Abstract
Despite its beneficial effects, radiotherapy still results in a range of side effects that negatively impact quality of life of patients. Exercise has been shown to counteract the side effects induced by cancer treatment. This narrative review aims to provide an up-to-date review of the effects of an exercise intervention in cancer patients during radiotherapy. A literature search was performed on PubMed to identify original articles that evaluated the effects of an exercise programme to alleviate treatment-related side effects in cancer patients undergoing radiotherapy with or without other cancer treatments. Benefits related to exercise training have been shown in breast, prostate, rectal, lung, head and neck cancer patients undergoing radiotherapy. Therefore, exercise should be considered as a concurrent treatment alongside radiotherapy to alleviate treatment-related side effects and facilitate effective recovery. Due to the onset and progress of treatment-related side effects throughout radiotherapy, a regular clinical evaluation seems strongly advisable in order to continuously adapt the exercise programme depending on symptoms and side effects. An exercise professional is needed to personalize exercise training based on the medical condition and tailor it throughout the intervention according to progress and the patient’s medical status. Future studies are needed to confirm the potential benefits of exercises observed on treatment-related side effects. Furthermore, because of the narrative design of this study, a systematic review is required to evaluate the strength of the evidence reported.
Introduction
Cancer is one of the leading causes of death worldwide, accounting for an estimated 9.6 million deaths in 2018. 1 Radiotherapy (RT) is an important modality for curative treatment in several cancers, either alone or in combination with chemotherapy, hormone therapy, immunotherapy and/or surgery.2–4 About 50% of cancer patients receive RT throughout the course of their illness. 5 Over the past century, progress in this field has resulted in prolonged survival and better control of disease- and treatment-related complications. 6
Despite its beneficial effects, RT still results in a range of side effects that negatively impact quality of life (QoL) of patients. 7 Fatigue is one of the most common symptoms reported by cancer patients undergoing RT, affecting more than 65% of patients.8,9 Cancer-related fatigue (CRF) is defined as ‘a distressing, persistent, subjective sense of physical, emotional and/or cognitive tiredness or exhaustion related to cancer or cancer-related treatment that is not proportional to recent activity and interferes with usual functioning’. 10 CRF progressively increases in severity over the course of RT. 11 This debilitating CRF sometimes persists for months or years after treatment completion, 12 hence the importance of limiting it as soon as possible. Furthermore, patients undergoing RT have frequently reported sleep disorders such as insomnia and excessive daytime sleepiness. 13 Psychosocial functions are also negatively impacted by 30% of RT patients. Depressive symptoms worsen during RT and may persist after RT completion. 14 Weight loss is common in cancer patients during RT and is associated with reduced physical function, muscle strength and overall survival.15–17 In addition, cancer cachexia, which refers to irreversible loss of skeletal muscle mass, is found in many cancers and leads to progressive functional impairment and treatment-related complications. 18 Other tumour-specific side effects related to RT may also occur. Radiation-induced mucositis is common and painful in head and neck cancer (HNC) patients, 19 along with reduction of lean mass (LM).20,21 Esophagitis or pneumonitis occurs in 20% of non-small-cell lung carcinoma (NSCLC) patients undergoing RT. 22 Similarly, gastrointestinal and genitourinary toxicity are common in prostate and rectal cancer undergoing RT.23,24
CRF and the other treatment-related symptoms experienced by the patients during RT reduce physical activity levels, leading the patient into a vicious cycle which is associated with a decline in QoL (Figure 1). 25 In addition, prolonged physical inactivity may cause a decrease in energy and loss of functional capacity. 26

Vicious cycle of fatigue in cancer patients.
Evidence supports the effectiveness of exercise training during anticancer treatment to improve physical function, to reduce CRF, anxiety and depressive symptoms and to increase health-related QoL and symptom control.27–29 A recent meta-analysis with 113 studies including several cancers has demonstrated that exercise is more effective to improve CRF than pharmacological intervention. 30
Whereas most of the studies evaluating exercise during active treatment were carried out during chemotherapy, 31 only a few studies have been focused on the effects of exercise intervention during RT in the last decade. This narrative review aims to provide an up-to-date review of the effects of an exercise intervention in cancer patients during RT.
