Abstract
Objective:
The majority of patients with COVID-19 showed mild symptoms. However, approximately 5% of them were critically ill and require intensive care unit admission for advanced life supports. Patients in the intensive care unit were high risk for venous thromboembolism and hemorrhage due to the immobility and anticoagulants used during advanced life supports. The aim of the study was to report the incidence and treatments of the two complications in such patients.
Method:
Patients with COVID-19 (Group 1) and patients with community-acquired pneumonia (Group 2) that required intensive care unit admission were enrolled in this retrospective study. Their demographics, laboratory results, ultrasound findings and complications such as venous thromboembolism and hemorrhage were collected and compared.
Results:
Thirty-four patients with COVID-19 and 51 patients with community-acquired pneumonia were included. The mean ages were 66 and 63 years in Groups 1 and 2, respectively. Venous thromboembolism was detected in 6 (18%) patients with COVID-19 and 18 (35%) patients with community-acquired pneumonia (P = 0.09). The major type was distal deep venous thrombosis. Twenty-one bleeding events occurred in 12 (35%) patients with COVID-19 and 5 bleeding events occurred in 5 (10%) patients with community-acquired pneumonia, respectively (P = 0.01). Gastrointestinal system was the most common source of bleeding. With the exception of one death due to intracranial bleeding, blood transfusion with or without surgical/endoscopic treatments was able to manage the bleeding in the remaining patients. Multivariable logistic regression showed increasing odds of hemorrhage with extracorporeal membrane oxygenation (odds ratio: 13.9, 95% confidence interval: 4.0–48.1) and COVID-19 (odds ratio: 4.7, 95% confidence interval: 1.2–17.9).
Conclusion:
Venous thromboembolism and hemorrhage were common in both groups. The predominant type of venous thromboembolism was distal deep venous thrombosis, which presented a low risk of progression. COVID-19 and extracorporeal membrane oxygenation were risk factors for hemorrhage. Blood transfusion with or without surgical/endoscopic treatments was able to manage it in most cases.
Introduction
As of 1 October 2020, over 30 million patients have been infected by the coronavirus disease (COVID-19) worldwide. 1 Common clinical manifestations included fever, cough, shortness of breath and ground-glass opacity on chest computed tomography. 2 In addition to the respiratory system, COVID-19 was also associated with cardiac injury,3,4 abnormal coagulation 5 as well as gastrointestinal symptoms 6 and might lead to complicated situation if patients were pregnant or immunosuppressed.7–9 Although mostly mild, approximately 5% of the COVID-positive patients were critically ill and require intensive care unit (ICU) admission, 2 where they could receive advanced life supports including mechanical ventilation, continuous renal replacement treatment (CRRT), extracorporeal membrane oxygenation (ECMO) and artificial liver support (ALS). ICU patients were high risk for both venous thromboembolism (VTE) and hemorrhage due to a combination of immobility and anticoagulants used during advanced life supports. Both adverse events could be clinically challenging and associated with poor prognosis. Up to now, there is no study comparing the incidence and consequence of hemorrhage and VTE in the ICU between patients with COVID-19 and patients with community-acquired pneumonia (CAP). Therefore, in this study, we report these results and provide prevention and treatments of the two complications.
Methods
Study population
This retrospective study included consecutive patients admitted to the ICU as a result of COVID-19 and CAP. Patients with COVID-19 (Group 1) were enrolled between 10 January 2020 and 26 March 2020 at our hospital, the First Affiliated Hospital, School of Medicine, Zhejiang University. If the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) caused pneumonia in the community settings, the pneumonia was also diagnosed as CAP. We selected patients with CAP (Group 2) between 1 December 2018 and 1 November 2019 in the same hospital. The duration was before the outbreak of COVID-19, excluding the SARS-CoV-2 as a pathogen. Inclusion criteria for both groups were (a) the patient was critically ill and (b) the patient was either admitted to the ICU directly, or to the general ward at first and then transferred to the ICU due to the deterioration of the pneumonia. The diagnosis of COVID-19 was confirmed by real-time polymerase chain reaction for SARS-CoV-2 RNA. Patients with CAP were diagnosed according to the guideline. 10 If the COVID-19 was suspected by clinical symptoms but not confirmed by positive SARS-CoV-2 RNA, the patient was excluded. The Clinical Research Ethics Committee of our hospital approved this study. Written informed consent was waived due to the urgent need to collect data.
