Abstract
Introduction:
Healthcare professionals’ awareness of adverse drug reaction reporting and pharmacovigilance practices differ by country. The study assesses healthcare professionals’ knowledge, practice, and potential barriers to pharmacovigilance-related practices and reporting adverse drug reaction.
Methods:
A cross-sectional investigation was conducted in government and private healthcare settings. The study included licensed physicians, pharmacists, and nurses. To examine knowledge, practice, and potential barriers to pharmacovigilance-related practices and adverse drug reaction reporting, a 22-item validated questionnaire was used.
Results:
The final analysis included 311 healthcare professionals. Most healthcare professionals, 59% (N = 182), mentioned encountering patients with adverse drug reactions during the last year. On the other hand, most healthcare professionals, 54% (n = 167), mentioned that they had not reported adverse drug reactions. A good proportion of respondents mentioned that it is essential to report adverse drug reactions (N = 288, 92.6%), availability of adverse drug reactions reporting forms in practice sites (N = 216, 69.5%), had awareness regarding how to report adverse drug reactions (N = 221, 71.1%), the necessity of reporting minor/less important adverse drug reactions (N = 265, 85.2%), and were trained on how to report adverse drug reactions (N = 201, 64.6%). Adverse drug reaction reporting program in the United Arab Emirates (N = 148, 47.6) was known to many healthcare professionals. Lack of time was the major impediment to reporting adverse drug reactions at 42.7% (N = 133). The predictor variable work experience does add to the model (p < 0.05) concerning association with filling of adverse drug reaction forms (Estimate = 0.380; SE = 0.452; p = 0.400), professional role (Estimate = 0.454; SE = 0.673; p = 0.500). In addition, the predictor variable practice setting adds to the model (p < 0.05) concerning the knowledge regarding the availability of adverse drug reaction reporting forms (Estimate = −1.229; SE = 0.298; p = 0.000), training on how to report adverse drug reactions (Estimate = −0.660; SE = 0.294; p = 0.025), and awareness regarding the adverse drug reaction reporting program in the United Arab Emirates (Estimate = −1.032; SE = 0.280; p = 0.000).
Conclusion:
Pharmacists had the most knowledge regarding adverse drug reaction reporting and pharmacovigilance. The underreporting of adverse drug reactions was documented among physicians and nurses. Lack of time was the most significant barrier to reporting adverse drug reactions, followed by uncertainty and complicated adverse drug reaction documentation forms.
Keywords
Introduction
Healthcare professionals (HCPs) are frontline stakeholders in detecting, monitoring, and reporting adverse drug reactions.1,2 However, in many countries, adverse drug reactions (ADRs) are underreported. 3 Studies from different parts of the world have attempted to identify the reasons for underreporting by surveying HCPs’ knowledge, attitudes, and pharmacovigilance practices.4–6 There is also a large body of work evaluating the pharmacovigilance practices of different HCPs.
HCPs’ awareness of pharmacovigilance and ADR reporting varies among countries. Some studies have documented lower awareness regarding the ADR reporting and monitoring system.5–7 Few studies have revealed a lack of understanding about how, where, and when to report an ADR.8–10 In contrast, studies have also documented good knowledge of ADR reporting.1,11 Furthermore, many studies have also reported lower awareness about the ADR reporting system among different HCPs in Uganda, 2 Bosnia, 3 Saudi Arabia, 12 Pakisthan,13,14 Nepal, 15 Zambia, 16 Italy, 17 and Kuwait. 18 Conversely, respondents exhibited intermediate knowledge regarding ADR reporting and pharmacovigilance in a study from Jordan. 19 It is vital to enhance the awareness of HCPs to improve ADR reporting. The lack of a proper reporting system may lead to underestimating the prevalence and severity of ADRs in the population and pose a major challenge for regulatory authorities to make appropriate decisions, such as creating an alert or drug withdrawal. Further, not identifying and reporting ADRs may significantly impact patient morbidity and mortality.5,6
Few studies have evaluated the association between demographic/profession-related variables and pharmacovigilance/ADR reporting knowledge, attitude, and practice (KAP).19,20 The findings of these studies vary, where some researchers found no significant associations between demographic parameters (age, gender, nationality, profession, years of experience, and practice setting) and attitude or knowledge. 19 Some research has found a substantial relationship between knowledge and occupation, gender, and professional standing. 15 Understanding the demographic/professional variables and ADR reporting knowledge, attitude, and practice could be beneficial in establishing targeted interventions and improving pharmacovigilance.