Methods
A systematic literature search was performed on PubMed to identify original articles that evaluated the effects of an exercise programme to alleviate treatment-related side effects in cancer patients undergoing RT. Exercise programme, which was defined as a subject of physical activity that is planned, structured and repetitive, and has as a final or an intermediate objective the improvement or maintenance of physical fitness, should be composed of either aerobic training or resistance training or combination of both. 32 The exercise programme had to be performed during a course of RT in any setting (in-hospital or home-based). We included studies in which cancer patients received RT for their primary tumour. A concurrent chemotherapy or hormone therapy or a previous surgery was not an exclusion criterion. Articles were excluded if they mixed different types of cancer in their cohort without distinction in the results if the RT was focused on metastases, if subjects had already completed RT or if only a portion of the included participants received RT. Included studies were limited to full text available published in English or French languages from inception to September 2019.
The search strategy used on PubMed included the following terms: ‘neoplasm’, ‘tumour’, ‘cancer’, ‘resistance training’, ‘exercise therapy’, ‘endurance training’, ‘endurance’, ‘physical exercise’, ‘strength training’, ‘aerobic’, ‘concurrent training’, ‘exercise therapy’, ‘physical training’, ‘strength exercise’, ‘resistance training’, ‘progressive resistive’, ‘exercise programme’, ‘strengthening programme’, ‘weight bearing exercise’, ‘aerobic exercise’, ‘radiotherapy’, ‘radical external beam’, ‘X-ray therapy’ and ‘radiation therapy’.
All variables reported in the included studies were analysed. In order to describe the interventions, studies involving the same patients were counted only once so as not to bias the results.
Synthesis of evidence on the impact of exercise intervention by cancer type
Twenty-nine studies were included in this study. The study selection is detailed in the PRISMA flow chart (Figure 2). The detailed characteristics of the included studies are shown in Table 1 and the summary of the outcome measures used is described in Supplementary Appendix 1.

PRISMA flow diagram.
Characteristics of included studies.
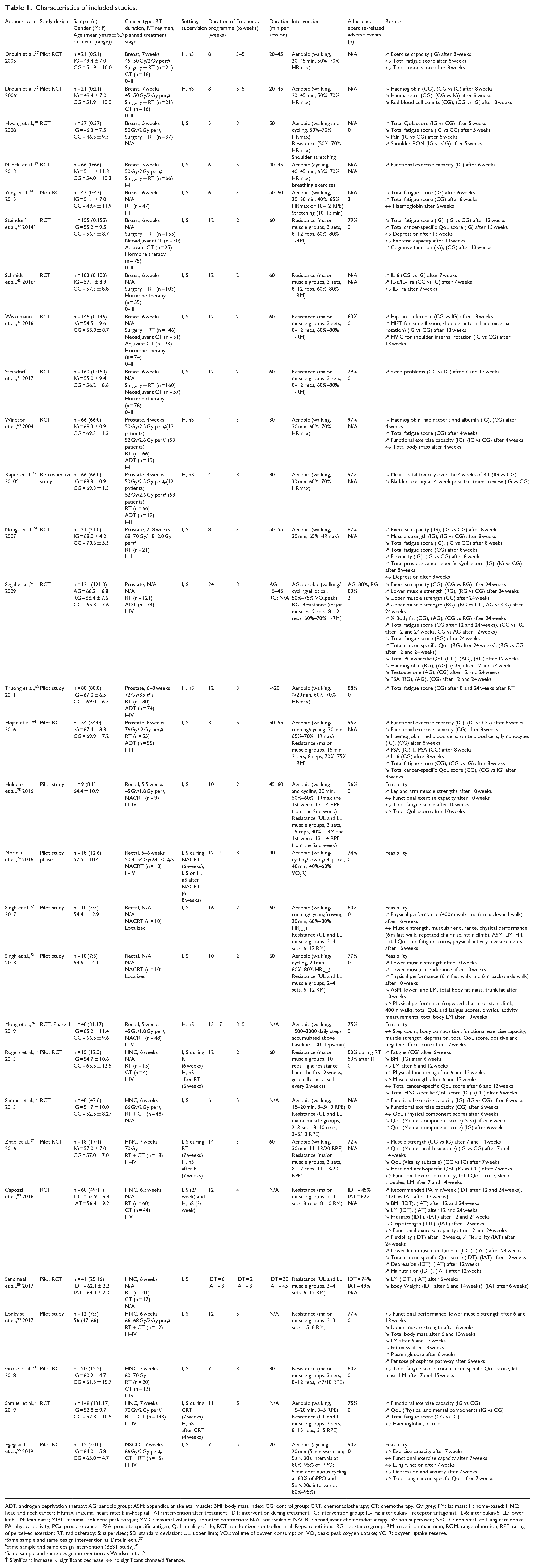
ADT: androgen deprivation therapy; AG: aerobic group; ASM: appendicular skeletal muscle; BMI: body mass index; CG: control group; CRT: chemoradiotherapy; CT: chemotherapy; Gy: grey; FM: fat mass; H: home-based; HNC: head and neck cancer; HRmax: maximal heart rate; I: in-hospital; IAT: intervention after treatment; IDT: intervention during treatment; IG: intervention group; IL-1ra: interleukin-1 receptor antagonist; IL-6: interleukin-6; LL: lower limb; LM: lean mass; MIPT: maximal isokinetic peak torque; MVIC: maximal voluntary isometric contraction; N/A: not available; NACRT: neoadjuvant chemoradiotherapy; nS: non-supervised; NSCLC: non-small-cell lung carcinoma; PA: physical activity; PCa: prostate cancer; PSA: prostate-specific antigen; QoL: quality of life; RCT: randomized controlled trial; Reps: repetitions; RG: resistance group; RM: repetition maximum; ROM: range of motion; RPE: rating of perceived exertion; RT: radiotherapy; S: supervised; SD: standard deviation; UL: upper limb; VO2: volume of oxygen consumption; VO2 peak: peak oxygen uptake; VO2R: oxygen uptake reserve.
Same sample and same design intervention as Drouin et al. 37
Same sample and same design intervention (BEST study). 45
Same sample and same design intervention as Windsor et al. 60
↑ Significant increase; ↓ significant decrease; ↔ no significant change/difference.
