Abstract
Backgrounds:
Papillary thyroid cancer is the most common pathological type of thyroid cancer. miR-96-5p, a member of the miR-183 family, constitute a polycistronic miRNA cluster. In breast cancer, miR-96-5p promotes cell invasion, migration, and proliferation in vitro by inhibiting PTPN9. Moreover, miR-96-5p was reported to function as an oncogene in many cancers. However, whether miR-96-5p is involved in the development of papillary thyroid cancers and its potential mechanism is still unknown. The present study aims to explore the relationship between miR-96-5p and GPC3 expression in the development of papillary thyroid cancers.
Methods:
Transcriptomic sequencing was carried out using six pairs of papillary thyroid cancer and adjacent normal tissues. Quantitative real-time polymerase chain reaction (PCR) experiments were performed to examine the expression of genes.
Results:
In total, there were 1588 up-regulated and 1803 down-regulated differentially expressed genes between papillary thyroid cancer and normal tissues. Gene ontology and Kyoto encyclopedia of genes and genomes analysis revealed that extracellular matrix structure and proteoglycans were mainly involved in papillary thyroid cancer. Among the cluster of proteoglycans, GPC3 was significantly down-regulated in papillary thyroid cancer and is a target of miR-96.
Conclusion:
miR-96-5p participates in the development of papillary thyroid cancer by regulating the expression of GPC3. Thus, targeting miR-96-5p may be a potential therapeutic approach for preventing and treating papillary thyroid cancer.
Introduction
Papillary thyroid cancer (PTC) is the most common pathological type of thyroid cancer, accounting for about 84% of thyroid cancers. 1 It has been reported that 6800 people died of thyroid cancer in China in 2015, with 90,000 new cases. 2 The incidence of thyroid cancer in women has increased more significantly, with an average annual rate of 20.1%, ranking fifth among the most common cancers. 2 Thyroid cancer is also the most common cancer in Chinese women before age 30. 3 Therefore, developing novel approaches to prevent and treat thyroid cancer in China is urgent. The prognosis of PTC is good, but it is prone to early lymph node metastasis and postoperative recurrence.4,5 At present, there are many kinds of tumor treatment methods, and the emergence of next-generation sequencing has greatly changed the mode of tumor diagnosis and treatment and has since entered a new era of personalized precision medicine.6,7 Nowadays, the application of next-generation sequencing technology to find new molecular targets for the diagnosis and treatment of thyroid cancer is still a hot research topic, which can provide valuable results for the prevention, diagnosis, and treatment of thyroid cancer.
MicroRNA (miRNA) is a class of non-coding small RNAs with a length of about 21 nucleotides, which cause the degradation of target mRNA or inhibit its translation through specific base pairing with the target mRNA.8,9 miRNAs play important roles in a variety of biological processes, such as proliferation, metastasis, and apoptosis.10–12 In PTC, multiple miRNAs have been identified as tumor suppressors, including miR-146b, miR-222, miR-135b, and miR-221. 13 miRNAs are not only dysregulated in PTCs but also have important implications for assessing the tumor invasiveness of PTCs. It has been reported that the up-regulation of miR-146b, miR-222, and miR-221 in PTC was closely related to tumor aggressiveness, while the down-regulation of miR-191 and miR-486 in PTC was associated with tumor prognosis and progression. 14 Moreover, the up-regulation of miR-221, miR-222, and miR-146b-5p is closely related to the infiltration and metastasis of thyroid cancer cells, and miR-613 and miR-16 may play a role in the tumor invasion of PTC with a certain inhibitory effect. 15 These studies inferred that miRNAs are closely related to the invasive and metastatic behavior of PTC. miR-96-5p, a member of the miR-183 family, constitutes a polycistronic miRNA cluster. 16 In breast cancer, miR-96-5p promotes cell invasion, migration, and proliferation in vitro by inhibiting PTPN9. 17 Moreover, miR-96-5p was reported to function as an oncogene in many cancers including non-small cell lung cancer. 18
Many proteoglycans in the tumor microenvironment have been reported to be key macromolecules that contribute to the biology of various types of cancer including proliferation, adhesion, angiogenesis, and metastasis, affecting tumor progress. Glypican-3 (GPC3) is a proteoglycan containing heparan sulfate on the cell surface. It can bind to proteins and inhibit dipeptidase activity. It may play a role in the control of cell division and growth regulation. Its main functions are negative regulation of epithelial cell proliferation and negative regulation of the classical Wnt signaling pathway.19–21 Deletion or mutation of GPC3 results in a lack of growth inhibition and apoptosis in some cells. Studies have reported that the down-regulation of GPC3 is associated with various tumors such as mesothelioma, breast cancer, lung cancer, and ovarian cancer.22–24 It has been demonstrated that GPC3 is a potential target of miR-96-5p in hepatocellular carcinoma cells and non-small cell lung cancer.18,25 However, whether miR-96-5p is involved in the development of PTCs and its potential mechanism is still unknown. Therefore, the present study aims to explore the potential relationship between miR-96-5p and GPC3 in the development of PTCs.
