Abstract
Introduction:
The occurrence of extended spectrum beta lactamase-producing uropathogens, especially in pregnant women can result in life-threatening condition and morbidity for both the mother and the newborn due to very limited drug options for treatment of these pathogens. The aim of this study was to determine the bacterial profile, associated factors, and their antimicrobial susceptibility patterns and to identify extended spectrum beta lactamase-producing bacterial uropathogens.
Methods:
A hospital-based cross-sectional study was conducted from July to September 2018 on a total of 177 pregnant women with and without symptoms of urinary tract infection at ALERT Hospital, Addis Ababa, Ethiopia. From these study participants, 72 have symptoms, whereas 105 have no symptoms. All urine samples were inoculated onto cysteine lactose electrolyte deficient medium and MacConkey agar. Colonies were counted to check the presence of significant bacteriuria. Pure isolates of bacterial pathogen were characterized and identified at species level by colony morphology, gram stain, and standard biochemical procedures. All Gram-negative isolates were put into Muller–Hinton agar plates for antibiotic susceptibility test by Kirby-Bauer disc diffusion technique. Extended spectrum beta lactamase was detected using double-disk synergy methods on Muller–Hinton agar. The data were double entered into epidemiological Information system and analyzed using Statistical Package for Social Science version 26.
Results:
The overall proportion of urinary tract infection among pregnant women was 14.7% (n = 26/177). Klebsiella pneumoniae was the predominant bacterial etiologic agent of urinary tract infection 26.9% (n = 7/26). The proportion of extended spectrum beta lactamase among Gram-negative isolates was 50% (n = 6/12). Among extended spectrum beta lactamase-producing isolates (100%), all are resistance to amikacin and gentamicin while intermediate level resistance rate of 66.7% was observed among trimethoprim–sulphamethoxazole. They were susceptible for some limited drugs, and these were Nitrofurantoin (83.3%) and Chloramphenicol (83.3%).
Conclusions:
Majority of extended spectrum beta lactamase-producing isolates exhibited co-resistance to other commonly prescribed antibiotics. This indicates that the option of treatment for these pathogens rapidly decreased from time to time which results serious life-threatening conditions, especially in mother and newborn unless the appropriate measure is taken.
Keywords
Introduction
The second most common infectious disease in the community is urinary tract infection (UTI), which can affect all age group across the life span. 1 As many as 35% of nosocomial infections is due to UTI, thus it is the common nosocomial infection and it is the second most common cause of bacteremia in hospitalized patients. 2 The infection is usually due to bacteria that transfer from the digestive tract to the opening of the urethra and start to multiply to cause infection. 3 Women are more susceptible to UTI than men are, and the main reasons for this are pregnancy, short urethra, absence of prostatic secretion, and easy contamination of the urinary tract with normal flora in the feces. 4 It was reported that up to 15% of women would have one episode of UTI at some time during their life. 5
The factors that contribute for high occurrence of UTI among pregnant women include ureteral dilatation, increased bladder volume, and decreased bladder tone, along with decreased ureteral tone which contributes to ureterovesical reflux and increased urinary stasis. In addition, pregnant women develop glycosuria, which encourage bacterial growth in the urine. 4
UTIs usually occur due to bacteria. Common bacterial pathogens include Gram-negative species such as Escherichia coli, Proteus, Klebsiella, Pseudomonas, Enterobacter, and Serratia spp. and Gram-positive organisms, including Enterococcus spp., group B streptococci, and Staphylococcus aureus. Among these, E. coli is the major causative organism of UTI. 6
There are three major types of UTI in pregnancy. They are asymptomatic bacteriuria, acute cystitis, and acute pyelonephritis. The clinical presentations of these conditions vary. Acute pyelonephritis is infection of the kidney and the pelvic ureter. It is a serious systemic illness affecting 1%–2% of all pregnancies and the most common non-obstetric cause of hospital admission during pregnancy. Without treatment it can cause preterm labor and maternal septicaemia. 6
Acute cystitis relates to infection of the urinary bladder. The major distinguishing feature of acute cystitis from asymptomatic bacteriuria is the presence of dysuria, urgency, and frequency. Most mothers may not be aware that they are having the infection because urgency and frequency are common symptoms in a normal pregnancy. 7
Unlike in non-pregnant women, in pregnant women asymptomatic bacteriuria is the most common complications, occurring in 4%–7% of normal pregnancies. It can result in a variety of negative obstetric outcome and medical conditions such as preterm labor, the development of acute and chronic pyelonephritis, preeclampsia, low birth weight, chronic renal disease, and prenatal mortality. 7
Nowadays, the most worrying issue in the worldwide is, increase in prevalence of extended spectrum beta lactamase (ESBL) producers among different bacterial strains and species, especially in hospitals and healthcare settings. Infections caused by ESBL-producing organisms have resulted in poor outcomes, reduced rates of microbial and clinical response, greater hospital expenses, and longer hospital stays. 8
Many of the isolates producing these enzymes are also resistant to quinolones, trimethoprim, and aminoglycosides, often plasmid has co expression of other resistance mechanisms. Choice in the treatment of ESBL-producing bacterial infections is extremely rare. 9
Increased extended spectrum beta lactamases–Enterobacteriaceae (ESBL-E) associated UTI during pregnancy have a negative impact on pregnant women and neonates, including low birth weight, intrauterine growth restriction, fetal death, premature rupture of the membranes, and neonatal infections. 10
So far many studies have been conducted concerning bacteriological profile and drug susceptibility of UTI among pregnant women in our country.11,12 However, to best of our knowledge, there is no documented study concerning magnitude of ESBL-producing Gram-negative uropathogens among pregnant women, especially in our study area. Laboratory detection of ESBL-producing Gram-negative bacteria will help pregnant to get effective treatment and reduces the subsequent complications of UTIs. Thus, the aim of this study was to determine the magnitude of ESBL-producing Gram-negative uropathogens among pregnant women attending anti natal care in ALERT Hospital.