Breast cancer undergoing RT
RT after breast-conserving surgery as well as after mastectomy and axillary dissection reduces the risk of recurrence and breast cancer (BC) death.33,34 Unfortunately, radiation in the breast area leads to physical and psychological impairments, including fatigue, pain, muscle strength loss and decrease in functional capacity. 35 Furthermore, some complications including shoulder tissue fibrosis and pulmonary and cardiac toxicities appear months or years after treatment completion. 35 These impairments have a negative impact on daily life activities and QoL. 35
Description of intervention
Nine studies have addressed the effects of exercise training in BC patients undergoing RT.36–44 The detailed characteristics of the BC studies are shown in Table 1. Drouin et al. (2006) is a secondary analysis of data from Drouin et al. (2005); they are therefore considered in the following analysis as a single article.36,37 Four studies include sample of the same population and perform the same type of intervention (BEST study); 45 these interventions are described only one time.40–43 All BC patients, except those in the study by Yang et al., 44 which did not mention the information, underwent surgery followed by RT.36–43 Time since surgery varies from 45 to 70 days.40–43 Four studies are randomized controlled trials (RCTs)36–43 and one is a non-RCT. 44 The intervention groups (IGs) are compared to stretching,36,37 relaxation40–43 or usual care control groups.38,39,44 The delivered total RT dose varies from 45 to 50 Gy in 2 Gy per fraction.36–39
Four studies provide a supervised outpatient hospital-based programme38–44 and one provides a home-based programme with a weekly phone call.36,37 Exercise interventions comprised either aerobic training,36,37,39,44 resistance training40–43 or a combination of the two. 38 Frequency of the intervention varied from two to five times a week over 5–12 weeks.36–44 During the aerobic training, patients walked36–38,44 or cycled38,39 for 20–45 min at mild to moderate intensity, defined by a maximal heart rate (HRmax) of 40%–70%36–39,44 or a rating of perceived exertion (RPE) of 10–12. 44 During the resistance training, patients in the BEST study protocol performed three sets of 8 to 12 repetitions at 60%–80% of repetition maximum (RM).40–43 The remaining study worked at 50%–70% HRmax but did not mention the volume. 38
Effects of intervention
Three studies reported minor adverse events (difficulty in breathing, fatigue and dizziness during aerobic training, 44 and shoulder tendonitis),36,37 but it is important to note that no study reported the onset or increased severity of lymphoedema following exercise during RT, even in resistance training targeting the upper limbs. This is in agreement with the literature beyond the scope of RT, which states that resistance training does not cause or increase the severity of lymphoedema. 46
Fatigue was evaluated in four studies.37,38,40,44 Improvement in overall fatigue was found in the IGs including resistance training, 40 aerobic training 44 or a combination of the two. 38 In the remaining study, the aerobic intervention did not significantly improve fatigue symptoms in the IG in comparison to the control group (CG), although a trend towards improvement in fatigue total score was observed in the IG. 37 However, patients in the IG significantly improved the affective meaning domain of fatigue, while the cognitive/mood domain of fatigue improved significantly and the sensory domain significantly worsened in the CG. 37 A recent meta-analysis including nine RCTs has investigated the effect of exercise on fatigue in BC patients undergoing RT and shows a significant reduction in fatigue in favour of the IG compared to the CG (standardised mean difference (SMD): –0.46, 95% confidence interval (CI) –0.79 to –0.14). 47 Subgroup analyses reported that supervised combined aerobic and resistance training was more effective for fatigue than home-based, aerobic or resistance interventions. 47 Nevertheless, robust conclusions cannot be drawn because some results are based on a small amount of data or a small number of studies. 47
