Abstract
Aim:
In the face of increasing tobacco consumption in Sub-Saharan Africa, it is crucial to not only curb the uptake of tobacco, but to ensure that tobacco users quit. Considering the minimal attention that tobacco cessation interventions receive in Sub-Saharan Africa, this review aims to describe studies that evaluated tobacco cessation interventions in the region.
Methods:
A search of studies published till December 2019 that evaluated tobacco cessation interventions in Sub-Saharan Africa and examined tobacco quit rates was conducted in PubMed-Medline, Web of Science and Scopus. Study designs were not limited to randomised control trials but needed to include a control group.
Results:
Of the 454 titles and abstracts reviewed, eight studies, all conducted in South Africa, were included. The earliest publication was from 1988 and the most recent from 2019. Five studies were randomised control trials, two were quasi-experimental and one was a case–control study. Populations studied included community-based smokers (four studies) and university students, while the relevant clinic-based studies were conducted in pregnant women, tuberculosis patients and HIV-infected patients. Sample sizes were 23 in the case–control study, 87–561 in randomised control trials, and 979 (pregnant women) and 4090 (three rural communities) in the quasi-experimental studies. Four studies included nicotine replacement therapy in the interventions while four utilised only psychotherapy without adjunct pharmacotherapy. Quit rates were evaluated by exhaled carbon monoxide levels (five studies), blood carbon monoxide, urinary cotinine levels and self-reported quit rates. Four studies (two each with and without pharmacotherapy) reported significantly better outcomes in the intervention versus the control groups while one study findings (without pharmacotherapy) were significant in women but not men.
Conclusion:
This review highlights that scant attention has been paid to tobacco cessation intervention in Sub-Saharan Africa. The heterogeneity of these studies precluded comparisons across interventions or populations. There is a need for evidence-based low-cost tobacco cessation intervention that target high-risk population in Sub-Saharan Africa.
Introduction
Tobacco is the leading preventable cause of mortality globally contributing to cancer, cardiac disease, stroke, chronic lung diseases and other non-communicable diseases. 1 It accounts for more than eight million deaths annually. In addition, tobacco smoking exacerbates tuberculosis and HIV infection leading to poorer outcomes. 2 Consequently, stopping tobacco use is among the single most effective lifestyle measure to improve health. Substantial evidence shows that smoking cessation reduces mortality from tobacco-related diseases and improves health. 3 Unfortunately, smoking is a powerful addiction and despite numerous quit attempts, many individuals who smoke frequently fail to stop smoking, during which time they are unfortunately losing life-years.1,4 Smoking cessation programmes are therefore necessary to provide the support required for smokers to quit. 5
The World Health Organisation (WHO) has emphasised the importance of providing support for tobacco cessation in its MPOWER measures, which is a practical tool proposed to reduce tobacco use; the ‘O’ is to ‘offer help to quit tobacco use’. 2 Effective tobacco cessation interventions (TCI) have shown to greatly increase the likelihood of successfully quitting tobacco. The probability of successful quitting can be doubled with the use of proven cessation medications and professional support. Therefore, providing access to and encouraging the use of tobacco cessation services should be a critical component of any tobacco control strategy. 2
In 2011, The Lancet Non-Communicable Disease (NCD) Action Group and the NCD Alliance identified tobacco control as the ‘most urgent and immediate priority’ intervention to reduce NCDs, 6 with this tenet echoed at the United Nations High-level meeting on NCDs in the same year. 7 To reduce the global smoking prevalence by 30% by 2025 from a 2010 baseline, countries were urged to fully implement the WHO Framework Convention on Tobacco Control (WHO FCTC). As described in the WHO FCTC Article 14, 1 tobacco cessation is a cost-effective healthcare intervention, and governments and healthcare providers need to provide resources and improve access to programmes to help tobacco users quit.
