Abstract
Background:
Currently, more than two-thirds of medical decisions are made based on laboratory test results. However, due to the compromised pre-analytical and post-analytical phases (or extra-analytical phase), the reporting of incorrect or inappropriate test results was still prevalent. Studies focusing on the prevalence of and factors associated with laboratory error remain very limited in Ethiopia.
Objective:
This study aimed to assess the magnitude of extra-analytical errors and associated factors in medical laboratory services in 13 public hospitals in Addis Ababa, Ethiopia, from January to April 2020.
Materials and Methods:
A hospital-based cross-sectional study was conducted in the medical laboratories of public hospitals in Addis Ababa from January to April 2020 to assess extra-analytical errors and associated factors. Data were collected from 2401 laboratory request forms ordered within the study period and 169 laboratory professionals working in public hospitals. The collected data were entered and analyzed using SPSS version 23 software. Data were analyzed using simple descriptive statistics, percentages, and frequencies and summarized using tables and text. Bivariate logistic regression analysis was conducted, and variables (with a p value below 0.25) were included in the multivariate logistic regression model. A p value <0.05 was considered a cutoff point in the final model.
Results:
In this study, of the errors detected, 60.3% occurred in the pre-analytical phase and 39.7% in the post-analytical phase. Extra-analytical errors in laboratory services were significantly associated with lack of written procedures for laboratory activities (adjusted odds ratio = 2.79, 95% confidence interval = 1.34–5.86), 1–2 years of work experience (adjusted odds ratio = 5.5, 95% confidence interval = 2.54–9.31), work experience (3–5 years) (adjusted odds ratio = 2.67, 95% confidence interval = 0.94–4.56), and education (diploma) (adjusted odds ratio = 6.30, 95% confidence interval = 2.17–12.26).
Conclusion:
This study discovered errors in the pre-analytical (60.3%) and post-analytical (39.7%) phases, and none of the requisition papers had complete data. This frequency may be affected by workload, a lack of written procedures, the number of years of work experience, and the level of education of the staff.
Introduction
Providing quality laboratory services or error-free laboratory services is an integral component of the modern healthcare system. 1 In spite of this fact, laboratories’ quality systems in Sub Saharan Africa such as Ethiopia remain weak and need more effort to improve. Accurate, reliable, and timely laboratory tests are required to diagnose illness; identify causative factors; monitor the effectiveness of treatment, and perform investigations for key diseases. Patient safety is influenced by the frequency and seriousness of errors that occur in the healthcare system. 2 The consequences of errors during the laboratory testing process can affect patient care in many ways, such as delay in reporting, unnecessary redraws, misdiagnosis, and improper treatment. 3 Previous studies report that, as a percent of total error, pre-analytical phase are 46% or 68.2% and post-analytical phase are 18% or 47%.4,5
The extra-analytical phase is one of the most challenging steps and is often overlooked by medical laboratory managers, who may be unfamiliar with the processes. The predominant feature of this phase was the lack of uniform standards and adequate indicators to ensure the excellence of phlebotomy and other sample reception procedures. 6 Due to the compromised pre-analytical and post-analytical events, the reporting of incorrect or inappropriate test results is still prevalent.7–9 Poor communication between laboratory professionals and clinicians is generally cited as a chief issue affecting quality during the pre- and post-analytical phases. 10 Appropriate monitoring of pre-analytical errors requires interdepartmental cooperation since several sources of these errors fall outside the direct control of laboratory employees.11,12
Quality in laboratory medicine is the assurance that all phases of the testing process are carried out correctly, confirming efficient decision-making and effective patient management. 13 Errors can have a negative impact on patient care by contributing to incorrect treatment, longer hospital stays, and dissatisfaction with healthcare services. Ethiopia is working to improve laboratory service quality, but errors remain extremely high. The previous studies were conducted at a single health facility. As a result, the purpose of this study was to evaluate the magnitude of extra-analytical errors and associated factors in medical laboratory services in 13 public hospitals in Addis Ababa, Ethiopia.
