Abstract
Objectives:
Medical oncologists and pharmacists at our institution established an integrated support program aimed at preventing unnecessary treatment interruption or dose reduction during oral targeted therapy with lenvatinib. Here, we evaluated the benefits of this program in managing patients with thyroid cancer receiving lenvatinib.
Methods:
We retrospectively evaluated thyroid cancer patients who received lenvatinib between May 2015 and March 2017. This descriptive study collected records in which pharmacists contributed to changing doctors’ prescriptions and categorized the interventions.
Results:
During the study period, 24 thyroid cancer patients were treated with lenvatinib. Among patients, the incidence of temporary interruption and dose reduction of lenvatinib due to adverse drug reactions was 100% (n = 24) and 83.3% (n = 20), respectively. There were 193 temporary interruptions of lenvatinib due to adverse drug reactions. A total of 501 outpatient pharmacy services were conducted by pharmacists in collaboration with oncologists, of which 125 were interventions (24.9%). In addition, pharmacists conducted 156 telephone follow-up services; 18 (11.5%) of these were to consult an oncologist about a patient’s confirmed problems and resulted in the decision to continue observation with no medical intervention while 41 (26.2%) resulted in the oncologist deciding to temporarily interrupt lenvatinib treatment after the report of an adverse drug reaction from the pharmacist.
Conclusion:
Pharmacist interventions in collaboration with medical oncologists improved lenvatinib therapy. Interventions for outpatients were conducted not only in outpatient clinics but also by telephone follow-up, clarifying the importance of continuous management for patients at risk of adverse reactions and misuse of oral medicine.
Keywords
Introduction
Thyroid cancer accounts for 1% of all cancer cases, and 90% of all endocrine tumors are thyroid cancers. The prognosis of thyroid cancer is favorable, with a 10-year survival rate of 85%. Further, the incidence of distant metastasis is less than 5%, and 10-year survival in patients with metastasis is 25%–42%.1,2 Lenvatinib is an oral multikinase inhibitor that inhibits vascular endothelial growth factor receptors 1–3, fibroblast growth factor receptors 1–4, platelet-derived growth factor receptor a, and RET and KIT signaling pathways.3–5 In the SELECT trial, an international randomized phase 3 study in patients with radioiodine-refractory thyroid cancer, lenvatinib significantly prolonged median progression-free survival to 18.3 months, compared to 3.6 months with placebo, and improved the overall response rate. 6 Accordingly, lenvatinib is now considered the standard therapy for thyroid cancer which is unresectable and refractory to treatment with radioactive iodine.
Nevertheless, a systematic review reported that more than half of patients treated with lenvatinib experienced proteinuria, fatigue, and decreased appetite and that 15%–25% had grade ⩾ 3 adverse effects, including thrombocytopenia, hypertension, and peripheral edema. 7 Indeed, the incidence of temporary interruption or dose reduction of lenvatinib in the SELECT trial 6 was 89.7% in the overall population and 93.3% in the Japanese patients. 8 These findings indicate that the continuation of lenvatinib requires suitable management of adverse drug reactions.
