Abstract
Objectives:
Research on the determinants of physical activity in mildly symptomatic patients with chronic obstructive pulmonary disease is lacking. This study examined the predictors of physical activity in patients with low-risk chronic obstructive pulmonary disease.
Methods:
A total of 41 male patients with chronic obstructive pulmonary disease belonging to Group A of the Global Initiative for Chronic Obstructive Lung Disease were included. Regarding the objective index, the physical activity (number of steps/day and the amount of Ex (metabolic equivalent × hours)/day) of the participants was measured with a tri-axis accelerometer. In addition, regarding the evaluation index, respiratory function and dynamic lung hyperinflation were measured by a spirometer, skeletal muscle mass was measured using bioelectrical impedance analysis, skeletal muscle strength (grip and lower limb muscle strength) was measured using a dynamometer, exercise capacity was measured by the incremental shuttle walking test, and health-related quality of life was measured.
Results:
Significant correlations were found between the number of steps per day and age (ρ = −0.501, p < 0.01), forced vital capacity predictive values (ρ = 0.381, p < 0.05), dynamic lung hyperinflation (ρ = 0.454, p < 0.01), grip strength (ρ = 0.318, p < 0.05), and walking distance in incremental shuttle walking test (ρ = 0.779, p < 0.01), but not skeletal muscle mass, lower limb muscle strength, or health-related quality of life. A multiple-regression analysis with the number of steps per day as the dependent variable extracted only walking distance in incremental shuttle walking test, yielding a moderate single-regression equation (steps/day = −934.909 + 11.052 × walking distance in incremental shuttle walking test, adjusted R2 = 0.548, p < 0.001).
Conclusion:
It was suggested that the amount of physical activity of patients with low-risk chronic obstructive pulmonary disease could be predicted by walking distance in incremental shuttle walking test.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is caused by chronic bronchial inflammation and destruction of the alveolar walls, resulting in airflow limitation and reduced respiratory function. Dynamic lung hyperinflation, especially during exertion, causes dyspnea and makes physical activity difficult for patients with COPD. 1 Physical activity is a strong predictor of prognosis in COPD patients, 2 and since physical inactivity is associated with exacerbations, 3 it is important to maintain physical activity.
Physical activity decreases with the intensity of dyspnea symptoms. 4 Because the intensity of symptoms is accompanied by a decline in exercise capacity, 5 it can be difficult to improve physical activity when the symptoms are severe. Therefore, it is desirable to maintain physical activity and manage the disease while the symptoms are mild and exercise capacity is preserved.
Even patients with early COPD with mild symptoms have been reported to have limitations in physical activity. 6 Factors limiting exercise in patients with mild COPD may be attributed to dysfunction of the lungs and airways, including airflow limitation, dynamic lung hyperinflation, and heterogeneity of gas exchange between alveolar and arterial blood. 7 Therefore, physical inactivity is thought to be caused by these exercise limiting factors.
Systematic reviews of the clinical and functional determinants of physical activity in COPD patients have reported a variety of factors, including dynamic lung hyperinflation, gas exchange capacity, exercise capacity, dyspnea, systemic inflammation, history of previous exacerbations, quality of life, and self-efficacy. The scientific evidence for most of these factors is extremely low or unknown. 3 Furthermore, research on the determinants of physical activity in mildly symptomatic COPD patients is lacking. 8
In this study, we comprehensively investigated the factors affecting the levels of physical activity in mildly symptomatic COPD patients who belonged to Group A of the Global Initiative for Chronic Obstructive Lung Disease (GOLD), which have not been clarified in previous studies.3,8 This study aimed to examine the predictors of physical activity on patients with low-risk COPD.
Methods
Participants
A total of 68 patients with stable COPD (attending three clinics and one hospital) participated in this study between August 2018 and January 2020, excluding 13 patients (10 in Group B, 2 in Groups C, and 1 in Group D) belonging to groups other than Group A of the GOLD assessment classification. 9 Of the 55 patients in Group A, 3 who met the exclusion criteria, 7 with missing values, and 4 female patients were excluded, and 41 male patients (mean age: 75.2 ± 6.4 years, mean forced expiratory volume in 1 s/forced vital capacity (FEV1/FVC): 61.1% ± 8.5%), were included from the analysis (Figure 1). The participants were independent in their daily activities and did not include those on home oxygen therapy. Those with evident dementia, serious orthopedic, or cerebrovascular diseases that would interfere with walking and those with unstable heart diseases were excluded.