Materials and methods
Study setting, design, and period
From January to April 2020, a hospital-based cross-sectional study was conducted to assess extra-analytical errors and associated factors in the medical laboratories of public hospitals in Addis Abeba, Ethiopia. The city has 14 public hospitals, five of which are run by the Addis Abeba Health Bureau (AHB), six by the Federal Ministry of Health, and three by the Ministry of Defense, all of which provide teaching, specialized care, and referral services. Since all the laboratories in these hospitals participated in the national basic laboratory strengthening programs, almost all the hospitals have similar workflows in the laboratories. No matter the sample collection sites, all the samples were collected by the laboratory’s professionals. 14 One of the 14 public hospitals was excluded from the study due to pre-testing of the questionnaire, while the other 13 were included.
Study subjects and inclusion criteria
All request forms ordering medical laboratory tests (laboratory request forms (LRFs)) for serology, hematology, parasitology, and chemistry departments were included in the study. Those requesting emergency, pathology, and microbiological tests were excluded. Moreover, employees who were unavailable were excluded. All medical laboratory professionals working in Addis Ababa’s public hospitals during the study period with at least 1 year of experience were invited to participate. Participation was voluntary.
Sampling method and sample size determination
Simple random sampling for laboratory professionals and systematic random sampling techniques for LRFs were applied. For quantitative data type, sample size was calculated for the laboratory request using the minimum sample size calculation formula for estimating a single proportion assuming p = 50%, 2% margin of error (d), and 95% confidence interval (CI); hence, no similar study was obtained in Ethiopia:
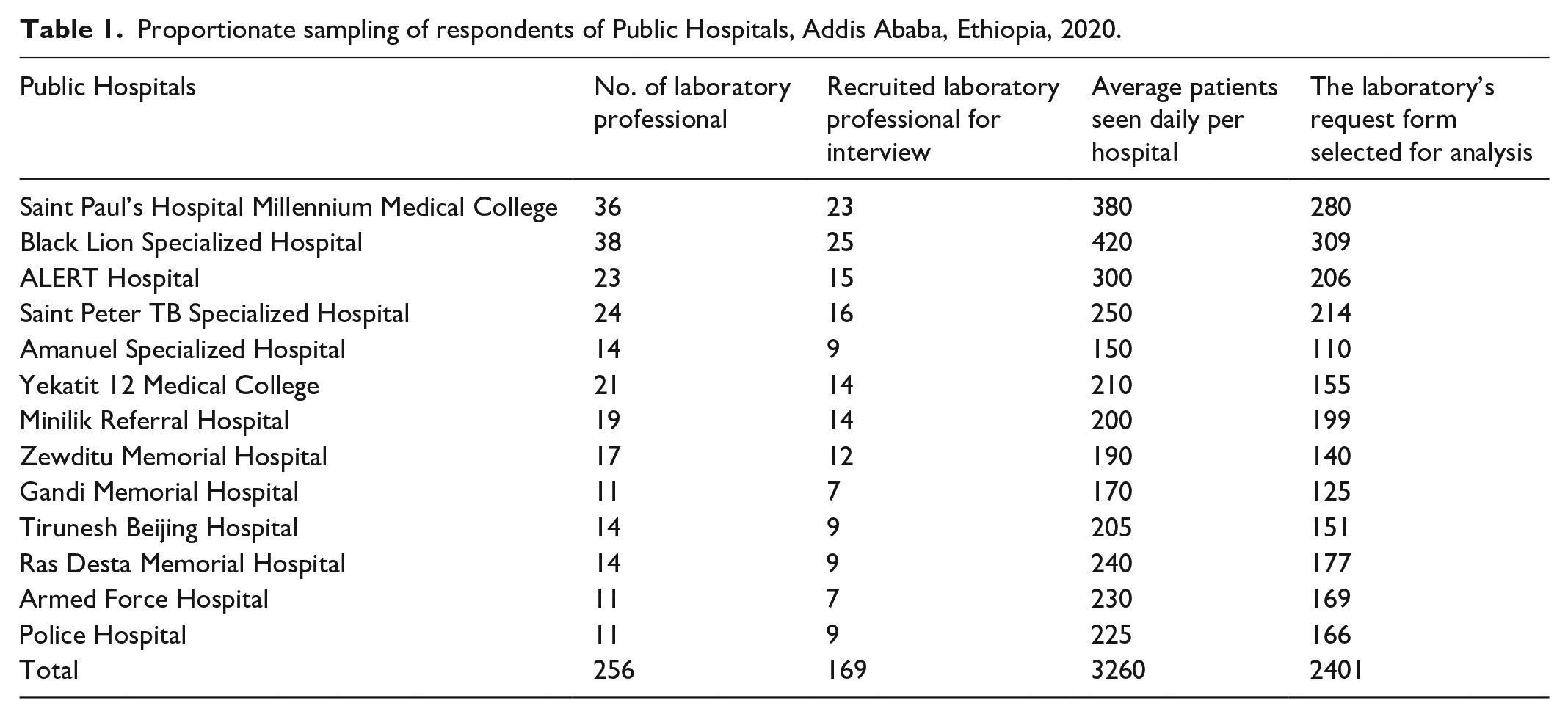
So, the final sample size for LRF was 2401 (Table 1).
