Abstract
Objectives:
Despite breast cancer treatment outcomes being relatively poor or heterogeneous among breast cancer patients, there was a paucity of data in the African settings, especially in Kenya. Hence, this study aimed to determine treatment outcomes among breast cancer patients at Kitui Referral Hospital.
Methods:
A hospital-based retrospective cohort study design was conducted among adult patients with breast cancer. All eligible breast cancer patients undergoing treatment from January 2015 to June 2020 in the study setting were included. Hence, a total of 116 breast cancer patients’ medical records were involved in the study. Patients’ medical records were retrospectively reviewed using a predesigned data abstraction tool. The data were entered, cleaned, and analyzed using SPSS (Statistical Package for Social Sciences) version 26 software. Descriptive analysis—such as percentage, frequency, mean, and figures—was used to present the data. Kaplan–Meier survival analysis was used to estimate the mean survival estimate across different variables. A Cox regression analysis was employed to determine factors associated with mortality.
Results:
The study showed that the overall survival and mortality rate was 62.9% (73) and 37.1% (43), respectively. The regression analysis showed that patients who had an advanced stage of disease had a 3.82 times risk of dying (crude hazard ratio= 3.82, 95% confidence interval = 1.5–9.8) than an early stage of the disease. Besides, patients with distant metastasis had 4.4 times more hazards of dying than (crude hazard ratio = 4.4, 95% confidence interval = 2.1–9.4) their counterparts.
Conclusion:
The treatment outcome of breast cancer patients was poor, and its overall mortality among breast cancer patients was higher in the study setting. In the multivariate Cox regression analysis, the tumor size was the only statistically significant predictor of mortality among breast cancer patients. Stakeholders at each stage should, therefore, prepare a relevant strategy to improve treatment outcomes.
Introduction
Breast cancer incidence and mortality are rising, being the second prevalent cancer type and the fourth-ranked deadly cancer. 1 Till the end of 2020, there were a total of 7.8 million breast cancer cases worldwide. 2 Although the prevalence is the highest in developed nations, breast cancer-related death is greater in developing countries, 2 with the highest burden in Sub-Saharan Africa. 3 In Kenya, it is the third deadly cancer next to esophageal and cervical cancer, which accounted for 9.9% of cancer death.1,4 In addition, breast cancer is associated with a considerable economic impact 5 and poor quality of life. 6
Various treatment modalities involving chemotherapy, radiation, surgery, or a combination of these are used to manage breast cancer depending on the tumor stage and biology. Breast cancer care has been shown to improve overall survival, disease-free survival, complete remission, and reduce morbidity and mortality.7,8 The number of cancer survivors continues to increase due to the advancement of early detection and treatment. 9 Juliane Farthmann et al. 10 reported that health-related quality of life and global health status increased 6 months after therapy.
Despite those significant benefits, breast cancer treatment outcome is poor.11,12 The condition is worse in developing countries due to the scarcity of healthcare resources and disparity in treatment and healthcare facilities. 13 In addition, patients seek care lately during the advanced stage, which reduces the overall survival. 14 The chance of survival is negligible with palliative care in metastatic breast cancer. 15 In addition, triple-negative breast cancer remains a challenge with no current target therapy and poor prognosis. 14 Although the Kenya National Cancer Screening Guideline promotes early detection, a previous study revealed that more than half of breast cancer patients were diagnosed at an advanced stage. 14 Little is known regarding the treatment outcome of breast cancer patients. 16 Hence, this study aimed to determine treatment outcomes and associated factors among breast cancer patients at Kitui Referral Hospital.
Methods
Study design, setting, and period
The study was conducted using a hospital-based retrospective cohort design among adult patients with breast cancer at Kitui Referral Hospital between November 2020 and January 2021. Kitui Referral Hospital was started in 1984 and upgraded to a county level referral center. It serves eight constituencies in Kitui County. The county hospital offers various treatment options in cancer management, including surgery and chemotherapy.
Study population
All adult breast cancer patients treated at Kitui Referral Hospital from January 2015 to January 2020 were eligible to the study population.
Eligibility criteria
Inclusion criteria
Adult patients (⩾18 years) with a histologically confirmed breast cancer diagnosis and treated at Kitui Referral Hospital.
Exclusion criteria
Breast cancer patients who were transferred for clinical management outside Kitui Referral Hospital.
Incomplete medical records.
Sampling technique
All eligible breast cancer patients undergoing treatment from January 2015 to January 2020 in the study setting were included. Hence, a total of 116 breast cancer patients were involved in the study.
