Abstract
Transforming the orthopedic landscape, hip arthroscopy pioneers a minimally invasive surgical approach for diagnosing and addressing hip pathologies. With its origins dating back to Burman’s 1931 cadaveric study, this groundbreaking technique gained clinical relevance in 1939 through Takagi’s report. However, the 1980s marked the actual emergence of hip arthroscopy for treating a wide range of hip disorders. Now, a staple in modern orthopedics, hip arthroscopy empowers patients with previously undiagnosed and untreated hip conditions, enabling them to obtain relief and reclaim their lives. By employing a compact camera and specialized tools, surgeons expertly navigate the hip joint, performing procedures from excising loose bodies and mending labral tears to addressing femoroacetabular impingement and tackling other intricate issues. This innovative approach has dramatically elevated patients’ quality of life, allowing them to embrace targeted treatments and resume daily activities without resorting to lifestyle alterations.
Keywords
Introduction
With an enhanced comprehension of hip pathologies and remarkable strides in imaging and surgical technologies, hip arthroscopy and hip endoscopy have emerged as prevalent orthopedic procedures in the U.S., accounting for an estimated 1.77 million surgeries annually. 1 As this trend is poised to rise, it is crucial to pause and examine current insights from recent literature.
Tracing back to Michael Burman’s pioneering description of hip arthroscopy in 1931, 2 numerous manuscripts have been published regarding hip pathologies and their treatment with this innovative technique. 3 Von Glinski et al., 4 in their article titled “The impact of the 30 most cited articles on hip arthroscopy,” highlighted the evolution of hip arthroscopy, emphasizing three cardinal topics: Labral Repair, femoroacetabular impingement (FAI), and complications and revisions. Complications undoubtedly hold a significant position in this discussion. In the systematic review by Weber et al., 5 minor complications were observed in 7.9% of cases, whereas major complications were noted in 0.45% of the instances. Predominantly, extra-articular fluid extravasation emerged as the chief major complication. However, instances of avascular necrosis and femoral neck fractures have also been reported.5,6
Anatomy
Delving into the fundamental bone anatomy of the hip joint, the acetabulum—part of the iliac bone—forms the socket, whereas the femoral head acts as the ball (Figure 1(a)). This unique configuration can lead to impingement resulting from excessive acetabular coverage or femoral head–neck junction abutment, known as FAI, a term introduced by Ganz et al. 7

Schematic representation of hip. (a) Normal hip, (b) hip with Pincer-type impingement (the red zone represents the acetabular over coverage), and (c) hip with cam-type impingement (the red zone represents the femoral head-neck junction abutment).
Femoroacetabular impingement