Treatment strategies
For Group 1, members of the multidisciplinary team would discuss the optimal treatment plan for each patient, which contains individualized antiviral, anti-shock and anti-hypoxia treatments and advanced life supports.
11
For Group 2, the treatments were according to the latest guidelines.10,12 Advanced life supports included intubation and mechanical ventilation, ALS, ECMO and CRRT. The ALS, which could clear cytokines and toxic molecules and supplement albumin and coagulation factors, was indicated if there was a high level of inflammatory cytokines or rapid deterioration.
13
ECMO was indicated if mechanical ventilation failed to provide sufficient oxygenation or the patient was not suitable for long-term ventilation. CRRT was employed for severe kidney injury. Complete blood count, prothrombin time (PT), activated partial thromboplastin time (APTT), international normalized ratio (INR), fibrinogen and
Data collection
We reviewed medical records to extract demographic information, laboratory results, computed tomography and ultrasound finds, treatment strategies and adverse events of each patient. The data we collected were documented in standardized data collection forms. For original data, please contact the corresponding author.
Statistical analysis
Categorical data were compared using the chi-square test or Fisher’s exact test. Normal and non-normal distributed continuous data were analyzed using Student’s t test and Mann–Whitney U test, respectively. Results were given as mean, median (interquartile range (IQR)) or number (percentage) wherever appropriate. To explore risk factors associated with hemorrhage, multivariable logistic regression model was used. Considering the total number of patients (n = 85) in our study and to avoid overfitting in the model, five variables were chosen for multivariable analysis, namely age, COVID-19, CRRT, ECMO and ALS. The choice was based on theoretical connection between the variable and hemorrhage. Anticoagulation was frequently used in CRRT, ECMO and ALS. Age was related to morbidity.16,17 COVID-19 might also contribute to abnormal coagulation.5,18 Statistical analysis was performed via SPSS 22.0 for Windows (SPSS Inc., Chicago, IL, USA). All tests were two-sided, and a P value less than 0.05 was considered statistically significant.
Results
Demographic information and comorbidities
A total of 34 patients in Group 1 and 51 patients in Group 2 were enrolled in this retrospective study. Their demographic information and comorbidities were summarized in Table 1. The mean age was 66 and 63 years in Groups 1 and 2, respectively. The majority of patients were male in both groups. Patients with CAP tended to smoke and have chronic obstructive pulmonary disease (COPD). There was no other statistical difference in demographics and comorbidities detected.
Characteristics of patients with COVID-19 and patients with CAP.

CAP: community-acquired pneumonia; COPD: chronic obstructive pulmonary disease.
Data are given as number (%) unless otherwise stated.
Coagulation test
Results of coagulation tests on admission to our hospital and to the ICU were summarized in Table 2. Platelet was within the normal range, and fibrinogen was elevated in both groups. Hemoglobin level, PT, APTT and INR were almost normal in Group 1. For Group 2, however, the four test results were slightly out of the normal range with statistical significance detected. In terms of
Coagulation tests.

CAP: community-acquired pneumonia; HBG: hemoglobin; PLT: platelet; INR: international normalized ratio; PT: prothrombin time; APPT: activated partial thromboplastin time; FBG: fibrinogen; IQR: interquartile range; ICU: intensive care unit; APACHE: Acute Physiology and Chronic Health Evaluation.
Data are given as mean unless otherwise stated.