Furthermore, tobacco control is increasingly considered a vital element for human development because tobacco use contributes to poverty on multiple fronts. These include the cost of purchasing tobacco, healthcare costs for treatment of tobacco-related diseases, and the loss of human capital from tobacco-attributable morbidity and mortality. 2 Consequently, curbing tobacco use has been recognised as important in promoting sustainable development and incorporated within the Sustainable Development Goals (SDGs) 2030 agenda. Comprehensive tobacco cessation measures are among the key initiatives required to achieve the SDG targets on tobacco control. 2
Nevertheless, tobacco use is rising in some countries, especially among vulnerable groups such as women and the youth. This is true in Sub-Saharan Africa (SSA), where the tobacco industry concertedly targets these vulnerable groups. 8 Therefore, in the face of increasing tobacco consumption on the continent, including in women and the youth, it is crucial to not only curb the uptake of tobacco, but it is also essential to ensure that tobacco users quit. There are highly effective and inexpensive TCI that are recommended even in resource-constrained settings such as SSA. 2 A small window of opportunity currently exists, particularly in developing regions, to reverse these trends and decrease the epidemic of tobacco-related morbidity and mortality, given the long delay between smoking uptake and the development of disease.3,9 Therefore, the aim of this systematic review was to examine studies that evaluated TCI in SSA countries.
Methodology
Sources of information and selection of eligible studies
The Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) framework was used for reporting this review, 10 while the Cochrane Handbook for Systematic Reviews of Interventions, version 6.0, was applied when conducting the review. 11 A search of PubMed-Medline, Scopus and Web of Science was conducted of literature published till 5 December 2019. Relevant studies that evaluated TCI in SSA and examined tobacco/smoking quit rates or reductions in tobacco use/smoking were evaluated for inclusion. Study designs were not limited to randomised control trials (RCTs) but needed to include a control group. Key search terms included ‘tobacco cessation’ OR ‘smoking cessation’ OR ‘quit smoking’ OR ‘quit tobacco’ OR ‘stop smoking’ AND ‘Africa’ OR ‘sub-Saharan Africa’. For example, the search string used in PubMed-Medline was as follows: (‘tobacco cessation’ OR ‘smoking cessation’ OR ‘quit smoking’ OR ‘quit tobacco’ OR ‘stop smoking’) AND ( ‘Africa’ OR ‘sub-Saharan Africa’).
Data collection, extraction, assessment and synthesis
Two authors (N.P., A.N. or N.P., M.K.) sequentially screened titles, abstracts and then full texts for inclusion (Figure 1). The literature was screened for any paper that evaluated a tobacco cessation programme in SSA. The outcome examined was tobacco/smoking cessation/reduction in the intervention and control groups following the evaluation of the intervention (differences in prevalence). The outcomes used were those defined in the included studies and comprised self-reported tobacco use/abstinence or biochemically verified changes (urinary cotinine, or blood or exhaled carbon monoxide (CO) levels). In multi-country studies, data pertaining to SSA needed to be specifically reported. Any disagreements regarding the included papers were resolved through discussion or reviewed by a third author (A.P.K.). The reasons for excluding studies were also recorded.

Preferred reporting items for systematic reviews and meta-analysis (PRISMA) diagram.
The data extracted from the selected studies included variables relating to the study design and setting, sample size, participant characteristics, the intervention, training of counsellors, assessments done, and outcomes evaluated. Data extraction was done by one author (N.P.), and another author (A.N.) verified the accuracy and validity of the extracted data. A risk of bias assessment was also conducted using the Cochrane risk of bias assessment tool for RCTs 11 and the Risk Of Bias In Non-Randomised Studies – of interventions (ROBINS-I) tool for the other included studies. 12
Statistical analyses
The heterogeneity of the included studies precluded comparisons across interventions or populations. Therefore, a meta-analysis could not be conducted. A qualitative description of the studies, interventions and outcomes are instead presented.
Results
There were 645 titles retrieved and after removal of duplicates, 452 titles remained (Figure 1). An additional two titles were retrieved after personal communication with the authors, resulting in a total of 454 titles being reviewed. Twenty-seven full texts were evaluated for eligibility and eight, including a conference abstract, were selected for this review. The methodological details of the latter study were obtained from a qualitative paper on the same study. 13
Study settings, designs, populations and sample sizes of included studies
All eight included studies were from South Africa with five studies conducted solely in the Western Cape Province of the country14–18 (Tables 1 and 2). A study each was conducted in Tshwane 19 and Klerksdorp 20 while a single study was multicentred with sites in Cape Town, Johannesburg and Durban. 21 Seven studies included both men and women, and a single study was conducted in pregnant women. 18
Overview of included studies that utilised nicotine replacement therapy (with or without psychotherapy).