Patients and methods
Study setting
The study was conducted at ALERT Hospital, which is found in Kolfe-Keraniyo subcity, Addis Ababa, Ethiopia. ALERT Hospital is one of the specialized tertiary referral hospitals in the country. It is located in Addis Ababa at 7 km south west on the way to Jimma. ALERT is a medical facility in Addis Ababa, specializing in Hansen’s disease, also known as “leprosy.” It was originally the All Africa Leprosy Eradication, Rehabilitation, and Training Center (hence the acronym), but the official name is now expanded to include tuberculosis: All Africa Leprosy and Tuberculosis Eradication, Rehabilitation, and Training Center. ALERT’s main mission was to provide training in multiple aspect of leprosy including prevention, treatment, and rehabilitation in an African context. There is currently a 240-bed teaching hospital, which includes dermatology, ophthalmology, surgery, an orthopedic workshop and a rehabilitation program. ALERT also provides anti natal care (ANC) and post-natal care service for women who were referred from different health institutions. It is estimated that the hospital offers ANC service for approximately 4800 pregnant women yearly. Daily in average 20 pregnant women visit this hospital. The 1st ANC timing is before 16 weeks of gestational age, 2nd visit between 24 and 28 weeks of gestation age, 3rd visit between 30 and 32 weeks gestational age, and the 4th visit between 36 and 38 weeks of gestational age.
Study design and period
A hospital-based cross-sectional study was conducted from July to September 2018 at ALERT Hospital, Addis Ababa, Ethiopia.
Inclusions and exclusions criteria
Inclusion criteria
Pregnant women attending antenatal care with or without signs and symptoms of UTI and who was willing to participate in the study.
Exclusion criteria
Pregnant women who have taken antibiotics for the last 10 days.
Sample size determination
The sample size was calculated using the single population proportion formula:
where n = sample size, Z = Z statistic for a level of confidence, p = expected prevalence or proportion, and d = precision.
By considering estimated prevalence, p = 12%, 12 5% precision (d = 0.05), and 95% level of confidence (z = 1.96),
The estimated sample size calculated to be 162. By adding 10% contingency the final estimated sample size was 177.
Sampling technique
A convenient sampling method was applied to recruit study participants. The study participants were selected based on the inclusion criteria, and all available study participants were included until sample size achieved during the study period.
Data and specimen collection
The questioner was translated from English to Amharic and also translated back to English. Training was given for data collectors. Pre-test was done on 5% of participants getting service other than the study hospital and some amendment was made based on the findings of the pretest. After obtaining written informed consent sociodemographic, pregnancy-related, clinical, and health facility-related data were collected from pregnant women.
Ten ml of first morning, clean-catch mid-stream urine (MSU) samples were collected. The participants were instructed how to collect the urine sample by cleansing the gentile with soap and water by using pre-labeled (time, date, identification code, age), leak proof, wide mouth, and sterile plastic universal containers (FL Medical, Italy).
Specimen transportation
The urine specimens then were delivered to microbiology laboratory in ALERT Hospital using an ice box and processed within 2 h.
Cultivation and identification of isolates
Using calibrated wire inoculating loop (0.001 ml), all urine samples were inoculated to cysteine lactose electrolyte deficient medium (Oxoid, England), and MacConkey agar (BD, USA). cultures was incubated in aerobic atmosphere at 37°C for 24 h. Colonies were counted to check the presence of significant bacteriuria (SB). All positive cultures from urine samples were characterized by colony characteristics, Gram stain and biochemical tests. Gram stain was done for all positive cultures with SB in order to categorize the bacteria as Gram-negative or Gram-positive by determining their reactions, cell shape, and arrangement. All Gram-negative bacteria were then identified at species level by the pattern of biochemical profiles using standard procedures. The Gram-positive bacteria were first differentiated using catalase test. All catalase test positive bacteria were inoculated on to Mannitol salt agar. In this study, all identified Gram-positive bacteria were Staphylococci. Coagulase test was performed for all Staphylococci. All coagulase negative isolates were then identified at species level by performing novobiocin susceptibility testing using standard procedures.
Antimicrobial susceptibility testing
Antimicrobial susceptibility testing was done by disk diffusion method. All the isolated Gram-negative bacteria were inoculated onto Muller–Hinton agar plates (100 mm plate) (pH 7.2–7.4) for antibiotic susceptibility test by Kirby-Bauer disc diffusion technique. Disc diffusion tests was performed and interpreted according to the recommendations of the Clinical and Laboratory Standards Institute (CLSI), 13 (Annex IV).