Diverse biological mechanisms have been proposed to explain CRF during RT, including inflammatory biomarkers. 48 The potential benefits of resistance training on inflammatory parameters were explored by measuring interleukin-6 (IL-6) and interleukin-1 receptor antagonist (IL-1ra). 43 The IL-6 and IL-6/IL-1ra ratio significantly increased from baseline to the end of RT in the CG, while inflammatory parameters remained unchanged throughout RT in the IG, where resistance training counteracted the increase in these parameters. 43 Increased levels of IL-6 and IL-6/IL-1ra were significantly associated with increased physical fatigue at the end of RT and 6 weeks later. These inflammatory parameters seem to mediate the positive effect of resistance training on physical fatigue during RT, but there are probably other biological mechanisms involved. 43
Another potential mechanism proposed to explain CRF is anaemia, 48 especially with concomitant chemotherapy, because the relationship between anaemia and CRF in BC patients undergoing only RT is controversial.49,50 A walking intervention showed beneficial effects for anaemia, with a slight non-significant increase in haemoglobin, haematocrit and red blood cell counts in the IG, whereas the CG experienced a significant decline in these three variables and all differences between groups were statistically significant. 36 These findings show that performing a walking endurance training in BC patients during RT may prevent a decrease in erythrocyte levels. A positive correlation was also observed between the change in peak oxygen uptake (VO2) and the post-intervention erythrocyte measures, supporting the relationship between erythrocyte levels and physical fitness during this period. However, these results need to be confirmed, due to the small sample size and the lack of information about dietary intake that may have an influence on erythrocyte levels. 36
QoL is frequently reduced due to cancer and treatment-related side effects in BC patients. 7 The benefits of exercise to improve QoL during cancer treatment are evident, as reported by a Cochrane review. 31 During RT, the benefits are less convincing. A recent meta-analysis investigating the effect of RT on QoL in BC patients showed a medium-sized but non-significant increase in QoL in favour of the IG with large statistical heterogeneity (SMD: 0.46, 95% CI –0.01 to 0.93). 47 However, this meta-analysis included studies with large heterogeneity in terms of exercise prescription (low-intensity mind-body exercises, resistance and/or aerobic training) and planned treatment (RT and/or chemotherapy). Among the studies included in our narrative review, one reports improvement in overall QoL in the IG and decreased QoL in the CG, the differences between groups being significant. 38 Another shows a significant improvement in overall cancer-specific QoL in IG after a 12-week resistance programme. 40 Comparison between groups demonstrated that the further perspective domain increased significantly more in the CG than in the IG, while the role function and pain domains were improved in the IG compared to the CG. 40 This positive result regarding pain is particularly interesting to note because pain is a factor mentioned by patients to explain the decrease in physical activity levels during treatment. 51 Patients are also often worried about worsening their pain with exercise. 40 This pain reduction is supported by another study that observes a significant decrease in pain in the IG compared to the CG. 38