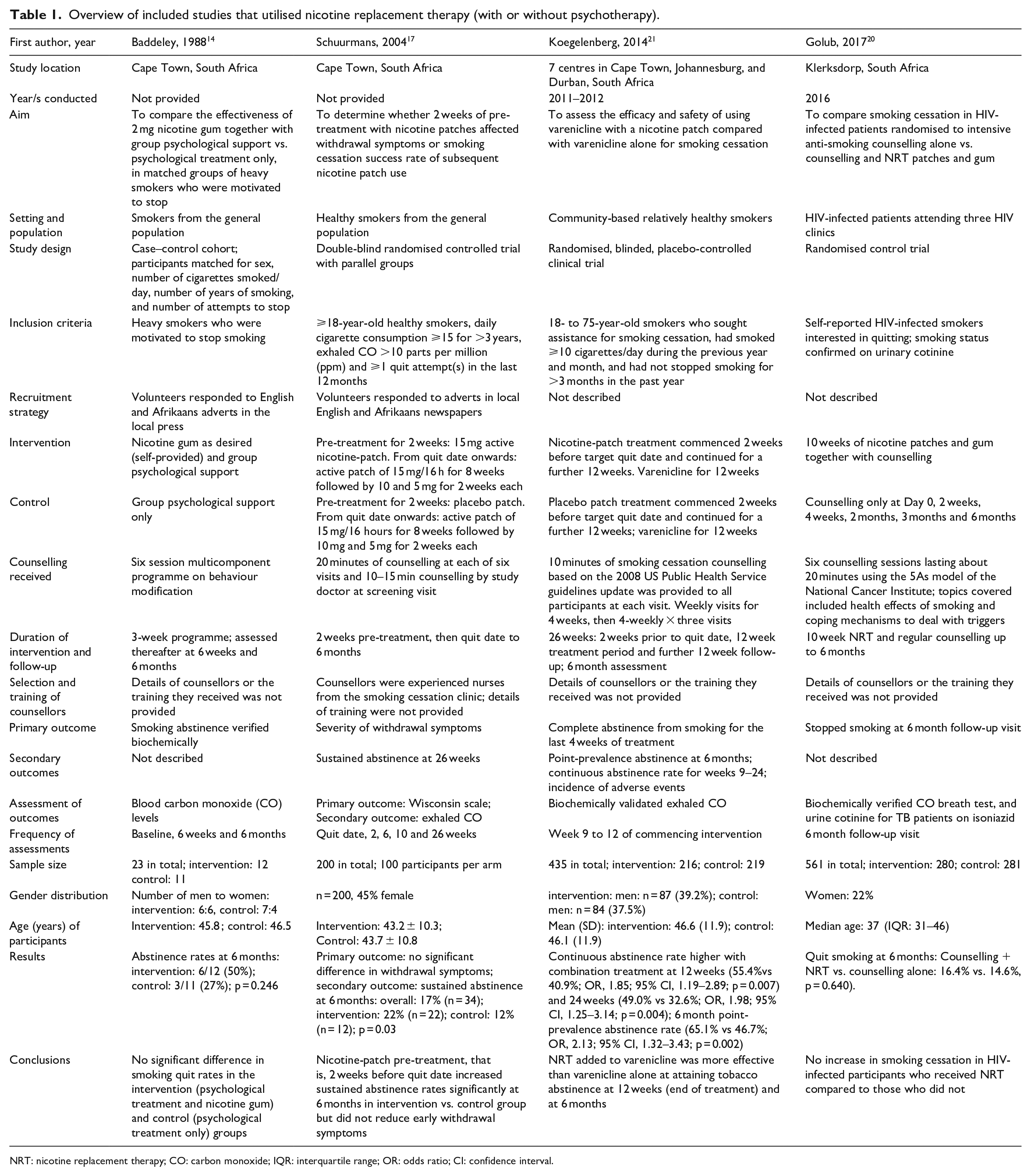
NRT: nicotine replacement therapy; CO: carbon monoxide; IQR: interquartile range; OR: odds ratio; CI: confidence interval.
Overview of included studies that utilised only psychotherapy without adjunct pharmacotherapy.