The following antibiotics were used: Ampicillin (10 μg, BD), Amikacin (30 μg, BD), Gentamicin (10 μg, BD), Meropenem (10 μg, Oxoid), Nitrofurantoin (300 μg, BD), Trimethoprim–sulphamethoxazole (1.25/23.75 μg, BD), Cefixime (5 μg, BD), Ceftriaxone (30 μg, BD), Cefotaxime (30 μg, BD), Ceftazidime (30 μg, BD), Chloramphenicol (30 μg, BD), and Amoxicillin-clavulanic acid (20/10 μg, BD).
ESBL detection
Screening method was done by detection of resistance to any of the following third-generation cephalosporin antibiotics: Cefotaxime, ceftazidime, and ceftriaxone by disk diffusion method. Organisms were screened for ESBL by using CLSI 13 recommendation (Annex IV). These CLSI 13 recommendation breakpoints indicative of suspicion for ESBL production were for ceftazidime (30 µg) ⩽ 22 mm, ceftriaxone ⩽ 25 mm, and cefotaxime ⩽ 27 mm.
Isolates which were suspected for ESBL production by ESBL disk screening method were undergone ESBL disk confirmation test by double-disk synergy method (DDST). In this test disks of third-generation, Cephalosporins (Cefotaxime, Ceftazidime, and Ceftriaxone) and Amoxicillin/clavulanate (Augmentin) are kept 20 mm apart, center to center, on inoculated Mueller–Hinton agar. A clear extension of the edge of the inhibition zone of cephalosporin toward Augmentin disk is interpreted as positive for ESBL production (Annex IV).
Quality assurance
Quality of laboratory test was maintained by following standard operating procedures for all laboratory test. Sample containers were appropriately labeled. Expiration date, evidence of freezing, unequal fill, contamination, and bubbles were cheeked for all media. Positive and negative control was performed for media used based on CLSI 13 recommendation. Quality control was done for each batch of the medium by using E. coli ATCC 25922 and/or S. aureus ATCC 25923 as standard strains. K. pneumoniae ATCC 700603 (ESBL positive) and E. coli ATCC 25922 (ESBL negative) control strains were used for ESBL detection. 13
In order to cheek the disc quality, representative discs were tested against the reference strains of S. aureus ATCC 25923, E. coli ATCC 25922 (ESBL negative), K. pneumoniae ATCC 700603 (ESBL positive), and P. aeruginosa 27853. The zone of inhibition was compared with standard value as recommended by CLSI 13 . All test results were recorded appropriately before entry to statistical analysis and prior to this data were double-checked.
Statistical analysis
The collected data were checked for completeness, errors, and missing values before data entry to statistical tool. Sociodemographic, clinical and laboratory data were double-entered into epidemiological Information system. Then, the data were exported to SPSS version 26 Statistical package for social science version 26, IBM, USA) for analysis. Descriptive statistics was done. Association between SB and sociodemographic, pregnancy-related, clinical, and health facility-related variables was checked by using bivariate logistic regression analysis. Then, those variables having p-value < 0.2 in the bivariate analysis was entered into multivariable logistic regression analysis to control the influence of possible confounding variables and to see the strength of association. Statistically significant level was declared at p-value < 0.05. Finally, the results were presented by text, tables and figures.
Results
Sociodemographic characteristics of the study subjects
A total of 177 pregnant women with and without symptoms of UTI, both admitted in ward and those who were in outpatient level were investigated during the study period. The age of the study participants was ranged from 19 to 40 years with majority 40.7% (n = 72/177) in age group of 25–29 years. The mean age was 27.71 ± 4.908 (Table 1). All most all of the study participants were married 98.9% (n = 175/177). Regarding to the residence of the study participants, 93.2% (n = 165/177) were from urban and 6.8% (n = 12/177) were from rural. Majority of the study participants followed Orthodox Christianity 49.7% (n = 88/177) and seconded with Muslim 43.5% (n = 77/177) (Table 1).
Socio-demographic characteristics of pregnant women attending ANC in ALERT Hospital, Addis Ababa, Ethiopia (July–September, 2018).

Proportion of UTI
Of the 177 cultured urine samples, SB was detected in 26 urine specimens. The overall proportion of UTI was 14.7 % ((n = 26/177), 95% CI = 9.6–19.8). Of the total 26 isolates, Gram-negative bacteria were prevalent 57.7% (n = 15/26) than Gram-positive bacteria 42.3% (n = 11/26).
High proportion of UTI (symptomatic and asymptomatic) were observed among pregnant women of age greater than 34 25.0% (n = 5/26). Out of pregnant women who were hospitalized, 30.8% (n = 8/26) had SB, whereas 69.2% (n = 18/26) had no SB, and there was a significant association between being outpatient or inpatient with UTI (AOR = 4.875, 95% CI = 1.628–14.598, p = 0.005) (Table 2). The proportion of bacteriuria in symptomatic and asymptomatic infection were 19.4% (n = 14/72) and 11.4% (n = 12/105), respectively.
