Abstract
Background:
To demonstrate the safety and performance of the Arrow EZ-IO Intraosseous Vascular Access System, particularly in the pediatric patient population, a retrospective observational study was conducted in 2021 and 2022.
Methods:
Following study design, IRB approval, and investigator selection, data were collected for all patients needing intraosseous access—adult and pediatric. The primary endpoint was the success rate for achieving intraosseous access; the secondary endpoint was the rate of adverse events. Following initial data collection, additional data were collected and a sub-set analysis was conducted to demonstrate the same in pediatric patients only, which is the focus of this report.
Results:
Data for 106 pediatric cases were collected. The success rate for achieving intraosseous access and infusion was 96.2%. There were three adverse events in two patients (1.9%); none serious or previously unreported. The mean duration of device use was 60 h (SD = 46). For 46 patients, the device was used for up to 48 h, and for another 45 patients, the device was used for a longer duration.
Conclusions:
This report is the first characterization of the safety and performance of the Arrow EZ-IO Intraosseous Vascular Access System when used in the pediatric population for longer dwell times (>24 h), with no serious complications reported. Performance and safety objectives were met. The results of this real-world evidence study are in alignment with findings from the clinical literature concluding that, for pediatric patients, the Arrow EZ-IO Intraosseous Vascular Access System is safe and effective for providing vascular access in urgent, emergent, and medically necessary situations, in which intravenous access is difficult or impossible to obtain. In addition, this study supports the use of intraosseous access for dwell times greater than 24 h.
Introduction
Background and rationale
Intraosseous (IO) vascular access involves inserting a catheter into the bone marrow cavity to administer fluids and/or drugs. In the 1920s, Drinker recognized the value of IO access for fluid/drug delivery into the circulatory system 1 and early use of the technique was primarily in pediatric patients.2 –4 In the early 21st century, improved IO devices enabled clinicians to gain IO access for systemic fluid/drug delivery as quickly as venous access and, depending on the anatomical site, at infusion rates comparable to those achieved through central venous catheters (CVC).5,6 One such device is the Arrow EZ-IO Intraosseous Vascular Access System (Teleflex Medical Incorporated, Morrisville, NC, USA). The device is indicated in the United States and Canada for IO access when vascular access is difficult to obtain in emergent, urgent, or medically necessary cases for up to 24 h.7,8 For patients ⩾12 years old, device use may extend to 48 h. In European Union countries, the device is indicated for up to 72 h. Historically, the complication rate for IO devices has been low. In a 2021 publication, Philbeck et al. described a study that examined the safety of EZ-IO access for 48 h in adult volunteers. 9 Of 121 subjects, there were no serious complications or unanticipated adverse events (AE). Subsequently, the US FDA expanded the indications for use to allow use of the device for up to 48 h in adult patients and pediatric patients age ⩾12 years when alternate intravenous (IV) access is not available or reliably established. However, in North America, the use for pediatric patients under the age of 12 years remains restricted to 24 h, effectively limiting the utility of the IO route for those patients requiring longer durations of use
A retrospective observational study was conducted in 2021 and 2022 to collect real-world data to demonstrate the safety and performance of the EZ-IO system for the general patient population. This article reports on the subset of data for IO use in pediatric patient subjects included in the study. The objective of the subset analysis was to understand the general safety and performance of the EZ-IO system in pediatric patients for whom it was determined that IO access was needed, particularly when the device remained in place for 24 h or more.
Methods
Study design
To determine and confirm the real-world safety and performance of the EZ-IO system, a retrospective observational study was designed. Following the completion of the study, it was realized more data were needed for pediatric patients (defined as patients ⩽18 years of age) needing IO vascular access. Patients’ identities, including names and screening identification, were de-identified. Other than the need for IO vascular access, there were no inclusion or exclusion criteria.
Setting and participants
The study was approved by the Advarra Institutional Review Board (review number Pro00070542; Advarra, Columbia, MD, USA) with an exemption from informed consent and continued review, and was conducted in accordance with the Declaration of Helsinki. 10 Health care professionals (HCPs), comprising physicians and nurses identified through a healthcare panel database, located in the European Union (n = 19), United Kingdom (n = 9), and United States (n = 1), served as investigators in the study. The HCPs’ typical roles within their respective organization included work in the emergency department, anesthesia, intensive care, and surgery, and had been previously trained on the use of the IO device. The training covered anatomical site selection, proper landmarking, needle size selection, needle insertion angle, features of the device, insertion pain mitigation, line flushing, line securement, line maintenance and monitoring, and needle removal. Following training, all had actual clinical experience using the device, and self-reportedly used the IO device on average 12 times a month (standard deviation (SD) = 20). Prior to the initiation of the study, investigators were also trained on the use of the electronic data capture system used to collect the data which, later, were reviewed for plausibility and analyzed by a biostatistician. Starting in August 2021 and concluding in August 2022, data from consecutive cases were extracted for pediatric patients for whom the EZ-IO was used in emergent, urgent, or medically necessary (as defined by the attending clinician) cases. Data were collected regarding the insertion, use, and removal of the device only. No additional follow-up data were collected.
