Abstract
Objective:
Upper extremity vascularized composite allotransplantation is an innovative treatment option for people with upper extremity amputations. Limited patient-relevant long-term outcomes data about transplant success may impede patients’ informed treatment decision-making. We assessed perceptions of what constitutes upper extremity vascularized composite allotransplantation success among individuals with upper extremity amputations.
Methods:
This multisite study entailed interviews and focus groups with individuals with upper extremity amputations and upper extremity vascularized composite allotransplantation candidates, participants, and recipients. We examined perceptions of transplant success and preferences for five upper extremity vascularized composite allotransplantation outcomes. Qualitative data were analyzed using thematic analysis; and quantitative data were analyzed using descriptive statistics.
Results:
In all, 50 individuals participated in interviews (61.7% participation rate), and 37 participated in nine focus groups (75.5% participation rate). Most were White (72%, 73%), had a mean age of 45 and 48 years, and had a unilateral amputation (84%, 59%), respectively. Participants conceptualized upper extremity vascularized composite allotransplantation success as transplant outcomes: (1) restoring function and sensation to enable new activities; (2) accepting the transplanted limb into one’s identity and appearance; (3) not having transplant rejection; (4) attaining greater quality of life compared to prosthetics; and (5) ensuring benefits outweigh risks. Participants rated their most important upper extremity vascularized composite allotransplantation outcomes as follows: not having transplant rejection, not developing health complications, grasping objects, feeling touch and temperature, and accepting the upper extremity vascularized composite allotransplantation into your identity.
Conclusion:
Individuals with upper extremity amputations maintain several conceptions of vascularized composite allotransplantation success, spanning functional, psychosocial, clinical, and quality of life outcomes. Providers should address patients’ conceptions of success to improve informed consent discussions and outcomes reporting for upper extremity vascularized composite allotransplantation.
Introduction
Upper extremity (UE) vascularized composite allotransplantation (VCA) is an innovative treatment option for individuals with UE amputations. To date, 56 UE VCAs have been performed on 37 patients in the United States (Organ Procurement and Transplantation Network (OPTN) as of January 2023), and more than 120 have been performed worldwide.
1
Having demonstrated good to excellent outcomes, UE VCA has expanded worldwide over the past two decades.1,2 UE VCA clinical
However, the outcomes assessments used by different VCA centers are not standardized, making it difficult to compare outcomes across different measures.7,8 Reporting requirements also vary by institution and country. 7 In the United States, the OPTN requires transplant centers to submit data, but does not mandate completing all data fields, resulting in a lack of complete, longitudinal functional outcomes data from which to examine the benefits and efficacy of UE VCA. 6 Internationally, about half of the transplant centers report VCA outcomes to the International Registry on Hand and Composite Tissue Transplantation (IRHCTT). 7
Moreover, psychosocial outcomes (i.e., recipient satisfaction with treatment) are assessed and reported inconsistently and not comprehensively. For example, the OPTN’s health-related quality of life (QoL) questionnaire (i.e., Short-Form 12) does not sufficiently address potential psychosocial benefits of VCA including the impact of UE VCA on a recipient’s body image and social functioning. 9
Providing patients comprehensive information about the UE VCA success rate may help them to make better informed treatment decisions. 10 Adults with upper limb loss report receiving insufficient information about treatment options from providers, which may be exacerbated by the paucity of reported long-term outcomes and qualitative accounts from UE VCA recipients.11,12 Information disclosure is essential for patients to set realistic expectations about treatment. 13 More accurate treatment expectations have been associated with improved outcomes for upper extremity conditions; however, patients with more accurate treatment expectations and a high level of disability at baseline (such as individuals with an UE amputation) may be more likely to be unsatisfied with outcomes.14–16 Information disclosure can allay patients’ uncertainty about the range of potential UE VCA outcomes (i.e., functional, psychosocial, health risks).17–19 Failure to meet a recipient’s expected outcomes in the past has led to psychosocial disorders, suicide, and requests from the recipient to re-amputate their transplanted limb. 20 Little is known about perceptions of transplant success among individuals with UE amputations. This article assessed perceptions of UE VCA success among individuals with UE amputations.