Association of independent variables with culture results among pregnant women attending ANC at ALERT Hospital (July–September, 2018).

COR: crude odds ratio; AOR: adjusted odds ratio; CI: confidence interval.
Variables having p-value < 0.2 in the bivariate analysis was entered into multivariable analysis.
Distribution of uropathogens
The percentage of each uropathogen isolated from MSU samples are presented as shown in Table 3. The predominantly isolated bacteria were K .pneumoniae 26.9% (n = 7/26) followed by S. aureus 23.1% (n = 6/26), E. coli 15.4 % (n = 4/26), and S. epidermidis 15.4 % (n = 4/26). Other UTI isolates were C. diversus 7.7% (n = 2/26), K. oxytoca 3.8% (n = 1/26), E. cloacae 3.8% (n = 1/26), and S. saprophyticus 3.8% (n = 1/26).
Percentage and types of bacterial isolates from culture positive pregnant women at ALERT Hospital from (July–September 2018).

The predominant bacteria K. pneumoniae 26.9% (n = 7/26) constitute the highest number (n = 4) among bacteria isolated from symptomatic pregnant women with SB, whereas E. cloacae, S. epidermidis, and K. oxytoca constitute the lowest (n = 1). S. aureus, the major isolates from Gram-positive bacteria, 23.1% (n = 6/26) constitute the highest number (n = 4) among bacteria isolated from asymptomatic pregnant women with SB, whereas E. coli and S. saprophyticus constitute the lowest (n = 1). Distribution of bacterial isolates in relation to symptom is shown in Figure 1.

Frequency of bacteria isolated from urine in relation to symptom at ALERT Hospital from July to September 2018.
K. pneumoniae 26.9% (n = 7/26) accounts the highest number (n = 5) among bacteria isolated from pregnant women in outpatient department with SB. On the other hand, S. aureus, S. epidermidis, and K. pneumoniae (n = 2) were the most common isolates among bacteria isolated from pregnant women in inpatient department with SB (Figure 2).
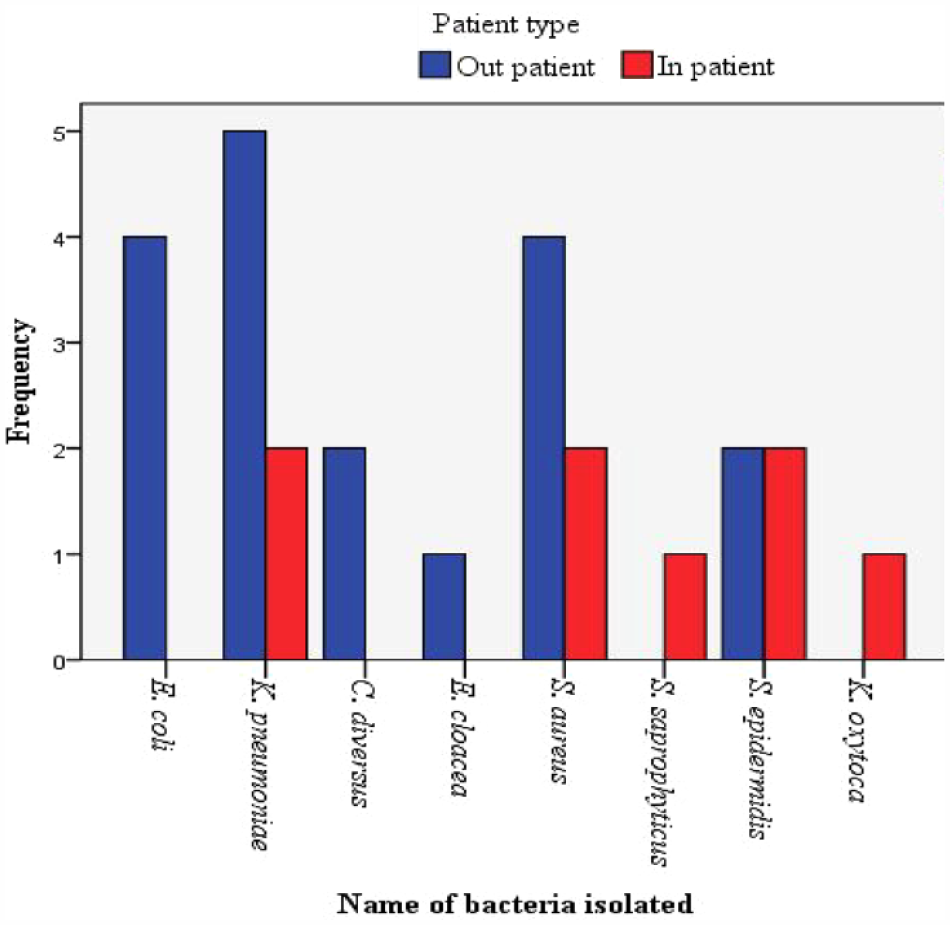
Frequency of bacteria isolated from urine accordingly patient type at ALERT Hospital from July to September 2018.