Variables
The primary endpoint for the study was the success rate for achieving IO access using the EZ-IO system. Success was determined by the ability to infuse fluids and drugs into the intraosseous space following IO needle placement. A secondary endpoint was defined as the rate of device-related AEs. The patient’s specific need for IO access was determined by the investigator. Considering this was a retrospective study, and the use of the subject device had already been completed and recorded for each case, there were no prescribed or protocol-directed interventions in the treatment and care of the patients. Clinical characteristics, including specific uses of the device, placement site, and duration of use, were also extracted from electronic medical records.
Bias
Selection bias was minimized by the completion of the study by the enrollment of consecutive cases over a defined period of time to include data for all cases in which the study device was used, regardless of the outcome. Observer bias was reduced by involving multiple investigator clinicians in the data collection process. Response bias was minimized by forming study questions on the case report form that used clear language and provided clear and relevant answer options that were not open-ended or leading, and by reporting consecutive cases regardless of outcomes.
Sample size calculation and statistical methods
The sample size was calculated using the Wilson score method which assumed a binomial distribution of use, based on a performance success acceptance criterion ⩾75%, and 94.8% expected performance success. In addition, using R software (The R Foundation, Indianapolis, IN, USA), a one-sided confidence interval of 95% and an 80% likelihood of meeting the acceptance criterion was used to calculate the sample size. It was determined that, with a sample size of 90 cases, the probability of meeting the predefined acceptance criteria (lower bound of a one-sided 95% CI ⩾75%) was 95%. Continuous variables were described as mean (SD) and median (interquartile range (IQR)). Categorical variables were represented as frequencies with proportions. A one-sided 95% lower bound was used as the statistical measure to assess the results compared to the acceptance criteria.
Results
Data were collected for 108 cases, but due to an irreconcilable error in the IO needle dwell time/length of use calculation, two cases were excluded, resulting in 106 cases available for analysis (see Figure 1).

The flow of inclusion and exclusion of cases included from analysis starting with an original observational study of Arrow EZ-IO in adult and pediatric patients requiring IO access.
Further details are presented in Table 1.
Detailed information on IO access was analyzed in 106 pediatric cases (unless otherwise noted).

Performance and safety
With the primary endpoint success rate for achieving IO access (device placement), there were four failures resulting in a success rate of 96.2%. Among the four failures, only one was likely related to the IO device: a case of excessive bleeding in a patient (1–9 years age group). The other three failures were the death of the patient during the placement attempt (1–9 years age group), patient condition instability, and clinician selection of a needle that was too short for correct placement (both 10–18 years age group). Three AEs occurred in two patients who were deemed potentially related to the study device. One patient experienced pain and extravasation. The pain was treated with analgesics and the extravasation was treated with a tight dressing. In one patient, infection was noted after 24.5 h of dwell time. The device was removed and the infection was treated with antibiotics. None of the AEs were considered serious. For more details, refer to Table 1.
Duration of use
Data on duration of use were missing for 15 cases. Among the 91 cases for which the EZ-IO duration of use was recorded, the mean duration was 60 h (SD = 46). The median (IQR) duration of use was 49 (37–74) h. In 45 of those cases (49.5%), the subject device was used for >48 h (see Table 2).
Duration of use by year group and information available on 91 cases.