Fifty results of
Advanced life support, VTE and hemorrhage
As summarized in Table 3, more ALS was used in patients with COVID-19, while patients with CAP received more intubation. Approximately a quarter of patients in both groups received ECMO or CRRT. VTE was detected in 6 (18%) patients with COVID-19 and 18 (35%) patients with CAP with no statistical significance (P = 0.09). Most of the VTEs in both groups were distal deep venous thromboses (DVTs). There was no proximal DVT detected. Internal jugular vein thrombosis was discovered in three patients, all of which were catheter related. Only one low-risk pulmonary embolism (PE) in a patient with CAP was detected. Details were listed in Table 4. The treatments of VTE were individualized. If patients were already receiving ALS, ECMO and/or CRRT that required anticoagulation, no extra anticoagulant was administered. If not, the treatments would follow the guidelines.14,15 In terms of hemorrhage, 21 and 5 bleeding events occurred in 12 (35%) patients with COVID-19 and 5 (10%) patients with CAP, respectively (P = 0.01). Details were listed in Table 5. Gastrointestinal bleeding was the most common in both groups. Compared with Group 2, more surgical/endoscopic treatments and more blood transfusion were needed to control the bleeding in patients with COVID-19. Except the intracranial bleeding that caused one patient’s death, all the remaining hemorrhages were restored by conservative treatments with or without surgical/endoscopic treatments.
Advanced life support, VTE and hemorrhage.

CAP: community-acquired pneumonia; ALS: artificial liver support; ECMO: extracorporeal membrane oxygenation; CRRT: continuous renal replacement treatment; VTE: venous thromboembolism.
Data are given as number (%) unless otherwise stated.
The patients with hemorrhage met the International Society on Thrombosis and Haemostasis major bleeding criteria.
VTE in the ICU.
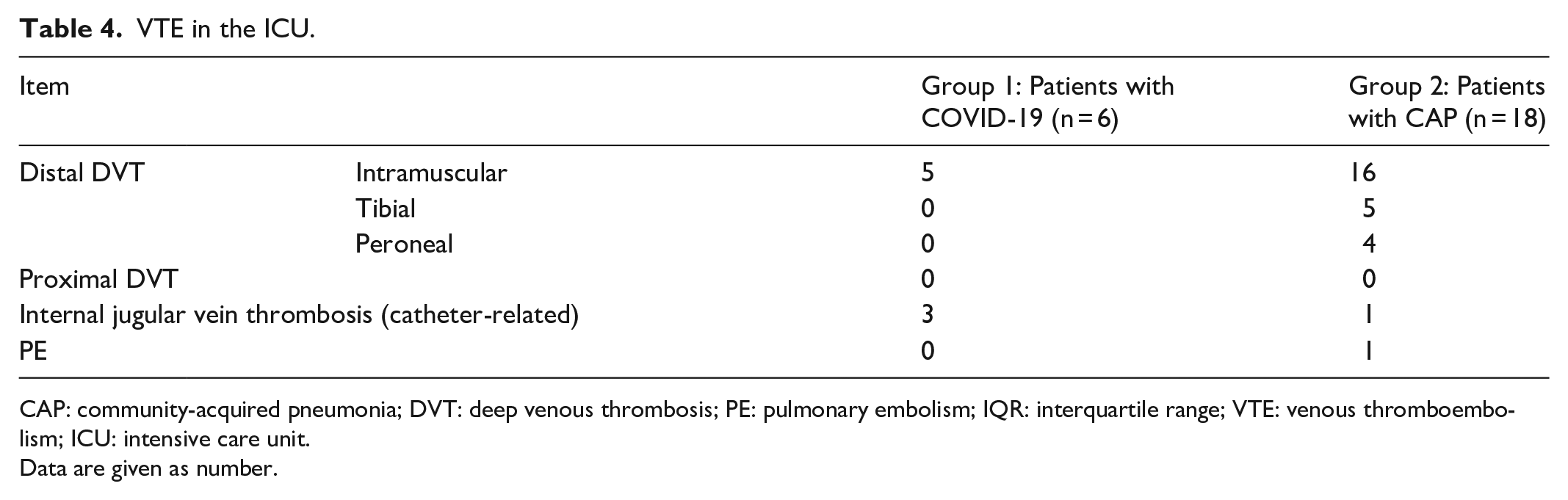
CAP: community-acquired pneumonia; DVT: deep venous thrombosis; PE: pulmonary embolism; IQR: interquartile range; VTE: venous thromboembolism; ICU: intensive care unit.