In Japan, hospital pharmacists face difficulties managing oral targeted therapy in the outpatient setting.9,10 The role of hospital pharmacists involves both dispensing and clinical services: licensure for pharmacy technicians has not been established and pharmacists are therefore required to cover both roles. Recently, the Japanese government provided a healthcare reimbursement fee for outpatient pharmacy service, 11 but at a level which does not cover outpatient cancer treatment. Our previous nationwide survey in Japan reported that community pharmacists lack confidence in educating patients about oral chemotherapy because they do not routinely dispense these drugs and are not currently familiar with how they are used, their dosing, or their side effects. 12 In 2007, in response to the increased demand for clinical pharmacy services for outpatient therapy, the Department of Pharmacy at the National Cancer Center Hospital East (NCCHE) established the first Japanese outpatient clinic in which pharmacists work directly with oncologists.13–16 Our previous retrospective survey on pharmacists’ outpatient services for oral targeted therapy with sorafenib for hepatocellular carcinoma showed that most sorafenib misuse occurred at home, and that a telephone follow-up program was effective in improving this misuse. 17 To reduce unnecessary treatment interruption or dose reduction in lenvatinib oral targeted therapy, medical oncologists and pharmacists at the NCCHE established an integrated support program conducted via pharmacists’ outpatient services. In this program, the pharmacist contacts the patient before the medical oncologist’s clinical examination, explains the treatment to the patient after the examination, and conducts telephone follow-up.18,19 (Figure 1) In the oncology field, several reports have shown that interventions by pharmacists in outpatient therapy prevent errors and enable suggestions and provision of more suitable medications.20–22 Because of the substantial heterogeneity in the outcomes of these reports, however, no comprehensive understanding of their benefit in outpatient settings has yet been obtained. 23
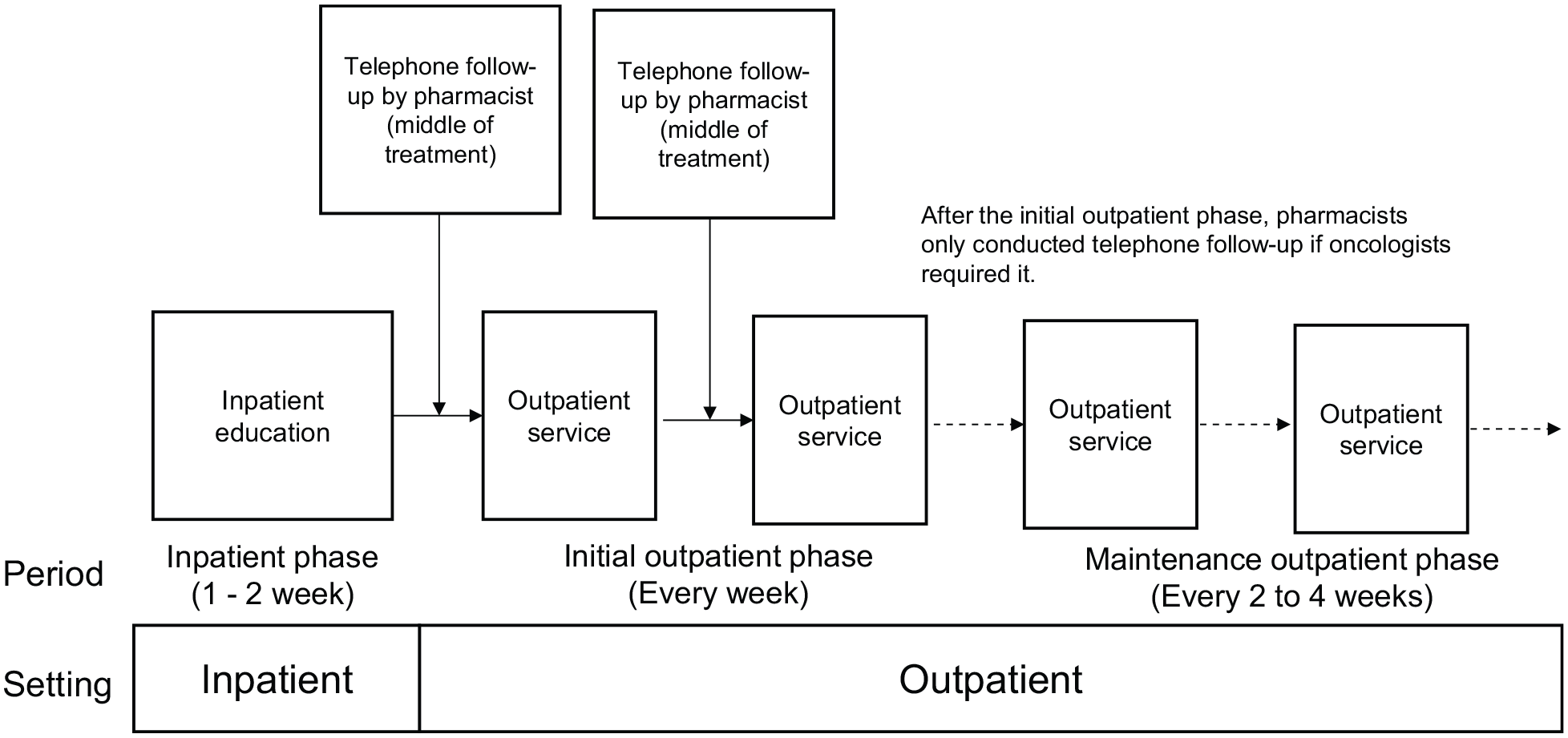
Treatment flow.