Methods and materials
Patient and tissue collection
A total of six patients diagnosed with papillary thyroid carcinoma were recruited. The inclusion criteria for papillary thyroid carcinoma patients in this study are as follows: PTC patients were diagnosed by fine-needle aspiration, and no radiotherapy or chemotherapy before surgery. The exclusion criteria are patients diagnosed with papillary thyroid carcinoma combined with other tumors; with a history of radiotherapy, chemotherapy, or radioactive exposure. Papillary thyroid carcinoma tissues and corresponding paracancerous (healthy tissue from papillary thyroid) tissues were collected during surgery from each patient. Once collected, the papillary thyroid carcinoma tissues and corresponding paracancerous tissues were quickly put into liquid nitrogen for freezing. This study was approved by the Clinical Research Ethics Committee of Shenzhen Hospital (Guangming), Chinese Academy of Sciences (No. LL-KT-2020190), and followed the Declaration of Helsinki. All participants have signed informed consent. Tumor tissue and paired normal tissue adjacent to the tumor were then dissected for RNA sequencing as previously described. 26
RNA sequencing analysis
In the present study, we adopted our previously published data from PTC patients, which has been submitted to the gene expression omnibus (GEO) database, with the accession number GSE129957, 26 and re-examined the transcriptome data to further explore the possible involvement of signaling pathways in PTC. HISAT/Bowtie2 tool was adapted to map the clean reads to the reference sequence. 27 RSEM software package is used for the quantification of the expression level of each gene. 28 Differentially expressed genes (DEGs) were identified between groups using the NOIseq 29 and PossionDis 30 algorithms. Gene functional annotation clustering was performed using Homo sapiens as background with NIH DAVID, following a modified Fisher’s exact test with Benjamini-Hochberg multiple corrections.31,32 In addition, enriched Kyoto encyclopedia of genes and genomes (KEGG) pathways were identified using KOBAS 3.0 with a hypergeometric test and Benjamini-Hochberg FDR correction.33,34
MRNA extraction and RT-qPCR
To examine the expression level of miR-96, miRNAs were isolated from tissues and cells using the minute microRNA isolation Kit (Tiangen Biotech, Beijing, China) following the manufacturer's instructions. Briefly, 0.5 µg of RNA was used for cDNA synthesization using One Step PrimeScript™ RT-PCR Kit (Takara, Dalian, China) according to the manufacturer’s instructions. Quantitative real-time PCR was then carried out using the ABI7500 real-time PCR instrument (Thermofisher, Minneapolis, USA). The primer sequences used in this study are shown in Table 1. Threshold cycle (CT) values were analyzed at the end of PCR. The fold change in miR-96 levels was then calculated.
Sequence of the primers for quantitative real-time RT-PCR.