Methods
Study design
In this cross-sectional study, we conducted interviews and focus groups to assess perceptions of transplant success and relative importance of UE VCA outcomes among individuals with an UE amputation. 21 Mixed-methods analysis helped to elaborate and clarify qualitative and quantitative findings.22,23 The findings reported herein were part of a larger study examining informed consent and treatment decision-making about UE VCA. 24
Setting
The study was conducted at Northwestern University (NU) in Chicago, IL; Walter Reed National Military Medical Center (WRNMMC) in Bethesda, MD; and Johns Hopkins University (JHU) in Baltimore, MD. David Rotter Prosthetics, LLC in Joliet, IL, and The Shirley Ryan AbilityLab in Chicago, IL, served as additional recruitment sites which aided NU recruitment. Institutional Review Boards approved and oversaw the respective study at each site: NU (STU00209718), WRNMMC (WRNMMC-EDO-2020-0432), JHU (00225728). NU hosted a single Institutional Review Board for WRNMMC. We used the Consolidated Criteria for Reporting Qualitative Research to guide quality reporting for our qualitative study. 25
Participants and recruitment
Eligible participants included English-speaking adults (age 18–65 years) with acquired UE amputations. Eligible individuals had either never pursued UE VCA or were UE VCA candidates (i.e., individuals who contacted a transplant program to express interest in pursuing UE VCA), UE VCA participants (i.e., individuals who began the UE VCA evaluation process), and UE VCA recipients who had already received a transplant. Adults who were cognitively impaired or born with UE limb differences were ineligible for the study. Research team members did not have pre-established relationships with study participants.
Patients at each study site were e-mailed/mailed recruitment letters, followed by a phone call 1 week later (⩽5 times) to screen for eligibility. Flyers describing the study were distributed through in-person and online communities (support groups, Facebook, and Reddit). Individuals recruited by flyers contacted the study team first and were screened by phone for eligibility. All participants provided informed verbal consent.
Data collection
In-depth interviews
We conducted in-depth interviews by telephone from July 2020 to March 2022 to assess participants’ perceptions of and preferences for UE VCA success. Target interview numbers were set a priori at
The interviewer started the interview by providing background information about UE VCA that covered the definition, purpose, and potential benefits of UE VCA. This analysis was based on four open-ended questions and one closed-ended ranking question from the larger study’s interview guide which elicited perceptions, information needs, and decision-making about UE VCA. One open-ended question asked participants to identify their personal definition of transplant success (“How do you define success (as it pertains to hand transplantation)?”). The concept of success emerged through inductive review of transcripts, and this question was incorporated into the interview guide after 22/50 interviews had been completed.
The other questions were present in the interview guide from the beginning. A closed-ended question asked participants to rank five transplant outcomes, identified inductively through five cognitive interviews, from most to least important: grasping objects; feeling touch and temperature; accepting the UE VCA into your identity; not having rejection; and not developing health complications. Three other open-ended questions assessed participants’ perceived benefits and risks of UE VCA: “What do you think are the benefits of upper limb transplantation?”; “What are some activities that you cannot do now, that you think might be possible to do with an upper limb transplant?”; and “What do you think are the risks of upper limb transplantation?”
Interviews concluded with sociodemographic questions (e.g., age, gender, race); a question about health literacy (“How often do you need to have someone help you when you read instructions, pamphlets, or other written material from your doctor or pharmacy?” anchored by “Never,” “Rarely,” “Sometimes,” “Often,” and “Always”; “Never” and “Rarely” responses were considered adequate health literacy) 28 ; and clinical information about the participant’s amputation (e.g., date, level, and type of amputation).