The results of exercise capacity and exercise functional capacity are reported in three studies in which a moderate aerobic intervention showed an increase in exercise capacity (peak VO2: +6.3%) 37 and functional exercise capacity (6-min walk test: +24 m) 39 in the IG, while no change in exercise capacity was reported after resistance training. 40
Isometric and isokinetic muscle strength in knee flexion and shoulder internal and external rotation were improved significantly in the resistance training group compared to the CG. 42 The patients pretreated with chemotherapy showed better benefits performing a resistance training than patients without previous chemotherapy. Concerning surgery-related aspects, strength gain in the upper limb on the operated side improved more than in the upper limb on the non-operated side. 42
Another symptom frequently reported by women with BC during treatment is sleep disturbance. 52 The side effects of RT such as fatigue, pain, depression and impaired physical functioning have been shown to play a role in the worsening of sleep disorders. 53 Steindorf et al. compared the effects of a supervised resistance training to relaxation on sleep disturbance in BC patients receiving RT. At the end of the RT and prolonged intervention, sleep problems decreased significantly in the IG compared to the CG. 41 Twelve months after the end of the treatment, a difference was still observed between groups but was not statistically significant. Previous chemotherapy, depressive symptoms, previous hysterectomy, higher body mass index (BMI), degenerative disorders and thyroid disorders have been identified as determinants of sleep problems at baseline. 41
Psychological impairments are frequently observed in BC patients who undergo RT. Some improvements in the Profile of Mood States, a psychological questionnaire assessing mood and affective states, occurred after a 7-week moderate walking programme. 37 Although the total score did not show any significant difference, the IG showed significant improvement in the domains of depression–dejection and anger–hostility, while the CG improved significantly in the confusion–bewilderment domain from pre- to post-RT assessments. 37
Summary
In conclusion, initiating an intervention exercise throughout RT in BC patients is feasible and safe based on the results of all the nine studies. This intervention may be helpful for CRF,38,40,44 exercise capacity, 37 exercise functional capacity, 39 muscle strength, 42 sleep disturbance, 41 pain 38 and QoL.38,40 An aerobic training may prevent a decrease in erythrocyte levels, 36 while a resistance programme seems to counteract the increase in inflammatory cytokines, 43 which play a crucial role in cancer-related cachexia. 54 Regular exercise training performed during RT may therefore alleviate treatment-related side effects and should be initiated at the same time as the RT-based treatment modality.
A supervised intervention seems to be more effective to reduce fatigue than a home-based setting, as does combined aerobic and resistance training compared to isolated aerobic or resistance training. 47 However, further research is needed to confirm these results, because they are based on a small amount of data (home-based: n < 50) and a small number of studies (one study with resistance training). Future RCTs should also determine the optimal type, frequency and timing of exercise to achieve the greatest benefit in this context.