The significance of FAI in contemporary orthopedic literature lies in its recognition as a cause of osteoarthritis in young adults. 8 Defining impingement subtypes precisely is not just academic; it also has direct therapeutic implications. Fortier et al. 9 quantified the incidence of FAI at 54% and noted that over 70% of these cases displayed mixed morphologies. When symptomatic, FAI can be effectively treated with hip arthroscopy, but correctly classifying the impingement type is crucial. Pincer-type FAI 10 (Figure 1(b)) occurs when the acetabulum overcovers the femoral head, predisposing labral crushing against the femoral neck and potential degeneration. Treatment involves an acetabular rim osteoplasty using a burr, typically performed from the capsular side, while preserving labrum circulation and the chondrolabral junction essential for healing. 11 Following acetabuloplasty, literature delineates various techniques for anatomically repositioning the labrum. The chosen technique frequently depends on the nature of the repair, including the perilabral or “loop” suture and translabral or “labral base” repair. 12
Cam-type FAI (Figure 1(c)) results from a mismatch between the acetabulum and proximal femur due to an abnormal flattening or dysplastic bump, causing chondrolabral junction damage as it rubs against adjacent cartilage.13,14 Surgical treatment encompasses osteoplasty to reshape the femoral-neck junction and dynamic and radiographic evaluation to restore femoral head sphericity and avoid over-resection. 15 Preoperative measurement of bone removal is necessary, with some computed navigation systems aiding surgeons in accurately estimating bone resection during surgery. 16
Recently, arthroscopists have shown increased interest in the labrum—a thick, fibrous rim encircling the acetabulum—enhancing hip stability and safeguarding cartilage (Figure 2). 17
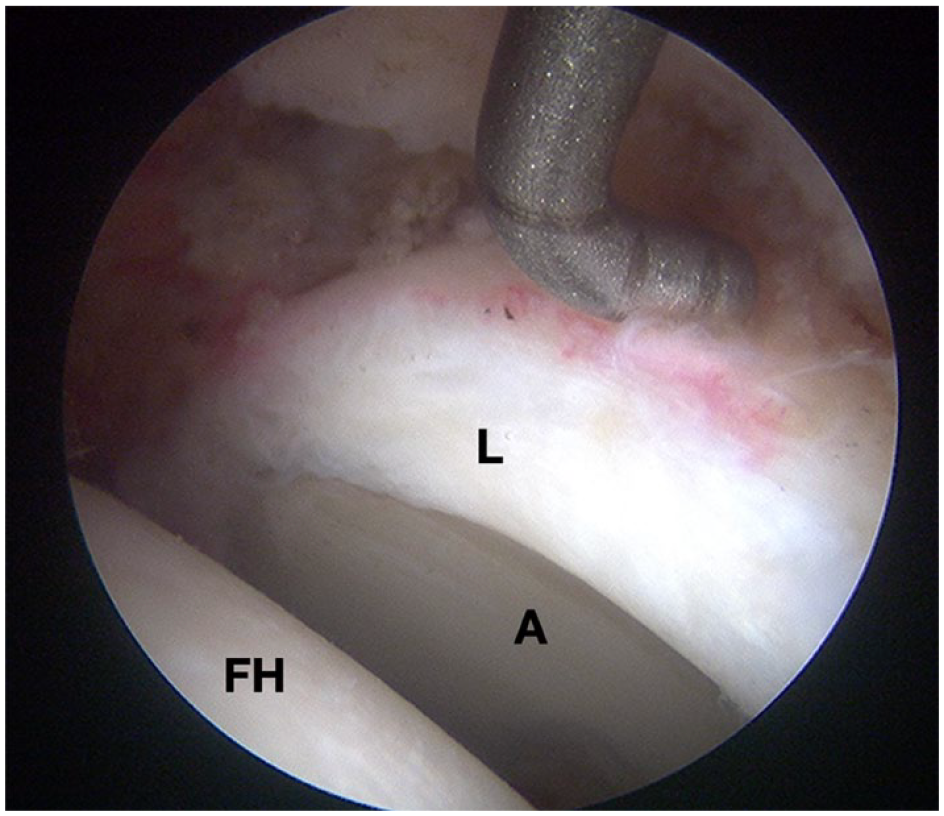
Intraoperative photographs (hip arthroscopy) of the left hip.
Labrum repair versus debridement versus reconstruction
The diagnosis of acetabular labral tears is complex and multifaceted. Advances in diagnostic procedures have enhanced accuracy in recent years; however, clinical presentation is often masked by its resemblance to intra-articular and extra-articular diseases. 18 Anterior hip pain in young patients may resemble symptoms associated with a wide variety of conditions, including FAI, iliopsoas impingement, internal snapping hip, and neuropathies intersecting the anterior hip joint, as described by Battaglia et al. 19 This symptomatic overlap, augmented by the limited specificity of clinical examinations, complicates the differentiation of anterior hip pain generators. Despite diagnostic advancements in recent years there is still a need to recognize the inherent complexities associated with accurately identifying labral tears among similar presenting pathologies.20,21
Although labral tear incidence is consistent across sexes and ages, symptomatic labral tears tend to be more common among women, particularly those aged 15–40. 22
Hip arthroscopy can treat symptomatic labral tears after conservative measures prove unsuccessful. Over time, labral tear repair techniques have evolved 23 (Figure 3), with labral debridement initially being the most common procedure for labral injuries during the early years of hip arthroscopy.

