Abstract
Objective:
Changes in the blood cell function, metabolism, and the coagulation system were associated with diabetes mellitus. This study aimed to determine the magnitude and associated factors of hematological abnormalities in adults with type 1 diabetes mellitus.
Methods:
A total of 204 medical charts of adults with type 1 diabetes mellitus who registered for follow-up at the University of Gondar Comprehensive Specialized Hospital were reviewed from June to August 2021. Data were collected using a structured data extraction checklist. To identify factors associated with hematological abnormalities, both bivariate and multivariate logistic regression analyses were done. Statistical significance was defined as a p-value of <0.05.
Results:
The overall magnitude of leukocytosis and anemia in adults with type 1 diabetes mellitus was 76.0% (95% confidence interval: 70.07–81.89) and 30.90% (95% confidence interval: 24.49–37.28), respectively. Neutrophilia and lymphocytosis were the common white blood cell abnormalities detected in 53.43% (95% confidence interval: 46.53–60.33) and 43.63% (95% confidence interval: 36.76–50.49) of the patients, respectively. Besides, thrombocytosis and thrombocytopenia were observed in 5.4% (95% confidence interval: 2.27–8.52) and 10.3% (95% confidence interval: 6.09–14.5) of the patients, respectively. Only being male (adjusted odds ratio = 2.28 (95% confidence interval: 1.46–5.29)) and duration of diabetes mellitus (⩾3 years) (adjusted odds ratio = 8.41 (95% confidence interval: 2.49–28.29)) were significantly associated with anemia and leukocytosis, respectively.
Conclusion:
Hematological abnormalities, particularly anemia and leukocytosis, are common in patients with type 1 diabetes mellitus. Therefore, preventive and control strategies for hematological abnormalities are essential in patients with type 1 diabetes mellitus particularly for male and patients with a long duration on diabetes mellitus to reduce the burden and related complications.
Introduction
Diabetes mellitus (DM) is a metabolic condition marked by persistent hyperglycemia as well as impaired carbohydrate, lipid, and protein metabolism. 1 It is a global public health issue that comes with a higher risk of vascular consequences from a variety of diseases, including metabolic, cellular, and blood abnormalities. 2 According to the 2019 International Diabetes Federation (IDF) study, there are approximately 463 million adults living with diabetes globally, with 19 million from Africa. Type 1 diabetes mellitus (T1DM) affects an estimated 1.1 million children and adolescents worldwide in 2019. 3
T1DM is an autoimmune disease that results from the destruction of beta cell in pancreatic islets leading to an increase in blood sugar levels. 4 It is the leading cause of DM in childhood, but it can occur at any age. 3 It develops through activation of the immune system against beta cell antigens and the initiation of proinflammatory responses. 4 Persistent hyperglycemia in patients with DM is associated with long-term damage and dysfunction of organs such as eyes, kidneys, nerves, heart, and blood vessels.1,5 An imbalance between hyperglycemic cell damage and defective endogenous reparative mechanisms bears the brunt of its multi-organ consequences. DM is characterized by inflammation and abnormalities in the hematopoietic compartments. 6
DM is directly associated with changes in the hematological parameters that affect the red blood cells (RBCs), white blood cells (WBCs), and the coagulation factors. 7 The effects of hyperglycemia in RBCs include change in the erythrocyte membrane, disturbance in the oxygen binding of hemoglobin (Hgb), and modification in mechanical characteristics. 8
Anemia is a common and under-recognized complication in patients with T1DM. 9 One in five (∼20%) patients with T1DM have Hgb levels below the normal reference range. 10 In Ethiopia, the pooled prevalence of anemia among patients with T1DM was 16.78%, according to a recent systematic review study. 11 DM patients with anemia may have an increased risk of adverse outcomes due to diabetic retinopathy, neuropathy, nephropathy, and cardiovascular disease. 12 The most well-known cause of anemia in DM is reduced erythropoietin production due to malfunction of erythropoietin-producing cells in the cortical interstitium, which can exacerbate microvascular problems.13,14 Other causes of anemia include systemic inflammation,15,16 suppression of erythropoietin release, medications, renal impairment, altered iron metabolism, and hyperglycemia.17,18
Studies revealed that patients with T1DM had higher neutrophil counts and thus correlate with an increased risk of vascular disease.19,20 Immoderate enlistment of neutrophils from the bone marrow and the return of marginated cells to the circulatory pond has been postulated for the increment of circulating neutrophils. 21 Increased mobilization of WBCs and secretion of proinflammatory cytokines, adhesion molecules, and oxidative stress are common in diabetic cardiac injury.22,23 On the contrary, adults newly diagnosed with T1DM had lower total WBC count and fewer neutrophil, basophil, monocyte, and lymphocyte counts. This could be due to abnormal neutrophil yield and maturation, peripheral consumption or damage, and tissue detainment. 24
Elevated platelet counts including increased mean platelet volume (MPV), platelet distribution width and reticulated platelets, as well as platelet dysfunction frequently occur in T1DM.25,26 Circulating platelets in patients with DM are hyperreactive with increased adhesion, activation, and aggregation activity. 27 However, thrombocytopenia in T1DM increases bruising or bleeding during insulin injections and finger-pricking.