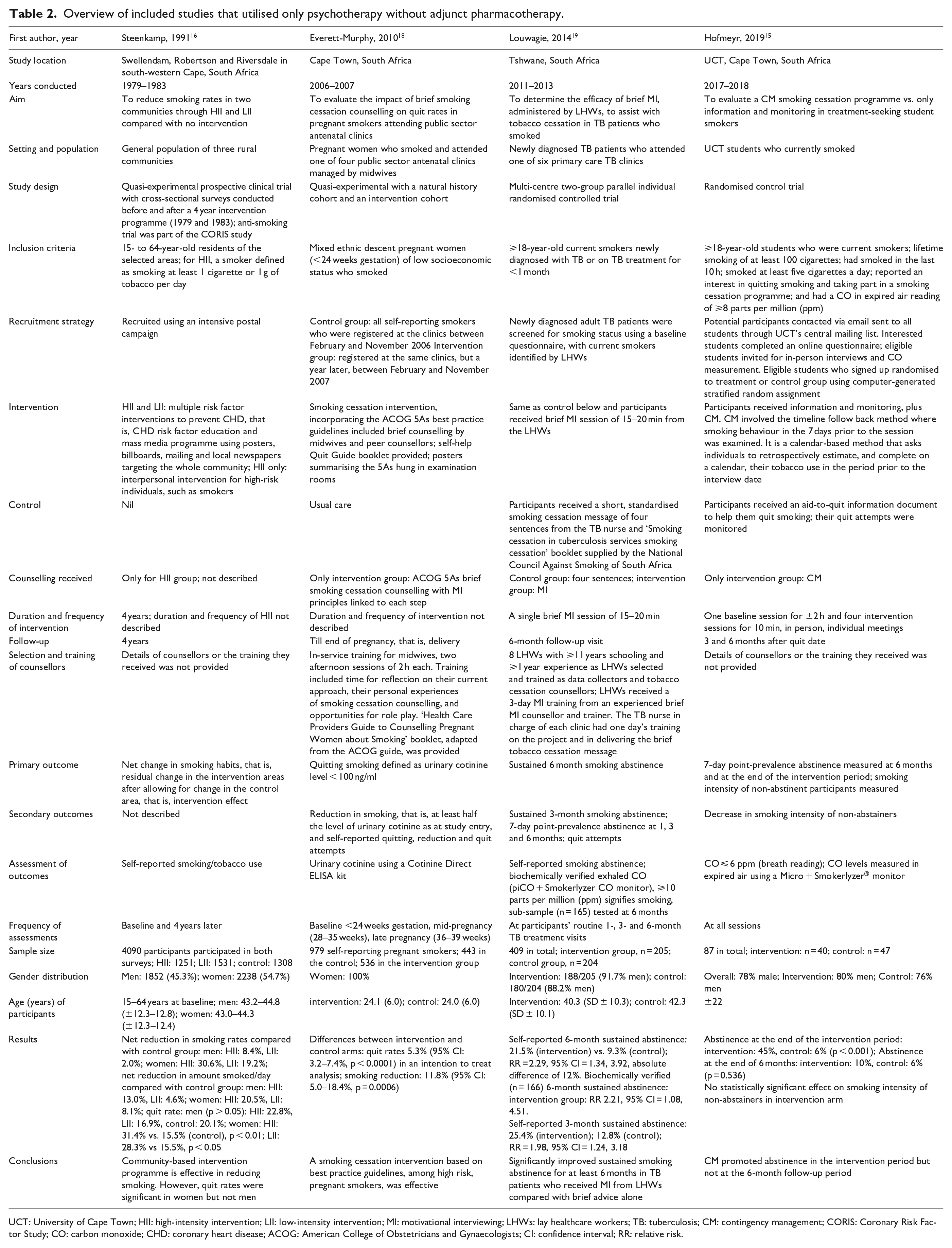
UCT: University of Cape Town; HII: high-intensity intervention; LII: low-intensity intervention; MI: motivational interviewing; LHWs: lay healthcare workers; TB: tuberculosis; CM: contingency management; CORIS: Coronary Risk Factor Study; CO: carbon monoxide; CHD: coronary heart disease; ACOG: American College of Obstetricians and Gynaecologists; CI: confidence interval; RR: relative risk.
Five studies were RCTs,15,17,19–21 two were quasi-experimental16,18 and one was a case–control study. 14 Participants or sites were randomly selected for seven of the eight studies; in the case–control study; however, participants self-selected for the intervention, that is, the use of nicotine gum. 14
Four studies targeted community-based smokers,14,16,17,21 three studies were conducted in patients attending public sector health clinics (one each in antenatal, 18 tuberculosis (TB) 19 and antiretroviral 20 clinics), and one study was conducted in students at the University of Cape Town (UCT). 15 Sample sizes were 23 in the case–control study, 14 87–561 in RCTs,15,17,19–21 and 979 (pregnant women) and 4090 (three rural communities in the CORIS Study) in the quasi-experimental studies.16,18
The sources of heterogeneity in the included studies comprise the wide range of interventions (discussed below), the different study designs and the specific population groups targeted for the interventions. These differences precluded comparability across studies.