Data collection tool and procedure
A data abstraction tool was prepared based on the standard reporting parameters of cancer treatment outcomes and previous research findings. The tool consisted of sociodemographics, clinical characteristics, and treatment outcome parameters. Then, the medical records of eligible patients were retrospectively reviewed from the day of diagnosis to the data collection time. Time from the date of cancer diagnosis to date of cancer-related death or last follow-up, time from date of the first radiographic metastasis seen to date of cancer-related death or last follow-up, and time from the date of primary cancer diagnosis to the first radiographic metastasis seen were also collected from patients’ medical records. The clinical outcome was then assessed.
Data quality control
A pretest was done in 5% of the sample size to ensure the reliability and validity of the data collection tool at Kitui Referral Hospital. The result of the pretest was used to make necessary modifications before the actual data collection.
Statistical analysis
The data were entered, cleaned, and analyzed using SPSS version 26 software. Percentage, frequency, mean, and figures were used to present the data. The Kaplan–Meier survival analysis was used to estimate the mean survival estimate across different variables. A Cox regression analysis was also employed to determine factors associated with mortality.
Operational definition of terms
Cancer-specific survival: the time period from the date of cancer diagnosis until the date of cancer-related death or last follow-up.
Cancer-specific survival after metastasis: the time interval from the date of the first radiographic metastasis to the date of cancer-related death or last follow-up.
Complete remission: no evidence of the disease after treatment using a repeat scanning.
Metastasis-free survival: the time period from the date of cancer diagnosis to the first radiographic metastasis.
Non-response: a failure to achieve partial remission.
Partial remission: a reduction in tumor volume of at least 50% compared to the imaging before the commencement of treatment.
Progressive disease: an increased tumor size despite treatment.
Results
Sociodemographic characteristics of the patients
A total of 116 adult breast cancer patients fulfilled the eligibility criteria. More than half of (55.2%, 64) of the study participants were below 60 years, and their mean age was 56 ± 15.75 years. In addition, more than half (61.2%, 71) of the study participants were married, 49.1% (57) of them had tertiary education, and 52.6% (61) of them were dependent on farming. Furthermore, 62% (72) of the study participants had no family history of cancer (Table 1).
Sociodemographic characteristic of breast cancer patients.

Clinical characteristics of breast cancer patients
Out of 116 patients, 55.2% (64) of them had metastasis. Of which, 22.6% of the patients had metastasis to the lungs, followed by bone and multiple metastases to the lung (13.8%) and bone (13.8%). Based on histopathology characteristics, most of the patients (97.4%, 113) had invasive ductal breast carcinoma. However, lobular carcinoma (0.9%, 1) and an unknown histological type of cancer (1.7%, 2) constituted the lowest percentage. In addition, 42.2% (49) of patients were clinically diagnosed with stage III breast cancer, and 5.2% (6) of them had unknown status (Figure 1).