Aims
This retrospective cohort study evaluated the benefit of outpatient services conducted by pharmacists in collaboration with oncologists for lenvatinib oral targeted therapy in patients with thyroid cancer.
Ethics approval
Ethical approval for this study was obtained from the National Cancer Center Institutional Review Board (# 2015-251). The Institutional Review Board asserted that informed consent to the study was not required due its retrospective chart review design.
Methods
Subjects and study design
We retrospectively evaluated thyroid cancer patients who received oral targeted therapy with lenvatinib between May 2015 and March 2017. We excluded patients who had previously received lenvatinib in the SELECT clinical trial. 6 All patients started on lenvatinib as inpatients, during which time they were educated about the drug and received an initial evaluation of their response to it. For this study, we focused on outpatient services and did not collect data on the inpatient education phase. After 1–2 weeks of inpatient education, the patients continued lenvatinib in an outpatient setting. In the outpatient clinic, pharmacists worked with patients in the same examination room as oncologists to evaluate adverse drug reactions and medication adherence, and to suggest medications for adverse drug reactions to oncologists. Before the oncologist’s outpatient examination, the pharmacist collected information on the patient’s status, such as adverse drug reactions, adherence, medicines requested by the patient, and medications that the patient took or forgot to take, and then provided the information to the oncologists. During the oncologist’s outpatient examination, the pharmacist suggested prescriptions and provided drug information to the oncologist when requested. After the oncologist’s outpatient examination, the pharmacist checked prescription orders for errors and provided medication counseling to patients regarding their prescriptions. At the mid-point of the patients’ outpatient treatment program, pharmacists telephoned patients and evaluated their adverse drug reactions and medication adherence. To standardize pharmacists’ assessments, oncologists and pharmacists developed a telephone follow-up checklist, and the results of the checklist were recorded in the electronic patient record system (Figure 2). In cases where patients encountered problems due to adverse drug reactions, the pharmacist contacted the oncologist to discuss strategies for managing the patient (Figure 3). Oncologists and pharmacists developed a collaborative flowchart for managing adverse drug reactions, including hypertension, proteinuria, diarrhea, nausea, constipation, oral mucositis, cough, skin disorders, and hand-foot syndrome.18,19 Doctors evaluated the reactions using NCI-CTCAE v4.0. Pharmacists referred to these evaluations and suggested supportive medicines and temporary drug holidays to enable recovery. All activities were recorded in the patient’s electric medical record. In the initial period of outpatient treatment, patients visited the oncologists once a week at the outpatient clinic. After the oncologist confirmed that the patient’s condition had stabilized, the visits were reduced to once every 2–4 weeks, at which time pharmacists also stopped the telephone follow-up, unless required by the oncologist to ensure safe administration of therapy. Pharmacists directly consulted with oncologists during the outpatient sessions, or called them using the in-hospital personal mobile phone system if they were elsewhere in the institution. In our previous study, 15 we investigated the benefits of pharmacist collaboration with oncologists for various types of anticancer agents for four months using pharmacy service records, in which pharmacists recorded their services in daily practice. In this study, however, we investigated the patients’ electric medical records retrospectively to clarify outpatient pharmacy activities. These included (1) pharmacists’ contributions to lenvatinib therapy at outpatient clinics, (2) pharmacists’ telephone follow-up services regarding lenvatinib at home, and (3) pharmacists’ interventions for lenvatinib usage and drug-related problems. The study did not use statistical calculation to justify the number of cases, and was conducted as a retrospective observational study which summarized pharmacy services and interventions using patient electric medical records. Data are described as a “service” for routine work in daily practice and as an “intervention” for activities which affected a patient’s treatment.

