Abstract
Dolutegravir is an integrase inhibitor and is recommended by the World Health Organization as the preferred first-line and second-line human immunodeficiency virus treatment in all populations. Excessive weight gain associated with dolutegravir-based regimens is an emerging issue; however, the long-term metabolic consequences of this effect have not been fully understood. Growing evidence shows that this leads to a higher incidence of hyperglycemia, hypertension, and metabolic syndrome, along with elevated cardiovascular risk. Dolutegravir-based regimens, also associated with greater adipocyte differentiation and greater expression of markers associated with lipid storage, continue to be a problem among patients living with human immunodeficiency virus. The mechanisms by which certain antiretroviral therapy agents differentially contribute to weight gain remain unknown. Some clinical investigators speculate that dolutegravir could interfere with central nervous system appetite regulation (melanocortin-4 receptor) and insulin signaling, or may have better penetration of adipose tissue where they could exert a direct impact on adipose tissue adipogenesis, fibrosis, and insulin resistance. This review summarizes our current understanding of weight gain and fat changes associated with dolutegravir and its possible secondary metabolic comorbidities.
Background
The introduction of highly active antiretroviral therapy has resulted in a reduction in morbidity associated with opportunistic infections as well as a significant decrease in mortality among persons with HIV (PWH). 1 However, metabolic disorders and their associated comorbidities, such as cardiovascular disease (CVD) and diabetes, continue to pose a challenge to our ability to provide effective long-term treatment to this population. 2 Traditional risk factors like smoking and dyslipidemia, as well as HIV-related inflammation and immune activation, are well-known causes of cardiometabolic comorbidities in ART-treated PWH. 3 However, obesity and weight gain after starting ART are becoming increasingly recognized problems in today’s HIV treatment paradigm. 4 Weight gain after starting antiretroviral treatment has become a concern in recent years. 5 Although weight gain may be a positive prognostic indicator in PWH who are underweight at the time of ART initiation,6–8 it may increase the risk of cardiovascular and metabolic diseases in those who are normal or overweight.6,9
There is growing evidence that people who take the integrase inhibitor dolutegravir (DTG) are more likely to gain weight after starting treatment. 10 DTG is an antiretroviral drug that belongs to the class of integrase strand transfer inhibitors (INSTIs) and was approved in Canada in October 2013. 11 The World Health Organization (WHO) recommends a DTG-based regimen as first-line ART due to its non-inferiority antiviral efficacy, high genetic barrier to resistance, and lower potential for adverse drug effects. 12 However, several studies have investigated the relationship between DTG use and weight gain in PWH.13–15 Recent studies, including two randomized trials in Sub-Saharan Africa, have shown that DTG causes significant weight gain in ART-naive patients who are starting DTG for the first time.10,15,16 In addition, switching from a protease inhibitors (PIs) or non-nucleoside reverse transcriptase inhibitors (NNRTIs)-based regimen to a DTG-based regimen results in weight gain in ART-experienced patients.14,17 In this review, the current understanding of weight gain and fat changes associated with DTG and its possible secondary metabolic comorbidities is summarized.
Methodology
This narrative review includes studies from research databases such as Scopus, PubMed, PubMed Central (PMC), Web of Science, Google Scholar, and Cochrane Library that were published in or translated into English. The keywords used were “dolutegravir,” “weight gain,” “metabolic comorbidities,” and “dolutegravir-associated weight gain.” Randomized controlled trials, retrospective studies, review articles, cohort studies, and observational studies were included. The review considered studies involving adult populations. There was no set time frame for study inclusion, although the author prioritized recently published papers. As this paper reviews studies of various designs, it is limited in its ability to directly compare the study outcomes.