Hematological abnormalities are a common complication and under-recognized burden in patients with DM. 7 Early diagnosis and treatment of these abnormalities including anemia helps to decrease morbidity and mortality and improves their quality of life in patients with DM. 11 However, there are limited studies that have assessed hematological abnormalities particularly among adults with T1DM in Ethiopia. Therefore, the study aimed to determine the magnitude of hematological abnormalities and their associated factors in adults with T1DM attending the University of Gondar Comprehensive Specialized Hospital.
Methods and materials
Study design and period
A retrospective cross-sectional study was conducted from June to August 2021.
Study area
This study was conducted at the University of Gondar Comprehensive Specialized Hospital. The hospital is located in Gondar town in Amhara regional state, which is located 743 km northwest of the capital Addis Ababa, Ethiopia. The hospital serves the people of Gondar and the neighboring areas. The town is situated at a height of 2133 m above sea level. The DM clinic is located inside the outpatient department (OPD) and constitute a large number of patients attending in the follow-up clinic. According to data recorded from the hospital chronic OPD, up to 2000 to 4000 patients with T1DM visit the DM clinic each year. In chronic illness clinic, about 20 health care providers, that is, 10 nurses, 6 general practitioner, 2 senior specialists, and 2 residents, work together to improve the outcome of patients with DM.
Population
All patients with T1DM who were registered in the OPD of the University of Gondar Comprehensive Specialized Hospital were used as source population. Patients with T1DM who had a registration list with each follow-up date in their medical records at the OPD clinic at the time of data collection and who fulfill the inclusion criteria were the study populations.
Eligibility criteria
The study included all medical records of patients with T1DM above the age of 18 for at least 3 months prior to the data collection and who had regular follow-up and comprehensive data. Meanwhile, patients with T1DM with a known hematological disorder, those who had received a blood transfusion in the previous 3 months, and pregnant women were excluded.
Sample size determination and sampling technique
The sample size calculation was not applicable because the study used a retrospective cross-sectional technique. This study included a total of 204 adults with T1DM who had a follow-up data for at least 3 months prior to the data collection period and had complete information for key factors.
Data collection tools
Data collection was carried out using a structured data extraction checklist. The data sources were the patient admission form, follow-up card, and DM registration book. The data extraction sheet consists of sex, age, body mass index (BMI), duration of DM, fasting blood glucose (FBS) level, type of medication, and hematological parameters. Hematological parameters including RBC, Hgb, hematocrit (Hct), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), mean cell volume (MCV), WBC, WBC differential count, and platelet count were collected. Data were collected by one supervisor and trained nurses. The data collectors and supervisors received training on how to retrieve records using the data extraction sheet.
Operational definitions
Anemia: According to World Health Organization (WHO) 28 altitude adjusted Hgb values <12 g/dL for females and <13 g/dL for males. According to WHO standard value, leucopenia, neutropenia, and lymphopenia is defined are total WBC count <4000 cells/μL, neutrophil count <1500 cell/μL, and lymphocyte count <600 cell/μL, respectively. 29 Leukocytosis, neutrophilia, and lymphocytosis are defined as a total WBC count above 11,000 cell/μL, neutrophil count > 7000 cell/μL, and lymphocyte count > 450 cell/μL, respectively.30,31 According to the WHO, underweight is defined as a BMI of <18.5 kg/m2, normal weight is defined as a BMI of between 18.5 and 24.9 kg/m2, overweight is defined as a BMI of 25 and 29.9 kg/m2, and obesity is defined as a BMI of ⩾30 kg/m2. 32 Hypoglycemia is defined as a blood glucose level of ⩽70 mg/dL. 33
Statistical analysis
Epi-data version 4.6.0.0 was used to enter the data. The data were then analyzed using the Statistical Package for Social Science (SPSS) version 23 software. The data were summarized using descriptive statistics. The normality of the data was checked by performing Shapiro–Wilk test. To assess the strength of the association between the dependent and independent variables, bivariable binary logistic regression tests were done. Variables with a value of <0.25 in the bivariate analysis were fitted into the multivariate logistic regression to adjust the confounding factors. The strength of association was determined by calculating crude odds ratio (COR) and adjusted odds ratio (AOR) with the corresponding 95% confidence interval (CI). p-value less than 0.05 was considered statistically significant.