Pharmacists, such as physicians and nurses, reported having more knowledge of pharmacovigilance than other HCPs.12,16,17,19,20 Despite under-reporting, HCPs had a good attitude towards ADR reporting/pharmacovigilance, as reported in a few studies.3,5,9,11,17–19 The most common reasons cited for under-reporting of ADRs in these studies are professional obligation, lack of understanding of how and where to report, insufficient clinical knowledge, insufficient time, lack of training, no availability of forms, and a fear of legal liability.4,10,11,17 Studies have also reported a lack of training, inadequate communication between physicians and other HCPs, and, most significantly, an effective reporting system.13,16,18 It is important to have a clear understanding of the barriers or factors that contribute to the underreporting of ADRs. By doing so, we can implement measures to address these issues and improve the overall reporting culture.13–17
In the United Arab Emirates (UAE), the Ministry of Health and Prevention (MOHAP) manages healthcare. Besides, National Health Authorities oversee healthcare systems in individual emirates of the UAE. The MOHAP works with each of these health authorities. The MOHAP is responsible for monitoring the safety of medications in the whole country. Ras Al Khaimah is in the Northern parts of the UAE, and pharmacovigilance activities are generally overseen by the respective branches of the MOHAP. The UAE National Pharmacovigilance Center is located in Abu Dhabi, the capital city of the United Arab Emirates. It operates under the MOHAP, evaluates and analyzes all reported ADRs, and submits the data to the WHO-Uppsala Monitoring Center and WHO for examination and recording.21,22
Unfortunately, underreporting is still common in the UAE despite several measures by regulatory bodies. Interestingly, only a few studies focused on assessing the pharmacovigilance practices among physicians, hospital pharmacists, and nursing staff in a single study. Therefore, conducting comprehensive investigations to explore and evaluate HCPs’ roles and contributions to pharmacovigilance activities is essential. Besides, earlier studies were less focused on assessing pharmacovigilance practices among HCPs in primary healthcare centers. Therefore, conducting comprehensive investigations to explore and evaluate HCPs’ roles and contributions to pharmacovigilance activities is essential. Earlier research studies have reported an association between underreporting ADRs and inadequacies in the knowledge, attitude, and practice among HCPs regarding pharmacovigilance practices in other parts of the world. Awareness regarding the pharmacovigilance system and ADR reporting varies among other countries.
Furthermore, the lack of knowledge, impediments to ADRs, and ADR reporting described in Middle Eastern studies may be comparable or different. Hence, it is essential to conduct similar research to generalize the findings of those studies. The data obtained from the study could help strengthen the ADR reporting system and pharmacovigilance activities in the study settings and by the local pharmacovigilance center. The study intends to evaluate HCPs’ knowledge and practice of pharmacovigilance, discover the potential barriers to pharmacovigilance-related practices, and identify the demographic characteristics associated with pharmacovigilance practices among HCPs.
Materials and methods
Study design, setting, and duration
An exploratory study was carried out at eight hospitals (four public and four private) and nine primary healthcare clinics (PHCs) in Ras Al Khaimah, a region in the Northern Emirates of the UAE. The study spanned 8 months from January 2021.
Study sample size and sampling technique
The overall sample size was estimated to be 310 using a formula for determining sample size based on a single proportion formula
Study inclusion and exclusion criteria
The study included licensed HCPs (nurses, pharmacists, and physicians) who are permanently employed in the study settings and consent to participate. The study excluded enrolling HCPs in administrative positions, residents, trainees, pharmacy technicians, and HCPs on extended leave. Further, the study did not include pharmacists who work in community pharmacy settings. The study also excluded HCPs who declined to participate and HCPs with less than 1 year of experience. Further, the study excluded HCPs who were not directly involved in patient care.
Development of survey instrument
Investigators developed a 22-item survey instrument in the English language to assess the HCPs’ ADR reporting and pharmacovigilance practice. The questions were prepared based on the extensive literature review on the research topic and input from the three external content experts experienced in pharmacovigilance research and practice. Content experts provided input on the relevancy and clarity of the suggested survey items. The questions were carefully identified, selected, reviewed, and amended based on the study’s objective. The survey item pool is organized according to the domains and constructs found. Ensure that each item is clear, short, and related to the targeted component of HCP’s knowledge, attitude, or practice in ADRs. Finally, the pilot questionnaire was tested on diverse HCPs (Physicians (n = 10), Pharmacists (n = 10), and Nurses (n = 10)) to evaluate the survey items for clarity, comprehension, and correctness. The instrument was amended accordingly based on the input using open-ended questions.
Validation of the survey instrument
The survey questionnaire was validated for both content and reliability before administration. The content was validated using a two-step process developed by Lynn 23 Ten panelists validated the questionnaire. The content validity index and the Kappa coefficient of the agreement were calculated based on the panelists’ quantitative judgments. The items with Cronbach’s coefficient α, above 0.75, were retained, and items below this acceptable standard were deleted, resulting in the retention of 22 items.
Identification and selection of the target population
HCPs satisfying the study’s inclusion criteria were selected from the identified study sites. A sample frame was created by combining lists of eligible HCPs collected from identified hospitals and PHCs. Stratified sampling was used to categorize respondents depending on their occupation. Random sampling methods were used within each stratum to guarantee representative and unbiased sampling.
Administration of survey instrument
The questionnaire was self-administered, and the anticipated duration for completing the questionnaire was expected to range from 10–15 min. The questionnaire was designed to collect both qualitative and quantitative data. The majority of the items (1–16 in total) reflecting pharmacovigilance practice or ADR reporting were scored on a three-point Likert scale ranging from “not sure” to “yes.” While four questions (17–20) were rated using a four-point Likert scale (“never” to “frequently”), 24 respondents were required to answer all questions in the questions section to advance within the questionnaire.
Data collection
The survey instrument was disseminated online and in hard copies in accordance with the preferences and accessibility of the specific HCPs being targeted. The survey instrument was distributed using a Google survey form for HCPs whose email IDs are available and active. The study’s investigators personally delivered and collected the hard copies of the survey instrument in case of nonavailability or accessibility of the email IDs. Upon receiving the questions via the Google survey form, the respondents were presented with the informed consent form page. They were directed to the main survey instrument only after voluntarily indicating their consent by checking a box. The respondents willingly participated in the study by signing the informed consent and completing the survey questionnaire in hard copy format. The study ensured the confidentiality of the survey participants throughout the research. A systematic approach was employed for follow-up and reminders after the initial distribution of the survey instrument. Reminders were sent out repeatedly across various communication channels, including tailored emails and phone calls. A non-response is recorded if the questionnaire is not returned within 3 weeks.