Antimicrobial susceptibility pattern of Gram-negative bacteria
The result of antimicrobial susceptibility pattern of bacterial isolates is shown on Table 4. In general, all Gram-negative isolates (n = 15) showed resistance rate of 100% to amikacin and gentamicin. On the other hand, all Gram-negative isolates showed sensitivity rate of 100% to meropenem. K. pneumonia which constitutes 46.7% of Gram-negative bacteria showed 100% resistance to ampicillin, amikacin, and gentamicin, but 100% susceptible to meropenem, nitrofurantoin, chloramphenicol, and cefotaxime.
Antimicrobial susceptible pattern of Gram-negative bacteria isolated from urine culture in pregnant women at ALERT Hospital, Addis Ababa, Ethiopia (July–September 2018).
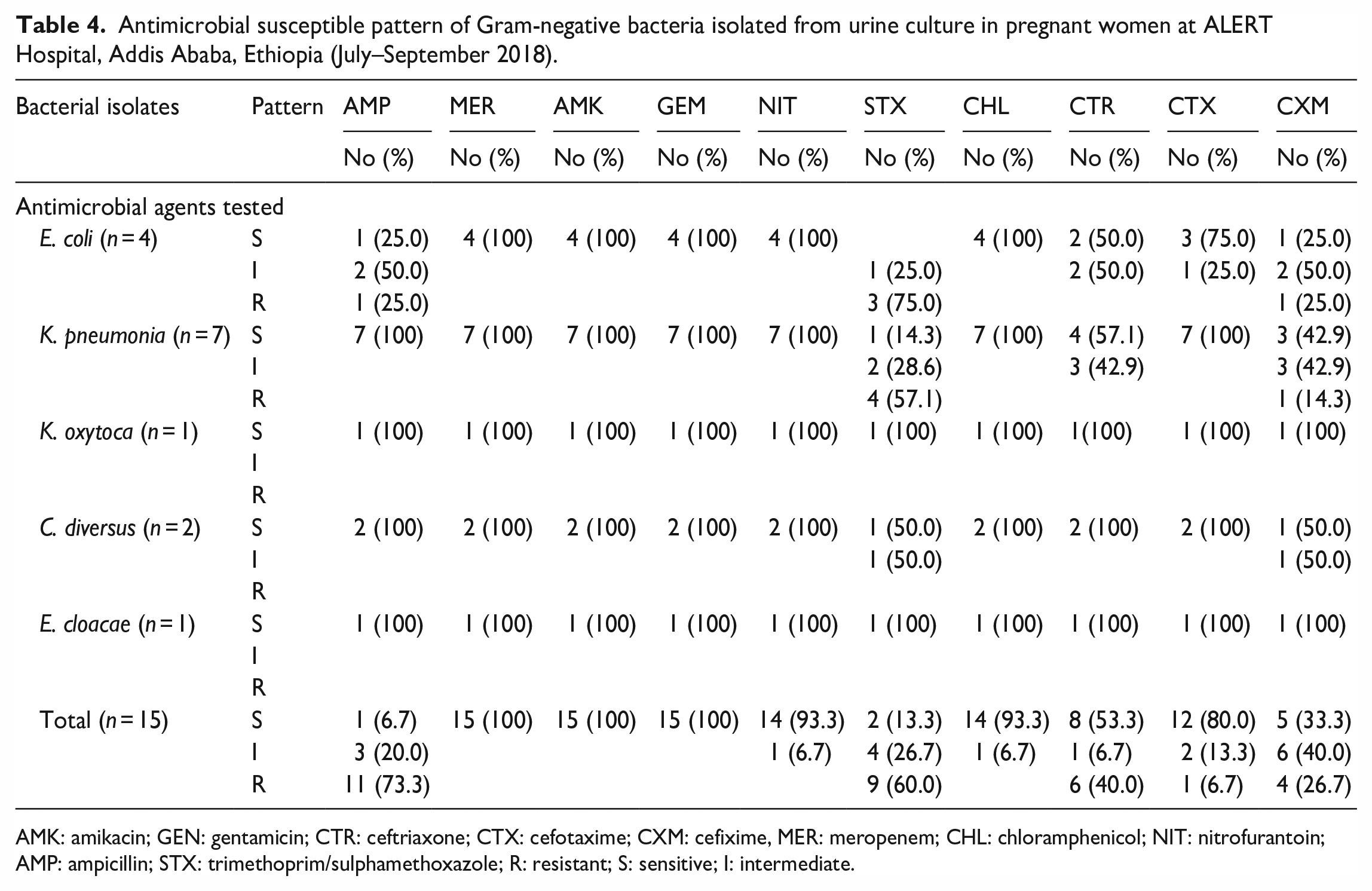
AMK: amikacin; GEN: gentamicin; CTR: ceftriaxone; CTX: cefotaxime; CXM: cefixime, MER: meropenem; CHL: chloramphenicol; NIT: nitrofurantoin; AMP: ampicillin; STX: trimethoprim/sulphamethoxazole; R: resistant; S: sensitive; I: intermediate.
Multiple drug resistance patterns of Gram-negative isolates
Among the total Gram-negative isolates (n = 15), multi-drug resistance (MDR) (resistance to two or more drugs) was observed in 80% (n = 12/15) of the isolates. Among all isolates of K. pneumoniae which is predominant from Gram-negative bacteria (n = 7), MDR were observed in 71.4%(n = 5/7) of the isolates (Table 5).