Interviews were conducted by female and male research team members (BK, KV, MD, and MN) trained by the principal investigator (EJG), an expert in qualitative research. Interviewers were data analysts, research coordinators, and occupational therapists with graduate (e.g., MS and/or PhD) and/or baccalaureate degrees. Interviewers informed study participants that they were members of the research team and were not UE VCA clinicians (except MN). Cognitive interviews (
Focus groups
We conducted focus groups among adults with UE amputations, and UE VCA candidates, participants, and recipients, from August 2021 through May 2022 as part of a larger study to assess participants’ feedback on the content and design of an educational website about UE VCA intended to bolster the informed consent process for UE VCA. 30 Target focus group numbers were set a priori at three focus groups per study site to reach thematic saturation. 31 We aimed to have an average of five participants per focus group to facilitate telephone communication dynamics. 32
The moderator provided background information about VCA at the start of each focus group that covered the definition of VCA, a description of the types of VCA organs, and an explanation that VCA organs are transplanted primarily from a deceased donor. We drew from one open-ended question and one closed-ended question from the larger study’s moderator’s guide to elicit participants’ conceptions of and preferences for UE VCA success (“When you hear the concept of ‘transplant success,’ what does that mean to you?”). Thereafter, the moderator displayed three definitions of UE VCA success, developed through inductive coding of prior in-depth interviews, and asked participants to choose their preferred definition (“Which interpretations fit your idea of transplant success better: (1) surgical success in terms of attaching the hand to the body, (2) hand function gained from the transplant, and (3) the relevant function gained form a transplant versus a prosthetic?”). Participants were asked to provide a rationale for their choice and debated choices during the focus group discussion. Focus groups were moderated by female and male research team members (EJG, MD, and MN) trained by the principal investigator (EJG). Focus groups had an average duration of 2 h and were audio-recorded and transcribed. Participants were compensated with a $35 gift card. The aforementioned sociodemographic and clinical information was collected immediately after each focus group via an online survey.
Qualitative analysis
All interview and focus group transcripts were analyzed together for themes and patterns that emerged from the data using the constant comparison method. 33 The research team established an initial codebook by developing deductive codes based on question topics from the in-depth interview guide (i.e., benefits, risks, information needs, decision-making, etc.).The research team then developed inductive codes by iteratively reviewing and open-coding 2–5 transcripts from each study site. The research team held analytic retreats to resolve coding discrepancies, iteratively adjust code definitions, and refine the codebook until reaching thematic saturation.34,35 After finalizing the codebook, two research team members at each study site (BK, JG-S, KV, MD, ML, and MN) independently coded transcripts using NVivo (QSR International; Burlington, MA,USA) to establish inter-rater reliability (kappa > 0.80). 36 Then, research team members coded all interview and focus group transcripts, and discussed coding discrepancies to reach consensus. Research team members analyzed all coded segments for each code together to develop code summaries that reflected broad themes related to participants’ perceptions of UE VCA success. 37
Statistical analysis
Descriptive statistics were performed on post-activity demographic surveys and the close-ended five item UE VCA outcomes ranking question. We calculated frequencies, means, and standard deviations (SDs). “Never” and “Rarely” health literacy question responses were considered adequate. We used IBM SPSS Statistics (Version 27; IBM Corp., Armonk, NY, USA) to analyze the quantitative data.
Results
Participant demographics
In all, 50 individuals participated in interviews (61.7% participation rate; completed/consented) and 37 individuals participated in nine focus groups (75.5% participation rate; completed/consented) were conducted. Some individuals participated in both data collection activities (NU:
Of the 50 interview participants, most were males (78%), White (72%), on average 45 years of age, had adequate health literacy (94%), and had a unilateral amputation (84%). The median number of years since participants’ first amputation was 9.9 years (SD 8.2) (range < 1–41 years).
Of the 37 focus group participants, most were males (65%), White (73%), on average 48 years of age, and had adequate health literacy (84%). More than half of the participants had a unilateral amputation (59%). The median number of years since participants’ first amputation was 10 years (SD 8.3) (range < 1–41 years). Tables 1 and 2 present participants’ sociodemographic characteristics.
In-depth interview participants’ demographic and clinical characteristics.

SD, standard deviation.
“Other” included people who identified as Hispanic or Mexican (
Percentages do not add up to 100 because some participants did not respond.
Percentages add up to greater than 100 due to more than one response from some participants.
Some participants had multiple surgeries for their amputation or multiple amputations.
Focus group participants’ demographic and clinical characteristics.

SD, standard deviation.
“Other” included one person who identified as Malagasy (
“Other” included one person in the process of getting a body-powered prosthesis (
Percentages do not add up to 100 because some participants did not respond.
Percentages add up to greater than 100 due to more than one response from some participants.
Some participants had multiple surgeries for their amputation or multiple amputations.
Themes
We identified five major themes regarding definitions of UE VCA success: (1) restoring function and sensation to enable new activities, (2) accepting the transplanted limb into one’s identity and appearance, (3) experiencing no rejection from surgical attachment of the donor limb, (4) attaining greater QoL from UE VCA compared to prosthetics, and (5) ensuring that the benefits outweigh the risks. Table 3 presents illustrative representative quotations for each theme.
Representative quotations by theme.