Algorithm of the participants analyzed in this study.
The participants were fully informed about the contents of this study, and their written consent was obtained. This study was conducted according to the principles of the World Medical Association Declaration of Helsinki, following approval from the Research Ethics Review Committee of the Kyoto Tachibana University, Japan (Approval No.: 17-07).
Procedures
This was a cross-sectional study. The participants’ physical activity, respiratory function, dynamic lung hyperinflation, skeletal muscle mass, limb muscle strength, and exercise capacity were assessed. Assessments other than physical activity were performed in 1 day. A tri-axis accelerometer (Active style PRO HJA-750C, OMRON, Kyoto, Japan) was used to evaluate physical activity. The tri-axis accelerometer (Active Style Pro) is an accelerometer developed in Japan that captures changes in body movement and posture employing original signal processing and has the feature of being able to accurately measure not only walking and running activities but also other activities that have not been captured by conventional accelerometers. 10 At the end of the evaluation, the participants were given an activity monitor and instructed to wear it vertically around the waist of their pants, except when sleeping and bathing. For the number of days of measurement, we requested a maximum of 1 month in order to ensure a minimum level of reliability of the measurement data; we adopted participants who had been measured for at least 3 days (2 weekdays and 1 weekend day). 11 Daily measurements were taken on days when there were more than 6 h of measurement time, avoiding the middle of the night when older adults are believed to be less active. 12 The adopted measurement data were divided by the number of days to calculate the average physical activity per day.
As recommended by the American Thoracic Society guidelines, pulmonary function was evaluated using a spirometer (AS-507; Minato Medical Science Co., Ltd., Osaka, Japan). 13 The predictive values of FVC (FVC % pred) and FEV1 (FEV1 % pred) were calculated using the formulas proposed by the Japanese Respiratory Society. 14 The FEV1 % pred was further classified by the GOLD assessment. 9
Dynamic lung hyperinflation was assessed according to the method described by Fujimoto. 15 We used a spirometer to measure the maximum inspiratory capacity after breathing to the rhythm of 20 breaths per minute on a metronome for 30 s (IC-20) and the maximum inspiratory capacity of breathing loudly and quickly to the rhythm of 40 breaths per minute on a metronome for 30 s (IC-40). Low amounts of IC-20 and IC-40 were evaluated as the strength of dynamic lung hyperinflation. Measurements were performed twice, and the better value was adopted.
A body composition analyzer (InBody270; InBody Japan Co., Ltd., Tokyo, Japan) was used to measure skeletal muscle mass. This measurement is performed using bioelectrical impedance analysis. 16 For the measurement, the participants were instructed to stand barefoot on the measurement table, place both feet on the electrodes, grasp the gripping electrodes with both hands, and maintain a stationary standing position until the completion of measurement. Muscle mass and skeletal muscle mass index (SMI) of the right and left upper and lower limbs, trunk, and whole body were measured. Body fat percentage was also measured at the same time. SMI was calculated by dividing the muscle mass of the extremities (kg) by the height (m) squared (SMI = (right upper limb muscle mass (kg) + left upper limb muscle mass (kg) + right lower limb muscle mass (kg) + left lower limb muscle mass (kg))/height (m) squared).
Limb muscle strength was assessed based on grip strength (GS) and knee extensor strength. A digital GS meter (Grip-D; Takei Kiki Kogyo Co., Ltd., Niigata, Japan) was used to measure GS. The width of the grip was adjusted such that the second joint of the index finger was almost at a right angle, in accordance with the new physical fitness test implementation guidelines proposed by the Ministry of Education, Culture, Sports, Science and Technology. 17 In a static standing position, the participants grasped the GS meter with both upper limbs hanging down and flexed their fingers with maximum effort. The GS was measured twice on each side, and the normalized value (%) obtained by dividing the maximum value of either side by the body weight (BW) was used as the representative value (GS/BW).