Stage of breast cancer patients during diagnosis.
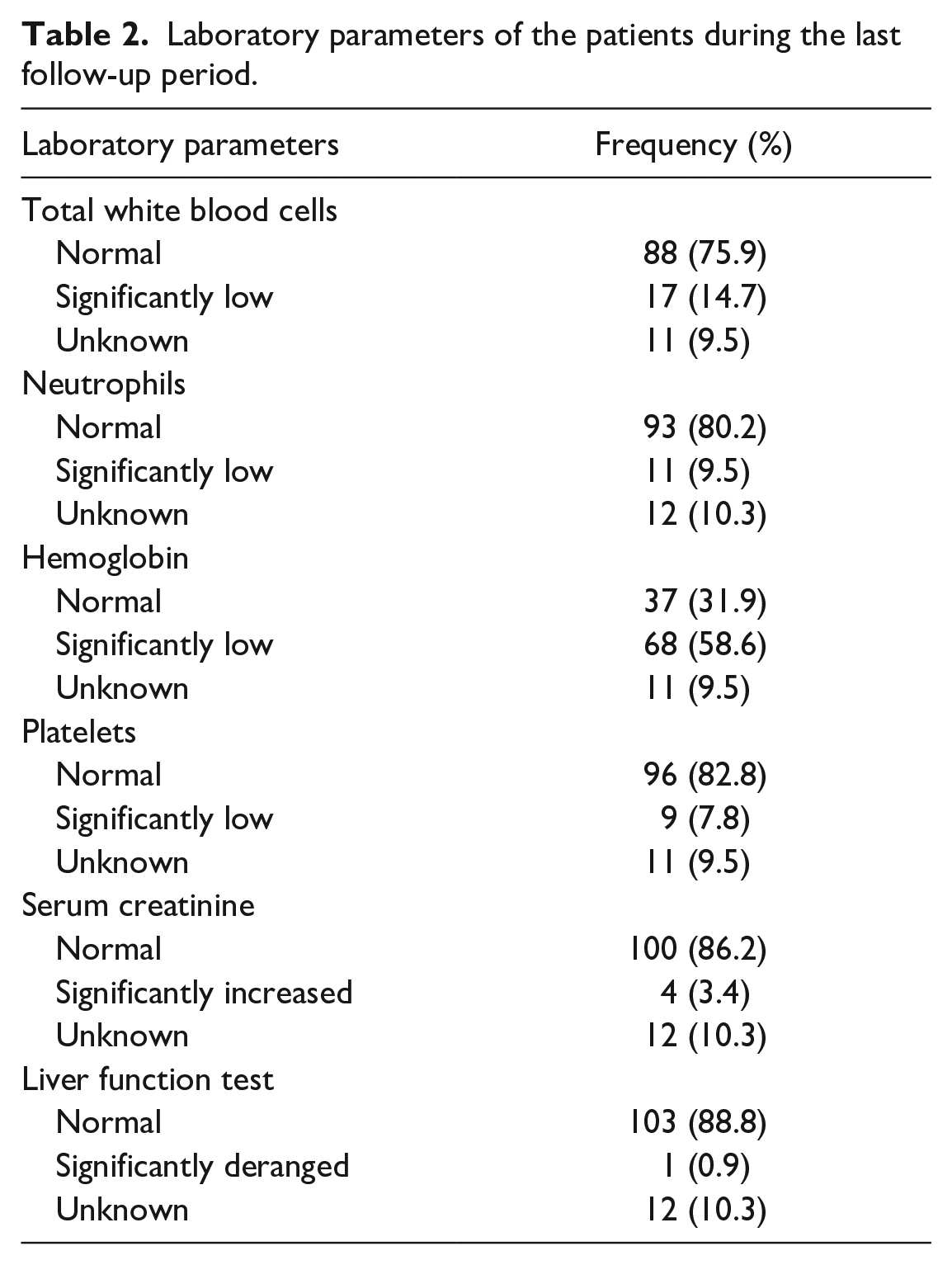
The majority of the patients had normal white blood cell, neutrophils, and platelets counts, and serum creatinine and liver function tests during the last follow-up period. However, more than half of the patients (58.6%, 68) had lower hemoglobin levels, as indicated in Table 2.
Laboratory parameters of the patients during the last follow-up period.
A relatively higher percentage of patients were tested positive for estrogen receptor (41.4%, 48), negative for progesterone receptor (41.4%, 48), and negative for human epidermal growth factor receptor 2 (HER2) (49.1%, 57). In addition, 41.01% (48) and 31% (36) of patients expressed progesterone and HER2 receptor-positive, respectively. Furthermore, 30.2% (35) and 14.7% (17) of the patients revealed triple-negative and triple-positive to hormone receptors, respectively (Figure 2).

Hormone receptor status among breast cancer patients during diagnosis.
Treatment regimens used for breast cancer patients
Various modes of therapy were used to treat breast cancer patients. Of these, a combination of surgery, chemotherapy, and hormonal therapy was the most common mode of treatment (45.7%, 53) employed, while hormonal therapy alone (0.9%, 1) and both surgery and chemotherapy (0.9%, 1) were the least treatment modalities used (Table 3). 4.3% (5) and 9.5% (11) of patients did not get cancer treatment and got symptomatic management, respectively. The frequently used combination chemotherapy was doxorubicin and cyclophosphamide (AC) (49.1%, 57), followed by AC + paclitaxel (12.1%, 14).
Treatment regimen among breast cancer patients.

Treatment outcome of breast cancer patients
This study showed that the overall mortality rate was 37.8% (44), while the overall rate of the censured event was 62.9% (73). Furthermore, a higher number of patients had partial remission (43.1%, 50) and disease progression (42.2%, 49) during the last follow-up period. Interestingly, only a few breast cancer patients had unknown and non-response remission status in the last follow-up period (Figure 3). The mean cancer-specific survival time was 25 ± 23.6 months. The mean cancer-specific survival time after metastasis was 5.6 months. However, the mean metastasis-free survival time was 9.4 months.

Status of remission of breast cancer patients during the last follow-up.
This study showed that 57.8% (67) of the patients had a regressed tumor after treatment. Nonetheless, 28.4% (33) and 13.8% (16) of patients showed progress and no tumor size changes, respectively. In addition, the study showed that the overall survival of the study participants during the follow-up period was 62.9% (73). Furthermore, the majority (82.8%, 96) of the patients survived during their first year of treatment, and the percentage of survival was decreased during the first to the fifth year of treatment (Figure 4).