Data analysis
Data was collected from all the study sites, compiled, and analyzed using the IBM Statistical Package for Social Sciences (SPSS) version 27 software (SPSS Inc., Chicago, IL, USA). Chi-square analysis evaluated the potential association between demographic factors and participants’ rankings. Ordinal logistic regression analysis evaluated bivariate associations between the independent and the outcome variables.
Ethical considerations
The regional MOHAP Research Ethics Committee approved this research (MOHAP/REC/2020/60-2020-F-P). All study participants received verbal or written information regarding the study. Written informed consent was obtained from all the study participants.
Results
Among the 970 HCPs contacted, 316 replies were obtained, resulting in a response rate of 32.5%. One hundred ninety-four responses were collected via Google Forms, while 122 were collected in paper format. Five responses were deemed invalid because most of the questionnaire’s items were incomplete. A total of 311 comprehensive responses were considered for the ultimate analysis.
Characteristics of the study participants
Pharmacists made up the majority of the HCPs that took part in this study, accounting for 35.7% (n = 111), followed by nurses (32.2%) (n = 100), and physicians (32.2%) (n = 100). Most participating HCPs were females, accounting for 56.3% (n = 175). About 29.9% (n = 93) of participants belong to the 31–35 age group, followed by the 25–30 age group, which is 25.1% (n = 78). The average age of study participants was 37.56 ± 10.1 years. Only 10.9% (n = 34) of the participants had specialized in their field, while 71.4% (n = 222) were graduates. The majority of participating HCPs, that is, 27.7% (n = 86), had 5 years of experience and 6–10 years of experience, while 18.3% (n = 57) had an experience of more than 20 years, with an overall mean experience of 12.4 ± 9.6 years. The vast majority of the HCPs that took part in the study were from Government hospital settings, that is, 61.7% (n = 192). The demographic details of the study population are documented in Table 1.
Study participants’ demographic overview.

Pharmacovigilance practices among HCPs
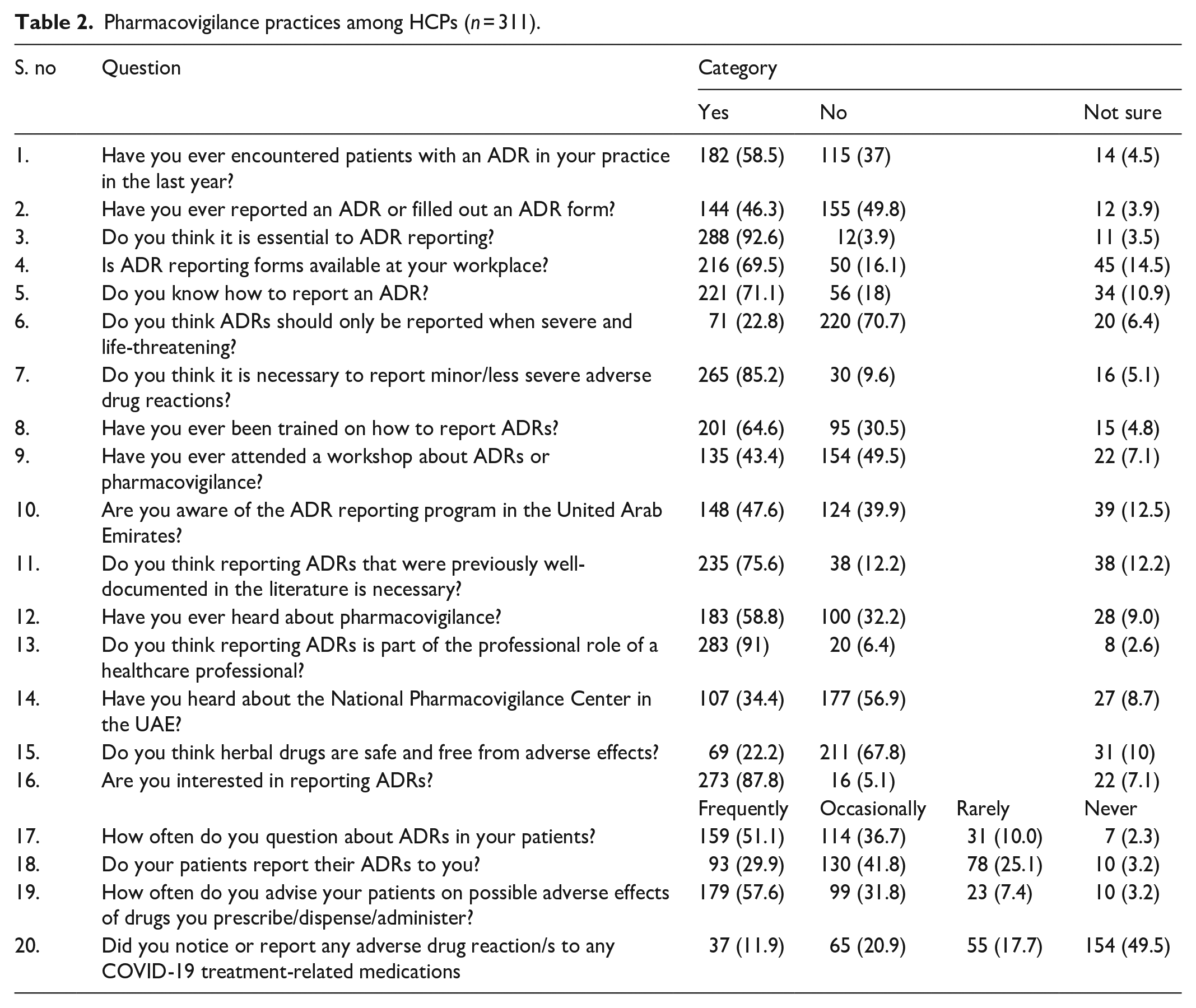
Most HCPs, 59% (N = 182), mentioned encountering patients with ADRs during the last year. While a small percentage of respondents, 4% (N = 14), were unsure about it. Furthermore, 54% of HCPs (N = 167) mentioned not reporting ADRs. A good proportion of the respondents mentioned that it is essential to report ADRs (N = 288, 92.6%), availability of ADR reporting forms in practice sites (N = 216, 69.5%), had awareness regarding how to report ADRs (N = 221, 71.1%), the necessity of reporting minor/less important ADRs (N = 265, 85.2%), and were trained on how to report ADRs (N = 201, 64.6%). At the same time, a greater percentage of HCPs (N = 220, 70.7%) did not agree that only severe and life-threatening ADRs should be reported. A considerable proportion of HCPs had an awareness