Multi-drug resistance pattern of Gram-negative bacterial isolates (n = 15) from pregnant women at ALERT Hospital, Addis Ababa, Ethiopia (July–September, 2018).

R0: no antibiotic resistance; R1: resistance to one drug; MDR: resistance to two and more drugs.
ESBL-producing Gram-negative bacilli
Fifteen members of Enterobacteriaceae have been isolated from 177 pregnant women. These isolates were K. pneumoniae 26.9% (n = 7/26), E. coli 15.4 % (n = 4/26), C. diversus 7.7% (n = 2/26), E. cloacae 3.8% (n = 1/26), and K. oxytoca 3.8% (n = 1/26). However, E. cloacae 3.8% (n = 1/26) and C. diversus 7.7% (n = 2/26) were excluded from further screening for ESBL though they were suspected because as this methods were not validated for these groups.
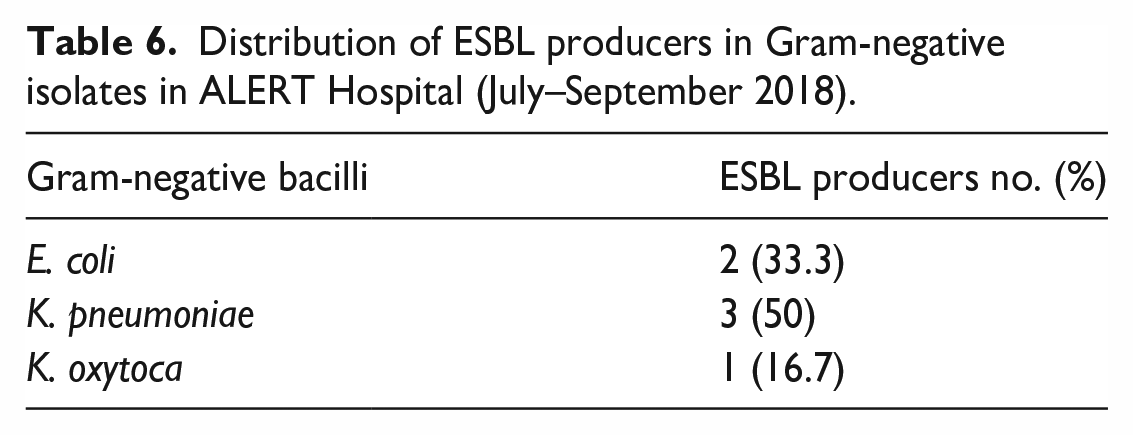
Therefore, 12 Enterobacteriaceae isolates were included, and all of them were suspected for ESBL by screening method. Hence, 12 Enterobacteriaceae were tested for ESBLs using the phenotypic confirmatory DDST according to European committee of antimicrobial susceptibility testing. The overall prevalence of ESBL-producing Gram-negative bacilli was 50% (n = 6/12). The most prevalent ESBL isolates were K. pneumoniae and E. coli accounting, 50% (n = 3/6) and 33.3% (n = 2/6), respectively. The least prevalent ESBL isolate was K. oxytoca, which accounts 16.7% (n = 1/6). Type of ESBL-producing Gram-negative bacilli isolates is shown in Table 6.
Distribution of ESBL producers in Gram-negative isolates in ALERT Hospital (July–September 2018).
Antimicrobial susceptibility pattern of ESBL-producing Gram-negative bacilli
The result of antimicrobial susceptibility pattern of ESBL-producing Gram-negative isolates is shown in Table 7. In general, among ESBL-producing isolates (100%), all are resistance to amikacin and gentamicin. All ESBL-producing isolates were susceptible for meropenem. K. pneumoniae, which accounted 50% of ESBL producers, was100% resistance to amikacin and gentamicin; 33.3% resistance to chloramphenicol, sulphamethoxazole-trimethoprim, and ampicillin. However, 100% susceptibility level was observed for meropenem.
Antimicrobial susceptible pattern of ESBL-producing Gram-negative bacilli at ALERT Hospital, Addis Ababa, Ethiopia (July–September 2018).

AMK: amikacin; GEN: gentamicin; CTR: ceftriaxone; MER: meropenem; CHL: chloramphenicol; NIT: nitrofurantoin; AMP: ampicillin; STX: trimethoprim/sulphamethoxazole; R: resistant; S: sensitive; I: intermediate
Discussion
The overall all proportion of UTI in this study was 14.7%. This was a similar finding to the study done in Khartoum North Hospital in Sudan (14.0 %). 14 Our result was also similar to the local findings reported previously in Mekelle (11.9%) 15 and Gondar (12%). 12 However, the finding in this study was lower than the study done in Sudan (22%). 16 The explanation for lower finding of our study than the study done in Sudan by Mohammad, 16 would be, the participant in the Sudan study was only hospitalized pregnant women which increases the prevalence of UTI due to the fact that the occurrence of nosocomial infection in admitted patients.