Proportionate sampling of respondents of Public Hospitals, Addis Ababa, Ethiopia, 2020.
Similarly, a sample size of laboratory professionals was determined using the single population proportions formula, assuming P = 50%, level of significance = 0.05, and marginal error (d) = 5% due to a lack of previous similar studies:

(Z α/2) = z-score at 95% CI = 1.96, the formula the sample size (n) was n = 1.962*0.5*0.5/0.052 = 384.
Based on public hospitals’ profiles, the total study populations were 256, which is less than the calculated sample size, and a correction factor was done following the finite population formula (nf); hence, the estimated sample size was
nf = n/1 + n/N = 256/(1 + 1/384) = 154
Allowing 10% for non-responses, the final sample size was 169. In the same manner, as with the LRF, the number of respondents from each hospital was allocated proportionally.
Data collection procedure
After getting permission to conduct the study from the chief executives of hospitals, the data were collected in close collaboration with the health facility’s quality officer and heads of laboratory units. The data collection material was pre-tested on one public hospital to ensure its completeness, consistency, and applicability and was modified accordingly. The researchers checked at least five questionnaires per day on the spot. The LRFs were assessed prospectively in real time. In addition, the quality of the samples was assessed according to predefined criteria on the observational checklist. Quality indicators were observed in the clinical chemistry, hematology, parasitology, and serology departments. A test requisition form (TRF) was used to check points in the pre-analytical phase for laboratory errors. An organization checklist was used, which covered the quality indicator items of the TRF, sample quality, and post-analytical phase. Regarding the TRF, name, age, sex, registration number, location/ward, the requesting physician’s name and signature, clinical and diagnostic information, the date of collection, and the date of request were assessed. For monitoring the sample quality, we have checked for hemolysis, lipemia, and insufficient or inappropriate vials in a prospective manner. Furthermore, in the post-analytical phase, quality indicators such as reviews, documentation, reporting, or communication of critical results, and whether established turnaround times (TAT) were met or not were checked. A structured questionnaire was used to gather sociodemographic, educational, and training information about the staff included in the study. The principle investigators were on hand to control the overall activities and assist the data collectors.
Data quality assurance
To ensure the quality of the data, experienced professionals evaluated the data collection tool for appropriateness and overall evaluation. The questionnaire was pre-tested at ABET Hospital with more than 5% of the sample size. The results of the pre-test were not included in the final analysis. The principal investigator instructed three laboratory technologists on how to collect data. The principal investigator double-checked the questionnaire and checklist for accuracy at the end of the day.
Statistical analysis
All collected data were coded and entered into the statistical software SPSS version 23 (IBM Corporation, USA). Descriptive statistics (frequency and percentages) were calculated, and summary results were presented in the form of text and tables. Variables with a p value less than 0.25 were included in the multivariate logistic regression model after bivariate logistic regression analysis. The strength of the association between independent variables and extra-analytical errors of laboratory services was measured using an odds ratio with a 95% CI. In the final model, a p value <0.05 was considered a cutoff point.
Results
Characteristics of the study participants
In our study, 169 laboratory professionals from 13 public hospitals were included, and the response rate was 100%. The results show that we have a good cross section of staff with regards to sex, education, work experience, discipline, and refresher training. Of note is the number of males who made up 56.8% of the participants: those who had a bachelor’s degree were 106 (62.7%), those who had work experience ranging from 6 to 10 years were 66 (39.1%), and those who do not attend refresher training were 123 (72.8%) (Table 2).
Sociodemographic characteristics and attending of training and continuing education programs of laboratory professionals working in a public hospital in Addis Ababa, Ethiopia (n = 169).