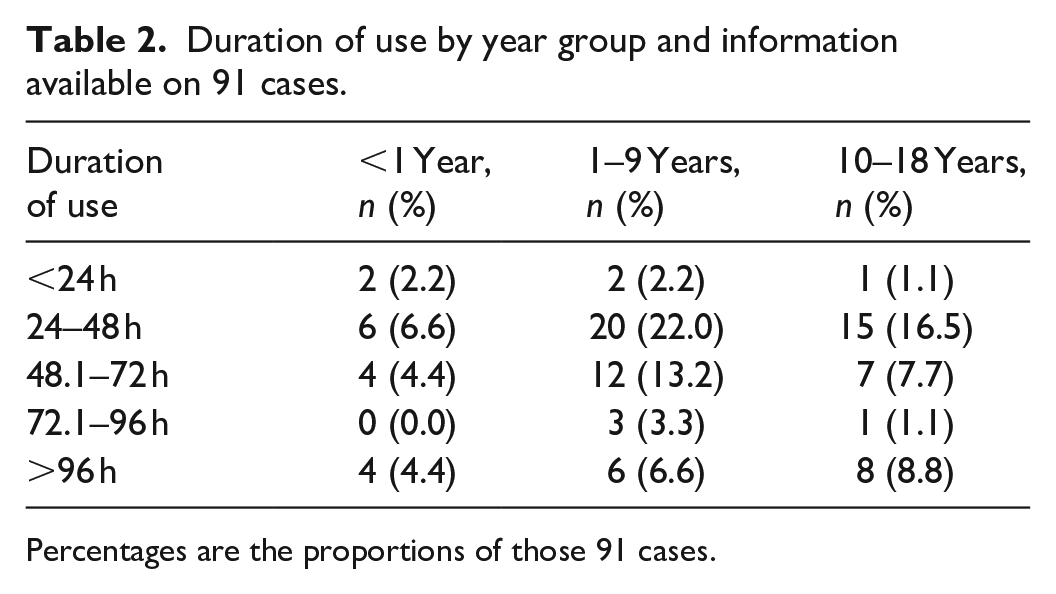
Percentages are the proportions of those 91 cases.
Discussion
Interpretation and generalization
While novel to some clinicians, IO access has been in use and described in the clinical literature for over a century. 1 Likewise, the Arrow EZ-IO System is no longer a “new” device—having been on the market since 2004, but the continued introduction of new data demonstrating or confirming that the device can be used safely and effectively makes IO access practical and acceptable across all age groups in a variety of clinical scenarios including pre-hospital, emergency department, and critical care settings. The primary advantage of IO access is the speed at which the device can be placed compared to other forms of vascular access. While the cost of the study device is generally greater than that of peripheral intravenous (PIV) catheters or CVC, often CVCs are placed primarily because the patient has difficult vascular access and PIV access cannot be obtained. A study by Dolister et al. concluded that, in those cases, if an IO device is placed instead of a CVC, substantial cost savings could be realized. 11 The American Heart Association, 12 National Association of EMS Physicians, 13 and American College of Emergency Physicians 14 —among other professional organizations—have policy statements advocating the use of IO devices for adults and pediatrics in a number of clinical situations including cardiac arrest, and particularly when peripheral IV is difficult or impossible to obtain.
Several clinical studies and case series describing the battery-powered IO system support IO access as a fast, safe, and effective alternative to peripheral IV or CVC for difficult vascular access. Placement and infusion success rates are generally >90%.15 –19 These include a 2022 study in which IO access attempts were made in 161 neonates and IO access was successful in 146 cases (91%). The first attempt needle-insertion success rate was 75%. 19 The battery-powered IO system was used in 88% of the cases in which the device was identified. In the present study of 106 cases, IO access and infusion were successful in 96% of cases—a result in alignment with or exceeding what has been reported in the clinical literature for the general population but not specifically for the pediatric population. Among placement sites typically used for IO access, placement success was consistent, and there was no obvious correlation between placement site and placement success or dwell time.
One site for IO access typically under-used and under-studied is the distal femur. Most references in the clinical literature simply state that the distal femur is a viable site for IO access but lack data to report its safety and performance. Schwindt et al.’s study reported four IO insertion attempts in the distal femur among the 146 neonates, and all were successful. 19 In 2024, Zitek et al. reported on 163 pediatric patients for whom IO access was used in a prehospital setting. Among those, there were 234 IO access attempts, with 82 attempts in the distal femur. The success rate of attempts in the distal femur was 89% compared to 85% in the proximal tibia. Complication rates were similar; 6% in the distal femur and 5% in the proximal tibia. 20 Burton and Chigaru conducted a retrospective medical chart review of pediatric critical care transport patients who underwent IO insertion from 2018 to 2020. The most common site of insertion for the 271 patients was the tibia (56%). Of note, 40 (14%) patients underwent more than two insertion attempts and, for these patients, the final successful insertion site was the distal femur. 21 In the present study, there were only nine placement attempts in the distal femur and they were all successful.
Regarding serious AEs with IO access, they are rarely reported in the clinical literature. A seminal 1985 study by Rosetti et al. reported a 0.6% serious complication rate with the most prevalent being infection, including osteomyelitis. 22 Later studies have also reported rare cases of osteomyelitis.23 –29 According to the manufacturer of the EZ-IO system, among millions of uses, there have been fewer than a dozen reports of osteomyelitis attributed to the use of the device (Christopher Ross, MD, Teleflex Medical Director, personal communication, January 2024). In 2021, Philbeck et al. reported a study specifically aimed at establishing the safety of the EZ-IO system when left in place for 48 h in adults. Among 121 subjects who were healthy or with a history of mild to moderate renal disease and/or controlled diabetes, there was no incidence of osteomyelitis or other serious AEs. 9 However, those findings are not directly generalizable to the pediatric population. In the present study, despite a substantial number of cases (n = 45, 49.5%) in which the EZ-IO remained in place and operational for more than 48 h, there were no serious AEs. There was one infection reported in a case after the device had been used for 24.5 h, but the infection was successfully treated with antibiotics.
