Abstract
Background:
To analyze the characteristics and factors influencing the diagnosis of unilateral isolated fungal sphenoid sinusitis.
Methods:
A retrospective analysis was conducted on the clinical data of patients who underwent surgery for unilateral isolated sphenoid sinusitis between June 2020 and May 2023. Data collected included variables such as gender, age, side of the specimen, symptoms, sinus computed tomography findings, pathological results, and complications. The patients were categorized into two groups: the sphenoid sinusitis group and the fungal sphenoid sinusitis group.
Results:
Among the 84 cases studied, there were 19 males (22.6%) and 65 females (77.4%). Inflammation was observed in 11 patients (13.1%), while fungi were detected in 73 patients (86.9%). Headache was reported in 74 cases (88.1%). Sinus computed tomography findings revealed calcified plaques/spots in 51 cases (60.7%), hyperosteogeny in 75 cases (89.3%), and bone destruction in 11 cases (13.1%). The thickness of the sinus wall ranged from 0.92 to 7.35 mm. The thickness ratio of the bilateral sinus walls ranged from 0.80 to 6.78. The chi-square test indicated significant differences between the two groups in terms of calcified plaques/spots, hyperosteogeny, the thickness of the lesion-side sinus wall, and the ratio of bilateral sinus walls (p < 0.05). Diagnostic tests using the ROC curve demonstrated that calcified plaques/spots, the thickness of the lesion-side sinus wall, and the ratio of bilateral sinus walls had moderate accuracy in diagnosing fungal sphenoid sinusitis.
Conclusion:
While headache is not a specific symptom of fungal sphenoid sinusitis, sinus computed tomography findings such as calcified plaque/spots, the thickness of the lesion-side sinus wall, and the thickness ratio of bilateral sinus walls are valuable for diagnosis.
Keywords
Introduction
Isolated sphenoid sinus disease (ISSD) encompasses a range of primary conditions confined to the anatomical boundaries of the sphenoid sinus, which can be congenital, inflammatory, or neoplastic in nature, with an incidence of 1%–2% among all sinus lesions. 1 Inflammatory lesions account for 80% of cases, neoplastic lesions for 7%, and other diseases for 13%. 2 The diverse and concealed anatomical location of the sphenoid sinus often leads to misdiagnosis or missed diagnosis, potentially resulting in severe complications such as orbital apex syndrome, blindness, encephalitis, and cavernous sinus thrombophlebitis. With the use of computed tomography (CT) and magnetic resonance imaging (MRI), ISSD can be detected and diagnosed early.
Fungal sinusitis, a common specific infectious disease of the paranasal sinuses, predominantly affects the maxillary sinus, followed by the sphenoid, ethmoid, and frontal sinuses, and is more prevalent in women over 50 years old.3,4 Most solitary lesions in the sinus are fungal ball (FB). Fungal sphenoid sinusitis is often missed due to the deep and concealed location of the sphenoid sinus, and nonspecific clinical symptoms, and is not detected until CT or MRI is performed. Diagnosis relies on clinical symptoms, imaging, and intraoperative findings, including pathology. Confirming the diagnosis preoperatively can assist surgeons in planning the surgical approach, thereby reducing operation time and patient discomfort. This study retrospectively examined the clinical data of patients with unilateral ISSD in our department, focusing on the influence of clinical symptoms and imaging on the diagnosis of isolated fungal sphenoid sinusitis (IFSS).
Patients and methods
General data
This retrospective study was approved and consented to by the Ethics Committee of Yantai Yuhuangding Hospital (No. 2024-179).
The study retrospectively analyzed clinical data from 84 patients diagnosed with unilateral isolated sphenoid sinusitis who underwent endoscopic sinus surgery at our center between June 2020 and May 2023. Data collected included variables such as gender, age, the side of the affected specimen, medical history, symptoms, sinus CT findings, complications, and pathological results. These patients were categorized into two groups based on pathological findings: the sphenoid sinusitis group and the fungal sphenoid sinusitis group. The inclusion criteria for the study were: (1) the presence of unilateral ISSD; (2) completion of endoscopic sinus surgery; (3) availability of preoperative sinus CT imaging data within the local imaging network; and (4) postoperative pathology indicating inflammation and fungi, including allergic fungal sinusitis. Exclusion criteria: (1) sphenoid sinus cyst, polyps, benign or malignant tumors, bone diseases, meningoencephalocele and other diseases, and invasive fungal sphenoid sinusitis; (2) no pathological data; (3) a history of previous sinus surgery.