regarding the term pharmacovigilance (N = 183, 58.8%), ADR reporting program in the UAE (N = 148, 47.6), reporting ADR as a professional role of HCP (N = 283, 91%), necessary to report well-documented ADRs (N = 235, 75.6%). In contrast, a more significant proportion of HCPs (N = 177, 56.9%) and (N = 154, 49.5%) were unaware of the presence of the UAE National Pharmacovigilance Center and attended the workshop on pharmacovigilance, respectively. A more significant percentage of respondents (n = 211, 67.8%) opined that herbal drugs are not safe and free of ADRs and were interested in reporting ADRs in the future (N = 273, 87.8%). A good percentage of the HCPs, 51.1% (N = 159), mentioned that they often question their patients’ ADRs and advise them regarding ADRs 57.6% (N = 179). While 41.8% (N = 130) of HCPs mentioned that their patients report encountering ADRs to them. Notably, a small percentage of HCPs, that is, 11.9% (N = 37), have noticed ADRs to COVID-19-related medications. Pharmacovigilance practices among HCPs are documented in Table 2.
Pharmacovigilance practices among HCPs (n = 311).
HCPs versus pharmacovigilance practices
Notably, the majority of the HCPs encountering patients with ADR were pharmacists (67.6%), followed by physicians (65%) and nurses (42%). A similar trend was observed in documenting and reporting ADRs, with pharmacists filing 66% of forms, contrary to physicians (39%) and nurses (38%). In contrast to pharmacists and physicians, around 85% of nurses were aware of ADR reporting forms and procedures for reporting an ADR. A large majority of the HCPs, 92% of nurses, 87.4% of pharmacists, and 76% of the physicians mentioned it is required to report an ADR, even if it is minor/less severe. Comparative data regarding PV practices by HCPs are presented in Table 3.
Cross tabulation of type of HCPs versus pharmacovigilance practices.

p < 0.05 is statistically significant.
**p < 0.01 is statistically highly significant.
ADRs noticed by HCPs
When the respondents were asked to list any two drugs that they have noticed ADRs in their current practice, a total of 312 ADRs to a total of 104 drugs were listed by 278 respondents. The most common therapeutic class of drugs involved in ADRs was anti-infective drugs. The highest percentage of ADRs was reported for Ceftriaxone, 7.69% (n = 24), followed by Diclofenac, that is, 7.37% (n = 23). Some of the examples of classes of drugs and types of ADRs noticed by the respondents are presented in Table 4.
Classes of drugs and types of ADR noticed by HCPs.

Barriers to reporting ADR
When participants were asked about the barriers in reporting ADR, 42.7% (n = 133) of HCPs found reporting to be time-consuming, while over 39.8% (n = 124) were not sure about whether it is an ADR and 33.7% (n = 105) indicated that ADR reporting forms were complicated. In addition, some respondents (12.8%) did not report because they were worried about the legal liability. The different barriers to reporting ADRs are depicted in Figure 1.