Extra-analytical practices in medical laboratories
Table 3 shows 118 (69.8%) of activities have written procedures, though 75% have specific criteria for rejecting specimens. Rejection of unlabeled, contaminated, and hemolyzed samples is over 97%, with lack of training being listed as the reason for not rejecting hemolyzed samples. Workload pressure is recorded as being the major factor in failing to record rejected specimens in a logbook or notify rejected samples or requests to the responsible person. Furthermore, 49 (29%) of laboratory professionals did not collect samples according to standard procedures and 77 (45.6%) did not reject an incomplete request.
Pre-analytical variable activities reported by laboratory professionals working in a public hospital in Addis Ababa, Ethiopia (n = 169).

SOP: standard operating procedure.
Among post-analytical quality indicators, 80 (47.3%) of laboratory professionals did not verify laboratory results, and this was mainly due to workload. Likewise, 90 (53.3%) results were reported outside of the agreed TAT, and 81 (90%) of the reasons were associated with workload, meaning the report failed to meet the agreed TAT. In all, 49 (28.9%) laboratory professionals did not document and record laboratory results, but workload still accounts for more than two-thirds of the reasons. In all, 53 (31.4%) laboratory professionals said they did not communicate or alert laboratories about critical results for responsible individuals, and workload is the main reason for this critical problem. Overall, 60.3% of all errors were pre-analytical phase errors, while 39.7% were post-analytical phase errors (Tables 3 and 4).
Post-analytical variables activity reported by laboratory professionals working in a public hospital in Addis Ababa, Ethiopia (n = 169).

SOP: standard operating procedure; TAT: turnaround time.
Request form completion rates
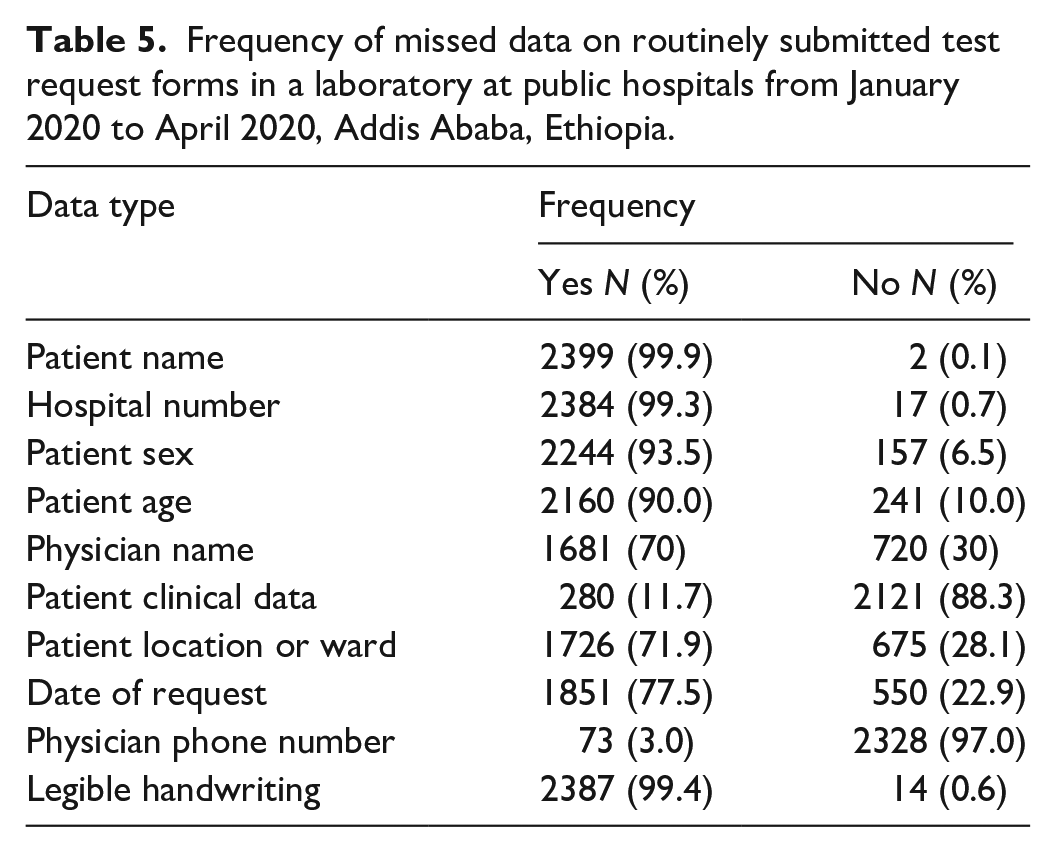
We discovered that there is no complete information (i.e., the requested forms do not have all the necessary data) on the laboratory requisition forms, such as clinical data 2121 (88.3%), physician phone 2328 (97%), physician name 720 (30%), patient location 675 (28.1%), and date of request 550 (22.9%) (Table 5).