Statistical analysis
Data were presented as Mean ± SEM. The differences were evaluated by Student’s
Results
DEG screening in PTC
The transcriptome data was generated from normal and PTC tissues. As shown in Table 2, each sample’s sequencing data generated 67 Mb clean reads (with a mapping rate of 94%–97%) after filtering low quality. The number of identified expressed genes was counted and their proportion and distribution to the total gene number in the database of each sample were calculated (Figure 1(a)). Besides, the correlation of gene expression level among samples was adopted to test whether the data are reliable. The correlation values between every two samples based on normalized expression result were drawn as a correlation heatmap (Figure 1(b)). The volcano plot showed the up- and down-regulated DEGs between normal and PTC tissues after DEG screening (Figure 1(c)). In total, there were 1588 up-regulated, and 1803 down-regulated DEG between PTC and normal tissues after DEG screening as shown in Figure 1(d).
The summary of the raw RNA sequencing data set.
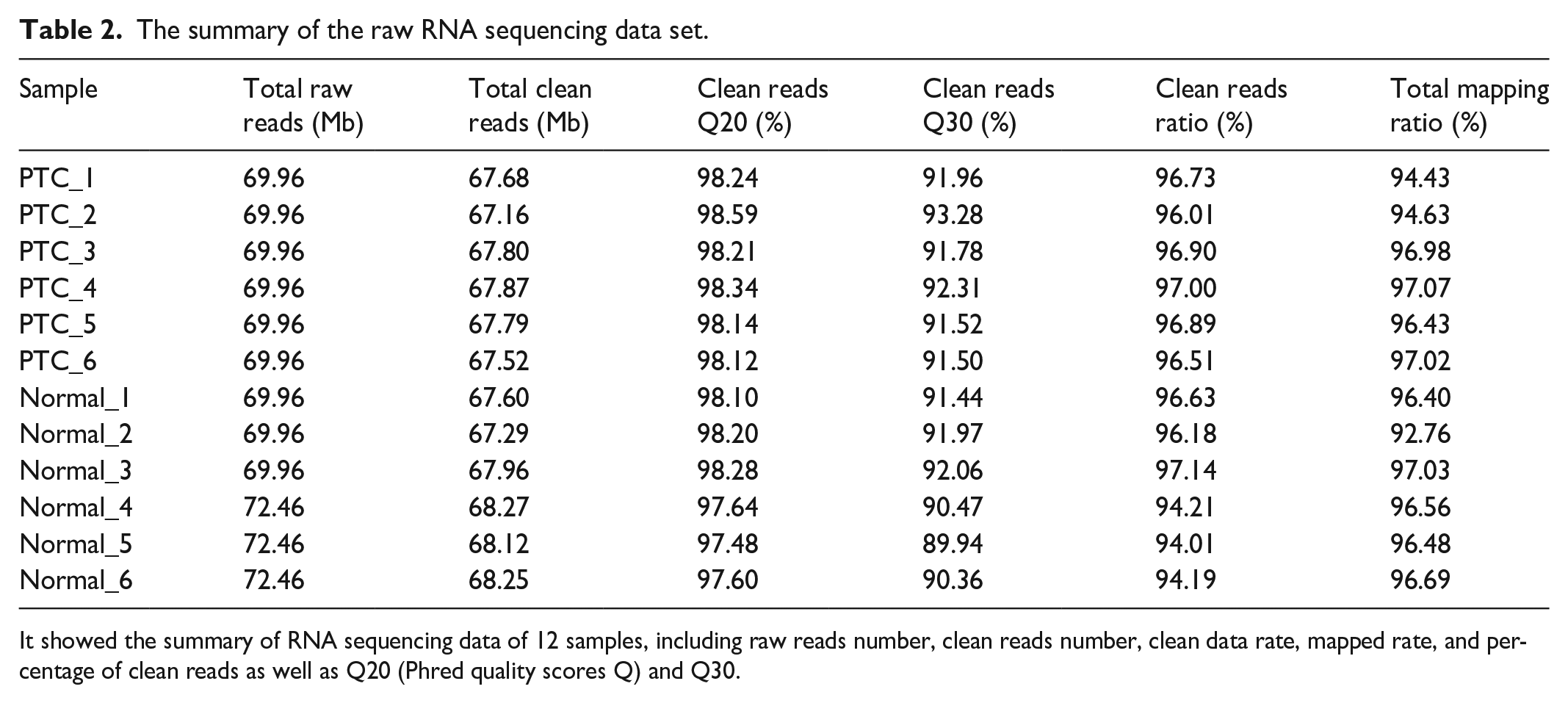
It showed the summary of RNA sequencing data of 12 samples, including raw reads number, clean reads number, clean data rate, mapped rate, and percentage of clean reads as well as Q20 (Phred quality scores Q) and Q30.