Percentage of survival rate among breast cancer patients.
Breast cancer patients with diabetes, distance metastasis, advanced-stage disease (stage IV), negative HER2 receptor status, tumors with no change, and hormonally treated patients had a shorter mean survival time than their counterparts (Table 4).
Mean survival time estimates among breast cancer patients.

CI: confidence interval; HER2: human epidermal growth factor receptor 2.
Statistically significant p-value < 0.05.
Predictors of mortality among breast cancer patients
In the multivariate Cox regression analysis, the size of the tumor was the only statistically significant predictor of mortality among breast cancer patients. Hence, patients with tumor progress and tumor with no change had 4.8 and 3.2 times more hazard of dying than regressed tumors, respectively (Table 5).
Predictors of mortality among breast cancer patients.

CHR: crude hazard ratio; AHR: adjusted hazard ratio; CI: confidence interval.
Statistically significant p-value < 0.05.
Discussion
This study provides data on breast cancer treatment outcomes and its associated factors at Kitui Referral Hospital. The study revealed that the majority of the patients (97.4%) had invasive breast ductal carcinoma, which is in line with other studies conducted in Kenya (84.2%–97.9%)17–20 and Ethiopia (95.63%). 12 Besides, a relatively higher number of patients had stage III (42.2%) and stage IV (25%) breast cancer during diagnosis. Furthermore, the study conducted at Aga Khan University Hospital, Kenya, reported that 67.5% of patients were diagnosed at the late stage of the disease. 21
In contrast, another study conducted at Kenyatta National Hospital reported that a higher number of patients (59.2%) were diagnosed at stage II. 17 Due to lack of awareness, rural residents, and limited health information, most patients in poor Sub-Saharan countries have presented to the hospital lately. However, some variation may be due to different literacy levels, health information, and inadequacy of healthcare resources among study settings.
A larger portion of this study participants revealed estrogen receptor-positive breast cancer (49.1%), followed by progesterone receptor-positive (41.4%) and HER2-negative (41.4%) breast cancer types. Similarly, a recent systematic review performed in five East African countries reported that 55% had estrogen receptor-positive (95% confidence interval (CI) = 47–62), 23% HER2-positive (95% CI = 20–26), and 27% triple-negative (95% CI = 23–32) breast cancer cases. 22 However, a relatively higher percentage of patients (30.2%) expressed triple-negative cases. A study conducted at Moi Teaching and Referral Hospital reported that 33% of the tumors were triple-negative, 59% were estrogen receptor-positive, and almost all tumors analyzed were HER2-negative. 23 In contrast to this study, Sayed et al. 19 reported that a higher number of patients had estrogen receptor-positive (72.8%) and progesterone receptor-positive (64.8%), while a lesser number of patients had HER2 (17.6%) and triple-negative breast cancers (20.2%) cases. This variation is associated with variation in gene expression among individuals.
More than half of patients (55.2%) had metastasis. Of which lung-only metastasis accounted for 21.6%, bone-only metastasis 13.8%, and multiple metastases 13.8%. In contrast, Wata et al. 16 found that less than half of the patients (46.1%) developed metastases. In addition, another study conducted in Kenya showed that 32% of women with breast cancer had metastases at diagnosis studies. 20 In contrast to this study, the major metastasis sites by Ekpe et al. 20 were bone (58%), lungs (57%), and liver (50%). In addition, the study in Kenya reported that bone-only metastasis occurred in 39.80% of patients, followed by multiple metastases (33.07%) and lung metastasis (10.94%). 24 This could be attributed due to the variation in cancer pathology among different patients.
In this study, both adjuvant (chemo-surgery) and hormonal treatment were frequently (45.7%) used, while hormonal therapy only (0.9%) and adjuvant therapy (0.9%) were used in a few patients. Similarly, Opanga et al. 17 reported that adjuvant chemotherapy (66.9%) was the commonly employed treatment modality. In addition, the study conducted at Kenyatta National Hospital revealed that a relatively higher number of patients used chemotherapy only (23.3%), and the lowest number of patients used chemo-radiation and surgery (1.9%). 25 Such variation may be due to the different treatment protocols for different breast cancer stages.