Telephone follow-up checklist.

Flowchart of how to manage patients’ adverse drug reactions: example for hypertension.
Data analysis
Descriptive statistics were used to examine the mean number and frequency of prescription modifications due to pharmacy services. Interventions for drug-related problems were categorized using the classification of Cipolle et al. 24 All calculations were performed using Microsoft Excel 2013.
Results
Patient characteristics
During the study period, 24 thyroid cancer patients were treated with lenvatinib. No patient declined to receive the outpatient service or telephone follow-up service. Eight patients (33.3%) were female, median age was 71 years, and all patients had an Eastern Cooperative Oncology Group (ECOG) Performance Status of 0 or 1. The histologic subtypes of thyroid cancer were papillary (n = 17), follicular (n = 3), poorly differentiated (n = 2), and anaplastic (n = 2). Median treatment period was 359 days (range 50–665 days). During the study period, the median initial education period was 12 days (range 6–29) and the median telephone follow-up period after patient discharge was 42 days (range 7–551). Among the 24 cases, the incidence of lenvatinib temporary interruption and dose reduction events due to adverse reactions to lenvatinib was 100% (n = 24) and 83.3% (n = 20), respectively. There was no treatment discontinuation due to lenvatinib adverse drug reactions (Table 1).
Patients’ characteristics.

ECOG PS: Eastern Cooperative Oncology Group Performance Status.
Total number of patients is 24.
Pharmacists’ contribution to lenvatinib oral targeted therapy at the outpatient clinic
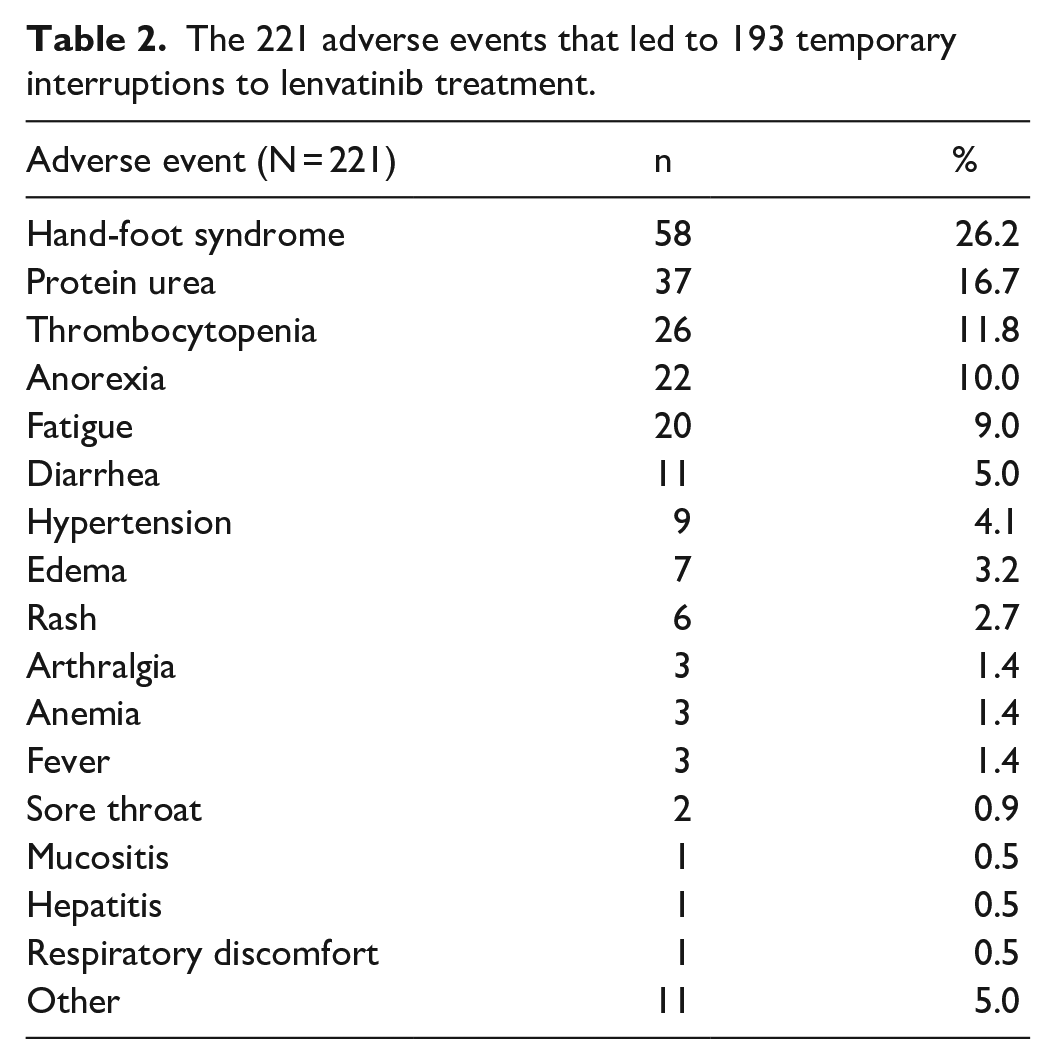
A total of 501 outpatient pharmacy services were conducted by pharmacists in collaboration with oncologists. There were 193 temporary interruptions to lenvatinib treatment, which occurred as a result of 221 adverse drug reactions, primarily hand-foot syndrome (n = 58), proteinuria (n = 37), thrombocytopenia (n = 26), anorexia (n = 22), and fatigue (n = 20) (Table 2). Among the 501 outpatient pharmacy services, 125 were interventions (24.9%), of which 44.8% (n = 56) concerned lenvatinib and 52.0% (n = 65) concerned supportive medications for adverse drug reactions due to lenvatinib, as solely identified by the pharmacist. The majority of interventions that were directly related to lenvatinib concerned lenvatinib misuse (n = 33) and inquiries from the pharmacist about the dose or schedule of lenvatinib (n = 19). Most prescription suggestions by pharmacists were for antihypertensives (n = 33) and thyroid hormone preparations (n = 10) (Table 3). According to the classification of Cipolle et al., 24 the 125 drug-related problems detected by pharmacists through the outpatient clinic were classified as additional drug therapy (n = 65), non-compliance (n = 41), and inappropriate drug use (n = 19).
The 221 adverse events that led to 193 temporary interruptions to lenvatinib treatment.
The 125 interventions among the 501 outpatient pharmacy services conducted by pharmacists.

Pharmacists’ telephone follow-up services for lenvatinib oral targeted therapy at home
During the study period, pharmacists conducted a total of 156 telephone follow-up services. Of these, 28 calls (17.9%) resulted in no action. Meanwhile, in 69 calls (44.2%), pharmacists observed drug-related problems and managed them using the developed flowchart; in 18 calls (11.5%), pharmacists consulted an oncologist about confirmed problems and decided to continue observation with no medical intervention; and in 41 calls (26.2%), oncologists decided to temporarily interrupt lenvatinib treatment after a report of adverse drug reaction from a pharmacist (Table 4). In accordance with the classification of Cipolle et al., 23 the drug-related problems detected by the pharmacists through telephone follow-up were classified as monitoring needed (n = 69), additional drug therapy (n = 18) and inappropriate drug use (n = 41).