Data quality assessment and differentially expressed gene screening in PTC. (a) Gene expression distribution of normal and PTC samples. (b) Heatmap of the correlation between normal and PTC samples with Pearson method. (c) Volcano plot for the samples with mRNA expression differences. Log2 (fold change) is plotted as the abscissa and log10 (Corrected
Gene ontology and KEGG pathway analysis in PTC
To further explore the associated functions of DEGs in PTC, we carried out gene ontology (GO) analysis to perform the enrichment and classification analysis (Figure 2). GO analysis identified enriched biological processes associated with “extracellular matrix organization,” “extracellular structure organization,” and “cell junction assembly” (Figure 2(a)). Moreover, enriched molecular functions were defined as associated with “extracellular matrix structural constituent,” “passive transmembrane transporter activity,” “signaling receptor activator activity,” and “glycosaminoglycan binding” (Figure 2(b)). In addition, identified enriched cellular component terms associated with “collagen-containing extracellular matrix,” and “synaptic membrane” (Figure 2(c)), indicating that extracellular matrix structure and components were mainly involved in PTC. KEGG pathway enrichment was further performed using the DEGs in PTC with the KOBAS method as previous literature reported.33,34 The DEGs were significantly enriched in the classifications of “extracellular matrix-receptor interaction,” and “proteoglycans in cancer” in PTC (Figure 2(d)). These results further implied that extracellular matrix components and proteoglycans were the major KEGG pathways involved in PTC. We subsequently plotted the 84 DEGs in PTC involved in the KEGG pathway of proteoglycans in cancer (Figure 3(a)), and the results suggested that GPC3, a proteoglycan containing heparan sulfate on the cell surface, is down-regulated. Then, by application of the mirDIP website, the potential miRNA, which could interact with GPC3 in PTC, was predicted (Figure 3(b)). The results demonstrated that miR-96-5p might be a potential miRNA in PTC, interacting with GPC3.

Functional analysis of DEGs by GO and KEGG classifications in PTC. (a–c) The comparison of GO enrichment between normal and PTC tissues. Top 20 significantly enriched GO terms were shown in biological process (a), molecular function (b), and cellular component (c). The enriched gene number as the abscissa and GO terms is plotted as the ordinate. (d) The comparison of pathway enrichment in PTC. The top 20 significantly enriched KEGG pathways were shown. The rich factor as the abscissa and KEGG terms is plotted as the ordinate.