Interventions of included studies
Studies that utilised nicotine replacement therapy
Four studies included pharmacotherapy (nicotine replacement therapy (NRT)) in the interventions14,17,20,21 (Table 1). The NRT offered varied across the four studies with two studies comparing NRT and counselling versus counselling alone while the other two studies compared different NRT regimens. These were as follows: (1) group psychological treatment only versus self-provided 2-mg nicotine gum together with group psychological support, 14 (2) intensive anti-smoking counselling alone versus counselling and nicotine patches and gum, 20 (3) 2-week pre-treatment with placebo versus 15-mg active nicotine patch followed by active patch for both groups, 17 and (4) using varenicline alone versus varenicline with a nicotine patch. 21
The intervention periods for the use of NRT were 3 weeks of self-provided nicotine gum used as desired, 14 10 weeks of nicotine patches and gum, 20 two additional weeks of nicotine patches, 17 and 14 weeks of nicotine patches. 21
All four NRT studies included psychological support using different counselling schedules. These included a multicomponent programme on behaviour modification, 14 10-min sessions based on the 2008 US Public Health Service guidelines 21 or 20 min using the 5As model of the National Cancer Institute. 20
All four NRT studies assessed smoking cessation biochemically; blood CO was tested in a single study 14 while the other three examined exhaled CO.17,20,21 Outcomes were assessed at baseline, during the intervention, immediately after the intervention period and after 6 months.14,17,20,21 Sustained abstinence at 6 months was significantly higher in the intervention versus the control groups in two studies17,21 and non-significantly different in the other two NRT studies.14,20
Studies that utilised psychotherapy only
The interventions in studies that utilised only psychotherapy without adjunct pharmacotherapy comprised (1) ‘interpersonal intervention’ (high-intensity intervention (HII)) and/or ‘mass media programmes’ (low-intensity intervention (LII)) versus no intervention, 16 (2) brief counselling and self-help quit materials versus usual care, 18 (3) brief motivational interviewing (MI) versus short, standardised smoking cessation message of four sentences 19 and (4) ‘contingency management’ (CM) versus an aid-to-quit information document 15 (Table 2). In the CORIS Study, the duration of the mass media programmes, that is, the LII was 4 years; however, the duration, frequency or details of the interpersonal intervention, that is, the HII was not described. 16 The smoking cessation intervention incorporating the American College of Obstetricians and Gynaecologists (ACOG) 5As best practice guidelines with brief counselling provided by midwives and peer counsellors was used in the study on pregnant women; the duration and frequency of the intervention was also not described. 18 Lay healthcare workers (LHWs) administered a single brief MI session of 15–20 min in the study in newly diagnosed TB patients. 19 In the UCT study, there was one baseline session for ±2 h and four sessions for 10 min. 15 The latter study used CM which involved the timeline follow back (TLFB) method, where smoking behaviour in the 7 days prior to the session was examined.
Smoking cessation was assessed by self-report in both the CORIS Study, 16 and in the study with TB patients. 19 However, a sub-sample of patients in the latter study had biochemically verified exhaled CO assessments at 6 months. Participants in the UCT study had their CO levels measured in expired air at all sessions. 15 Urinary cotinine levels were assessed in the pregnant women study at three time-points: baseline, mid-pregnancy and late pregnancy. 18
In the CORIS Study, the quit rates after 4 years were significantly higher in women exposed to both the HII and the LII compared with the control group; however, these findings were not significant in men. 16 Quit rates and reductions in smoking were significantly higher in the intervention versus the control group in pregnant women. 18 Sustained smoking abstinence for at least 6 months was significantly higher in TB patients who received MI from LHWs compared with brief advice alone. 19 In the UCT students exposed to CM, abstinence was not significantly higher than the control group at 6 months. 15
In summary, there was no significant difference in smoking cessation in the intervention and control groups in two NRT studies14,20 while two NRT studies reported significantly higher tobacco abstinence at 6 months in the intervention groups.17,21 In studies that utilised only psychotherapy without adjunct pharmacotherapy, smoking cessation was significantly higher in the intervention versus the control groups in pregnant women 18 and TB patients 19 but not in students. 15 The community-based intervention programme was effective in reduce smoking overall but quit rates in women only. 16
Risk of bias
Tables 3 and 4 describe the risk of bias for the RCTs and the non-RCTs, respectively, among the included studies. Among the RCTs, two studies each were at high risk of bias for blinding of participants and personnel (performance bias)15,19 and incomplete outcome data (attrition bias)17,21 while a single study was at high risk of bias for blinding of outcome assessments (detection bias) 19 (Table 3). The overall risk of bias was moderate for the non-RCTs (Table 4).14,16,18
Risk of bias assessment for the included randomised control trials with the supporting evidence.