Prostate cancer undergoing RT
RT is one of the primary treatments for localized and locally advanced prostate cancer (PCa) with or without combined androgen deprivation therapy (ADT). 4 Sexual dysfunction, gastrointestinal and genitourinary disorders have been reported along with fatigue as potential side effects of RT. In addition, ADT is associated with adverse effects including loss of muscle mass and increased body fat, 55 fatigue, 56 sexual dysfunction, 57 increased risk of cardiovascular disease and bone fractures. 58 PCa survivors show a high receptivity for health programmes and therefore represent a target population to make long-term lifestyle changes by initiating regular physical activity during treatment. 59
Description of intervention
Five prospective60–64 studies and one retrospective 65 study performed exercise programmes with PCa patients during RT. Kapur et al. 65 retrospectively analysed patient data from the study by Windsor et al.; 60 these are therefore considered as a single article in the following description of the exercise intervention. The detailed characteristics of the prostate studies are shown in Table 1. Three studies included PCa patients undergoing RT with or without ADT;60,62,63,65 one included PCa patients undergoing RT, all of whom were currently receiving ADT; 64 and one included patients undergoing RT without ADT. 61 The delivered total RT dose varies from 50 to 76 Gy in 20–38 fractions.60,61,63–65 Four studies were two-armed, comparing an aerobic training group60,61,63,65 or an aerobic and a resistance training group 64 to a CG. The remaining study compared three groups: an aerobic, a resistance and a CG. 62 Three of the studies were outpatient hospital-based programmes and supervised,61,62,64 while the other two were home-based.60,63,65 The length of the intervention ranged from 4 to 24 weeks. Training frequency was three times per week in four studies at moderate intensity60–63,65 and five times per week in one study. 64 These sessions lasted between 15 and 55 min.
Effects of intervention
Exercise was associated with high adherence (82%–100%)60–64 and satisfaction 63 and was safe with no adverse events reported, except in one study that mentioned three adverse events related to exercise, including one serious (acute myocardial infarction) during an aerobic session. 62
Fatigue was the most measured variable among studies, being evaluated in five studies.60–64 A significant increase in fatigue was observed in the CG after intervention compared to baseline in all studies,60–64 whereas the fatigue score of the IG showed no change over time in four studies60,62–64 and a decrease in one study. 61 Comparison of 24 weeks of aerobic or resistance training to the CG showed that both IGs mitigated fatigue in the short term, and these improvements were clinically relevant. Resistance training also generated longer-term improvements (at 24 weeks) in fatigue. 62 These findings were confirmed by a pooled analysis of five studies with seven different interventions that showed a significant fatigue reduction in favour of the IGs. 66 Interestingly, physically active patients showed lower levels of fatigue compared with patients physically inactive before, during and after RT. 60
It has previously been demonstrated that RT may alter cytokine responses, this being associated with radiation toxicity. 67 Moderate-intensity aerobic and resistance training decreased levels of inflammatory markers after RT in PCa patients. 64 Indeed, results have shown that an increase in IL-6 levels related to the RT and ADT is significantly reduced in the IG compared to the CG. This emphasizes the crucial role of physical exercise to reduce pro-inflammatory cytokines such as IL-6, which mediate cancer cachexia. 64
QoL was reported in four studies.61–64 A difference between groups in overall cancer-specific-QoL favoured the IG after 12 and 24 weeks of resistance training 62 and 8 weeks of a combination of the two. 64 PCa-specific QoL score improved after 8 weeks of aerobic training 61 while a significant decline was observed after 12 weeks of aerobic or resistance training with a return to baseline after 24 weeks. 62 In contrast to these results, a recent meta-analysis investigated the effects of an exercise programme during RT for PCa on QoL. The pooled results of three studies did not report significant improvement in QoL in favour of the exercise, possibly due to the large heterogeneity between studies. 66