Evidence of weight gain with dolutegravir
Emerging evidence from ART initiation studies suggests that DTG may play a role in weight gain. In Sub-Saharan Africa, two randomized controlled trials compared DTG and efavirenz (EFV) in ART-naïve PWH. The NAMSAL study randomized 613 ART-naïve PWH to either DTG or EFV, both combined with emtricitabine (FTC) and tenofovir disoproxil fumarate (TDF), and the ADVANCE study randomized 1053 ART-naïve PWH to DTG or EFV at the standard dose of 600 mg, both combined with FTC and TDF, and a third arm of DTG combined with FTC and tenofovir alafenamide (TAF). In both studies, participants randomized to DTG gained more weight than those on EFV.18,19 In a Brazilian cohort study of 495 participants, investigators observed that those starting ART with DTG had a mean increase in body mass index (BMI) of 1.02 kg/m2, whereas those who used DTG after the therapeutic change had an increase of 0.56 kg/m2 over 96 weeks. 20 Likewise, the randomized controlled trials compared DTG/abacavir (ABC)/lamivudine (3TC) and bictegravir (BIC)/FTC/TAF in treatment-naïve patients. At 96 weeks, patients on DTG/ABC/3TC had a mean weight gain of 2.4 kg compared to those on BIC/FTC/TAF (3.6 kg). 21
In addition to ART initiation trials, numerous ART switch trials also show an increase in weight after PWH switches to DTG from alternate ART regimens (NNRTIs, PIs, and non-DTG INSTIs).13,17,22 In the retrospective observational study, the investigator assessed weight change over 18 months in patients who switched from EFV/TDF/FTC to an INSTI-containing regimen or a PI-containing regimen versus those on EFV/TDF/FTC. Patients who switched to an INSTI-containing regimen gained an average of 2.9 kg compared to 0.9 kg among those who continued on EFV/TDF/FTC, while those who switched to a PI regimen gained 0.7 kg. Among INSTI regimens, those who switched to DTG/ABC/3TC gained the most weight at 18 months. 17 This was supported by another study that assessed 460 virally suppressed adolescents who transitioned to DTG, showing an increased rate of BMI change by 0.8 kg/m2 per year in the year after DTG started to a rate of 1.2 kg/m2 per year. 23
Several subsequent studies have supplemented these preliminary findings. For example, in the retrospective observational study, the investigator reported that a DTG-based regimen was significantly associated with greater weight gain than other INSTI- or PI-based regimens. 13 Likewise, the African Cohort Study (AFRICOS) found a 1.3 kg weight gain and a 0.44 kg/m2 increase in BMI among participants in the first year after the transition to DTG + 3TC + TDF. 24 A retrospective observational cohort study of HIV patients who have been shifted from EFV/TDF/3TC to DTG/TDF/3TC and those who have been maintained on EFV/TDF/3TC have been on a specified regimen for at least 1 year. Patients on a DTG-based regimen had a mean weight gain of 3.88 ± 2.021 kg in 1 year compared to those on an EFV-based regimen (2.26 ± 2.39 kg). 22
By contrast, two recent studies of DTG in treatment-naive patients did not report weight change. The SPRING-2 study compared DTG versus raltegravir (RAL), while the FLAMINGO trial compared DTG versus darunavir (both studies also included nucleoside reverse transcriptase inhibitors (NRTIs)).25,26 Likewise, the SINGLE trial compared DTG/ABC/3TC versus fixed-dose EFV/TDF/FTC in treatment-naïve patients. At 48 weeks, the incidence of weight gain recorded as an adverse event was 6 of 414 subjects on DTG/ABC/3TC versus 3 of 419 subjects on EFV/TDF/FTC. 27
In general, these studies support more weight gain with DTG (especially when switching from NNRTIs), but the data are inconsistent and many must be interpreted cautiously because they originate from observational cohorts and/or retrospective analyses. Further details on some studies can be found in subsequent sections (Table 1).
Summary of studies assessing dolutegravir-induced weight Gain.