Interpretation and measurement of sinus CT
High-resolution CT scans of the paranasal sinuses were conducted, capturing both coronal and axial views. The window level was set at either 200 or 400 HU, while the window width was adjusted to 2000 or 4000 HU, as referenced in studies.5,6 The CT scans were independently analyzed by three observers: two otorhinolaryngologists (one intermediate and one advanced), and one advanced imaging specialist. The axial view was specifically utilized to measure the thickness of the lateral wall of the bilateral sphenoid sinus at a consistent location. The measurements obtained by the three observers were averaged to ensure accuracy and reliability.
Statistical methods
The data were analyzed using IBM SPSS 26.0 statistical software. To compare the factors influencing the diagnosis of fungal sphenoid sinusitis, both the chi-square test and ROC curve analysis were employed. A p-value of less than 0.05 was considered statistically significant.
Results
In the study involving 84 patients (Table 1), 19 (22.6%) were male and 65 (77.4%) were female. The distribution of lesions was nearly equal, with 43 (51.2%) right-sided and 41 (48.8%) left-sided. The predominant symptom was headache, reported by 74 patients (88.1%), followed by purulent nasal discharge in 32 (38.1%; Figure 1), ocular symptoms (periorbital pain, orbital pain, blurred vision, decreased visual acuity, diplopia, ptosis, epiphora, etc.) in 24 (28.6%), nasal obstruction in 22 (26.2%), hyposmia in 10 (11.9%), and blood in nasal discharge in 6 (7.1%). Notably, 54 patients (64.3%) experienced two or more symptoms, such as headache combined with nasal discharge in 27 (32.1%), headache with nasal obstruction in 17 (20.2%), and headache with ocular symptoms in 24 (28.6%). Sinus CT scan revealed calcified plaques/spots in 51 cases (60.7%), hyperosteogeny in the sinus wall in 75 cases (89.3%), bone destruction in 11 cases (13.1%), enlargement of the sinus ostium in 21 cases (25.0%), and polyp blockage of the sphenoid ostium in 6 cases (7.1%; Figure 2). In addition, 5 patients (6.0%) had diabetes mellitus. Pathological analysis identified inflammation in 11 cases (13.1%) and fungi in 73 cases (86.9%; Figure 3). The thickness of the lesion-side sinus wall ranged from 0.92 to 7.35 mm, with a median of 3.55 mm, while the contralateral sinus wall ranged from 0.59 to 4.89 mm, with a median of 1.35 mm. The thickness ratio of the bilateral sphenoid sinus walls varied from 0.80 to 6.78, with a median of 2.76. Chi-square test analysis indicated statistically significant differences in sinus CT findings specifically in calcified plaques/spots, hyperosteogeny, the thickness of the lesion-side sinus wall, and the thickness ratio of bilateral sphenoid sinus walls between the fungal and inflammatory groups (p < 0.05). However, no significant differences were observed in gender, side, symptoms, and diabetes (p > 0.05). Diagnostic test evaluations showed that the area under the ROC curve was 0.849 for calcified plaques/spots, 0.823 for the thickness of the lesion-side sinus wall, and 0.712 for the thickness ratio of the bilateral sphenoid sinus walls. These findings suggested that the diagnostic performance of calcified plaques/spots, the thickness of the lesion-side sinus wall, and the thickness ratio of the bilateral sphenoid sinus walls were notably accurate. The diagnostic cut-off points were determined to be 3.03 mm for the thickness of the lesion-side sinus wall and 3.02 for the ratio of the bilateral sphenoid sinus walls (Table 2, Figure 4).
The characteristics of the two group patients.

Note: There were indicated statistically significant differences in sinus CT findings specifically in calcified plaques/spots, hyperosteogeny, thickness of the affected sinus wall and the ratio of bilateral sphenoid sinus walls between the fungal and inflammatory groups (P<0.05).