Ethical consideration
The study was carried out after the Ethical Review Committee of the University of Gondar School of Biomedical and Laboratory Sciences, College of Medicine and Health Sciences, gave ethical clearance. A letter of approval was also provided by the head clinical director of the University of Gondar Specialized Referral Hospital. A consent to participate in the study was waived because it was a retrospective study from medical records. To maintain confidentiality, the data collected from the participants was coded.
Results
Sociodemographic and some clinical characteristics
A total of 204 medical charts of patients with T1DM were reviewed. The mean ± SD age of the patients were 39.451 ± 15.4 years, with the majority of age group being 18–25 years. Of the participants, more than half (104; 51.0%) of them were females. Out of the study participants, about 120 (58.8%) were urban dwellers. The mean (± SD) FBS level of the patients was 172.4 ± 83.26 mg/dL with 20 (9.8%) of the patients had evidence of hypoglycemia. Regarding the duration, 58 (28.4%) patients with T1DM had ⩾3 years duration of illness since diagnosis (Table 1).
Sociodemographic and clinical characteristics of T1DM patients attending the outpatient clinic of University of Gondar Comprehensive Specialized Hospital, Northwest Ethiopia, 2021.

DM: diabetes mellitus; BMI: body mass index; FBS: fasting blood glucose; INH: inhaled human insulin.
Laboratory findings
The altitude adjusted Hgb level for all patients with T1DM ranges from 4.2 to 18.2 g/dL, with a mean value of 14.3 ± 2.24 g/dL. The mean (± SD) value of WBC count was 12.86 ± 3.32 × 103 cells/μL with a range of 2–23 × 103 cells/μL. Furthermore, the range of platelet count was 94–650 × 103 cells/μL, with a mean of 269.59 ± 99.76 × 103 cells/μL (Table 2).
Hematological parameters of adults with T1DM attending at the University of Gondar Comprehensive Specialized Hospital, Northwest Ethiopia.

IQR: interquartile range; RBC: red blood cell; MCV: mean corpuscular volume; Hgb: hemoglobin; MCHC: mean corpuscular hemoglobin concentration; Hct: hematocrit; MCH: mean corpuscular hemoglobin; SD: standard deviation; WBC: white blood cell.
Magnitude of hematological abnormalities
The overall magnitude of anemia among adults with T1DM was 30.90% (95% CI: 24.49–37.28); from them 42 (66.7%) were male patients (Figure 1). Thrombocytosis was detected in 5.4% (95% CI: 2.27–8.52), whereas thrombocytopenia was observed in 10.3% (95% CI: 6.09–14.5) of the patients with T1DM. The common hematological abnormalities in adults with T1DM were leukocytosis which was observed in 76.0% (95% CI: 70.07–81.89) of the patients. Besides, neutrophilia and lymphocytosis were detected in 53.43% (95% CI: 46.53–60.33) and 43.63% (95% CI: 36.76–50.49) of the patients (Figure 2).

The distribution of anemia by sex among adults with T1DM at the University of Gondar Comprehensive Specialized Hospital.

White blood cell abnormalities among adults with T1DM at the University of Gondar Comprehensive Specialized Hospital.
Factors associated with anemia
In this study, binary logistic regression model was used for the analysis of factors associated with anemia in adults with T1DM. In the bivariate logistic regression, variables that had a p-value < 0.2 were sex, age, duration of DM, BMI, and FBS. Only being male (COR = 2.86 (95% CI: 1.54–5.33)) was significantly associated with anemia in adults with T1DM in the bivariate logistic regression. Also, in the multivariate logistic regression analysis only being male (AOR = 2.28 (95% CI: 1.46–5.29)) was significantly associated with anemia in patients with T1DM (Table 3).
Bivariable and multivariable analysis of predator variables of anemia among adults with T1DM attending at the University of Gondar Comprehensive Specialized Hospital, Northwest Ethiopia.

Ref: reference category; BMI: body mass index; DM: diabetes mellitus; AOR: adjusted odd ratio; COR: crude odd ratio; CI: confidence interval.
Statistically significant at p < 0.05.
Factors associated with leukocytosis
To examine factors associated with leukocytosis in patients with T1DM, bivariate and multivariate logistic regression analyses were done. Only duration of DM ⩾ 3 years (AOR = 8.41 (95% CI: 2.49–28.29)) was statistically associated with leukocytosis (Table 4).