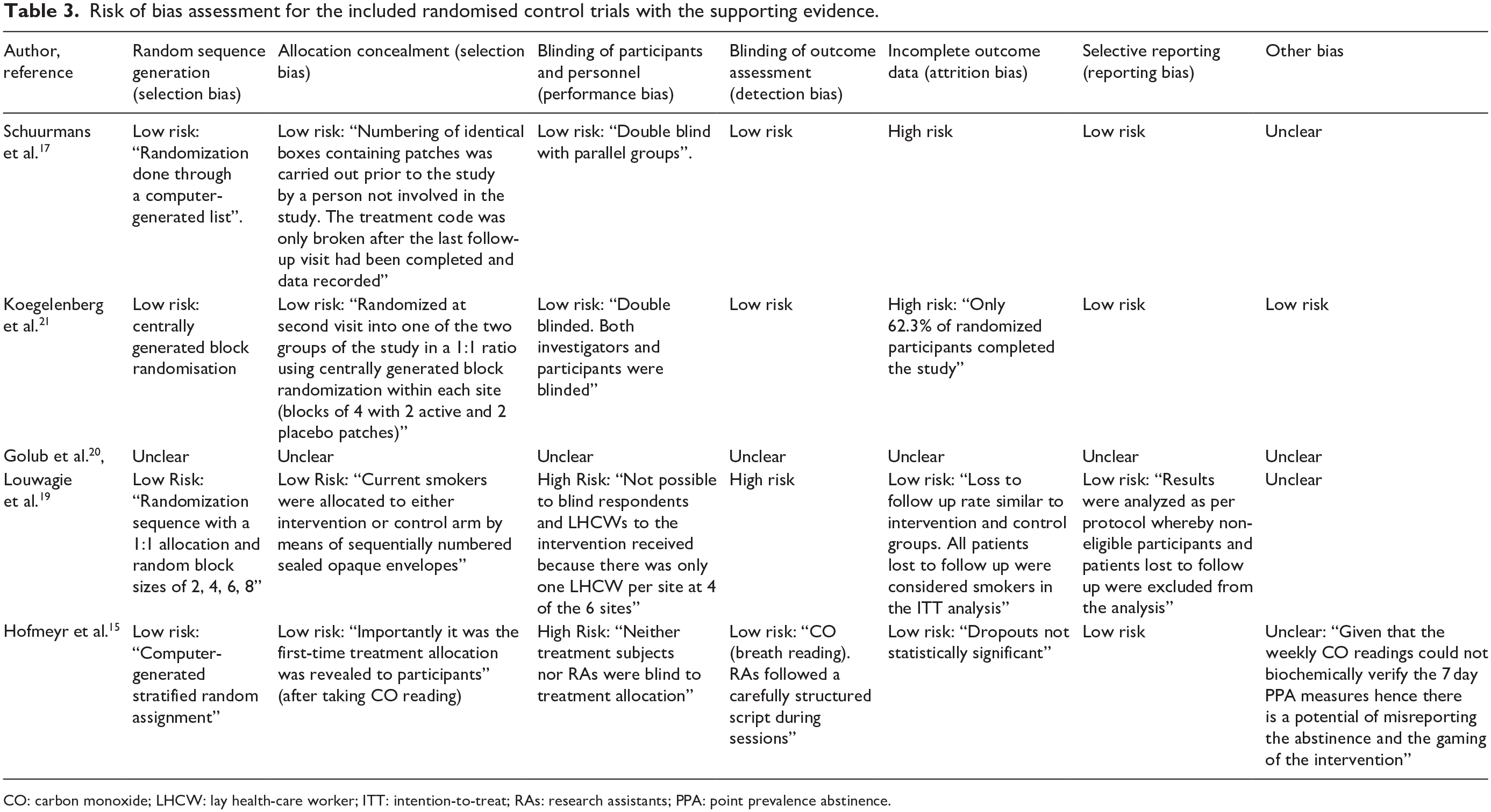
CO: carbon monoxide; LHCW: lay health-care worker; ITT: intention-to-treat; RAs: research assistants; PPA: point prevalence abstinence.