Data are given as number.
Hemorrhage in the ICU.
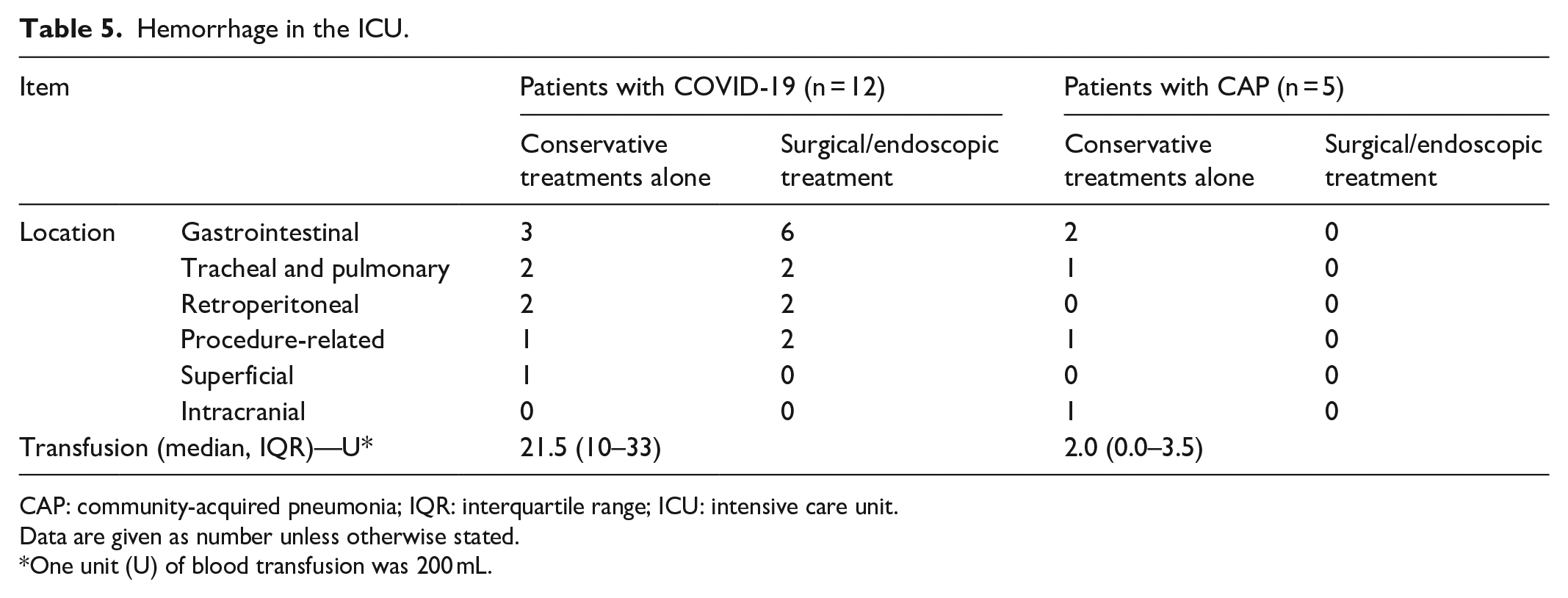
CAP: community-acquired pneumonia; IQR: interquartile range; ICU: intensive care unit.
Data are given as number unless otherwise stated.
One unit (U) of blood transfusion was 200 mL.