The knee extensor muscle strength was measured using a manual muscle strength measuring device (μ-TasF1; Anima Co., Ltd., Tokyo, Japan). The sensor pad was fixed to the anterior surface of the distal lower leg with a belt in an end-seated lower leg drooping position. In addition, the participants performed an isometric knee extension exercise with maximal effort for about 3 s, according to the method described by Kato et al. 18 Two measurements were taken on each side, and the knee extensor muscle strength was normalized by dividing the maximum value (%) of either side by the BW, which was used as the representative value.
Exercise capacity was assessed by the incremental shuttle walking test (ISWT). The measurements were performed according to the method described by Singh et al. 19 We measured the walking distance in incremental shuttle walking test (ISWD), transcutaneous oxygen saturation (SpO2), and pulse rate (with a pulse oximeter; Radical Docking Station RDS-1; Masimo, Irvine, CA, USA) at rest before and immediately after the exercise. In addition, we evaluated breathlessness and leg fatigue at rest using a modified Borg scale 20 before and immediately after the exercise. The ISWT was only measured once because a single measurement is sufficient to obtain reliable results. 21
Basic information about the participants was also assessed, including age, height, weight, body mass index, dyspnea (modified Medical Research Council (mMRC) dyspnea scale), 22 health-related quality of life (St. George’s Respiratory Questionnaire; SGRQ), 23 use of bronchodilators, and number of exacerbations.
Statistical analysis
To determine the sample size, the power analysis software G-power ver. 3.1.9.2 was used, and the sample size was determined to satisfy the 26 participants calculated with an effect size of 0.5, 0.05 α error, and 0.8 power for the correlation analysis. Statistical analyses were performed to analyze the correlation between the average number of steps per day and each evaluation index, as well as the correlation between the average level of physical activity per day and each evaluation index. After confirming normality using the Shapiro–Wilk test, normally distributed indices were analyzed using Pearson’s correlation coefficient, and non-normally distributed indices were analyzed using Spearman’s rank correlation coefficient. In addition, multiple-regression analysis using the stepwise method was conducted with the average number of steps per day and the average daily activity as dependent variables and the indices with significant correlations as independent variables to examine the factors influencing the physical activity. We created a multiple-regression equation from the multivariate analysis results that showed good prediction accuracy. Statistical analysis software SPSS version 24 was used, and the statistical significance level was set at 5%.
Results
The characteristics of the participants are shown in Table 1. In pulmonary function tests, most of the participants had an airflow limitation of GOLD I (46.3%) and GOLD II (48.8%); 7.3% of them experienced an exacerbation in the past year. Table 2 shows the level of physical activity. The number of valid days of measurement (mean ± standard deviation) was 25.2 ± 6.6 days. There was a large variation in the average number of steps taken per day, but the largest percentage (26.8%) of respondents took between 2000 and 3000 steps. In terms of the amount of Ex (metabolic equivalents (METs) × hours) per day, participants who took 2–4 Ex accounted for the highest percentage (51.2%), while those who took <2 Ex accounted for 24.4% (Figure 2). Regarding the amount of Ex per week, 87.8% of the participants had >10 Ex per week.
Attributes of the participants GOLD grade, mMRC, GOLD stage, bronchodilator use, and exacerbations are indicated by the number of participants; others are indicated by mean ± standard deviation.

BMI: body mass index; mMRC: modified medical research council dyspnea scale; FVC: forced vital capacity; FEV: forced expiratory volume; FEV/FVC: forced expiratory volume/forced vital capacity; GOLD: Global Initiative for Chronic Obstructive Lung Disease; IC: inspiratory capacity; SMI: skeletal muscle mass index; GS/BW: percentage of grip strength/body weight; QS/BW: percentage of quadriceps strength/body weight; ISWT: incremental shuttle walking test; LAMA: long-acting muscarinic antagonist; LABA: long-acting β-agonists; SGRQ: St. George’s Respiratory Questionnaire; ICS: inhaled corticosteroid.
Percentage of predicted value.
IC-20: inspiratory capacity at a rhythm of 20 beats per minute.
IC-40: inspiratory capacity at a rhythm of 40 beats per minute.