Frequency of missed data on routinely submitted test request forms in a laboratory at public hospitals from January 2020 to April 2020, Addis Ababa, Ethiopia.
Sample quality of the pre-analytical quality indicators
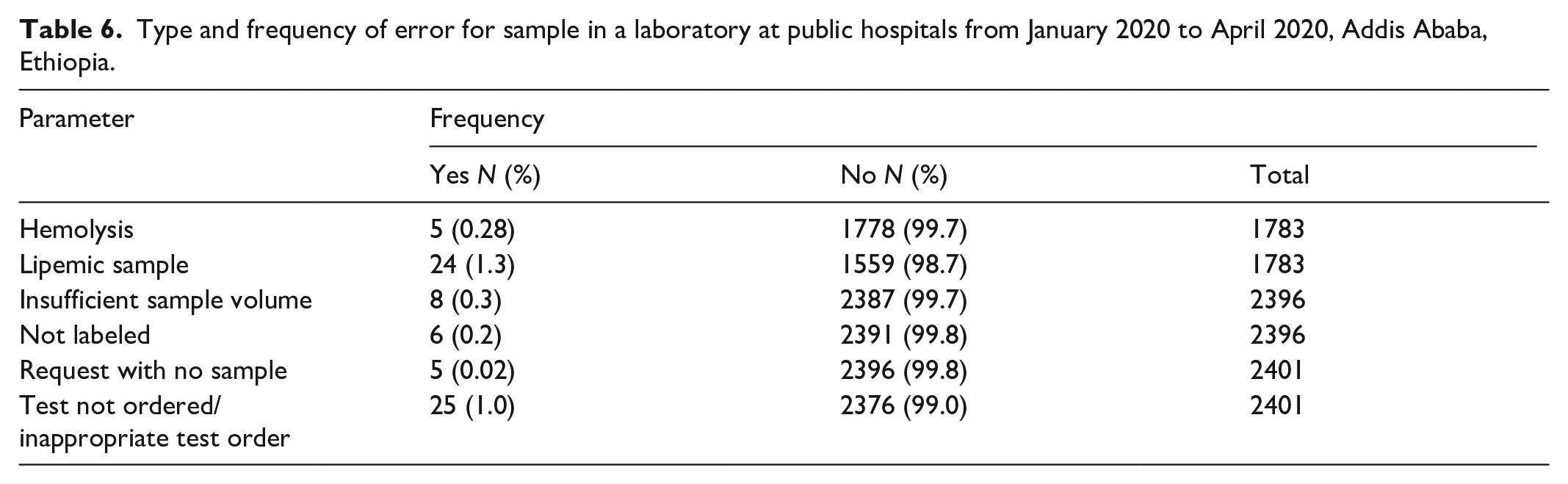
During the current study, 2396 laboratory samples were submitted to laboratories for various laboratory tests. Urine, chemistry, serology, parasitology, coagulation, and hormone specimens were received in the laboratory section with magnitudes of 298, 769, 933, 118, 197, 22, and 59, respectively. According to the current research, the most common reasons for sample rejection were lipemia 24 (1.3%), insufficient sample volume 8 (0.3%), hemolysis 5 (0.28%), and lack of labeling 5 (0.2%) (Table 6).
Type and frequency of error for sample in a laboratory at public hospitals from January 2020 to April 2020, Addis Ababa, Ethiopia.
Quality indicators of the post-analytical phase
During the post-analytical phase, various types of errors were discovered, such as the failure to record results 1741 (73.3%), the highest percentage, followed by 11 (73%) critical results that were not notified to the concerned physicians (Table 7).
Frequency of errors in the post-analytical phase of laboratories at public hospitals from January 2020 to April 2020, Addis Ababa, Ethiopia.

TAT: turnaround time.