We used multivariable logistic regression model to explore risk factors with hemorrhage. Due to the amount of patients (n = 85 in total) and previous findings, we included five variables, namely age, COVID-19, ALS, ECMO and CRRT. Multivariable logistic regression showed increasing odds of hemorrhage with ECMO (P < 0.01, odds ratio (OR): 13.9, 95% confidence interval (CI): 4.0–48.1) and COVID-19 (P < 0.05, OR: 4.7, 95% CI: 1.2–17.9).
Discussion
The COVID-19 pandemic, which is caused by the virus SARS-CoV-2, has put huge pressure on the medical system worldwide. The treatments for this disease are three-fold. For general population, it is important to keep social distancing, wash hands, cut travel 19 and wear masks. For patients with mild and moderate COVID-19, Fangcang shelter hospitals, which are large, temporary hospitals built by converting public venues into health-care facilities, are necessary to isolate them from their families and communities and provide medical care. 20 For severe and critically ill patients, dedicated tertiary hospitals are needed to provide intensive care and advanced life supports. Approximately 5% patients with COVID-19 are critically ill and require ICU admission. 2 With strong life supports and treatments, the mortality of COVID-19 can be decreased to a low level.
In this study, we compared the demographics, results of coagulation tests as well as the incidence and consequence of hemorrhage and VTE between the two groups. One particular interest of our study was the VTE and hemorrhage, the two common complications occurring during critical care.
VTE included DVT and PE. The incidence of DVT could be over 30% among patients in the ICU despite thromboprophylaxis.21,22 DVT would lead to swollen extremities and was closely related to PE in the short term. It also might result in post-thrombotic syndrome in the long term. Our results showed that most of the VTE detected in the two groups belonged to distal DVT, especially intramuscular venous thrombosis, which was considered to have a low risk of progression.
21
Proximal DVT and PE were rare. The sole PE detected was stratified as low risk.
15
A few catheter-related internal jugular vein thromboses were detected. Its incidence would be high if patients received thoracic or cardiovascular surgery.23,24 Among patients without surgery in ICU, symptomatic deep vein thrombosis related to catheters was as low as 1%.
25
The high incidence and low mortality risk of VTE in this study may be explained by use of a pneumatic compression device and prophylactic anticoagulation, close monitoring including
Hemorrhage was another major concern in the ICU. Except intubation and mechanical ventilation, other advanced life supports including ALS, CRRT and ECMO usually required anticoagulation, which might cause hemorrhage. The stress ulcer incurred by the pneumonia and procedures such as catheterization in central veins and femoral arteries might also lead to bleeding. In our study, 12 (35%) patients with COVID-19 and 5 (10%) patients with CAP suffered hemorrhage. Approximately half of the bleeding in patients with COVID-19 needed surgical/endoscopic treatments. The intracranial bleeding resulted in one death, while the remaining patients survived this adverse event, which suggested that a combination of conservative treatments and surgical/endoscopic treatments could deal with the bleeding in most cases.
To explore the risk factors for bleeding, we used the multivariable logistic regression model. Five possible factors, namely age, COVID-19, ALS, ECMO and CRRT, were analyzed. Its results showed only ECMO (OR: 13.9, 95% CI: 4.0–48.1) and COVID-19 (OR: 4.7, 95% CI: 1.2–17.9) were risk factors of hemorrhage in patients in the ICU.
Age was frequently related to adverse events and poor prognosis. Previous studies reported that age was a risk factor for mortality in patients with COVID-19. 17 Our study showed that age did not contribute to the hemorrhage, suggesting that the hemorrhage could happen in any patient.