ΔIC: difference between IC-40 and IC-20.
Borg (chest): modified Borg scale for dyspnea in the 6-min walk test.
Borg (leg): modified Borg scale for lower limb fatigue in the 6-min walk test.
Exacerbation: number of exacerbations in the last year.
The number of participants who responded is 39.
Physical activity of the participants (n = 41).

METs: metabolic equivalent.
Ex: METs × hours.
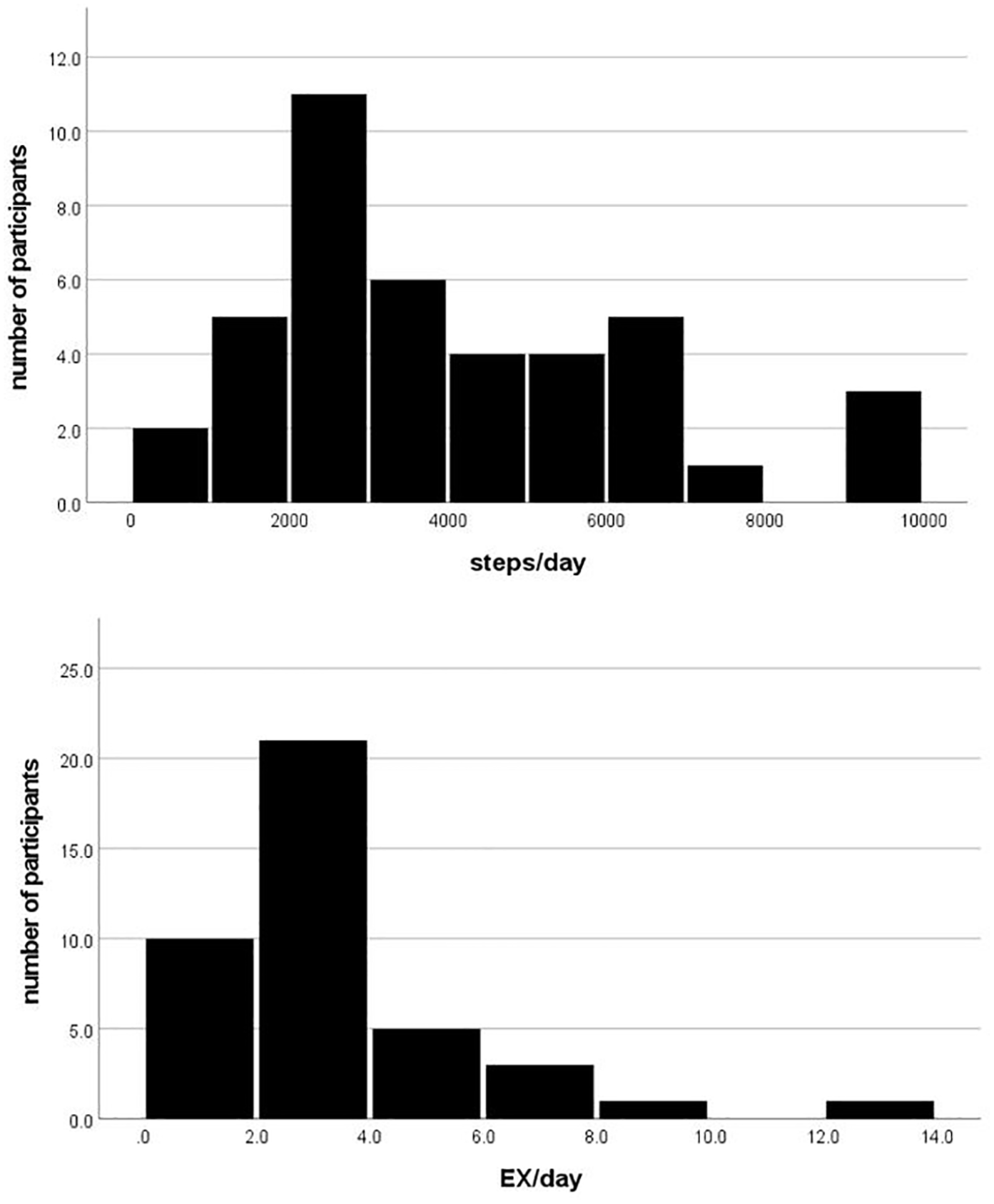
Average daily physical activity and number of participants. The upper graph shows the average number of steps per day and the number of participants; the lower graph shows the average Ex amount and the number of participants.