Factors associated with extra-analytical errors in laboratory services
The findings of the logistic regression analysis revealed that the absence of written procedures for laboratory activities, less than 5 years of work experience, and diploma holders were significantly associated with additional analytical errors in laboratory services. Laboratory professionals with 1–2 years of experience were five times more likely to make extra-analytical errors than those with >10 years of experience, while laboratory professionals with 3–5 years of experience were found to be 2.67 times more likely. Diploma holders were six times more likely to make extra-analytical errors than those with a degree or master’s degree. Furthermore, laboratory professionals who did not have written procedures for laboratory activities were 2.79 times more likely to produce extra-analytical errors than those who did have written procedures for laboratory activities (Table 8).
Logistic regression analysis shows the association between extra-analytical errors, and covariates in public hospital laboratories, Addis Ababa, Ethiopia, 2020.

Significant association at α p value < 0.05.
1, shows reference category; CEP: continues education program; PLA: have written procedure for laboratory activities; RT: refreshing training; TP: training in the procedure.
Discussion
Extra-analytical errors are a common problem in the laboratory that compromise patients’ monitoring and healthcare services. And the majority of laboratory professionals work under a high workload without continuing education and training. Against this background, this study focused on determining the frequency of extra-analytical errors and their associated factors at public hospital laboratories in Addis Ababa, Ethiopia. In this study, patient name (99.9%) and hospital number (99.3%) were relatively well-documented parameters that appeared on all request forms. These findings were comparable to those from Ethiopia,15–18 India, 19 Nigeria,20,21 and Ghana. 22 It was very expected that all clinicians would give these parameters more attention than others.
This study showed that 6.5% of the studied LRFs did not describe the patient’s gender. This error rate is higher than previous reports in Nigeria 1.1% 20 and 1.2% 21 but lower than Ghana 32.7% 22 and Ethiopia 10.3% 16 and 15.7%. 17 LRFs with no age of patients were observed in 10% of all forms inspected, which was similar to previous Ethiopian studies of 11.6%. 17 These errors were higher than reported error rates in other Ethiopian studies of 0.6%, 15 and 2.1%, 18 India 1.32% 19 and Nigeria 1.2% 21 but lower than studies done in Nigeria 48.3% 20 and Ghana 25.6%. 22 Reference values for some biochemical parameters differ with gender and age, 23 underlining the need for their presence in request forms. This dissimilarity could be attributed to the workload on physicians, sample size, site of data collection, carelessness among physicians, and lack of checking by the concerned bodies, or inappropriate orientation about the effect of not complete information on LRFs on the quality of the patient result.
The physician’s phone number was missing from 97% of LRFs. The name of the physician was missing from 30% of the laboratory forms. This is higher than reported error rates in other African studies.19–21 Giving proper attention to filling out the physician’s phone number for immediate communication of critical results is crucial. The patient’s location/ward was missing in 28.1% of the request forms in this study, but the study was done in Ethiopia at the UOG Hospital 15 and at St. Paul’s Hospital, 16 Nigeria, 21 India 24 had very low with 1.8%, 3.6%, and 0.4%, respectively. Other similar studies conducted in Nigeria 20 and Ghana 22 indicated that 20.1% and 47.8% of LRFs lack location or ward, respectively. On the other hand, this study revealed that 22.9% of the forms did not have the date of the request, which was similar to studies done in Niger 21.5%, 25 and India 23.13%. 19 Whereas Ambachew et al. 15 reported a rate of error almost two times higher than the current finding, 46.9%. This result was higher than the study done at St Paul’s Hospital Millennium Medical College, 6.4%. 16
In this study, 88.3% of LRFs omitted patient clinical data, which was consistent with findings from Ethiopia (98%, 15 70.0%, 16 72.8%, 17 and India 89.25%. 19 Similar to the study conducted in Nigeria, 26 0.28% of the samples in this study were rejected due to hemolysis. Previous Ethiopian15–17 and Indian27,28 studies, however, revealed lower proportions than the current findings. These differences could be attributed to a variety of factors, including training status, sample size, workload, and sample collection, which explains the majority of the site differences.
Excessive TAT (62%), which was significantly higher than previous studies conducted in other regions of Ethiopia of 3.5% 16 and 8.6%, 15 caused errors in the post-analytical phase. Numerous factors, such as workload, a lack of training, a staffing shortage, and the absence of a location or ward in LRFs, could be the causes for this high prevalence in this study. Ninety (53.3%) of the 169 laboratory professionals surveyed did not submit results within the TAT due to workload (90%) and a lack of wards/locations (10%), respectively. In contrast, research in Hawassa 29 and Addis Ababa 2 discovered that 32.9% and 80% of study data, respectively, were not released within the predetermined TAT. This discrepancy could be due to differences in the study’s organizational structure, workload, sample size, and workflow approach.