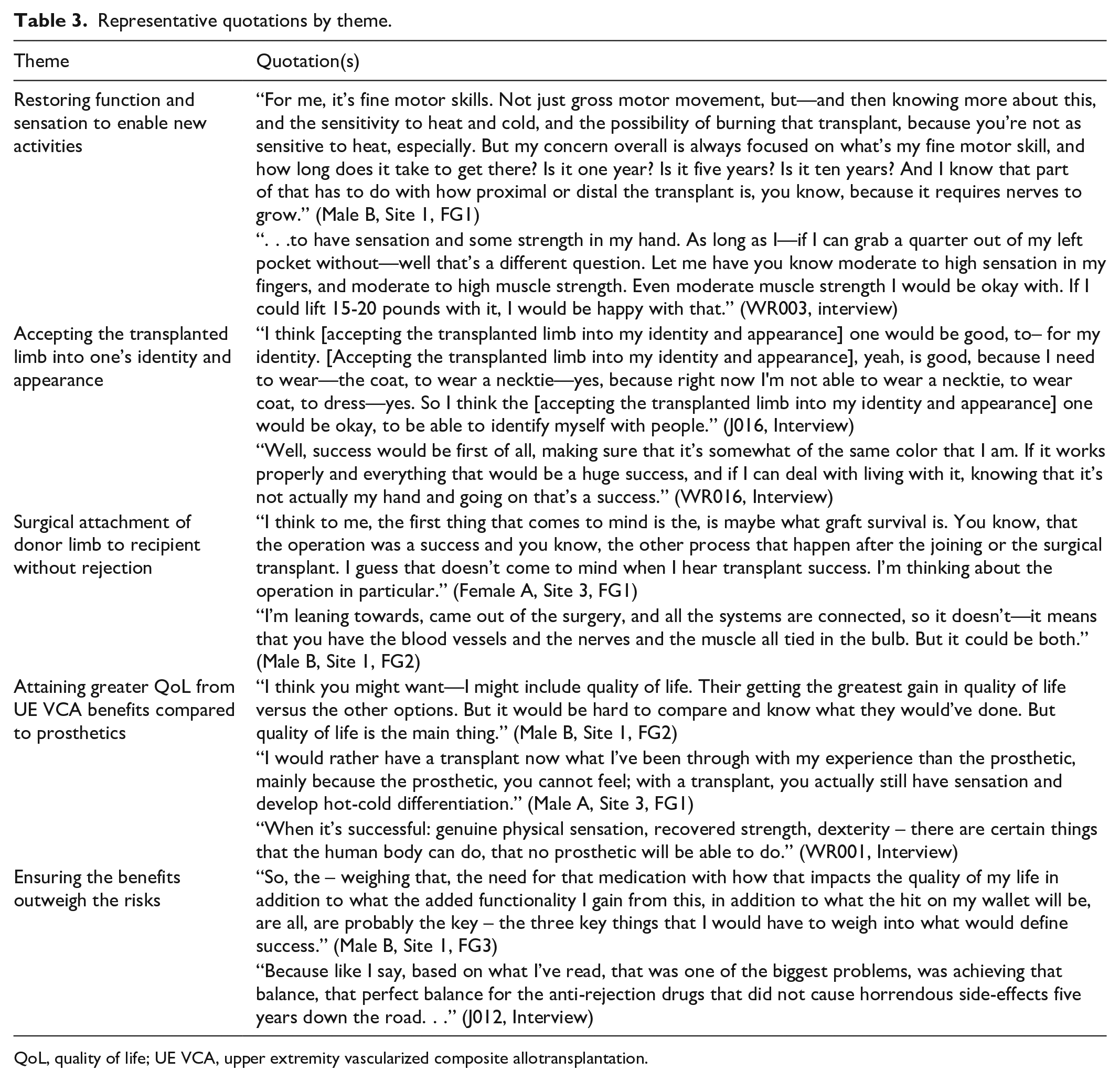
QoL, quality of life; UE VCA, upper extremity vascularized composite allotransplantation.