In this study, 30.8% of admitted pregnant women had SB, and there was a significant association between being outpatient or inpatient with UTI (COR = 3.284, 95% CI = 1.248–8.641, p = 0.016). Multivariable analysis showed that hospitalization was an independent risk factor for UTI infections. In this study, hospitalized pregnant women are almost five times more likely to develop UTI infections than non-hospitalized (AOR = 4.875, 95% CI = 1.628–14.598, p = 0.005). The proportion of UTI in hospitalized pregnant women increases due to the fact that the occurrence of nosocomial infection in hospitalized patients. This was supported by study done in Tikur Anbessa University Hospital. 17
In this study, the proportion of bacteriuria in symptomatic infection was 19.4% which was higher than previous studies reported from Ethiopia11,18 and study done in Sudan (7.3%). 16 The possible explanation for this is most of the study participants in the present study (93.8%) were in the 2nd and 3rd trimester of pregnancy. The risk to acquire bacteriuria during pregnancy increases with gestational age from 0.8% in the 12th week to 1.93% at the end of pregnancy. 19 On the other hand, the prevalence of (11.4%) bacteriuria in asymptomatic infection was in line with previous local report from Ethiopia 20 and elsewhere in the world such as in Ghana, 21 but lower than other findings in Sudan (14.7%). 16 The difference from latter study may be due to difference in study populations. From 72 pregnant women who complained of symptoms that suggest UTI, only 14 (19.4%) were found to have culture confirmed SB. Symptomatic patients whose urine culture did not show significant growth might be due to other less frequent UTI causing microorganisms, such as parasites, viruses and fungi. 22
The predominant etiological agents in our study were Gram-negative organisms 57.7%, whereas Gram-positive accounts relatively small proportion 42.3%. A previous study done in Mekelle showed similar finding. 15 Reports from Sudan also showed the same findings. 16 This could be due to the presence of unique structure in Gram-negative bacteria (pilus adhesions) which help for attachment to the uroepithelial cells and prevent bacteria from urinary lavage, allowing for multiplication and tissue invasion resulting in invasive infection and pyelonephritis in pregnancy.
In our investigation K. pneumoniae was the first most common bacterial etiologic agent of UTI which accounted (26.9%). This finding was in contrast with other study in which E. coli was the predominant bacteria. 23 Bacterial etiologies of UTI can show geographic variation and may even vary over time within a population. 24 The second dominant isolate was S. aureus (23.1 %). This was not supported by report from Addis Ababa, Tikur Anbessa Hospital, 17 in which K. pneumoniae was the second dominant isolate. Both E. coli and S. epidermidis were the 3rd common bacteria isolates (15.4%).
The present study revealed that Gram-negative isolates that had shown a higher proportion rate of resistance to the commonly prescribed antibiotics. Antimicrobial resistance levels for the Gram-negative isolates range from 0% to 100%, which was also described by study done in Tikur Anbessa Hospital. 17 All Gram-negative isolates showed resistance to amikacin and gentamicin (100%). Different finding was reported from a study done in Gonder University Hospital 11 in which resistance rate of 7.4% was observed. The level of resistance reported from study in Tikur Anbessa Hospital (70.9 %) was higher than report from Gonder University Hospital, but still it is lower than our investigation. 17 The possible explanation for lower findings from previous studies in our country as compared to the present study is amikacin and gentamicin are injectable drugs, which were not easily available. So, the selective pressure which was created by misuse and over use of these drugs was reduced in earlier time in our country. However, our result showed that the custom of drug administration changed time to time which resulted production of resistance to these drugs.
Ampicillin with 73.3% resistance rate was the second drug that showed high level of resistance following amikacin and gentamicin in this study. Higher findings were reported from Gonder 11 with 100% resistance rate. However, slightly lower result (70%) was investigated in a study at Tikur Anbessa Hospital. 20 The discrepancy might be due to difference in drug prescribing practice and etiologic agent across different geographic area.
Among the drugs cefotaxime, Nitrofurantoin and Chloramphenicol with resistance rate of 6.7% had a better susceptibility for most of Gram-negative bacteria than other drugs tested. Our finding was supported by previous studies done in Ethiopia by Getachew et al. 12 and Tazebew et al. 18 in Tanzania 25 and Iran. 26
Meropenems had 100% susceptibility for all Gram-negative bacteria isolated in our study. This was supported by study done by Agamy et al. 27 However, this result was higher than what had been found in other study which was 26.08%. 28 Since these drugs were not sold in our country and also they were expensive, this probably had restricted their procurement and indiscriminate use, therefore making the organisms susceptible to it.
In our study, overall MDRs were observed among 12 (80%) of Gram-negative isolates. This finding was lower than what has been reported from previous study at Tikur Anbessa, which was 89.1%. 17 The higher record reported from Tikur Anbessa was due to high prevalence of ESBL-producing isolates (78.57%). This is explained by the fact that many of the isolates producing these enzymes are also resistant to quinolones, trimethoprim, and aminoglycosides, often plasmid has co expression of other resistance mechanisms. 9
In this study, ESBL producers were found in 50% of Gram-negative bacilli which was similar with study done at Tikur Anbessa Specialized Hospital where the overall gastrointestinal colonization rate of ESBL-producing Enterobacteriaceae in hospitalized patients was 52%. 29 Our finding was higher than previous study done in Harar (33%). 30 In addition, our finding was also higher than studies from Nigeria (20%), 31 Northwestern Nigeria (34.3%), 32 and elsewhere such as Iran (40.8%). 33 On the other hand, higher results were reported from Tikur Anbessa Hospital in Addis Ababa (78.57%), 34 Southern Terai of Nepal (63.31%). 35 The finding of higher results from study done in Tikur Anbessa Hospital by Legese et al. 34 could be due to incorporation of large proportion of hospitalized study participants (80%) as compared to our study (14.7%). This is true due to hospitalization is the risk factor for colonization of ESBL-producing bacteria.