In some cases, ineffective critical value notification or failure to provide notification within the target time frame can be fatal. 30 In this study, 15 (0.6%) of the total sample perceived critical results. In all, 11 (73%) of these were not communicated to potential physicians. Critical results can be quickly communicated to the requesting doctor by telephone number or other communication systems. Failure to hand over critical results in a clinically acceptable time frame is more important than some other errors because easily treatable patients may die. In contrast, only 15(0.48%) critical results were not communicated to the considering physicians in a study conducted at the UOG (University of Gondar) Hospital. 15 This might be due to a lack of awareness among laboratory staff, unwritten parameters on LRFs such as patient address, ward or location, and attending physicians’ name and phone number, or the absence of a communication system. In this study, 53 (31.4%) of the 169 laboratory professionals polled did not notify or communicate critical results due to workload and a lack of a communication system (83% and 17%, respectively). So a poor extra-analytical phase management system directly affects the analytical phase of laboratory services as well as patients and healthcare services.
Sushma et al. 31 found that providing appropriate training could reduce the frequency of error occurrence. In this study, 84 (49.5%) of 169 laboratory professionals interviewed said they had not received refresher training before performing the procedures. The lack of training could be one of the causes of observed errors. This is confirmed by a study done in India, which indicated that after formal training of laboratory personnel, and medical and nursing staff, pre- and post-analytical errors significantly decreased. 28 Well-documented standard operating procedures (SOPs) help laboratories establish a well-functioning quality management system and client-centered services that contribute to healthcare services.32,33 This study discovered that more than one-fourth of laboratories lacked a written procedure for every laboratory task. As a result, patients and healthcare services, as well as the additional analytical phase of laboratory services, are directly impacted by the lack of procedures for each laboratory activity. Moreover, extra-analytical errors in laboratory services were statistically significantly associated with the absence of written protocols for laboratory activities.
Limitation
There were some additional analytical steps that were not covered, including drug intake by the patient, diet, timing of sample collection, and tourniquet application. Likewise, since the data were collected from 13 different hospitals, slight personal differences in the criteria for the assessment of sample quality or the established TAT might be another drawback.
Conclusion
This study discovered errors in the pre-analytical (60.3%) and post-analytical (39.7%) phases, and none of the requisition papers had complete data. The absence of written procedures for laboratory activities, a lack of work experience, a high workload, and not having a first degree or higher were potentially associated with extra-analytical errors in the laboratory. To minimize extra-analytical errors, it is critical to have an adequate number of laboratory professionals, educate or inform clinicians to fill out all necessary patient information on LRF, maintain a well-documented SOP in the working area, and apply rejection practice to incomplete LRFs.
Supplemental Material
sj-docx-1-smo-10.1177_20503121221148062 – Supplemental material for Magnitude of extra-analytical errors and associated factors in medical laboratories in thirteen Public Hospitals in Addis Ababa, Ethiopia
Supplemental material, sj-docx-1-smo-10.1177_20503121221148062 for Magnitude of extra-analytical errors and associated factors in medical laboratories in thirteen Public Hospitals in Addis Ababa, Ethiopia by Cheru Degfe and Ousman Mohammed in SAGE Open Medicine
Footnotes
Acknowledgements
The authors greatly acknowledge Addis Ababa University for funding the research. We wish to extend our deep appreciation to all laboratory professionals who are working at the selected public hospitals for their unreserved cooperation during data collection.
Authors’ contribution
Both authors made significant contributions to the manuscript. Both authors have read and given their final approval of the version of the manuscript submitted for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical consideration
The study was approved by the departmental research and ethical review committee (DRERC) of the department of medical laboratory science with protocol number DRERC/482/19/MLS and the research ethics review committees of the Addis Ababa Health Bureau and each of the five federal public hospitals before the study was commenced. Moreover, written informed consent was obtained from all subjects before the study. During data collection, names of professionals and health facilities were not mentioned. And each participant was informed about the purpose of the study, the right to refuse to participate in the study, and the confidentiality of the information gathered.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Addis Ababa university. However, they have no role in manuscript writing, editing, approval, or decision to publish.
Informed consent
Written informed consent for patient information to be published in this article was obtained from all subjects.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