The 5-year overall mean survival rate was 62.6%. It is relatively higher than the study conducted in five Sub-Saharan African countries, which reported that the overall 3-year survival rate was 50%. 26 Another study on Ugandan breast cancer patients also reported that the cumulative 5-year survival was 51.8%. 27 In addition, the overall survival for years 1, 2, and 5 were decreased with time with 82.9%, 71.6%, and 63.8%, respectively. This percentage is lower than a study in Brazil that showed that years 1, 2, and 5 overall breast cancer survival was 95.5%, 83.7%, and 87.3%, respectively. 28 This discrepancy could be attributed to lower screening rates, as the county has not been able to implement a sustained screening program due to financial, logistically reasons, lack of access to the improved infrastructure of pathology, radiology, surgery, radiation therapy, and medical oncology services and lower patient advocacy level.
During the 5 years of the treatment follow-up period, 37.1% of patients died. In contrast, other studies in Sub-Saharan African countries revealed a mortality rate between 23% and 41%.26,27 However, the majority of the patients had partial remission and progression of disease after treatment in this study setting. However, in India, 76% of the patients had either complete or partial responses after treatment. 29 The discrepancy could result from improved infrastructure in India compared to the rural areas of Kenya.
Furthermore, the study found that the mean cancer-specific survival period was 25 months, the mean metastasis-free survival was 15 months, and the cancer-specific survival after metastasis was 9.9 months. These findings are slightly lower than in the study conducted in Malaysia, where the mean cancer-specific survival was 68.1 months. 30 In contrast, cancer-specific survival after metastasis was found to be 16 months in the United States. 31 Furthermore, the study conducted in Egypt showed that the mean survival time was 84.6 ± 1.7 months. 32 This may be due to variations in the quality of healthcare.
Considering the associated factors, the size of the tumor was the only statistically significant predictor of mortality among breast cancer patients. Breast cancer patients diagnosed at the advanced stage of the disease had 3.82 times more hazards of dying (crude hazard ratio (CHR) = 3.82, 95% CI = 1.5–9.8) than an early stage of the disease. This finding is in line with similar studies (hazard ratio (HR) = 1.37; 95% CI = 1.32–1.42; p = 0.001). 18 This could be due to challenges, particularly with access to timely and adequate essential services. 33 In contrast to other studies,34,35 age and the presence of comorbidity were not statistically significant. In addition, a study conducted by Wang et al. 24 reported that age, race, marital status, grade, tumor subtype, tumor size, surgery of primary cancer, and a history of radiotherapy or chemotherapy were independent prognostic factors. These variations may be due to the different histopathology of breast cancer and other sociodemographic characteristics of the patients.
Limitation of the study
Although this study was the first of its kind to report treatment outcomes among breast cancer patients, it has the following limitations: smaller sample size, the data obtained might be affected by documentation due to the retrospective nature of the study, and generalizability may not be feasible as the study was conducted in a single setting. In addition, there were some incomplete data. Therefore, a multicenter prospective study ought to be conducted to describe the clinical pattern and treatment outcome of breast cancer in Kenya.
Conclusion
More than half of breast cancer patients survived after 5 years of the initial diagnosis, and more than one-third have died. Most patients were diagnosed with invasive ductal breast and had stage III breast cancer during the initial diagnosis. Relatively, more number of patients had estrogen receptor-positive, progesterone receptor-negative, and HER2 negative breast cancer types. In addition, the mean cancer-specific survival time and mean cancer-specific survival time after metastasis was 25 ± 23.6 and 5.6 months, respectively. Furthermore, the tumor size was the only statistically significant predictor of mortality among breast cancer patients in the multivariate Cox regression analysis as it is seen from the study that most of the patients had poor prognoses. Hence, countrywide awareness creation and screening programs should be performed.
Supplemental Material
sj-docx-1-smo-10.1177_20503121211067857 – Supplemental material for Treatment outcomes and its associated factors among breast cancer patients at Kitui Referral Hospital
Supplemental material, sj-docx-1-smo-10.1177_20503121211067857 for Treatment outcomes and its associated factors among breast cancer patients at Kitui Referral Hospital by Mwendwa Dickson Wambua, Amsalu Degu and Gobezie T Tegegne in SAGE Open Medicine
Footnotes
Acknowledgements
The authors thank all the oncology staff members of Kitui Referral Hospital about their assistance in the completion of this project.
Data availability
The data sets used during this study are available from the corresponding author on a reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical clearance was obtained from the US International University-Africa Institutional Review Board (approval no. USIU-A/IRB/073-2021). The Ethics committee waived the requirements for informed consent since the data were collected retrospectively from the medical records of patients.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
The name and other identities of the patients were not recorded during the data collection to ensure the confidentiality of the data. Informed consent was not sought for this study since the data were collected retrospectively from the medical records of patients. The Ethics committee waived the requirements for informed consent.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