The area under the curve of fungal sphenoidal sinusitis.

The test result variable(s): calcified plaques/spots, hyperosteogeny in the sinus wall, thickness of the affected sinus wall, erosion of the sinus wall, enlargement of the sinus ostium has at least one tie between the positive actual state group and the negative actual state group. Statistics may be biased.
The calcified plaques/spots, the thickness of the affected sinus wall, and the ratio of bilateral sinus walls had moderate accuracy in diagnosing fungal sphenoid sinusitis.
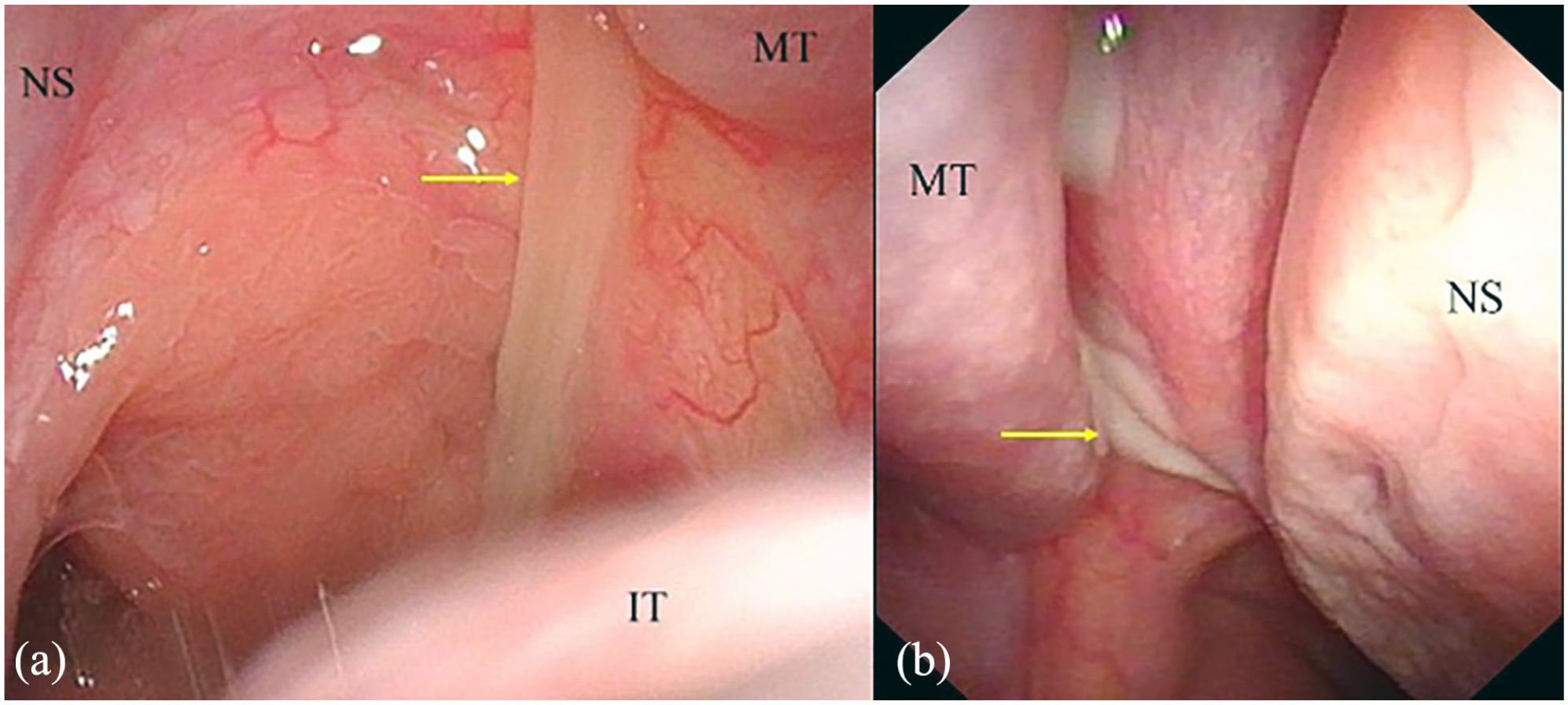
(a) Yellowish purulent discharge in the right olfactory cleft and (b) white purulent discharge in the left olfactory cleft.