Telephone follow-up interventions by pharmacists.

Pharmacists’ interventions for lenvatinib usage and drug-related problems
There were 193 temporary interruptions to lenvatinib treatment during the study period. Among these, pharmacists identified 33 lenvatinib misuse events (17.0%) in which patients made treatment-related decisions without consulting an oncologist or pharmacist. All lenvatinib misuse events were related to lenvatinib adverse drug reactions. Pharmacists also identified four lenvatinib mistake events in which patients misunderstood the oral targeted therapy schedule. In addition, pharmacists identified drug misuse events for antihypertensives (n = 3) and a lipid-lowering drug (n = 1). Pharmacists identified three drug misuse events out of a total of 156 calls during the telephone follow-up service period.
Discussion
In the outpatient clinic, 96.8% (121/125) of interventions conducted by pharmacists involved lenvatinib (56 interventions for lenvatinib and 65 interventions for supportive medications for adverse drug reactions due to lenvatinib). In our study, we focused on lenvatinib for pharmacist intervention, because pharmacists had authority to provide interventions for this agent under a treatment program approved by oncologists. In contrast, we did not include medications for other diseases, such as cardiovascular disease, osteoporosis, and diabetes, as these were prescribed by doctors who worked in different hospitals, and for which our pharmacists did not have authority to change treatment. Although lenvatinib significantly improved progression-free survival in the SELECT trial, 6 a sub-analysis revealed that patients with shorter dose interruptions had a greater degree of benefit than those with longer interruptions. 25 This sub-analysis highlights the importance of the timely management of lenvatinib toxicities to minimize dose interruptions and maximize lenvatinib efficacy. 25 In this study, from 501 outpatient pharmacy services, pharmacists made 125 interventions in patient treatment, most of which concerned lenvatinib and the suggestion of supportive medicine for adverse reactions to lenvatinib. The data clarified that there were cases of oral targeted therapy in which patients had self-assessed lenvatinib discontinuation; doctors had missed a restart or temporary drug holiday in managing adverse drug reactions; and lenvatinib misuse with a fatal outcome. Among the 156 telephone follow-up services conducted by pharmacists, 17% resulted in an oncologist’s decision to temporarily interrupt lenvatinib treatment after a report of adverse reaction from a pharmacist. To evaluate characteristics of the drug related problems, we categorized the interventions using classification of Cipolle et al., 24 and these results clarified that pharmacists improved inappropriate treatment and non-compliance, and suggested supportive medicines to continue the treatment. These findings show that collaboration between pharmacists and oncologists at outpatient clinics improves medication use and drug safety. In addition to the 125 interventions conducted in the outpatient clinic across a median of 359 days, pharmacists conducted 41 telephone follow-up services across a median of 43 days in which they consulted patients on whether or not to continue lenvatinib treatment at home. Several studies have already reported the benefits of telephone services26,27 or mobile application tools 28 in supporting patients under anticancer treatment. Our present findings also support the benefits of telephone service. Although we did not confirm the effects of the remote service on depression in or motivation of patients receiving cancer treatment, other studies have demonstrated benefits for psychological symptoms,29,30 and given the higher frequency and severity of adverse drug reactions in our patients, our intervention also likely benefited psychological symptoms in our patients.26,27,29,30 In the SELECT study, 6 the incidence of overall adverse drug reactions was 97.3%, and included hypertension (67.8%), diarrhea (60.9%), anorexia (51.7%), weight loss (47.1%), nausea (41.0%), fatigue (39.8%), stomatitis (36.8%), proteinuria (32.6%), and hand-foot syndrome (31.8%). In the international phase II study, 31 grade 3 and 4 adverse events were hypertension (10%), proteinuria (10%), diarrhea (10%), weight loss (7%), fatigue (7%), hand-foot syndrome (2%), and anorexia (2%). Our study demonstrated not only an improvement in adverse drug reactions but also a decrease in temporary interruption, treatment discontinuation and misuse of lenvatinib, all of which are related to clinical outcome. Pharmacist suggestions for supportive medicines were mostly for antihypertensive