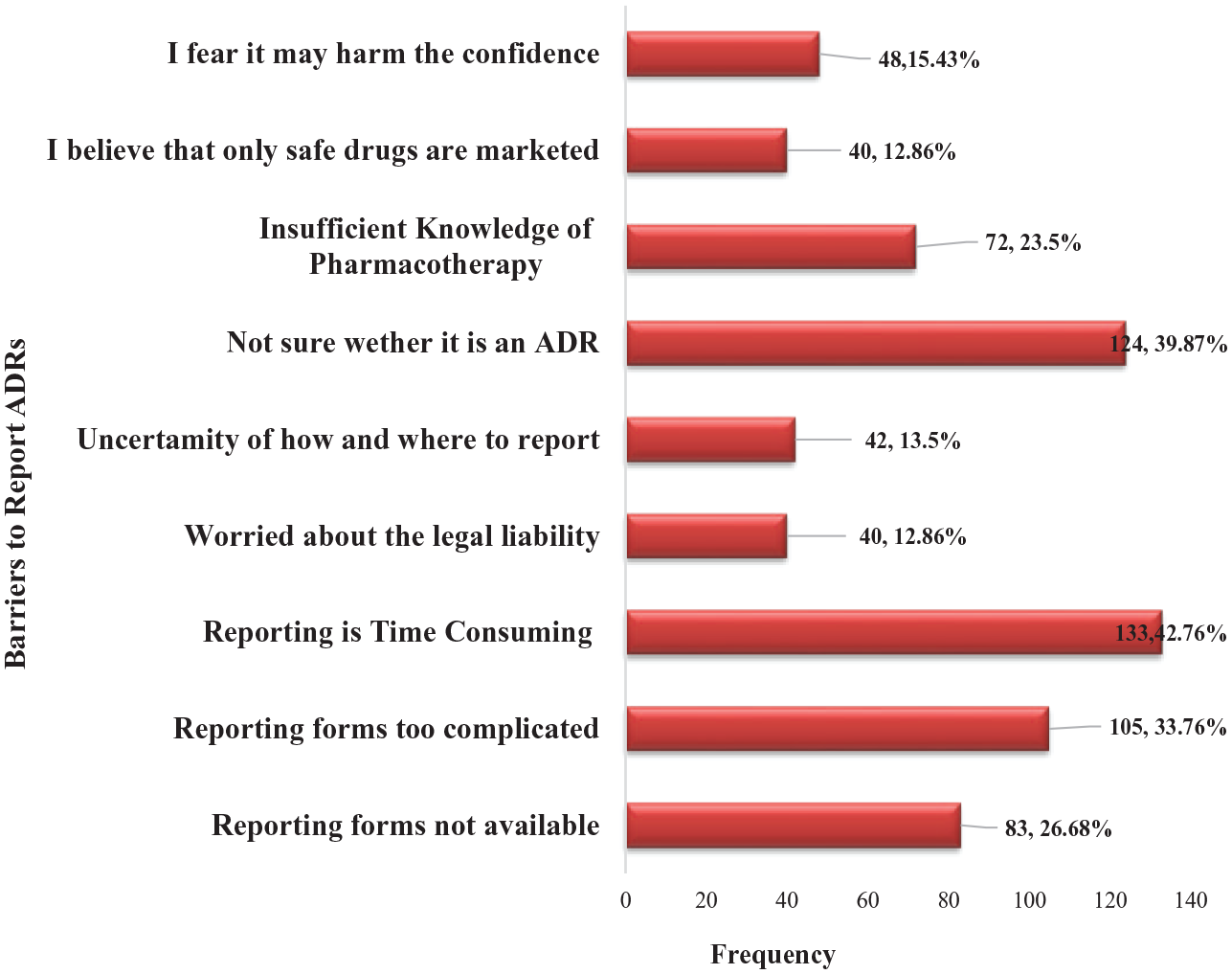
Barriers to reporting ADR.
Ordinal logistic regression analysis for pharmacovigilance practices
The predictor variable work experience does not add to the model concerning association with filling of ADR forms (Estimate = 0.380; SE = 0.452; p = 0.400), Professional role (Estimate = 0.454; SE = 0.673; p = 0.500). The Ordinal Logistic Regression Analysis for pharmacovigilance practices and work experience is presented in Table 5. The predictor variable practice setting adds to the model (p < 0.05) concerning the knowledge regarding the availability of ADR reporting forms (Estimate = −1.229; SE = 0.298; p = 0.000), training on how to report ADRs (Estimate = −0.660; SE = 0.294; p = 0.025), and awareness regarding the ADR reporting program in the UAE (Estimate = −1.032; SE = 0.280; p = 0.000) as presented in Table 6. At the same time, the predictor variable continuing medical education (CME) adds to the model (p < 0.05) concerning reporting of ADRs (Estimate = 0.963; SE = 0.479; p = 0.044) and the safety of herbal drugs (Estimate = 1.228; SE = 0.416; p = 0.003), as presented in Table 7.
Ordinal logistic regression analysis for pharmacovigilance-related practices and work experience.

p < 0.05 is statistically significant.
**p < 0.01 is statistically highly significant.
Ordinal logistic regression analysis for pharmacovigilance-related practices and practice setting.

p < 0.05 is statistically significant.
**p < 0.01 is statistically highly significant.
Ordinal logistic regression analysis for pharmacovigilance-related practices and continuing medical education.

p < 0.05 is statistically significant.
