antineoplastic therapy: A scoping review
Supplemental material, sj-docx-1-smo-10.1177_20503121251339581 for Clinical pharmacists and nurses in the management of oral mucositis in head and neck cancer patients during antineoplastic therapy: A scoping review by Palizhati Kaisha, Xinxin Li, Yiqun Tang and Yufen Zheng in SAGE Open Medicine
Footnotes
Acknowledgements
We appreciate the support of China Pharmaceutical University and Jiangsu Cancer Hospital for their support in the completion of this article.
Ethical considerations
Not applicable.
Author contributions
Palizhati Kaisha: The author participated in the conception and design of this study, data search and analysis, and manuscript writing of this study. Participated in every revision during the manuscript correction phase. Xinxin Li: This author participated in the design, data analysis, and manuscript writing of this study and also contributed to the manuscript writing. Participated in every revision during the manuscript correction phase. Yufen Zheng: This author helped with acquisition, analysis, and interpretation of data; and revising the article. Guided every revision during the manuscript correction phase. Yiqun Tang: This author helped with design and supervision of the study, data interpretation, literature search, critical review, and final approval of the manuscript. Guided every revision during the manuscript correction phase.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