Logistic regression analysis of leukocytosis and associated variables among adults with T1DM attending at the University of Gondar Comprehensive Specialized Hospital, Northwest Ethiopia.
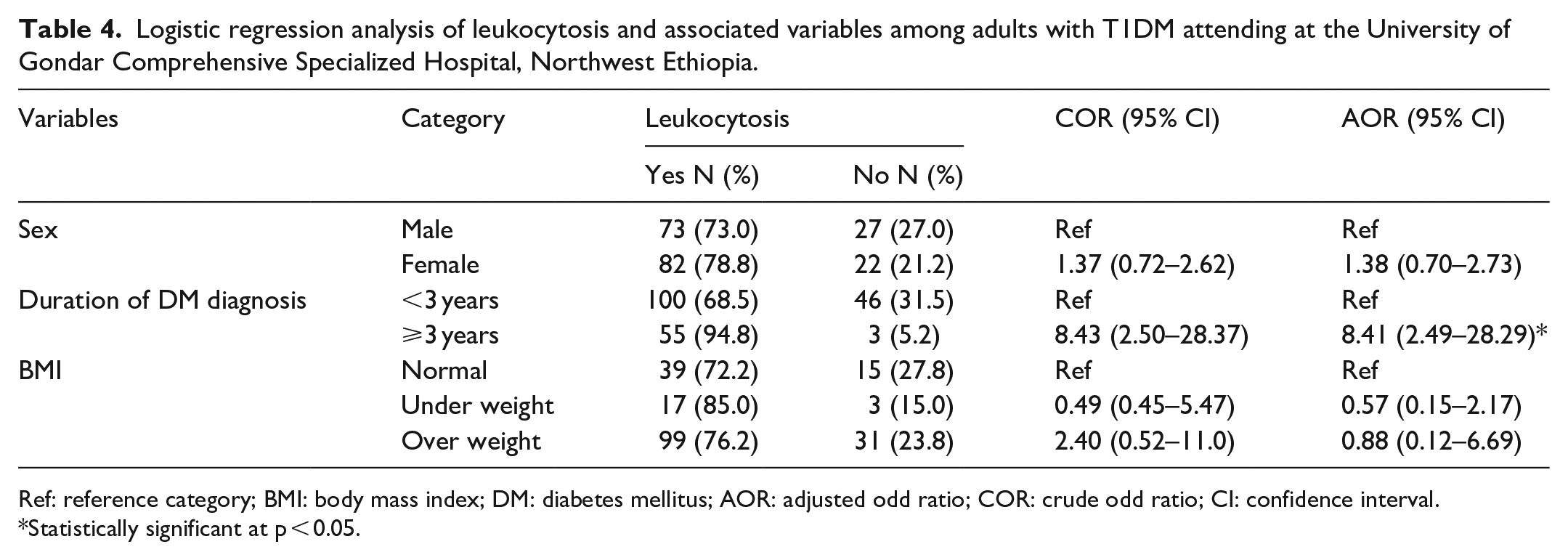
Ref: reference category; BMI: body mass index; DM: diabetes mellitus; AOR: adjusted odd ratio; COR: crude odd ratio; CI: confidence interval.
Statistically significant at p < 0.05.
Discussion
In the adult population, hematological changes, such as RBCs, WBCs, and platelet function and morphology changes, are common in T1DM, demonstrating their tight link with different components of metabolic syndrome, such as insulin resistance.24,34,35
The most frequent hematological abnormalities in the present study were leukocytosis, which was detected in 76.0% (95% CI: 70.07–81.89) of the patients. Although the percentage of patients with leukocytosis is not stated, previous research has shown that leukocytosis is a common symptom in patients with T1DM. Findings from studies conducted in Ethiopia, 36 Nigeria, 37 and Bangladesh 38 confirm that the mean value of total WBC count was higher in patients with DM than in non-diabetic controls. The increase in WBC count mainly reflects the elevated neutrophil and lymphocyte count in these studies. The reason for the high WBC count could be related to the increased oxidative stress caused by the high levels of hyperglycemia in patients with DM. In the state of hyperglycemia, angiotensin and cytokines stimulate the formation of polymorphonuclear and mononuclear WBCs. 39
In the current study, the magnitude of anemia in patients with T1DM was 30.9% (95% CI: 24.49–37.28). This indicates anemia as a moderate public health problem in adults with T1DM in the study area according to the WHO 28 anemia classification for public health importance (20.0–39.9%). The results of this study are similar with those of the previous study conducted at the General Hospital of Gelemso (30.2%) 40 and Debre Tabor General Hospital (29.1%) 41 and Egypt (36%), 42 but higher than the studies performed at Dessie Referral Hospital (16.3%), 43 Fenote Selam Hospital (21.9%), 44 and current systematic review in Ethiopia (16.78%) 45 and Australia (14%). 46 The reason might be only patients with T1DM aged 18 years and older are included in this study; therefore, with advanced age, decreased renal function, inflammation, bone marrow suppression, and malnutrition may occur which can increase the development of anemia. 47 However, this prevalence was lower than the finding from Saudi Arabia (55.5%). 16 The possible explanation for the difference could be attributed to variations in the age of the study population, nutritional status, and the study design.