Risk of bias assessment for the non-randomised included studies.

Low risk of bias (the study is comparable to a well-performed randomised trial); moderate risk of bias (the study provides sound evidence for a non-randomised study but cannot be considered comparable to a well-performed randomised trial); serious risk of bias (the study has some important problems).
Discussion
To our knowledge, this is among the first studies to examine TCIs in SSA. As illustrated by the included studies, there is a wide range of behavioural and pharmacological TCIs. Numerous approaches were utilised in the TCIs and these differed in terms of intensity, cost (although not examined in this review) and effectiveness. The NRT studies ranged from the self-selection of nicotine gum in a case–control study 14 to the use of patches and gum in an RCT 20 and pre-treatment with nicotine patches in two RCTs.17,21 Similarly, the studies with psychotherapy as their focus incorporated different techniques and principles. These included the ACOG 5As best practices, brief MI and CM. The different aims, interventions and designs of the included studies precluded comparisons across the studies and a meta-analysis from being conducted.
Of the TCI studies in SSA included in this review, all were conducted in a single country, that is, South Africa. The dearth of TCI studies in SSA is of concern considering that tobacco cessation support services complement other tobacco control initiatives and contribute to decreasing the prevalence of tobacco use. 2 The prevalence of adult tobacco smoking in SSA is significant with the prevalence ⩾15% in seven countries in 2017. These were Lesotho (21%), Sierra Leone (19%), South Africa (17%), Madagascar (16%), Mauritius (16%), Seychelles (16%) and Botswana (15%). 2 Therefore, there is an urgent need in the region for evidence-based TCI for tobacco users who wish to quit.
This is particularly relevant when considering that assisting tobacco users to quit is one of the most cost-effective preventive primary healthcare services. Indeed, of the four studies in this review that utilised only psychotherapy without adjunct pharmacotherapy, three were found to be effective and may be adapted to low-resource settings. These interventions consisted of brief counselling using the ACOG 5As best practice guidelines in pregnant women, brief MI in TB patients, and ‘interpersonal intervention’ (not described) and/or ‘mass media programmes’. Notably, in the study in UCT students, which was not found to be effective, students also received monetary incentives at each assessment if their exhaled CO was ⩽6 ppm. Such an intervention is unlikely to be cost-effective nor sustainable in low-resource settings.
Notably, psychological support using different counselling schedules was a component of all included studies that used NRT, emphasising the importance of psychotherapy in tobacco cessation programmes. Two of these studies tested the utility of psychotherapy with or without NRT and reported no significant differences in quit rates between groups. This possibly further highlights the role of psychotherapy in tobacco cessation. The other two NRT studies tested different NRT regimes and reported significant findings. One study tested the use of an additional 2 weeks of active nicotine patch (14 weeks vs 12 weeks) and the other study the use of varenicline with or without nicotine patches. However, the findings of the latter two studies are unlikely to influence tobacco cessation public health policies in most SSA countries because of the high cost and unsustainable financial implications of pharmacological treatment. Furthermore, out-of-pocket expenditure on pharmacological treatments for tobacco cessation is not feasible for the poor majority residing in SSA because they are not cheap nor affordable. 22 Therefore, tobacco cessation medications are likely to be reserved for upscaling of TCIs when resources are available. SSA countries should follow a stepwise approach when developing their TCIs taking cost and effectiveness of different cessation interventions into consideration. 2
The counselling offered in most studies included in this review comprised intensive face-to-face therapy, which is usually affordable for middle- and high-income countries only. 2 However, in resource-constrained setting, a more pragmatic approach is necessary. This may include the integration of brief advice into primary healthcare setting as an initial step in encouraging tobacco cessation. Importantly, tobacco cessation should urgently be integrated into healthcare programmes such as TB and HIV/AIDS because of worse outcomes reported in smokers as well as family planning and maternal health because of poorer outcomes in pregnant women.23,24 A study each in this review was conducted in patients with TB, HIV and pregnant women; a rollout of low-cost TCIs in these vulnerable groups will likely yield high returns.