According to the regression analysis, the ALS was also not related to hemorrhage. The ALS system was useful for clearing cytokine storm and exchanging plasma, which could filter toxic metabolites, balance fluid volume and provide serum albumin and coagulation factors. 13 Although heparinization was needed for the system, massive plasma (approximately 2000 mL) could be exchanged into the patient and protamines would be used to neutralize heparin, which contributed to hemostasis. In terms of CRRT, there were three widely used anticoagulants, namely, unfractionated heparin, low-molecular-weight heparin and citrate. Heparin and citrate could be regionally administered. Compared with heparin, high-quality evidence demonstrated that regional citrate had a bleeding rate less than 5% in critically ill patients and had a low risk of circuit loss, filter failure and heparin-induced thrombocytopenia.26–29 Therefore, it was considered a satisfactory anticoagulant. With the developments of anticoagulants in CRRT, our regression analysis excluded the CRRT as a risk factor for bleeding in critically ill patients with pneumonia.
ECMO was another vital life-saving technique. Venovenous ECMO was able to provide oxygenation in respiratory failure,30,31 and venoarterial ECMO could be a complementary strategy for high-risk PE,32,33 cardiogenic shock 34 and lung transplantation. 35 Complications including limb ischemia, brain injury and hemorrhage were prevalent.36,37 Thrombocytopenia, acquired von Willebrand syndrome and anticoagulation during the procedure might explain its bleeding tendency.38,39 Reduction from standard to low anticoagulation regimens decreased incidence of bleeding in half to approximately 20%. 40 Short-term heparin-free operation may be performed to control fatal bleeding. 11 Although the bleeding was common, days on ECMO was not associated with a decrease in the platelet count, and death due to hemorrhage accounted for approximately 3% reasons for discontinuation of ECMO.38,41 Our results also showed that ECMO was a risk factor for bleeding and most patients would survive this adverse event with a combination of conservative treatments and surgical/endoscopic treatments.
The regression results indicated that COVID-19 was a risk factor for bleeding as well. The COVID-19 tended to affect multiple organs that expressed angiotensin-converting enzyme 2 (ACE2), such as the lungs, heart, gastrointestinal tract and kidney. 42 In severe patients with COVID-19, abnormal coagulation and low level of platelets were observed.2,5 Pathology results also showed a reduction of blood cells in the all three classes in bone marrow. 43 In critically ill patients, end-stage disseminated intravascular coagulation might take place and lead to bleeding. 44 All these might explain the probability of hemorrhage in patients with COVID-19.
This study had three main limitations. First, it was a retrospective study, which indicated the level of evidence could be improved further. The baseline of the two groups was not the same in some aspects. More smoking, more COPD, lower hemoglobin and higher
Conclusion
VTE and hemorrhage were common in both groups. The predominant type of VTE was distal DVT, which presented a low risk of progression. COVID-19 and ECMO were risk factors for hemorrhage. Blood transfusion with or without surgical/endoscopic treatments was able to manage it in most cases.
Footnotes
Author contributions
Conception and design were performed by H.Z. and C.Q. Analysis and interpretation were performed by C.Q., G.W., J.X., W.Y, Z.W., Y.H., T.C., J.Z., X.H., J.H., J.F. and H.Z. Data collection and final approval of the article were performed by C.Q., T.L, G.W., J.X., W.Y, Z.W., Y.H., T.C., J.Z., X.H., J.H., J.F. and H.Z. The article was written by H.Z., T.L and C.Q. Critical revision of the article was performed by G.W., J.X., W.Y, Z.W., Y.H., T.C., J.Z., X.H., J.H., J.F. and H.Z. Statistical analysis was performed by C.Q., T.L, G.W., J.X., W.Y, Z.W., Y.H., T.C., J.Z., X.H., J.H. and J.F. C.Q. and H.Z. obtained funding. H.Z. took the overall responsibility.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study received special anti-COVID-19 fund by the Education Department of Zhejiang Province (Y202043454).
Ethical approval
Ethical approval for this study was obtained from the First Affiliated Hospital, School of Medicine, Zhejiang University (approval number: IIT20200438A).
Informed consent
Written informed consent was waived due to the urgent need to collect data. Approved within the ethnical approval (APPROVAL NUMBER: IIT20200438A).