Thrombocytopenia was the other hematological abnormality in the current study which was observed in 10.3% (95% CI: 6.09–14.5) of patients. Similarly, a Taiwan study showed that DM increases the severity of thrombocytopenia in dengue-infected patients. 48 This can easily cause bruising or bleeding during insulin injections. However, thrombocytosis was detected in 5.4% (95% CI: 2.27–8.52). This is similar to a study done in Ethiopia which report that platelet counts were significantly higher in patients with DM than in the controls. 36 Besides, studies have shown that there is no significant difference in the mean platelet count between children with T1DM and healthy controls.25,35 Possible mechanisms for the increased platelet count in DM are related to hyperglycemia, insulin deficiency and resistance, and associated metabolic cellular abnormalities and therefore increase the risk of thrombotic and vascular complications. 49
Regarding factors associated with different hematological abnormalities, being male was 2.28 times more likely to develop anemia as compared to female (AOR = 2.28 (95% CI: 1.46–5.29)). The result is comparable with the studies conducted at Gelemso General Hospital 40 and current systematic review in Ethiopia. 11 The possible reason could be that male patients with DM are more commonly affected by low testosterone levels and hypo-gonadotropic hypogonadism than females. Thus, the low testosterone level reduces the production of RBCs and may contribute to anemia. 50 In the present study, the duration of DM (⩾3 years) was significantly associated with high WBC count (AOR = 8.41 (95% CI: 2.49–28.29)). Similar to this study, previous study found the relationship between longer duration of diabetes and a high WBC count. 51 Clinically elevated WBC counts are observed in DM patients with a long duration of disease, thus may relate to the susceptibility of patients with DM to infection. 52
Limitations of the study
The study has some limitations. The data in this study may have been inadequate, inconclusive, or inaccurate because it was based on secondary data from routine medical records. Data on several potentially key predictor variables, such as the patient’s behavioral features and other comorbidities, were not analyzed, which could potentially limit the study findings. Furthermore, the study may lack representativeness due to inadequate sample size and we cannot report the cause-and-effect relationship of hematological abnormalities and microvascular complications due to the cross-sectional nature of the design.
Conclusion
This finding indicates high magnitude of hematological abnormalities of adults with T1DM including leukocytosis, neutrophilia, lymphocytosis, anemia, and platelet disorders (thrombocytopenia and thrombocytosis). Anemia is moderate public health problem among adults with T1DM in the current study. Moreover, hematological abnormalities were associated with male sex and duration of diabetes of the patients. Therefore, appropriate prevention and intervention strategies are required to halt the burden and microvascular complications related to hematological abnormalities.
Supplemental Material
sj-docx-1-smo-10.1177_20503121221094212 – Supplemental material for Hematological abnormalities among adults with type 1 diabetes mellitus at the University of Gondar Comprehensive Specialized Hospital
Supplemental material, sj-docx-1-smo-10.1177_20503121221094212 for Hematological abnormalities among adults with type 1 diabetes mellitus at the University of Gondar Comprehensive Specialized Hospital by Solomon Getawa and Tiruneh Adane in SAGE Open Medicine
Footnotes
Acknowledgements
We would like to thank the staff members of the University of Gondar Comprehensive Specialized Hospital chronic illness clinic for their cooperation during data collection.
Author contributions
All authors contributed significantly to the conceptualization and design, data acquisition, data analysis, and interpretation, as well as drafting the initial manuscript and critically reviewing it for final approval of the version to be submitted. All authors agreed to submit to the journal and to take full responsibility for the work.
Availability of data and materials
The data sets used and/or analyzed during the study are available from the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study was conducted after approval by the Ethical Review Committee of School of Biomedical and Laboratory Sciences, College of Medicine and Health Sciences, the University of Gondar Ref. No: SBMLS/2438/12. Also, a letter of permission was granted from the University of Gondar Specialized Referral Hospital chief clinical director.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Since it was a retrospective study from medical records, a written informed consent to participate in the study was waived off by the ethical review committee. The information obtained from the participants were coded to ensure confidentiality.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