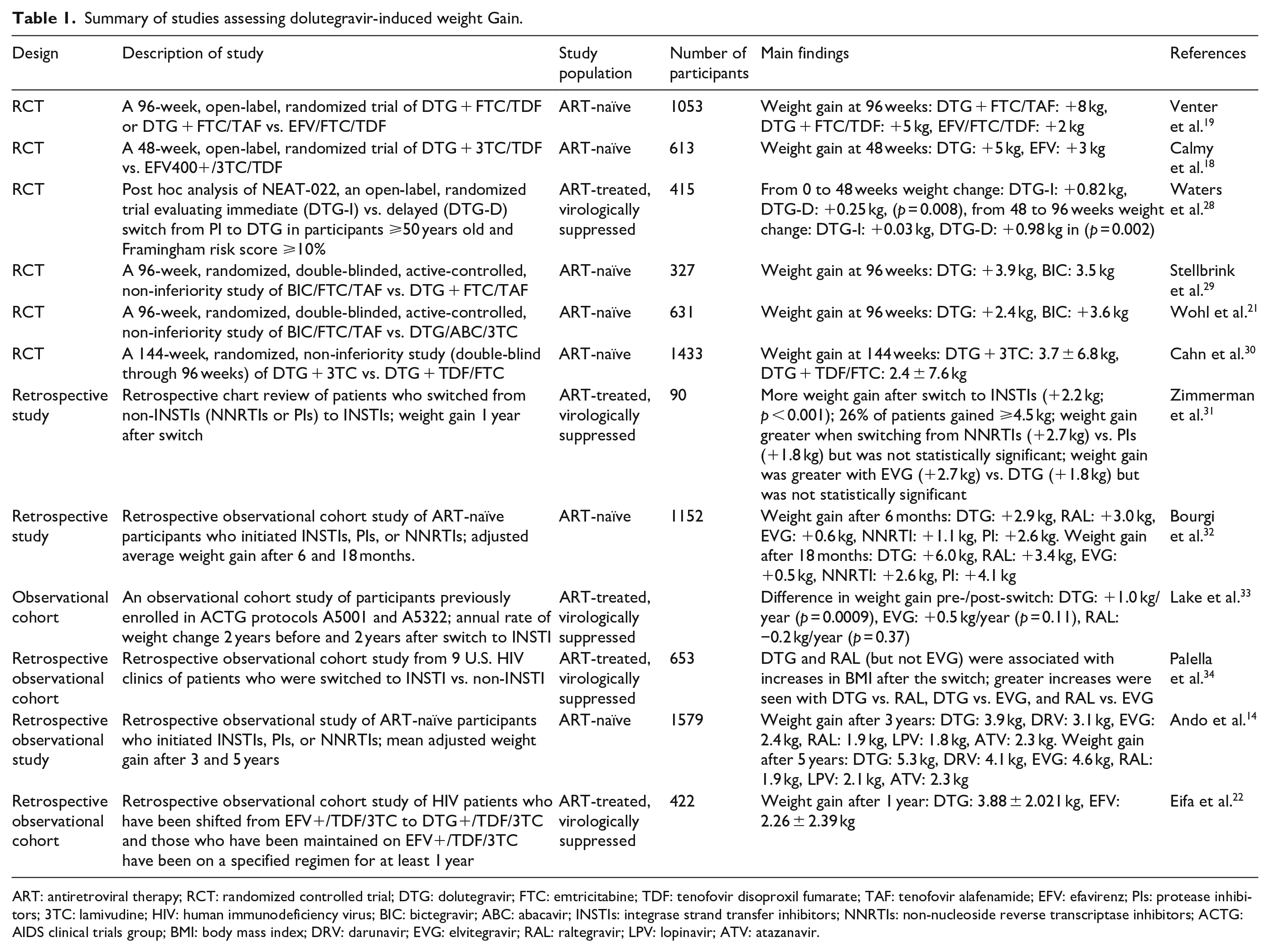
ART: antiretroviral therapy; RCT: randomized controlled trial; DTG: dolutegravir; FTC: emtricitabine; TDF: tenofovir disoproxil fumarate; TAF: tenofovir alafenamide; EFV: efavirenz; PIs: protease inhibitors; 3TC: lamivudine; HIV: human immunodeficiency virus; BIC: bictegravir; ABC: abacavir; INSTIs: integrase strand transfer inhibitors; NNRTIs: non-nucleoside reverse transcriptase inhibitors; ACTG: AIDS clinical trials group; BMI: body mass index; DRV: darunavir; EVG: elvitegravir; RAL: raltegravir; LPV: lopinavir; ATV: atazanavir.