In the correlation between average daily steps and physical characteristics, a significant inverse correlation between the average steps per day and age was found. Moreover, a significant correlation between the average steps per day and FVC % of pred, the IC-20 and IC-40, GS/BW, and ISWD was found for pulmonary function, dynamic hyperinflation, muscle strength, and ISWT, respectively. However, there were no correlations between the average daily steps/day and skeletal muscle mass and SGRQ. In the correlation between the average daily Ex and physical characteristics, a significant inverse correlation between the average daily Ex and age was found. The average daily Ex was significantly correlated with FVC % of pred, IC-40, and ISWD for pulmonary function, dynamic hyperinflation, and ISWT, respectively. However, no correlation was found between the average daily Ex and skeletal muscle mass, muscle strength, or SGRQ (Table 3).
Correlation with the amount of physical activity (n = 41).

BMI: body mass index; mMRC: modified medical research council dyspnea scale; FVC: forced vital capacity; FEV: forced expiratory volume; FEV/FVC: forced expiratory volume/forced vital capacity; SMI: skeletal muscle mass index; GS/BW: percentage of grip strength/body weight; QS/BW: percentage of quadriceps strength/body weight; ISWT: incremental shuttle walking test; SGRQ: St. George’s Respiratory Questionnaire.
Notation is shown as Spearman’s rank correlation coefficient ρ.
Percentage of predicted value.
IC-20: inspiratory capacity at a rhythm of 20 beats per minute.
IC-40: inspiratory capacity at a rhythm of 40 beats per minute.
ΔIC: difference between IC-40 and IC-20.
Borg (chest): modified Borg scale for dyspnea in the 6-min walk test.
ΔBorg chest: difference between Borg chest pre and Borg chest post.
Borg (leg): modified Borg scale for lower limb fatigue in the 6-min walk test.
ΔBorg leg: difference between Borg leg pre and Borg leg post.
Ex: METs × hours.
p < 0.05, **p < 0.01.
Multiple-regression analysis using the stepwise method with average daily step count as the objective variable and age, FVC % of pred, IC-40, GS/BW, and ISWD as explanatory variables was performed. The analysis of variance (ANOVA) results were significant (F = 49.5, p < 0.001). Only ISWD was extracted as an influencing factor. In a single-regression equation using ISWD with the use of unstandardized coefficients, a prediction equation for the number of steps with moderate accuracy could be created (steps/day = −934.909 + 11.052 × ISWD, adjusted R2 = 0.548, p < 0.001). The variance inflation factor (VIF) was less than 10, and there was no problem with collinearity. The results of multiple-regression analysis using the stepwise method with average daily Ex as the objective variable and age, FVC % of pred, IC-40, and ISWD as explanatory variables were also performed. The results of the ANOVA were significant (F = 19.3, p < 0.001). Similarly, the results showed that ISWD was extracted as an independent influencing factor and a significantly useful model (adjusted R2 = 0.314, p < 0.001). The VIF was less than 10, and there was no problem with collinearity (Table 4).
Results of multiple-regression analysis with physical activity as the dependent variable (n = 41).

VIF: variance inflation factor; FVC: forced vital capacity; GS/BW: percentage of grip strength/body weight; ISWD: walking distance of incremental shuttle walking test.
Percentage of predicted value.
IC-40: inspiratory capacity at a rhythm of 40 beats per minute.
Ex: METs × hours.
Discussion
In this study, we examined the factors relating to physical activity in stable COPD patients who belonged to Group A using physical ability as an index. The results suggest that physical activity can be predicted by ISWD.