(a) Intraoperative photographs (hip arthroscopy) of the left hip. (b) Intraoperative photographs (hip arthroscopy) of the right hip. The chondral–labral junction shows a tear and fraying (arrow) at the junction and synovitis at the capsule and labrum as viewed from the lateral portal.
Labral debridement involves resecting the damaged labrum segment using a radiofrequency device, a shaver, or arthroscopic forceps while preserving as much healthy labral tissue as possible 24 (Figure 4(a)). Labral repair aims to restore the inherent stability, lubrication, and pressure distribution capacities of the labrum within the joint, which are compromised by the tear. The labrum is meticulously visualized during hip arthroscopy and anatomically re-anchored using sutures and anchors. Concurrently, the acetabular rim undergoes trimming to rectify pincer impingement or to create a vascularized bed, optimizing the conditions for labral healing. 24 McGovern et al. 25 conducted a multicenter cohort analysis on anchor allocation and quantity during labral repair, finding that 41.5% of hips had a labral tear size of 3 h, and the most common repair location was the 12- to 3 o’clock position using the clockface diagram. Anchor usage varied depending on tear size, with the authors concluding that at least two anchors were needed for tears spanning >2 h in the clockface diagram to restabilize the labrum and restore the hip’s suction seal 25 (Figure 4(b)).

(a–c) Schematic representation of labral tears treatments in hip arthroscopy. (a) Labral debridement using a shaver, (b) labral tear repaired using anchors, and (c) labral reconstruction secondary a complete damage of the labrum.
Recently developed partial or complete labral reconstruction or augmentation techniques are treatment options for irreparable or non-functioning labrum tears. Ayeni et al. 26 published the first reports of labrum reconstruction in 2014. According to a recent publication by Maldonado et al., 27 labral reconstruction is highly valued among high-volume hip arthroscopists, with reconstruction more commonly recommended in revision rather than primary surgeries, allografts being the preferred graft choice, segmental reconstruction favored over circumferential reconstruction and labral tissue excision preceding reconstruction rather than augmentation (Figure 4(c)).
Chondral damage
Cartilage, a vital hip structure, is a highly specialized connective tissue facilitating load transmission with a low friction coefficient by providing a smooth, lubricated articulation surface. 28 Early diagnosis and treatment of articular cartilage injuries are essential, as they are recognized causes of osteoarthritis. A previous study by O’Connor et al. 29 reported a 70% sensitivity in detecting cartilage defects later identified during arthroscopic procedures.
Chondroplasty
Chondroplasty or debridement is the treatment of choice for type I or II damage according to the Outerbridge classification 30 (Figure 5(a)). This procedure aims to remove partial thickness or loose lesions or flaps, eliminating potential mechanical blocks in the hip and preventing the formation of loose bodies. Typically performed with a soft tissue shaver, a radiofrequency probe may also be used. 31 Chondroplasty or debridement studies are scarce as they are often conducted alongside other intra-articular procedures. However, Bedard et al. 32 found that chondral damage increased the risk of conversion to THA at 2 years.

(a and b) Intraoperative photographs (hip arthroscopy) of the left hip. (a) The femoral head shows cartilage damage (arrow), fraying of the labrum, and synovitis at the capsule as viewed from the lateral portal and (b) the chondral–labral junction shows an area of exposed subchondral bone (arrow).
Microfractures
Since the early 1980s, microfractures have been employed for cartilage repair. This technique involves creating “microfractures” in the subchondral bone perpendicular to the articular surface, granting access to marrow-based progenitor cells and growth factors. A marrow clot forms at the base of a prepared chondral lesion, enabling pluripotent cells to proliferate and differentiate into cells with chondrocyte-like morphology, generating cartilaginous repair tissue that fills the chondral defect. 33 Microfracture is an effective arthroscopic treatment for full-thickness degenerative and chondral lesions, although the resulting fibrocartilage has biomechanical properties inferior to hyaline cartilage. 33 Despite this, microfracture is relatively low-cost and easy to perform.