Body composition and fat changes with dolutegravir
Dolutegravir causes changes in the structure of fat cells that may promote obesity and insulin resistance, according to studies of cells taken from HIV-positive people and monkeys given the drug. 35 However, it is unclear how INSTIs may affect metabolism and lead to weight gain. 36 A study from France found that DTG has direct effects on adipose tissue, which could lead to weight gain. 35 To see whether INSTIs affect fat cells directly, DTG was given to human adipose stem cells from HIV-negative women in the laboratory before and during the differentiation process into adipocytes. Dolutegravir was linked to increased production of collagen types linked to adipocyte fibrosis and obesity. 35 As well, INSTI treatment led to elevated levels of collagen, fibronectin, and the myofibroblast marker, that is, smooth muscle α actin (αSMA).35,37 In adipocytes, DTG upregulates the expression of collagen proteins and induces adipose tissue fibrosis by promoting a profibrotic phenotype in adipose stem cells and adipocytes. 38
Dolutegravir exposure was associated with greater adipocyte differentiation and greater expression of markers associated with lipid storage. 38 Cells exposed to DTG had higher levels of lipid accumulation, lower levels of leptin and adiponectin, and a lower uptake of glucose compared to control samples in vitro studies.35,39 Besides, DTG also promotes lipid accumulation when added to already-differentiated mature adipocytes. These results indicate that INSTIs (particularly DTG) enhance adipogenesis and lipogenesis in differentiated adipose stem cells and mature adipocytes. 35 However, another study found that DTG has no significant effect on human adipose cell differentiation. 40 Consequently, further laboratory-based studies may be needed to understand the mechanism of the association between adipose tissue and DTG.
Risk factors for weight gain and fat changes with dolutegravir
Weight gain is more likely in women, black people, and people taking TAF as a DTG companion drug.5,33,41 However, in AFRICOS, there was also a difference in weight gain between men and women, with women having a greater weight gain 1 year post-DTG + 3TC + TDF switch, but this difference was not significant after the first year. 24 According to some studies, regimens with NNRTI anchors appeared to have greater weight gain than PIs anchors when transitioning to DTG-based regimens 33 ; however, there are conflicting data. 42 In AFRICOS, there is no clear evidence of an overall increase in the rate of weight gain following a switch to a DTG-based regimen. 24
There were more women in the NAMSAL study who had a 10% change in weight from baseline in the DTG + 3TC/TDF arm than in the EFV/3TC/TDF arm, a difference not seen in men. Men on DTG + 3TC/TDF, on the other hand, were more likely than men on EFV/3TC/TDF to develop obesity, which was not observed in women. Surprisingly, there was no difference in overweight or obesity incidence between men and women on DTG after 48 weeks, even though more women gained 10% of their body weight. 18 In the ADVANCE study, women on DTG + FTC/TAF gained more weight than men on DTG + FTC/TDF. 43 Recent studies have shown that TAF is associated with body weight gain.5,44 Especially, the combination of TAF and INSTIs has been reported to be associated with an increase in body weight both in treatment-naïve patients 5 and treatment-experienced patients. 45 As for TAF use, the pathophysiology of weight gain is not elucidated at this time but may include, among persons switching from a TDF-based regimen, the absence of weight gain suppression exerted by TDF use, involving much higher plasma levels of tenofovir. 46 The mechanism by which TDF suppresses weight gain is by suppression of appetite or weight gain with potential mitochondrial toxicity. 47
Following ART initiation, both lean and fat mass increased, and a lower CD4+ T-cell count, a higher baseline HIV-1 RNA, and older age were associated with treatment-emergent obesity. 41 HIV patients with long-term viral suppression gained significantly more weight after switching from daily, fixed-dose EFV/TDF/FTC to an INSTI-containing regimen compared to those who remained on EFV/TDF/FTC. Those who switched to the DTG/ABC/3TC regimen gained the most weight. 17 A baseline CD4 cell count of <200 cells/mm3 and a lack of ART were associated with a greater weight change from baseline. 48 Indeed, HIV infection itself and virus activity may also play a role in weight changes. ART-naïve patients and those with the lowest CD4 T-cell counts were at higher risk of weight gain, supporting the hypothesis that weight gain might be explained, at least in part, as a “return to health” phenomenon, the result of successfully suppressing viral replication, controlling inflammation, and normalizing resting energy expenditure. 49