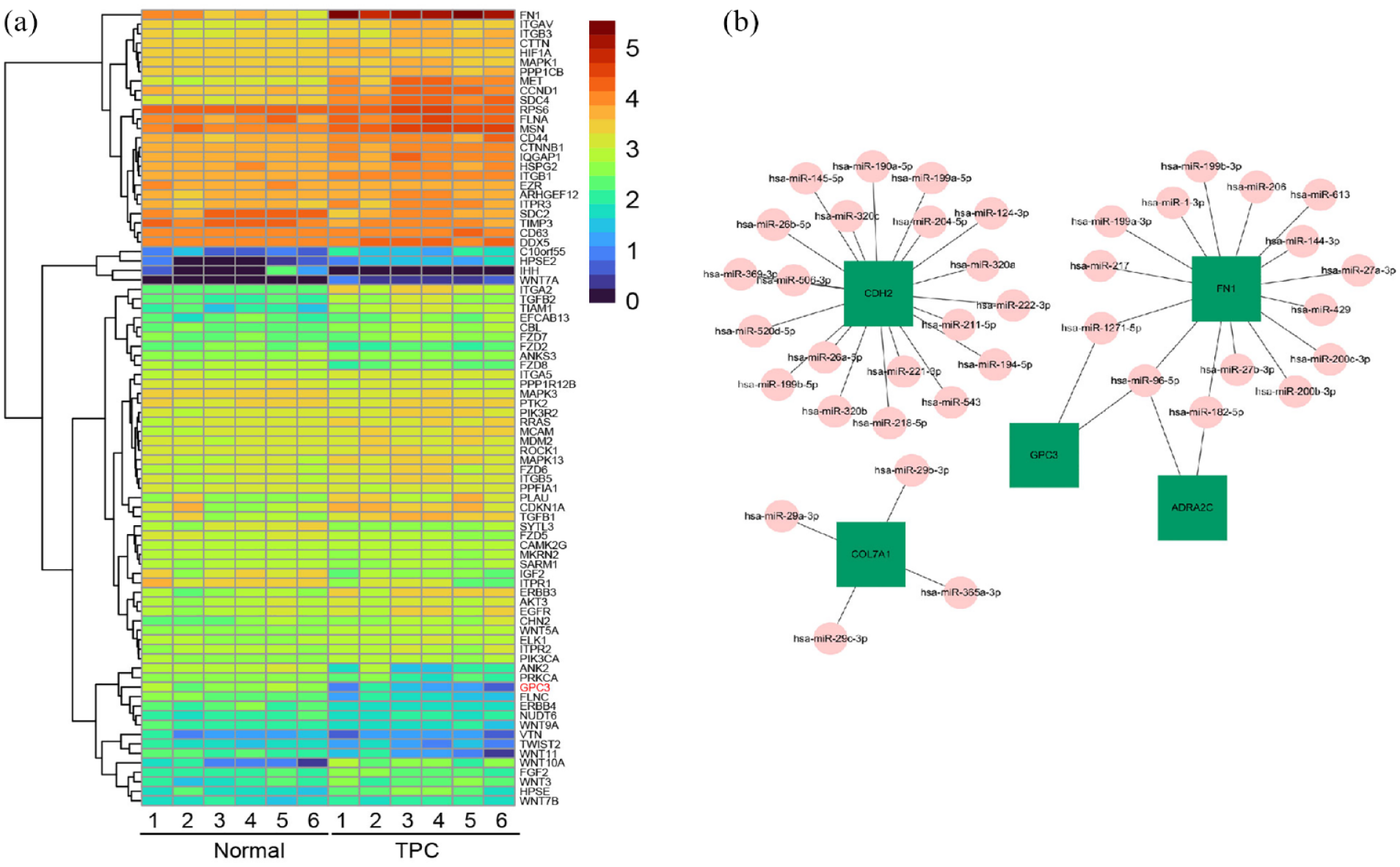
GPC3 is down-regulated and the predicted potential miRNA in PTC. (a) Heatmap of DEGs involved in KEGG pathway of proteoglycans in cancer. (b) Prediction of candidate gene interaction with potential miRNA using mirDIP website.