agents, including diuretics. Use of thyroid hormone preparations or analgesics not aimed at adverse drug reactions to lenvatinib but rather due to the thyroid cancer itself were also observed. In the SELECT trial, the incidence of temporary interruptions to lenvatinib treatment was 82% in the overall population and 80% in the Japanese population, while the incidence of dose reduction was 67% in the overall population and 90% in the Japanese population.6,8 Although all patients experienced temporary interruption to lenvatinib treatment in this study, there was no treatment discontinuation due adverse reactions to lenvatinib. These data suggest that temporary drug interruption and dose reduction are necessary to manage adverse drug reactions of lenvatinib oral targeted therapy. Further, while pharmacists identified only three lenvatinib misuse events using telephone follow-up services, they identified 33 such events in the overall period. This suggests that adherence should be monitored throughout the entire treatment period. Our previous report 15 described the benefits of these services for patients who received anticancer agents for all cancer types, but was conducted from June to September 2016 only, and the telephone follow-up service was limited to sorafenib and lenvatinib oral targeted therapy conducted at the National Cancer Hospital East. While our previous study 15 reported a telephone triage service, this study clarified the benefits of long-term care.
This study had a number of limitations, primarily based around the size of the patient population and workplace restrictions. First, the study was conducted at a single center using a retrospective design, and calculation and justification of the sample size was not done. Second, even though the impact of telephone follow-up was significant, it was conducted after the initial outpatient phase only if the oncologist required it, and impact after the initial phase of treatment could not be evaluated. Third, the provision of a telephone service with a single pharmacist at one facility is difficult, and this type of service may not be feasible for many institutions. Finally, the study was restricted to data showing which patients had misused an oral anticancer agent, despite the fact that we did not use valid criteria to evaluate patient medication adherence. Allowing for these limitations, we believe that the information in this study will be useful to others.
Even though the study data were limited to a specific targeted therapy, it clarified the benefits of an outpatient pharmacy service on lenvatinib, which has various adverse drug reactions due to multikinase inhibition. Further study is needed to prove the importance of support for cancer treatment, which requires the balancing of treatment effects and adverse drug reactions. In particular, the use of outpatient cancer treatment and oral anticancer agents has increased, 11 and patients require more outpatient services and remote interventions, such as telephone follow-up services, to control adverse drug reactions due to anticancer agents.
Conclusion
This retrospective study provides evidence that outpatient pharmacy services contribute to improving lenvatinib therapy and that telephone follow-up is useful for identifying severe adverse drug reactions in patients at home. The study also clarified the importance of managing adverse drug reactions and controlling the use of oral targeted medicines administered at home. The application of these data will aid in the safety monitoring of patients on oral anti-cancer agents, and may reduce the occurrence of non-adherence events and adverse drug reactions associated with anticancer treatment in outpatient settings.
Footnotes
Acknowledgements
The authors are grateful to Tomohiro Enokida, MD, PhD; Takao Fujisawa, MD; Yuri Ueda, MD; and Susumu Okano MD, PhD (Division of Head and Neck Medical Oncology, National Cancer Center Hospital East) for their collaboration with pharmacists in developing the outpatient pharmacy service program with the pharmacy department and for their support in improving the study design. The authors also thank Philip E Johnson, MS, RPh, FASHP, FHOPA, for his assistance in writing the paper.
Author contributions
All authors contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the National Cancer Center Institutional Review Board (# 2015-251).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
The National Cancer Center Institutional Review Board determined that Informed consent was not sought and waived to obtain written informed consent for the study due to its retrospective chart review design.
Trial registration
In-hospital registration number is #2015-251.