**p < 0.01 is statistically highly significant.
Discussion
This study intends to evaluate HCP’s knowledge and practice of ADR reporting and pharmacovigilance and determine potential barriers to identifying, monitoring, documenting, and reporting ADRs. Most HCPs (59%) mentioned encountering patients with ADRs during practice. In agreement with our findings, Gupta et al. 4 reported that most participants (64.4%) had encountered ADRs in their patients. Another study reported that 69.6% of the survey respondents encountered patients with ADRs. 5 The percentage of encountering ADRs was slightly higher in these two studies. The possible reasons for this could be the presence of a regular ADR reporting and monitoring system. In addition, previous experience of the HCPs concerning the identification and monitoring of ADRs might have contributed to better recognition of ADRs. Interestingly, a pilot study conducted in 2014 in two of the hospitals included in our study reported that a significant number of the physicians mentioned that only around 10% of their patients report their ADR. 7
In our study, the majority (54%) of the study respondents have not reported ADRs. In contrast, most HCPs (86.1%) had encountered an ADR during their practice, whereas 71.3% had reported it, as reported in a Saudi-Arabia-based study. 25 In contrast with our findings, a study reported that only 22.8% of HCPs reported ADRs to a pharmacovigilance center. 4 Meanwhile, only a tiny percentage of surveyed HCPs reported ADRs as reported in studies from Bosnia (15.4%), Sri Lanka (18.2%), and Turkey (13.1%).3,26,27 A good percentage (92.6%) of our study respondents agreed that it is essential to report ADRs; this is comparable to the findings of another study, which reported that a higher percentage (97%) of HCPs agreed that reporting ADRs is necessary. 4
The knowledge of the availability of ADR forms at practice sites was significantly higher among pharmacists compared to physicians and nurses; this could be because most pharmacists have attended pharmacovigilance-based workshops, and there is a presence of a pharmacovigilance reporting system in UAE, with separate PV officers have been nominated to each emirate to strengthen and facilitate the reporting. In contrast to our findings, 37.5% of health personnel have encountered the ADR reporting form. 1 A research found that just 34.7% of participants were acquainted with where to access the ADR reporting form, which contradicts our findings. 5 The finding indicates that it is essential to create awareness among physicians and nurses regarding ADR documentation and reporting.
A noticeable proportion of our study respondents (71.1%) reported knowing how to report ADRs. In contrast, a South African study found that 60.5% of respondents did not know how to report an ADR, and 51.5% indicated that their clinical education level made it difficult to determine whether an ADR occurred. 8 Around 64% of the study respondents mentioned being trained to report ADRs. However, our study did not assess the training required to report ADRs. Whether HCPs received formal training, in-house or from the MOHAP, or whether it indicates the formal reporting of ADRs in patients’ case notes or electronic records was unknown. In comparison, a study reported that 53.5% of healthcare workers received training to report ADRs. 4
Only 56.9% of our study population had awareness regarding the national pharmacovigilance system. A Saudi Arabian study reported high awareness (88.9%) regarding the responsible regulatory agency. 25 However, pharmacists’ knowledge of a national pharmacovigilance system was high. This could be due to the training they received or the workshops they attended. Other studies have also reported a higher knowledge of pharmacovigilance and ADR terminology among pharmacists.12,28 A recent national-based survey study conducted in UAE also reported a high percentage (93.3%) of knowledge regarding PV and ADR reporting among hospital pharmacy practitioners. 29 In contrast to our findings, the research found that 88.7% of physicians were unaware of the National Pharmacovigilance Centre. 6 It is essential to know about the National Pharmacovigilance Center, its roles, and responsibilities as it helps to enhance the PV reporting system.30–35 According to Saudi Arabian research, 59.1% of HCPs were ignorant of the Saudi Food and Drug Authority’s (SFDA) National Pharmacovigilance Center (NPC), and 36% assumed the Ministry of Health was responsible for receiving and assessing ADR reports. 11 A study conducted in Italy found that many nurses lacked awareness of several aspects of the pharmacovigilance system. Specifically, 58.1% (n = 331) were uninformed of the system itself, 63.5% (n = 362) did not know where to obtain the reporting form, 71.6% (n = 408) were unfamiliar with how to fill it in, and 65.8% (n = 375) were unsure about to whom and how to send it. 17 Similarly, 75.2% of healthcare workers knew of India’s National Pharmacovigilance Program. 4
On a positive note, a more significant percentage of HCPs did not agree that only severe and life-threatening ADRs should be reported and felt it necessary to report even minor and less severe ADRs. Similar to our observations, a study by Kiguba et al. 36 documented that 54.4% of the study respondents reported, “It is only needed to report serious or unexpected ADRs.” 36 Assuming that ADR is well known and minor, it might contribute to underreporting, as reported in a community pharmacy-related study from the Netherlands. It should be stressed that even though ADR is well known, it should be reported. 37 A significant proportion of our study population cited that they were trained in reporting ADRs; this could be the reason for a higher degree of awareness regarding the many components of ADR reporting. In contrast to our findings, according to one study, only 53.5% of healthcare personnel were instructed to report adverse responses. 4 According to another study, 95.7% of respondents were dissatisfied with their ADR reporting training. 6
Proper ADR training is crucial in determining the timely identification and reporting of ADRs. At the same time, improper training is considered a barrier or limiting factor.13,16 All HCPs should be trained, and the training should be reinforced regularly to strengthen the reporting system. A pilot study conducted at a couple of the same study sites reported that many physicians were interested in getting trained on ADR reporting, which is a very appreciative and encouraging factor. 7 Furthermore, in our study, the number of pharmacists who had received training and attended the pharmacovigilance workshops was significantly higher than that of the physicians and nurses. This indicates the necessity of training the nurses and physicians to strengthen the pharmacovigilance system.