Nevertheless, numerous barriers exist to implementing such support. Although tobacco users frequently encounter healthcare providers, they do not receive cessation advice despite the opportunities provided. 24 Barriers to delivering cessation advice among healthcare providers may include their smoking behaviour, lack of knowledge, awareness or motivation, inadequate counselling skills, other urgent priorities and insufficient time due to overburdened clinics. To overcome these barriers, governments need to prioritise the integration of brief cessation advice in primary healthcare settings by incorporating it into healthcare policies and programmes. Equally crucial is to ensure that the training and skills required to impart such advice is provided. In addition, a practical solution to address the multiple demands placed on senior healthcare professionals, who are frequently overburdened and in short supply in SSA, is to shift tobacco cessation counselling to lower-level healthcare workers. 25
It is essential to monitor and evaluate all tobacco cessation strategies and programmes to ensure the adoption of best practices. Therefore, the implementation, efficacy and cost-effectiveness of such programmes will need to be carefully reviewed, that is, both the use of lower-level healthcare workers and the integration of brief tobacco cessation advice into TB, HIV and antenatal services.
This underscores the need for existing healthcare systems to be strengthened to implement tobacco cessation promotion and tobacco dependence treatment initiatives. Unfortunately, SSA countries have a poor record in implementing TCI as illustrated by the following: 2 (1) tobacco use status was routinely recorded on medical records in only three SSA countries (Kenya, Nigeria and Seychelles). (2) Smoking cessation support was offered in some primary healthcare facilities in 10 SSA countries, but the cost was only partially covered in four countries and not covered at all in three countries. (3) NRT was available in 19 SSA countries; in six of these countries, a prescription was required. However, the cost was covered fully or partially in only eight of these countries. (4) Seven SSA countries have a national tobacco cessation strategy and 10 countries have national tobacco cessation clinical guidelines. (5) Only four SSA countries have national toll-free quit lines. 2
This highlights that governments need to invest in promoting cessation, by developing evidence-based cost-effective national strategies and guidelines and allocating adequate resources for programme implementation. In keeping with the findings of this review, they need to promote and provide counselling for those that stop smoking, and implement mass communication programmes that encourage quitting. 1 The latter is important because a lack of knowledge about quit strategies and peer-pressure, among other factors, also contribute to continued smoking or a failure to quit. 4 Moreover, while not reflected in the current review, a plethora of evidence from high-income countries demonstrates the benefits of pharmacotherapy in aiding successful tobacco cessation. Given that the use of pharmacotherapy increases the likelihood of successfully quitting tobacco, 2 SSA governments should strive to improve the availability, accessibility and affordability of cessation medicines.
For optimal effect, governments need to implement such programmes in conjunction with other demand-reduction tobacco control policies. 2 These include higher tobacco taxes, smoke-free spaces, prohibitions on tobacco advertising, promotion and sponsorship, large pictorial health warnings on tobacco packages, and anti-tobacco mass media campaigns. Such messages encourage quitting and create supportive environments.
The limitations of this study are that heterogeneity across studies precluded a meta-analysis from being conducted. The strengths are that three databases were used to search for relevant studies.
Conclusion
This review highlights that scant attention has been paid to TCI in SSA. All included studies were conducted in South Africa only. Furthermore, some of the interventions in the included studies cannot easily be introduced in resource-constrained settings and overburdened healthcare systems. For example, the long duration of the counselling sessions is not feasible for implementation. In others, the lack of accessibility and affordability of NRT in most SSA countries makes such interventions currently impractical and unattainable for most SSA tobacco users. However, NRT and other pharmacological aids to quit smoking can approximately double the chance that an individual will successfully quit.
It is essential to monitor and evaluate all tobacco cessation strategies and programmes to ensure the adoption of best practices. Currently, the limited availability of quality data prevents the implementation of tailored services in SSA. Additional studies are required that examine the effectiveness of best-practice cessation interventions in settings of graded resource availability, that is, from brief advice in primary healthcare to combination pharmacotherapies. Furthermore, there needs to be close collaborations between governments, academic institutions, non-governmental organisations and other stakeholders for the implementation and monitoring of optimal TCI services.
Supplemental Material
PRISMA-2009-checklist--2 – Supplemental material for A narrative systematic review of tobacco cessation interventions in Sub-Saharan Africa
Supplemental material, PRISMA-2009-checklist--2 for A narrative systematic review of tobacco cessation interventions in Sub-Saharan Africa by Nasheeta Peer, Ashika Naicker, Munira Khan and Andre-Pascal Kengne in SAGE Open Medicine
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