Restoring function and sensation to enable new activities
The most pervasive perception of UE VCA transplant success was the benefit of restoring function and sensation in the transplanted limb. Because many daily activities of living are challenging to perform without an upper limb, participants (
Participants (
Accepting the transplanted limb into one’s identity and appearance
Some participants (
Experiencing no rejection from surgical attachment of the donor limb
Several participants (n = 10 interviewees; and 16 focus group participants across 8 focus groups) conceived UE VCA success in surgical terms of joining the donor limb to the recipient’s arm and recovery occurring without adverse reactions or rejection. Participants described successful surgical attachment using parlance such as the graft being “taken” or the body accepting the new arm. Conversely, some participants expressed that surgical attachment alone, without regained function after rehabilitation, would be an overall failure of the transplant process.
Attaining greater QoL from UE VCA compared to prosthetics
Several participants (
Ensuring the benefits outweigh the risks
Success was defined by some participants (
Ranking UE VCA outcomes
Most interview participants (
Ranking transplant success outcomes by importance (
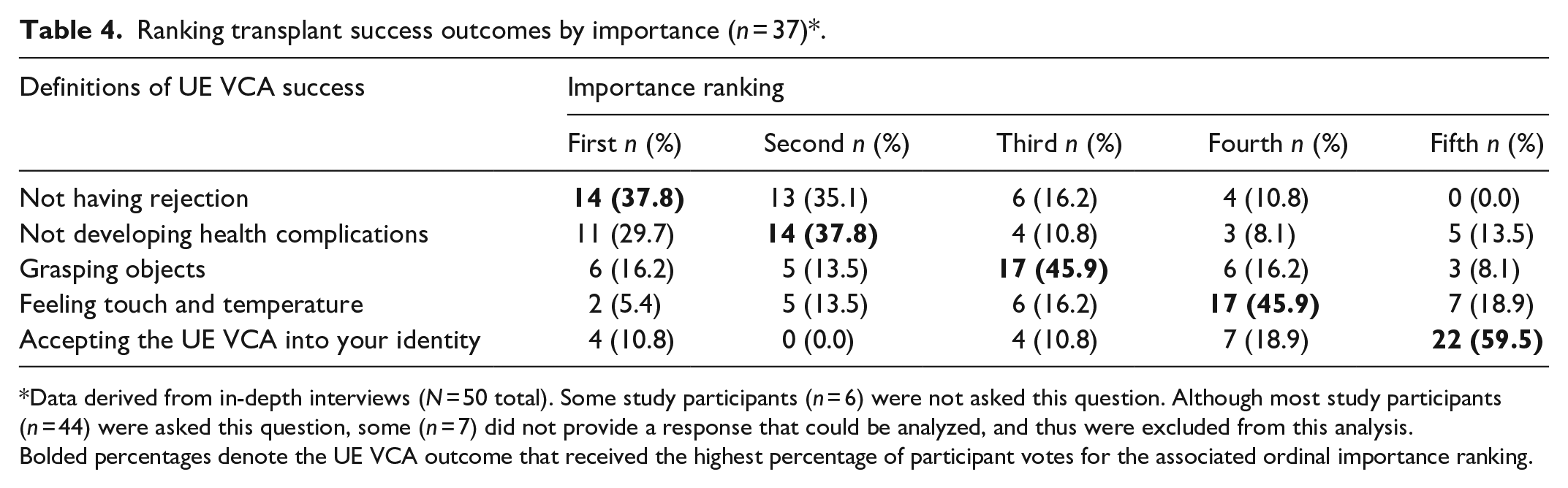
Data derived from in-depth interviews (
Bolded percentages denote the UE VCA outcome that received the highest percentage of participant votes for the associated ordinal importance ranking.

Participants’ relative importance vote percentage (first–fifth) by UE VCA Outcome (
Discussion
Principal results
This mixed-methods study assessed patient definitions of UE VCA transplant success spanning functional, psychosocial, clinical, and QoL conceptions of success. Our findings demonstrate that individuals with UE amputations may prefer certain UE VCA outcomes compared to others, specifically outcomes that avoid health risks of UE VCA.
Comparison with prior work
Participants’ leading preferences for outcomes of not having rejection and not developing health complications corroborate prior research studies in which patients with UE amputations recognized the inherent risks and potential failures of UE VCA.18,38 Having gone through prior amputation surgery and lived with an UE amputation, many individuals with an UE amputation prioritize avoiding further adverse health events and further burdens for their caregivers. 39 Patient education can clarify concerns about transplant rejection. While many study participants regarded limb rejection as a “failure” of the UE VCA procedure, clinical research shows that acute rejection is common for UE VCA recipients (e.g., 85% of recipients experienced ⩾1 acute rejection episode within the first year post-transplant) and is treatable. 6
Future implications/research
Patient-centered conceptions of UE VCA success, identified in our study by participants who are accurately informed about VCA, can be used to create patient-relevant UE VCA outcomes measures (i.e., measures which assess the specific functional, psychosocial, clinical, QoL outcomes identified by study participants). Having access to long-term outcomes data about UE VCA will help to better inform individuals about this treatment.