Potential mechanisms of weight gain and fat changes with dolutegravir
The mechanism of weight gain attributable to DTG is unknown, but some mechanisms have been postulated. One possible explanation is the rapid drop in viral load seen with DTG-based regimens and the correlation of virologic suppression with lower energy expenditure. 50 A long-term viral infection depletes fat stores. Body fat stores are replenished when people recover from a severe infection. 51 Another possible mechanistic explanation is possible differences in the effects of ART regimens on systems that regulate energy homeostasis and food intake, as well as insulin resistance. For example, DTG has been shown in vitro to inhibit the activity of the melanocortin-4 receptor (MC4R). Melanocortin-4 receptor is involved in human energy homeostasis, and MC4R-knockout mice are severely obese when both alleles are nonfunctional and moderately obese when only one allele is compromised. 52 In the general population, women have higher circulating leptin levels, 53 subcutaneous adipose tissue leptin mRNA expression, 54 subcutaneous adipose tissue metabolic rates, 55 and more hypothalamic-to-subcutaneous adipose tissue neuronal connections than men, 56 and leptin stimulates the production of pro-opiomelanocortin peptides (POMCs), which reduce food intake and body weight through the MC4R receptor.57,58 Furthermore, leptin receptor expression on POMC neurons may be required for fat distribution modulation in women but not in men. 59 Early implications of DTG weight gain: animal and human studies of MC4R/POMC deficiency suggest that pharmacologic MC4R agonism induces weight loss and that people with POMC deficiency benefit more than people with MCR4 deficiency. 60 Data from PWH are contradictory61,62 in their reporting of sex differences in leptin insufficiency or resistance or do not report sex-stratified results, but it is reasonable to speculate that women may have a differential or more exacerbated response to DTG-induced MC4R functional insufficiency. This is also a proposed mechanism for the weight gain associated with antipsychotics and merits further study with DTG. 63 Clearly, there is no single process that explains DTG-associated weight gain.
The most influential study negating that DTG is causative in weight gain came from the ADVANCE clinical trial. 64 Previous research has shown that EFV is metabolized through the cytochrome P450 2B6 enzyme (CYP2B6) pathways and that slow metabolizers exhibit significant side effects closely associated with elevated drug levels, such as metabolic changes, central nervous system symptoms, and hepatic injury.65,66 Afterward, researchers found that slow metabolizers of EFV who switched to INSTI-containing regimens gained the most weight. 67 Griesel et al. 64 showed that medium and fast metabolizers of EFV had the same weight trajectory as the DTG arm once the slow metabolizers were removed from the analysis, and a subsequent study found that increasing EFV levels in the cohort was associated with greater weight loss. 67
Metabolic comorbidities of dolutegravir-induced weight gain
Weight gain soon after beginning treatment reduces the risk of death in people who were previously underweight. This is an illustration of the “return to health” effect.6,7 Prior research, however, indicates that weight gain following ART initiation increases the risk of diabetes and CVD.6,9 For example, a cohort study report showed that a 1 kg/m2 increase in BMI after initiating ART could have a 12% increased risk of developing diabetes and an 18%–20% increased risk of developing CVD, regardless of the pre-ART BMI, 6 but little is known about whether weight gain with DTG has different effects in terms of comorbidity risk. Several recent studies have suggested that, despite weight gain from DTG, there may not be an equal increase in clinically significant metabolic parameters,68,69 although the data are conflicting.70–72 The prevalence of metabolic syndrome was estimated to be around 10 cases per 1000 person-years. 48 A higher prevalence of metabolic syndrome in PWH than in the general population has previously been reported, 73 with an estimated incidence in developed countries ranging between 8 and 14 cases per 100 person-years.74–76 The ACTG A5001 and A5322 trials found that weight gain was associated with lower high-density lipoprotein (HDL) levels and higher levels of low-density lipoprotein (LDL), total cholesterol (TC), triglyceride (TG) levels, and fasting blood glucose (FBG) among PWH on an INSTIs-based regimen. 33 However, in the SCOLTA prospective cohort study, blood levels of TC, TC/TG, and FBG did not differ between weight gainers and non-gainers, indicating that the metabolic impact of weight gain was minimal. 48