CT showed: (a) Thickening of the lateral wall of the right sphenoid sinus and patchy high-density shadow in the sinus; (b) The wall of the right sphenoid sinus thickened, the green line showed the thickness of the sinus wall; (c) The wall of the left sphenoid sinus thickened and the sinus ostium enlarged; (d) The wall of the right sphenoid sinus thickened and spotty high-density shadow was found in the sinus. The green line showed the value of the enlarged ostium of the sphenoid sinus.

(a) and (b) showed a lot of fungal mycelia in the sphenoid sinus lesions (hematoxylin–eosin staining, ×100).

The area under the curve of CT findings of patients with fungal sphenoidal sinusitis.
Discussion
The sphenoid sinus, due to its concealed anatomical location, poses significant challenges for detection via anterior rhinoscopy. Its proximity to critical structures, including the brain, pituitary gland, internal carotid artery, cavernous sinus, orbital apex, and multiple cranial nerves such as the optic nerve, increases the risk of severe complications if misdiagnosed or diagnosed late. Fungi, particularly Aspergillus, 7 are commonly found colonizing both normal and diseased nasal cavities and sinuses. Aspergillus can be cultured in over 95% of patients with chronic sinusitis. 8 Previous studies have identified various factors that lead to the stenosis or obstruction of the nasal ostium, creating an abnormal environment within the sinus cavity. This environment, characterized by secretion retention, hypoxic conditions, decreased pH, increased sugar content, and increased viscosity, facilitates fungal growth. 9 Fungal sinusitis exhibits a variable course potentially persisting from months to years. 10
Fungal sphenoid sinusitis often presents with nonspecific symptoms, with headache being the most prevalent.11,12 This headache typically manifests as pain in various regions, including the posterior occipital area, the top of the head, the forehead, the deep eyeball, and the deep mastoid process. The timing, location, and intensity of the headache can vary significantly among individuals, leading to frequent misdiagnoses as angioneurotic headaches or migraine. Knisely et al. 13 reported that the incidence of headache in such cases ranges from 33% to 81%. In this study, the incidence of headache was notably high, reaching 86.3% in the fungal group, and 100% in the inflammatory group. Other common symptoms include nasal obstruction, purulent nasal discharge, blood in the nasal discharge, and ocular symptoms. Interestingly, approximately 18% of patients were asymptomatic and were only diagnosed following imaging findings. 14 Preoperative nasal endoscopy may not always reveal positive findings; however, some cases exhibit edema or polypoid changes in the mucosa of the sphenoid sinus recess and yellow viscous secretions in the sinus. Ocular symptoms, which may include epiphora, visual field changes, diplopia, ocular motility disorders, and visual impairment are potentially linked to direct optic nerve stimulation by inflammation, local compression, nutritional disorders, and ischemia. In this study, the incidence of nasal obstruction was 26.2%, purulent nasal discharge 38.1%, blood in nasal discharge 7.1%, hyposmia 11.9%, and ocular symptoms (periorbital pain, orbital pain, blurred vision, decreased visual acuity, diplopia, ptosis, epiphora, etc.) 28.6%. Notably, 64.3% of patients experienced two or more symptoms, with combinations such as headache and nasal discharge (32.1%), headache and nasal obstruction (20.2%), and headache and ocular symptoms (28.6%) being common. Despite the high incidence of headaches, other symptoms provided limited diagnostic assistance. Consequently, the evaluation of diagnostic tests indicated low accuracy for headaches alone. Therefore, combining nasal endoscopy with sinus CT is essential and beneficial for accurate diagnosis.