Compared to physicians and nurses, pharmacists were much more aware of the pharmacovigilance system (>80%). A survey revealed that pharmacists and pharmacy technicians had the highest level of awareness regarding pharmacovigilance, with 60.5% of pharmacists and 40% of pharmacy technicians demonstrating this understanding. However, according to the same study, most healthcare workers (62.5%) were ignorant of pharmacovigilance. 6 Another study found that while most HCPs (72.5%) had heard of pharmacovigilance, just three (5.2%) accurately grasped the concept. Twelve (15.0%) indicated acceptable ADR understanding, whereas 37 (46.2%) revealed a favorable attitude toward ADR reporting. 1 According to a study conducted in South India, 62.4% of healthcare personnel correctly defined pharmacovigilance. 4 While in a Saudi Arabian study, it was 42%. 25 Interestingly, the survey participants heard the term “pharmacovigilance” (35.5%) for the first time in a study. 5 A Study from Cyprus reported that only 13% of the pharmacists, 2% of the nurses, and 20% of the physicians knew about pharmacovigilance. 10 A recently published study from Sri Lanka reported a high (90%) awareness regarding the term ADR by the HCPs. 26 Awareness of the accurate definition of ADR or PV is essential since it helps to guarantee patient safety and the proper reporting and monitoring of ADRs.
A significant proportion of our study population (91%) agreed that reporting ADRs is a professional responsibility of HCPs. In comparison, studies from other countries have documented almost similar observations.25,28 In an Indian study, 70% of respondents opined that reporting ADR is their professional responsibility. 38 Based on a study, over 97.5% of participants believe that they should report ADRs. Among them, 89% consider it a professional duty, while more than 70% believe that it should be obligatory. The aggregate mean score for positive or preferred ADR reporting methods was 24.6%, with pharmacists achieving the highest score. 8 In addition, a study conducted in Kuwait revealed that a significantly lower proportion of physicians (78.0%) compared to pharmacists (88.0%) viewed ADR reporting to be a professional obligation (N = 248/318 vs 147/167, p < 0.01). 18 Similar observations have been documented in studies from other parts of the world. 4
When we asked our respondents whether they noticed any adverse effects of COVID-19-related medications, a small percentage of respondents mentioned that they had noticed adverse effects of COVID-19-related medications; since three of our study sites were catering services to COVID-19 patients on an inpatient and outpatient basis, the HCPs working in these hospitals/PHC might have reported the same. Another highlight of our study was that the HCPs were asked to report the ADRs they had recently encountered in their practice. It is overwhelming that a significant proportion of HCPs reported different ADRs to various classes of drugs that they encountered. For example, anti-infective drugs followed by anti-inflammatory drugs were the most commonly associated drugs involved in ADRs. This observation of our study provides a basis for further Intensive monitoring of Anti-infective and anti-inflammatory drugs in healthcare settings. Only a few studies have tried to acquire this data type through survey-type studies. 7 Although there is a chance of duplication in the reported number of ADRs, all the HCPs working in a single department might have documented and reported the same ADR. In addition, the causality and severity of these ADRs may also be questionable. However, the study did its best to accumulate the pharmacovigilance-related data.
Some of the barriers cited in this study were consistent with similar studies, such as reporting being time-consuming, reporting forms being too complicated, being unsure whether it is an ADR, reporting forms not being available, worrying about legal liability, etc.13,16 A research found that 50.4% of HCPs thought that the reporting form was excessively difficult. In comparison, 58.1% thought that reporting ADRs was time-consuming. Insufficient clinical knowledge was a major constraint in reporting ADR (64.9%). 11 Studies conducted in Pakistan and Zimbabwe have also documented similar barriers.13,16 These studies, however, have revealed additional impediments, such as inadequate communication between physicians and other healthcare providers, and practitioner fears that the information presented may be incorrect. Studies have also documented the lack of training and awareness of reporting procedures and the national ADR reporting system as reasons for underreporting.19,27,29,38 Identifying barriers to reporting ADRs is essential to eliminate the barriers through pharmacovigilance-related awareness programs and workshops.
A significant association was documented between some of the variables and pharmacovigilance-related items. The practice settings of HCPs were a significant predictor of the availability of reporting forms, training received, and awareness about the National Pharmacovigilance Center. HCPs working in PHCs reported a higher pharmacovigilance-related awareness than HCPs working in government hospitals and private settings. In contrast, the working experience was not associated with any variables. Higher job experience (10 years) (AOR = 0.36, 95% CI: 0.13–0.97) was significantly associated with reporting ADR and practice in an Ethiopian study. 9 In our study, pharmacists had a significantly higher awareness than other HCPs. In contrast, a study found that nurses were much more knowledgeable about ADRs than other cadres (p < 0.001). 1 A separate study showed a statistically significant disparity (p = 0.004) in the overall score among other professions, with pharmacists (n = 18) obtaining a mean rank score of 34.08, doctors (n = 24) achieving 22.17, and nurses (n = 8) attaining 16.19. 16
Another significant observation was that continuing medical education was associated with some of the items related to pharmacovigilance practices, such as reporting or filling out ADR forms and awareness regarding the safety of herbal drugs. Unlike our results, a study conducted in Jordan identified no connections between attitude, knowledge, age, gender, and professional experience. 19 Few studies have shown a link between demographic factors and KAP for pharmacovigilance/ADR reporting. In a few research, Rabayah et al. 19 discovered no correlations (p > 0.05) between demographic factors (age, gender, nationality, occupation, years of experience, practice setting) and attitude or knowledge. 19 While some research has shown a significant (p < 0.05) relationship between knowledge and occupation, others have found a relationship between gender, professional position, and knowledge. 15 A study by Khan et al. 27 reported that the profession of HCPs and a lack of training were predictors of poor ADR reporting (p < 0.05).