The impact of DTG on insulin sensitivity is still subject to debate. Some researchers have not observed any impact of DTG on the homeostatic model assessment of insulin resistance or glycemia.25,77 Nonetheless, other researchers have found that DTG promoted insulin resistance and lowered circulating adiponectin levels.71,78 Adiponectin expression was low in the subcutaneous adipose tissue of DTG-treated macaques. 35 In addition, despite DTG’s proadipogenic effect in vitro, the drug can impair adipocyte function, resulting in insulin resistance and low leptin and adiponectin secretion. 40 Furthermore, long-term exposure to DTG blunts insulin-stimulated glucose transport and induces insulin resistance in adipocyte-differentiated adipose stem cells (ASCs). 35 In addition, DTG increased reactive oxygen species production and induced mitochondrial dysfunction characterized by increased mitochondrial mass and decreased membrane potential in proliferating ASCs and, to a lesser extent, in adipocytes. 35 Surprisingly, increased levels of oxidative stress have been linked to insulin resistance in vitro and in obese individuals.79,80
Fibrosis is a major feature of adipose tissue dysfunction, and it has been linked to metabolic disorders.35,81 Excess visceral fat is linked to several cardiometabolic risk factors, including high blood pressure, LDL, insulin, and glucose resistance, as well as a decrease in HDL. 82 A large study of over 49,000 people who were followed for 5 years after starting treatment discovered that those who had normal body weight when they started treatment had a higher risk of CVD the more weight they gained after starting treatment. 6 Obesity is associated with adipose tissue expansion, which can result from either adipocyte hypertrophy or hyperplasia. 81 Obesity plays a role in the development of non-alcoholic fatty liver disease (NAFLD). 83 Bischoff et al. 84 showed that the use of INSTIs increases the occurrence of hepatic steatosis and the progression to non-alcoholic steatohepatitis (NASH) in the context of increased body weight. Non-alcoholic fatty liver disease is a risk factor for liver cancer and promotes the development of diabetes and CVD. 85 In general, the impact of DTG on adipose tissue and its possible secondary metabolic comorbidities are described in Figure 1.

Impact of dolutegravir on adipose tissue and its possible metabolic comorbidities.
This work represents a narrative review. As a result, it has a limited scope and does not provide a comprehensive review of the subject matter. The authors did not systematically review the literature, and there were no direct comparisons between studies. Therefore, the material included and the conclusions drawn are subjective, non-comprehensive, and subject to the bias of the author.
Conclusion and future perspectives
There is mounting evidence that DTG-based regimens cause more weight gain and treatment-emergent obesity than other ART regimens, although data are inconsistent, and more randomized studies accounting for diet and lifestyle factors are needed. It is unclear whether DTG-based regimens cause lipid accumulation or increase the risk of cardiometabolic co-morbidities. Therefore, further studies are needed to confirm these findings in larger, multicenter cohorts and investigate the effects on cardiometabolic disease risk factors. There remains much to be learned, both about the mechanisms of DTG-associated weight gain and its health impacts. Future studies are needed to provide more definitive answers to these questions.
Footnotes
Acknowledgements
Not applicable.
Author contributions
Mohammed Jemal is involved in the conceptualization of this review, prepared manuscript draft and writing-up, manuscript approval and validation, manuscript editing, language editing, and design, as this author is the sole author of this manuscript, involved in all aspects of this paper.
Data sharing statement
Supporting data are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Not applicable.
Informed consent
Not applicable.