Autologous chondrocyte implantation
As orthobiologics advance, management options for partial thickness disruptions to full-thickness defects (type III or IV in Outerbridge classification) (Figure 5(b)) have expanded. Autologous chondrocyte implantation (ACI) is a technique developed to address large chondral lesions that microfracture cannot treat. ACI involves harvesting the chondral defect, growing it in vitro, and removing the damaged cartilage, like microfracture. 34 The harvested chondrocytes, now mixed with a bioabsorbable matrix, are then implanted back into the cleared defect. 35 Although positive outcome reports exist, more research is needed to validate its routine use for hip cartilage defects.
Matrix-induced autologous chondrocytes implantation
Matrix-induced autologous chondrocyte implantation (MACI) represents an evolution of ACI, where cultured chondrocytes are implanted onto an absorbable scaffold. Once transplanted, the cartilage defect is filled. Thier et al. 36 published a surgical technique using two different MACI products (NOVOCART Inject/Chondrosphere), concluding that both products effectively treated full-thickness cartilage defects of the hip in terms of pain relief and hip function improvement. 37
Autologous matrix-induced chondrogenesis
Autologous matrix-induced chondrogenesis (AMIC) is a single-step procedure that treats cartilage defects by covering the cartilage defect with a collagen matrix patch after a standard microfracture procedure. 36 The collagen patch stabilizes the fibrin clot and provides an environment conducive to cartilage formation. 38 Fontana et al. 39 reported significant improvements after a 5-year follow-up of 2–4 cm2 chondral lesions treated arthroscopically with the AMIC technique. Thorey et al. 40 assessed the effectiveness of AMIC on mid-size acetabular chondral lesions in young patients. They found that after the procedure, there was a significant improvement in HOOS, mHHS, and VAS pain scores over a 2-year period, highlighting AMIC’s potential in aiding patients’ return to recreational sports. However, a systematic review by Gao et al. 41 highlighted the relative scarcity of rigorous, randomized controlled studies that compared AMIC with more established interventions like microfracture and ACI.
Osteochondral autograft transplantation
The osteochondral autograft transplantation (OAT) or mosaicplasty involves harvesting osteochondral plugs from the non-weight-bearing surface of a joint to insert in a chondral defect prepared by drilling the defect’s size. 42 It depends on the location of the defect on the femoral head and whether an open or arthroscopic approach is used. In their study, Nam et al. utilized osteochondral plugs from the knee and the lower part of the femur head to address chondral defects stemming from post-traumatic hip dislocation. Hart et al. presented a unique case in which a femoral head defect caused by a screw penetration was treated using OAT harvested from the neighboring lateral femoral condyle. Highlighting the potential of OAT in managing femoral head avascular necrosis (AVN), Gagala et al. emphasized its effectiveness, especially during the precollapse stage. Anthonissen et al. provided evidence of OAT’s capability in addressing significant trauma-induced femoral head defects. Güngör et al.’s research involved two patients with FAI who received osteochondral OAT and femoral head osteochondroplasty using osteochondral cylinders from the ipsilateral knees. Meanwhile, Kaymaz et al. 43 documented a marked improvement in a patient’s Harris Hip Score over 18 months following OAT, noting that the autograft was harvested from the ipsilateral femoral head.
Platelet-rich plasma
Although results have varied, platelet-rich plasma (PRP) has been utilized for various intra-articular hip pathologies to enhance healing and improve hip arthroscopy outcomes. PRP consistently exhibits anti-inflammatory and procoagulant properties. 44 Mullins et al. 45 conducted a systematic review to assess the effectiveness of biological agents in treating cartilage defects linked to FAI. Results showed that PRP lacks strong evidence supporting its post-operative benefits for hip FAI, both HA and cell-based methods might offer positive results, though the evidence is limited.
Peripheral compartment
Hip preservation surgery has rapidly evolved, with a growing emphasis on extra-articular arthroscopic solutions for peri-articular hip issues.46,47 However, conservative treatments are often effective, and surgery is recommended only when they fail. We categorize them into greater trochanteric pain syndrome (GTPS), snapping hip syndrome (SHS), sub-spine impingement (SSI), and deep gluteal syndrome (DGS).