GPC3 was significantly down-regulated in PTC and is a target of miR-96
GPC3 is well-known to be negatively involved in the control of cell division and growth regulation. Our results showed that the normalized reads count of GPC3 is identified to be significantly decreased in PTC (483.67 ± 81.09 vs 37.67 ± 19.77), compared with normal tissues (Figure 4(a)). Quantitative RT-PCR was recruited to further confirm the down-regulation of GPC3. The results demonstrated that GPC3 is significantly down-regulated in PTC patients (1.57 ± 0.26 vs 0.65 ± 0.07), compared with normal tissues (Figure 4(b)). We next used ENCORI website analysis to assess the miR-96-5p binding site of the

GPC3 is significantly down-regulated in PTC and is a target of miR-96. (a) The normalized reads count of GPC3 in PTCs and normal tissues. (b) Quantitative RT-PCR results of the mRNA expression of
Discussion
PTC is one of the most common malignant tumors in the endocrine system. 36 With the development and progress of molecular biological diagnosis technology in recent years, the incidence of PTC is increasing year by year, 37 reaching as high as 28/100,000. 38 In recent years, the incidence of PTC in women has increased more significantly, ranking fifth among the most common cancers. 2 It is the most common cancer in Chinese women before the age of 30 as well. 3 In the present study, we explored the candidate DEGs and miRNAs related to the occurrence, development, and prognosis of PTC by conducting transcriptome sequencing of PTCs and adjacent normal tissues, analyzed the candidate DEGs and miRNAs, and verified them.
miR-96-5p, a member of the miR-183 family, constitutes a polycistronic miRNA cluster. 16 In breast cancer, miR-96-5p promotes cell invasion, migration, and proliferation in vitro by inhibiting PTPN9. 17 miR-96-5p has also been reported to be involved in the promotion of colorectal cancer cell proliferation and predicted poor prognosis. 39 Moreover, miR-96-5p was reported to function as an oncogene in many cancers including non-small cell lung cancer. 18 In this study, our data suggested that miR-96-5p was significantly up-regulated in PTCs. It is known that most miRNAs bind to target gene mRNAs in an imprecise manner, 9 and one miRNA may be involved in the regulation of more than 200 mRNAs. Therefore, we checked whether there are other unknown targets of miR-96-5p in papillary thyroid carcinoma.
Moreover, our results demonstrated that GPC3 was significantly down-regulated in PTCs, while miR-96-5p was significantly up-regulated in PTCs. It has already been reported that miR-96-5p can promote the invasion and metastasis of PTC by targeting and regulating the expression of CCDC67, SDHB, and FOXO1.40–42 According to our study, the expression of miR-96-5p is negatively correlated with the expression of GPC3 in PTCs. This result is consistent with the data from the ENCORI website analysis. Coincidently, GPC3 has also been reported to be a potential target of miR-96-5p in hepatocellular carcinoma cells and non-small cell lung cancer,18,25 In addition, miR-96-5p promotes the invasion and metastasis of PTC by regulating the target gene
Although we have demonstrated the negative relationship between the expression of miR-96-5p and GPC3, and the possible involvement in the development and occurrence of PTC, the limitation should be acknowledged in this study as well. Only six patients have been recruited in this study; the sample size is still not enough to make a solid conclusion. More patients will be further recruited for the confirmation of the conclusion in the future. Moreover, we only demonstrated the up-regulation of miR-96-5p and down-regulation of GPC3. The relationship between miR-96-5p and GPC3 is still not clear, the dual-luciferase experiment is needed to verify GPC3 is a target of miR-96-5p. This is also needed for further exploration in the future.
Conclusions
miR-96-5p participates in the occurrence and development of papillary thyroid carcinoma by regulating the expression of GPC3. Therefore. this study explored the relationship between the expression of miR-96-5p and GPC3 and the occurrence, development and prognosis of PTC. This study provided a valuable basis for the targeted therapy of PTC.
Footnotes
Acknowledgements
Not applicable.
Authors’ contributions
The authors’ contributions were as follows: H Hu and Y Lun were responsible for the concept and design of the study; H Hu, Q Chen, S Du, and S Ding were involved with experimental and analytical aspects of the manuscript; H Hu, and Y Lun performed data interpretation, presentation and writing of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the “Special Fund for Economic Development of Shenzhen Guangming District-2020 Sanitation Project,” project number 2020R01033.
Consent for publication
The paper has been read and approved by all authors. All authors approved the submission of this paper for publication. All authors confirmed that neither the manuscript submitted nor any part of it has been published or is being considered for publication elsewhere.
Informed consent
Written informed consent was obtained from all subjects before the study. All participants had signed the informed consent forms.
Trial registration
Not applicable.