CT scans play a crucial role in diagnosing fungal sinusitis due to the presence of dense interdigitating fungal hyphae, which typically manifest as high-density intrasinusoidal lesions. 14 These lesions are primarily composed of calcium and metal types, 15 with the incidence of calcium-type lesions in fungal sphenoid sinusitis reaching as high as 95%.15,16 Two theories explain the mechanism behind the high-density shadows observed in fungal maxillary sinusitis: the aerogenesis theory and the odontogenic theory. In contrast, fungal sphenoid sinusitis is attributed to the formation of calcium salt precipitations, characterized by patchy and gritty high-density calcifications. Additional CT findings include sinus wall hyperosteogeny and sclerosis, erosion of the sinus wall, and irregular surfaces. Imaging studies have demonstrated high sensitivity and specificity4,17–19 indicating potential mucosal and bony inflammatory factors. Previous research has linked the pathogenesis of fungal sinusitis to congenital mucosal immunity. In this study, CT scans revealed calcified plaques/spots in the sinuses in 51 cases (60.7%), hyperosteogeny in the sinus wall on the lesion side in 75 cases (89.3%), bone destruction in 11 cases (13.1%), and enlargement of the sinus ostium on the lesion side in 21 cases (25.0%). Our findings indicate that the high-density shadow of sinus lesions and hyperosteogeny of the sinus wall are statistically significant for diagnosing IFSS.
Recent studies have shown that sinus wall hyperosteogeny is associated with inflammatory stimuli. The incidence of bone destruction in the sinus wall is relatively low, ranging from 3.6% to 17% as reported in previous studies.20,21 Literature indicates that CT-scan hyperdensities account for approximately 60%–100% of FB cases. 4 Given that inflammatory stimuli can lead to sinus wall hyperosteogeny, we compared the thickness of the lesion-side sinus wall and the thickness ratio of the bilateral sphenoid sinus walls between non-fungal and fungal groups, finding statistically significant differences. Furthermore, diagnostic tests revealed that the accuracy of CT scans in diagnosing IFSS was higher than that of headache symptoms alone, enhancing the utility of CT in diagnosing IFSS.
Pathological examination serves as the definitive basis for diagnosis. During surgery, the lesions exhibited grayish-brown caseous material, mucopurulent secretions on the surface, and inflammatory exudation in the surrounding mucosal tissue. In our center, hematoxylin and eosin (HE) staining is predominantly utilized, with Gomori staining (hexamine silver staining) being rarely employed. In this study, HE staining was performed in all cases.
Surgery is the primary treatment choice. 22 All cases in this study underwent endoscopic sinus surgery to enlarge the ostium of the sphenoid sinus, effectively remove fungal masses, and repeatedly irrigate the sphenoid sinus with physiological saline. Postoperatively, nasal spray corticosteroids were administered to reduce mucosal edema around the nasal cavity and sphenoid sinus ostium, alongside oral mucolytics and saline irrigation of the sinonasal cavity. Notably, no antifungal therapy was administered, and no recurrences were observed during follow-up.
This study has several limitations. It is a single-center retrospective study, with written informed consent exempted by the Ethics Committee of Yantai Yuhuangding Hospital (No. 2024-179). The sample size was not calculated or justified, and the number of samples was not large enough. In addition, image measurement had some defects, and certain influencing factors could not be fully addressed, potentially introducing bias into the results. Future large-scale, multicenter, prospective studies are necessary to better guide the diagnosis and treatment of IFSS.
Conclusions
Headache is a common symptom experienced by most patients with unilateral isolated sphenoid sinusitis. However, our research indicates that headache is not a specific symptom of fungal sphenoid sinusitis. Instead, sinus CT findings are more diagnostically valuable. Specifically, the presence of calcified plaque/spots within the sinus, the thickness of the lesion-side sinus wall, and the thickness ratio of the bilateral sinus walls are significant diagnostic markers. We have determined that a sinus wall thickness of 3.03 mm on the lesion-side serves as a diagnostic cut-off point. In addition, a ratio of 3.02 for the bilateral sphenoid sinus walls is also indicative of the condition.
Footnotes
Acknowledgements
None.
Author contributions
Yan Wang: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Writing – original draft, Writing – review and editing. Xiumei Chen: Investigation, Resources. Dajian Li: Investigation, Resources. Yu Zhang: Investigation, Resources. Yan Sun: Investigation, Resources. Xicheng Song: Investigation, Resources.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
The research has been performed in accordance with the principles stated in the Declaration of Helsinki. Privacy and the interests of the patients are not compromised. This retrospective study was approved and consented to, the written informed consent was exempted by the Ethics Committee of Yantai Yuhuangding Hospital (No. 2024-179).
Trial registration
Not applicable.