Greater trochanteric pain syndrome
GTPS involves chronic pain over the lateral aspect of the greater trochanter (GT) radiating down the lateral thigh, typically affecting females over 40. Causes include external snapping hips, gluteus medius/minimus tendinopathies, and trochanteric bursitis. Common symptoms include pain during lying on the affected side, prolonged standing or walking, and transitioning from sitting to standing. Conservative treatments, such as cortisone injections, iliotibial band (ITB) stretching, and gluteal strengthening, are typically the first approach. If unsuccessful, surgical measures like endoscopic ITB release, bursectomy, and gluteal tendon repair are recommended, 48 with literature supporting the effectiveness of endoscopic ITB release. 48
Snapping hip syndrome
SHS, often asymptomatic but occasionally painful, has two types: internal, caused by the iliopsoas tendon snapping over the iliopectineal eminence, and external, caused by the ITB sliding over the GT. 49 Ballet dancers, gymnasts, and soccer players have increased susceptibility due to repetitive movements. 50 Conservative treatments include pain relief, tendon lengthening through stretching exercises, NSAIDs, and steroid injections. When conservative measures fail, surgical treatments like endoscopic lesser trochanter release and central release may be required. Coulomb et al. 51 found a higher recurrence rate in endoscopic central release than in peripheral release. For external SHS, endoscopic ITB release or endoscopic gluteus maximus tendon release is preferred, but more comparative studies with extended follow-ups are needed.
Subspinal impingement
Subspinal impingement (SSI) results from anterior inferior iliac spine (AIIS) hypertrophy due to AIIS avulsion or rectus femoris tendon injury. Patients usually report an initial trauma followed by acute symptoms, progressive activity-related pain, and restricted motion. 52 Nwachukwu et al. 53 observed improvement outcomes after decompression in 33 SSI patients undergoing arthroscopic treatment. Xu et al. 54 compared mini-open and arthroscopic decompression, finding similar outcomes. Recognizing the diagnosis, understanding the coexistence with FAI, and knowing treatment options is crucial.
Deep gluteal syndrome
DGS, previously known as “piriformis syndrome,” describes nondiscogenic sciatic nerve entrapment in the deep gluteal space. Kay et al. 55 identified iatrogenic causes (30%), piriformis syndrome (26%), and trauma (15%) as the most frequent DGS causes. Patients often experience persistent or intermittent deep pain in the posterior hip, buttock, and thigh. 56 Due to the numerous structures involved, diagnosis can be challenging, necessitating consideration of all available diagnostic tools (e.g., provocative tests, CT, MRI, electrodiagnostic studies, diagnostic injections).
Challenges and considerations
In this comprehensive review, we have meticulously analyzed multiple studies on hip arthroscopy; we present an encompassing perspective of its progress over several decades. This review highlights advancements in surgical techniques and their consequential enhancements in patient outcomes.
The inherent variability in the designs and methodologies of the studies incorporated may impact the range of our synthesis. The heterogeneity characteristic of hip pathologies poses a significant challenge, potentially underrepresenting the aggregate data. The efficacy of arthroscopic interventions must be interpreted with caution.
We must also admit that the relative recency of specific arthroscopic interventions has yielded sufficient long-term outcome data, which is imperative for a comprehensive assessment of these techniques’ durability and effectiveness. This is particularly important in biological therapies—such as PRP and autologous matrix-induced chondrogenesis—where, despite the promising preliminary evidence, the empirical foundation remains in the initial stages of development.
Conclusion
Orthopedic professionals must take an action-oriented approach to address hip pathologies, from labrum tears and chondral damage to SHS and DGS. Begin with conservative treatments, including physical therapy, pain relief, and medications, and when necessary, consider advanced surgical techniques such as hip arthroscopy, labral repair or reconstruction, and chondroplasty. Utilize microfractures, ACI, MACI, and AMIC for cartilage repair. Leverage endoscopic and arthroscopic procedures for peripheral compartment issues, paying close attention to patient-specific factors and concurrent pathologies. Staying up to date with the latest research and techniques ensures optimal patient outcomes and long-term relief in managing a wide range of hip pathologies.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Informed consent
Not applicable.