Regarding the factors that influence physical activity in COPD patients, Gimeno-Santos et al. 3 conducted a systematic review in 2014 and stated that the determinants of physical activity are not yet clear. In particular, it has been reported that physical activity (a complex behavior) and exercise capacity or physical fitness (a set of attributes) are correlated only to a moderate extent and constitute different concepts. 24 This is supported by the fact that some determinants of exercise capacity are not associated with physical activity. 3 Similarly, in this study, significant correlations were found between the average number of steps per day and age, FVC % of pred, dynamic lung hyperinflation, and GS but not lower limb muscle strength or skeletal muscle mass. No correlation was found between the average daily activity and limb muscle strength or skeletal muscle mass. Considering these results, the relationship between physical activity and determinants of exercise capacity is not strong, as shown in previous studies.3,24
In contrast, ISWD, a measure of exercise capacity, showed a significant moderate correlation with physical activity. Many previous studies on physical activity and exercise capacity have also reported a significant correlation;1,25,26 however, the relationship is not strong. Fastenau et al. 27 examined the association between physical activity and 6-min walking distance in patients with mild to moderate COPD according to the GOLD assessment and found that there was no significant association and that physical activity and exercise capacity differed. However, in this study, ISWD was able to predict the average number of steps taken per day with moderate accuracy. The participants in the Fastenau et al. 27 study had a mean mMRC of 1.5 (mean MRC: 2.5), and considering that the participants in other previous studies1,25,26 had a more severe GOLD stage than our participants, it is likely that many of them had more severe dyspnea than the participants in our study. Hanania and O’Donnell 28 reported that dyspnea has psychological effects, such as depression and anxiety, and the anticipation of dyspnea itself has a significant impact on the emotions and behaviors of COPD patients, causing them to limit their physical activity in order to reduce the symptoms. Therefore, we speculated that some of the participants in the study by Fastenau et al. 27 and other previous studies1,25,26 limited their physical activity, regardless of their exercise capacity, and that the relationship between physical activity and the 6-min walking distance may not have been observed. In contrast, the participants in this study had an mMRC of 0 or 1 and mild dyspnea. Therefore, it is possible that fewer people were psychologically affected, and this can be supported by the fact that there was no significant correlation between SGRQ, a measure of health-related quality of life, and physical activity and that the health-related quality of life was relatively high. 29 Therefore, it is conceivable that the physical activity of low-risk COPD patients could be predicted by the exercise capacity in ISWD.
In respiratory function, FVC % pred and dynamic lung hyperinflation correlated weakly with physical activity.
In patients with severe COPD, the worsening of dynamic lung hyperinflation due to chronic inflammation and exacerbation may cause respiratory dysfunction30,31 and significantly impact physical activity. However, in this study, COPD patients with mild dyspnea were included, and the IC was below the 200 mL threshold for change in IC affecting dynamic lung hyperinflation of Gatta et al. 32 Therefore, the effect of dynamic lung hyperinflation may have been minimal. Therefore, it is possible that the correlation was weak with physical activity. However, the results of this study suggest that lung volume during exercise is related to the maintenance of physical activity even in low-risk COPD patients, and the assessment of FVC % pred. and dynamic lung hyperinflation should be monitored closely.
The results of this study indicate that the ISWD in low-risk COPD patients is moderately related to physical activity. In order to evaluate physical activity with a pedometer, 3 days of evaluation (2 weekdays and 1 weekend day) 11 are required, which is time-consuming. However, ISWD can be evaluated in a short period of time, allowing for rapid evaluation. Since COPD is a chronic progressive disease, appropriate management is required in the early phase of the disease. Increased physical activity is recommended for the management of low-risk COPD patients. 22 Therefore, it is suggested that the assessment of ISWD can screen the physical activity of patients in the early phase of the disease and may be useful as a screening test for physical activity in low-risk COPD patients.
One of the limitations of this study is that it was a cross-sectional study; hence, the causal relationship between physical activity and ISWD is unclear. Therefore, a longitudinal study is required in the future.
Conclusion
In conclusion, the physical activity of low-risk COPD patients can be predicted by ISWD. Hence, ISWD can be used as a screening test for physical activity, suggesting that it may be useful for disease management.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was conducted following approval from the Ethics Review Committee of Kyoto Tachibana University, Japan (Approval No. 17-07).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI (Grant No. JP21K10782).
Informed consent
Written informed consent was obtained from all subjects before the study.