Concerning the effects on physical fitness of exercise intervention during RT, four studies report enhancement of at least one component of it.60–62,64 Improvement in exercise capacity and exercise functional capacity was shown after a moderate walking aerobic training (mean shuttle test distance: +67.5 m (IG) vs –11.5 m (CG) 60 and metabolic syndrome (METS): +2.6 (IG) vs –0.2 (CG)) 61 and a programme combining aerobic and resistance training 64 (6-min walk test: +30 m (IG) vs –24 m (CG)) in the IG compared to the CG. In Segal et al., the resistance group showed significant benefits for cardiovascular fitness compared to the CG. Surprisingly, resistance training preserved aerobic fitness as much as the aerobic training. 62 The authors hypothesize that this unexpected result may be because eight participants in the resistance group performed vigorous aerobic training in addition to their resistance training. 62 In addition, after an 8-week supervised walking programme, differences between groups post-intervention were reported in muscle strength and flexibility in favour of the IG. 61 Muscle strength and body fat percentage also improved after resistance training compared to the CG. 62
Exercise training does not appear to prevent the reduction of blood parameters in men with PCa undergoing RT. Indeed, one study reported a significant decrease in red blood cells, white blood cells, haemoglobin and lymphocytes for the IG and the CG after treatment, 64 and another showed a significant reduction in haemoglobin levels in the middle (12 weeks) and at the end of the intervention (24 weeks) in all groups (aerobic, resistance and CG). 62 Similarly, haemoglobin and haematocrit levels declined for both groups after 4 weeks of RT. 60
Bladder and rectum toxicities are common treatment-related symptoms in PCa receiving RT and may be disabling for patients, negatively affecting their QoL. A retrospective analysis of toxicity data from a previously published study 60 examined the potential effect of a 4-week aerobic training on the reported acute bladder and rectal toxicities in localized PCa patients undergoing RT. 65 The authors report a difference in rectal toxicity score over RT between groups, with lower rectal toxicity in the IG, suggesting that an aerobic programme may reduce the severity of rectal toxicity in PCa patients undergoing RT. The non-significant effect of exercise on bladder toxicity could be explained by the fact that urinary symptoms were present due to the PCa itself at the start of the RT. Therefore, the urinary symptoms measured were the consequence not only of the RT treatment, but probably also of the PCa itself. 65
Summary
An exercise programme performed during RT in PCa patients is associated with high adherence,60–64 satisfaction 63 and seems globally safe. A worsening in CRF, one of the most common side effects reported in PCa patients, is prevented with exercise,60–64 and the level of fatigue reported was inversely associated with the physical activity level. 60 Exercise training also improved exercise capacity, 61 exercise functional capacity,60,64 and muscle strength,61,62 and decreased levels of pro-inflammatory cytokines. 64 Its effect on QoL has not yet been established. Exercise training may also reduce the severity of rectal toxicity, but further research is needed to confirm these results. 65 Therefore, performing an exercise training with PCa patients undergoing RT appears to minimize treatment-related side effects and facilitate effective recovery.
Rectal cancer undergoing RT
National Comprehensive Cancer Network guidelines recommend long-course neoadjuvant chemoradiotherapy (NACRT) as a standard treatment for locally advanced rectal cancer before surgery. 68 This neoadjuvant treatment significantly reduces rates of local recurrence and treatment-related toxicity compared to adjuvant chemoradiotherapy, while overall survival is similar between groups. 69 Despite these benefits, NACRT may lead to various side effects such as diarrhoea, hand-foot syndrome, cardiotoxicity and haematologic toxicity. Physical side effects are also observed after NACRT, including increased fatigue and decreased cardiovascular fitness, exercise capacity and muscular strength.70,71
A reduction in VO2 at the ventilator threshold and VO2 at peak exercise occurs after NACRT. 70 Low cardiovascular fitness reflecting a poor physiological reserve is associated with increased risk of post-operative complications, mortality and recovery time. 72 It has been shown that it is possible to improve preoperative cardiorespiratory fitness by performing a 6-week aerobic training between NACRT completion and surgery. 70 However, as NACRT leads to impaired physical fitness, it would be interesting to start this exercise training as soon as the neoadjuvant treatment begins.