Our study’s main highlight was that it attempted to document ADRs encountered by HCPs in their daily practice. The study emphasizes educating all HCPs on ADR monitoring, documenting, reporting, and awareness of pharmacovigilance. Moreover, it is crucial to enhance the collaboration among academic institutions, pharmaceutical manufacturers, drug regulatory agencies, and HCPs in order to raise awareness about the provision of drug interaction (DI) services and ADR reporting protocols.
The study had certain constraints. The cross-sectional design restricted the capacity to establish causative linkages or observe variations over time. The sample size was calculated using a single proportion formula and did not include a power analysis. Recall bias was another limitation, particularly regarding the accuracy of participants’ recall of prior experiences or practices connected to ADR responses. While attempts were made to construct a valid survey instrument, inherent limitations in self-reported measures may have affected the instrument’s validity. The study was conducted in Ras Al Khaimah, a region in the Northern Emirates of the UAE. Therefore, the findings of this study cannot be extrapolated to other provinces in the country.
Nevertheless, Ras Al Khaimah is one of the most developed regions, offering state-of-the-art healthcare facilities. Therefore, it is anticipated that the outcomes in the other sections would be quite similar. Furthermore, the selection of the study sites was purposeful because of their easy reachability and accessibility. However, it is unlikely that the results would have been very different in other hospitals as the pharmacovigilance-related practices are most likely uniform.
Because the information was self-reported, healthcare providers may not have documented actual pharmacovigilance or ADRs reporting methods. However, this is one of the common limitations addressed in most survey-based studies. The cross-sectional nature of this study may preclude establishing a causal relationship between ADR reporting and explanatory variables. Reaching all the planned study respondents was an added challenge due to the current COVID-19 situation. However, the study achieved its target sample size with constant follow-up, reminders, and personal visits to collect the questionnaires.
Conclusions
The study provides a comprehensive overview of HCPs’ knowledge, attitude, and practice regarding pharmacovigilance practices in the UAE. While a significant proportion of HCPs acknowledged encountering patients with ADRs, there is a significant difference in the reporting process, with more than half of the study respondents admitting that they do not report ADRs. The study emphasizes positive factors, such as most respondents acknowledging the significance of ADR reporting and being aware of reporting processes. Among all HCPs, pharmacists had the most knowledge regarding ADR reporting and pharmacovigilance. Lack of time was the most significant barrier to reporting ADRs. Many HCPs indicated limited knowledge of the UAE National Pharmacovigilance Center and reported not attending pharmacovigilance training sessions. Strengthening these components can help to create a more knowledgeable and engaged healthcare staff in pharmacovigilance. In addition, variables such as practice setting and CME hours were significant predictors of pharmacovigilance and ADR reporting components. Although HCPs’ understanding and attitudes about ADR reporting are encouraging, the detected shortcomings indicate the need for focused interventions, training programs, and standardization.
Supplemental Material
sj-docx-1-smo-10.1177_20503121241249908 – Supplemental material for Exploring pharmacovigilance practices and knowledge among healthcare professionals: A cross-sectional multicenter study
Supplemental material, sj-docx-1-smo-10.1177_20503121241249908 for Exploring pharmacovigilance practices and knowledge among healthcare professionals: A cross-sectional multicenter study by Abdulkader Hayek, Sathvik B Sridhar, Syed Arman Rabbani, Javed Shareef and Tarun Wadhwa in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121241249908 – Supplemental material for Exploring pharmacovigilance practices and knowledge among healthcare professionals: A cross-sectional multicenter study
Supplemental material, sj-docx-2-smo-10.1177_20503121241249908 for Exploring pharmacovigilance practices and knowledge among healthcare professionals: A cross-sectional multicenter study by Abdulkader Hayek, Sathvik B Sridhar, Syed Arman Rabbani, Javed Shareef and Tarun Wadhwa in SAGE Open Medicine
Footnotes
Acknowledgements
We thank the Medical Directors and heads of the Nursing and Pharmacy Departments of Shaikh Saqr Hospital, Ibrahim bin Hamad Obaidallah Hospital, Omran Hospital, and Shaam Hospital, Ras Al Khaimah, UAE, for their excellent cooperation and support in conducting our research. We thank Mrs. Khadija Humaid, Senior Pharmacist, RAK PHC, for the kind and timely help. We sincerely thank all the Physicians, Pharmacists, and Nurses from Primary Healthcare clinics and other research sites for their kind support. We thank the Dean of RAK College of Pharmacy and the President of RAK Medical and Health Sciences University for all the support rendered during the research.
Author contributions
AK: Writing research proposal, data collection, writing original draft preparation. SBS: Conceptualization, research proposal review, supervision, data collection, statistical analysis, draft preparation, review, editing reviewing, and editing. SAR: Data collection, Survey Instrument development, draft review and editing. JS: Conceptualization, Survey Instrument development, Data collection, draft review and editing. TW: Data collection, Survey Instrument development, draft review and editing.
Availability of data and materials
Not applicable.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The regional Ministry of Health and Prevention, Research Ethics Committee, approved this research (MOHAP/REC/2020/60-2020-F-P).
Consent for publication
Not applicable.
Informed consent
All study participants received verbal or written information regarding the study. Written informed consent was obtained from all the study participants.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