Prior research has shown that minimizing the gap between treatment expectations and outcomes may lead to better patient satisfaction with treatment. 15 Given this notion, our study findings should be used to inform and validate UE VCA outcome measures so that they align with patients’ expectations and are relevant to patients’ treatment goals.
Currently, the OPTN and IRHCTT use objective and self-reported functional outcomes measures for UE VCA. Objective functional tests cover abilities such as grip strength, fine motor coordination, and temperature sensation which are important to study participants. 9 Furthermore, VCA recipients’ ability to complete activities of daily living and recreational activities are assessed by DASH, reflecting our study participants’ functional desires. 40
Although our study findings support the types of outcomes assessed by these functional metrics, standardized collection and mandated reporting of functional outcomes are needed to assess the long-term success of UE VCA. 6 In addition, larger sample studies are needed to determine if there is a functional advantage of UE VCA when compared to prosthetics or no remediation. 41 Psychosocial outcomes are lesser explored and reported. Psychosocial outcome measures should be tailored to VCA by capturing aspects of appearance, identity, and social functioning, as the Hand Transplant Scoring System does. 42 Future research should qualitatively assess whether receiving an UE VCA alleviates feelings of conspicuousness and/or social stigma experienced with limb loss. 43 Future qualitative accounts from UE VCA recipients should speak to the psychosocial and health-related QoL aspects of post-transplant life discussed by study participants.
Strengths
As a multisite study recruiting from three metropolitan areas, and online communities for individuals with UE amputations throughout the United States, our sample’s geographic diversity fosters transferability of study findings. Participants included civilians and military personnel, and reflected subgroups of individuals with UE amputations who varied by amputation level, laterality, and time since amputation, all of which are important factors considered for UE VCA candidate selection and treatment decisions. In addition, our study reports on a large number of people with UE amputations, a population that is difficult to recruit into research.
Limitations
As some participants lacked awareness and knowledge of UE VCA, their opinions were less informed about UE VCA. Given that a subset of participants engaged in both in-depth interviews and focus groups, perspectives may have been duplicated. Furthermore, challenges with administering the five-item UE VCA outcomes ranking question by phone led to incomplete responses which lowered the sample size.
Conclusion
Our study found that individuals with UE amputations held multiple conceptions of transplant success, pertaining to functional, psychosocial, clinical, and QoL outcomes. Participants preferred outcomes of avoiding health risks over functional or psychosocial gains from receiving an UE VCA. Providers should use patient conceptions of success to report outcomes in a patient-centered manner and to provide more relevant information to patients.
Footnotes
Author contributions
EJG conceived and designed the research study and participated in performing the research, analyzing/interpreting the data, and writing the manuscript. MD and JG-S participated in performing the research, analyzing/interpreting the data, and writing the manuscript. BK, KV, MN, ML, and WL participated in data collection and analyzing/interpreting the data. TR, WA, and SF participated in data collection. All authors reviewed and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by award #W81XWH-19-2-0033/RT180041 from the Department of Defense to EJ Gordon, Initiating Principal Investigator (PI); #W81XWH-19-2-0034/RT180041P1 to M Levan, Partnering PI; #W81XWH-19-2-0035/RT180041P2 to G Brandacher, Partnering PI; and #W81XWH-19-2-0036/RT180041P3 to S Tintle, Partnering PI. The opinions and assertions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Department of Defense, the U.S. Government, the Uniformed Services University, or the Henry M. Jackson Foundation for the Advancement of Military Medicine.
This study used Research Electronic Data Capture (REDCap), a secure online data capture application supported at the Feinberg School of Medicine by the Northwestern University Clinical and Translational Science Institute. This work was also supported in part by the Northwestern Medicine Enterprise Data Warehouse. The funders had no role in study design; collection, analysis, and interpretation of data; writing the manuscript; and the decision to submit the manuscript for publication.