This study revealed that K. pneumoniae was the most ESBL-producing isolates 50%. This was in line with study done in Uyo, Nigeria (50%). 31 Lower results were obtained by study done in Northwestern Nigeria (40%) 32 and local study at Tikur Anbessa Specialized Hospital (32%). 29 E. coli was the second most ESBL-producing isolates, which account 33.3%. Greater result was reported from Northwestern Nigeria (50%) 32 and previous study at Tikur Anbessa Hospital (68%). 29 K. oxytoca accounted 16.7% of ESBL producers, which disagree with study from Uyo in Nigeria where 6% of ESBL producers were K. oxytoca. 31 The variation on ESBL positivity might be due to the number of isolates studied, variation in institution to institution, geographic location, and also country to country. The prevalence of ESBL production is high in the referral centers where the patients are referred from the peripheral centers and where the antibiotic use is profuse. The higher prevalence of ESBL producers in our country compared to western countries can be explained by, western countries have strict infection control policies and practices, efficient and effective antibiotic audit systems, shorter average hospital stays, better nursing barriers, and other important healthcare measures, which substantially decrease the chances of acquisition and spread of ESBL strains. The uncontrolled use of 3rd generation cephalosporin antibiotics at our study hospital could be a leading contributory factor to the high ESBL proportion observed in this study.
Among ESBL-producing isolates (100%) all were resistance to amikacin and gentamicin, whereas intermediate level resistance rate of 66.7% was observed among sulphamethoxazole-trimethoprim. Therefore, using these drugs for the infection of ESBL-producing isolates can result in failure of treatment. Thus, the problem of ESBLs have great clinical importance, but most clinicians not give good concern yet. The choice of antimicrobial agents effective against ESBL- producing species is currently limited and cause serious therapeutic problems in the future. 36 In our study, most of them were susceptible for some limited drugs, and these were Nitrofurantoin (83.3%) and Chloramphenicol (83.3%) as compared to other tested drugs. Ampicillin (33.3%) was also an option which had lowered resistance level of 16.7%; however, it may not be satisfactory as needed since it had wide range of intermediate level (50.0%) with a tendency of becoming resistant. Meropenem was the 1st choice of drug for ESBL-producing bacteria since all isolates responded to this drug which had susceptibility level of 100%. However, meropenem is not easily accessible, available, and very costly.
Strength and limitation of the study
Strength of the study
❖ This study shows the magnitude of ESBL-producing Gram-negative uropathogens among pregnant women.
❖ To best of our knowledge, there was no documented study conducted similar to this in the study area.
❖ It suggests treatment options for ESBL-producing Gram-negative bacteria, especially in the study area
Limitations of the study
❖ Unavailability of certain drugs used for antimicrobial susceptibility testing
Conclusion
This study shows that overall proportion of UTI in pregnant women was 14.7%. In our investigation, K. pneumoniae was the predominant bacterial etiologic agent of UTI. ESBL-production showed high distribution in Gram-negative isolates, especially in K. pneumoniae. In this study, ESBL producers were found in 50% of Gram-negative isolates.
The finding from this study indicates that the highest level of resistance of Gram-negative isolates was observed for amikacin and gentamicin with 100% rate of resistance for each. Most of the Gram-negative isolates showed high resistance level for sulphamethoxazole–trimethoprim and ampicillin. MDR were observed among most of Gram-negative isolates.
Recommendations
➢ Genotypic characterization of ESBL-producing isolates, which causes UTI in pregnant women should be carried out, especially in our study area.
➢ Training about ESBL detection methodologies, especially DDST should be given for all laboratory personnel to help them to be familiar with this easy but valuable methods which aids them to do when it is requested.
➢ Further and extended investigation on ESBL-producing organisms in pregnant women that includes hospitals, different geographical zones, and other population in larger sample size are needed to confirm these findings and assess possible risk factors.
Footnotes
Acknowledgements
The authors would like to acknowledge Addis Ababa University for funding the budget to conduct the experiments and ALERT Hospital for providing all the necessary laboratory reagents, media, and other laboratory facilities. They would like to thank all the study participants for their willingness during sample collection.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Department Research and Ethics Committee (DERC) of Department of Microbiology, Immunology and Parasitology, Addis Ababa University (DERC committee’s reference number: DERC/17/18/02-C). This study was conducted in accordance with the Declaration of Helsinki.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: No external funds were obtained; only institutional support from Addis Ababa University and ALERT Hospital.
Informed consent
Written informed consent was obtained from all subjects before the study.