Among the most prevalent complications resulting from IO access are needle/catheter dislodgement, extravasation/infiltration, incorrect needle/catheter length, and broken or defective needle/catheter. 30 In a 2022 study by Hoskins et al. in which an electronic health record database was mined for cases in which IO access was used in the pediatric population, 1012 cases were analyzed. In that study, 29 (3%) of subjects experienced at least one AE. Those included lower extremity thrombosis, fracture, cellulitis/abscess, pulmonary embolism, and osteomyelitis. 31 In the present study, there were three AEs deemed potentially related to the study device, including one case each of pain, extravasation, and infection. Pain is an expected side effect from IO placement and infusion and, in this case, the patient was treated with analgesics. The case of extravasation was treated by tightening the dressing, and the infection was successfully treated with antibiotics. Overall, similar to the Hoskins study, the AE rate was 1.9%; but none were serious. This compares favorably with results from other published studies reporting AEs related to IO access. The overall low incidence of AEs prevented any meaningful correlation between AE and placement sites.
For the pediatric population, evidence supporting the lifetime/duration of use for extended periods is scant at best. In 2013, Oksan and Ayfer reported a 25-patient study in which the device remained in place and was safely used for 24 h in three pediatric patients. 32 The present study is the first known describing IO needle use and dwell time >24 h in a substantial number of pediatric patients. In 46 (51.2%) cases, the device was used for up to 48 h. In 23 cases (25.6%), the device remained in place from 48.1 to 72 h, and in the remaining 22 cases (24.2%) the device was used for >72 h. Considering this was an observational retrospective study, there were no prescribed or protocol-directed interventions in the treatment and care of the patients. Clinicians treated the patients and used the study device, including determining the duration of device use, using their judgment, as is usual in the practice of medicine. Despite the longer dwell time, there were no serious AEs. While the authors of this study do not advocate leaving the device in place for longer than indicated, for this cohort of patients, the data demonstrate the general safety of the device when left in place for durations not previously reported for pediatric patients.
Limitations
A limitation of this study was that data were not collected as part of a randomized controlled trial (RCT). However, data from real world evidence (RWE) have the advantage of representing all patient populations in whom a device is used, without regard to pre-determined inclusion and exclusion criteria required for RCTs. Findings are not necessarily applicable or restricted to a certain cohort examined, as in a traditional RCT. Data were collected from patient’s charts and reviewed as they were received, but there was no mechanism to monitor or confirm their validity. It is not known how each investigator identified cases in which IO access was used, though it was possibly by searching patient databases for relevant procedure codes. Data regarding the number of attempts required to obtain IO access, specific fluids or drugs infused, or clinical outcomes were not collected. Data on duration of use were missing for 15 cases. The high success rate found in this study should be interpreted with care due to the methodological limitations of potential selection and reporting bias.
Conclusions
This report is the first characterization of the safety and performance of the Arrow EZ-IO Intraosseous Vascular Access System when used in the pediatric population for longer dwell times (>24 h); with no serious complications reported. Performance and safety objectives were met. The results of this RWE study are in alignment with findings from the clinical literature concluding that, for pediatric patients, the Arrow EZ-IO Intraosseous Vascular Access System is safe and effective for providing vascular access in urgent, emergent, and medically necessary situations, in which IV access is difficult or impossible to obtain. In addition, this study supports the use of IO access for dwell times greater than 24 h.
Footnotes
Acknowledgements
None.
Author contributions
TP contributed to: Study conception and design; Acquisition, analysis, and interpretation of data; Drafting of the manuscript; Critical revision of the manuscript for important intellectual content; Final approval of the manuscript; Accountability for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. JM contributed to: Study conception and design; Acquisition, analysis, and interpretation of data; Critical revision of the manuscript for important intellectual content; Final approval of the manuscript; Accountability for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. MC contributed to: Acquisition, analysis, and interpretation of data; Critical revision of the manuscript for important intellectual content; Final approval of the manuscript; Accountability for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. CR contributed to: Study conception and design; Acquisition, analysis, and interpretation of data; Critical revision of the manuscript for important intellectual content; Final approval of the manuscript; Accountability for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TEP, CR, and JGM are employed by Teleflex, the manufacturer of the study device. MC was a consultant, paid by Teleflex to participate in the study described herein and assist in the development of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study described within the manuscript was funded by Teleflex, the manufacturer of the study device.