Description of intervention
Five studies have investigated the feasibility and preliminary efficacy of exercise intervention during and after NACRT.73–77 The detailed characteristics of the rectal studies are shown in Table 1. Four were prospective studies with a single group design,73–75,77 and one study was an RCT comparing an IG to a CG. 76 Intervention started at the beginning of the NACRT and is sometimes prolonged between treatment completion and the surgery.74–77 The delivered total RT dose varies from 45 to 54 Gy in 25–30 fractions.74–76 Four studies provided a supervised outpatient hospital-based programme73–75,77 and one provided a home-based programme with follow-up telephone calls. 76 Exercise interventions combined aerobic and resistance training in three studies73,75,77 and included only aerobic training in two studies.74,76 Supervised exercise sessions were performed two to three times a week for 10–16 weeks.73–75,77 During the aerobic training, patients performed walking, running, cycling, rowing or elliptical training for 20–40 min at moderate intensity, defined by HRmax73,75,77 at 50%–80%, by volume of VO2 reserve at 40%–60% 74 or by an RPE of 13–14. 75 During the resistance training, patients performed two to four sets per exercise at 6–12 RM intensity73,77 or three sets of 15 repetitions per exercise at 40% 1-RM. 75 The home-based intervention was composed of aerobic training based on targeted step counts (1500–3000 daily steps accumulated above baseline). 76
Effects of intervention
Results show that performing exercise training during and after NACRT is feasible, with good recruitment and retention rates.73–77 Patients reported a high level of satisfaction with the walking home-based programme during and after NACRT and would recommend it to other patients diagnosed with rectal cancer. 76 Exercise adherence was good to excellent (74%–96%). It was shown that adherence to supervised aerobic training during NACRT was better for patients who were women, younger, married, with better mental health, fewer diarrhoea symptoms, and higher anticipated enjoyment, support and motivation, although the results were not statistically significant. 78 Another study investigated perceived barriers to exercise during and after NACRT. 79 The most common perceived barriers were side effects from NACRT (88%), fatigue (76%) and diarrhoea (71%) during NACRT, and lack of motivation (79%), fatigue (57%) and feeling sick (50%) post-NACRT. 79 No major adverse events related to exercise were reported. Nevertheless, Singh et al. observed bowel activation due to exercise in some patients after the radiation session. 73 They decreased the intensity during the aerobic training to reduce the bowel activation. Other studies did not report this side effect.
After an aerobic training, a small decrease in VO2max (–1.3 mL/kg/min) was observed from pre- to post-NACRT. 74 This suggests that aerobic training during NACRT may prevent a decline in cardiorespiratory fitness. 74 Indeed, a greater decrease in cardiorespiratory fitness (–2.5 mL/kg/min) was noticed without an exercise programme during NACRT in rectal cancer patients. 70 Similarly, a clinically significant increase in functional exercise capacity was shown in two other studies (+46 m to the 6-min walk test 75 and –27.5 s to the 400-m walk test) 77 after exercise intervention. After a walking programme, a non-significant reduction in daily step count from pre- to post-treatment was reported for both groups, but the reduction was less in the IG. 76
A significant improvement in leg and arm muscle strength (+39.2% and +34.9%, respectively) was shown after 10 weeks of aerobic and resistance training (during and after NACRT), 75 while an increase in muscle strength and endurance was reported only in the lower limbs after a 10-week aerobic and resistance training. 73 Although not statistically significant, a 16-week intervention also improved muscle strength by 9%–29% and preserved LM at pre-surgery. 77
Total QoL and fatigue scores did not change throughout the intervention in three studies, suggesting a conservation of these parameters in rectal cancer patients undergoing NACRT.73,75,77
Summary
The current findings show that initiating an exercise intervention during and after NACRT is feasible, safe and well tolerated.73–77 It improves muscle strength and physical performance and prevents a decline in cardiovascular fitness, LM, QoL and fatigue.73–75,77 It is important to point out these findings because patients with better physical fitness could reduce their risk of poor post-operative outcomes and increase recovery. However, it is necessary to remain cautious in view of these results, because four studies were a single group design with a small sample (ranging from 9 to 18). Future RCTs are warranted to determine the efficacy of an exercise training during NACRT.
HNC undergoing RT
RT is delivered alone or in combination with surgery and/or chemotherapy in approximately 75% of all HNC patients. 80 These treatments are associated with acute and late toxicities such as dysphagia, mucositis, trismus, dry mouth, loss of taste, pain, nausea, vomiting and fatigue both during and after treatment that negatively affect the capacity to eat and QoL.81,82 Weight loss is frequently reported in HNC patients during chemoradiotherapy (CRT), with 51% of patients losing more than 5% of their body weight. 15 Muscle mass loss accounts for 72% of this. 20 Following 7 weeks of CRT, Jackson et al. reported a loss of 6 kg in LM 21 despite sufficient intake. 83 This loss is associated with a decline in physical function and QoL, and survival reduction.16,17 Moreover, HNC patients are more sedentary than other cancer patients, which may accentuate this decline in LM. 84
Description of intervention
Eight trials evaluated the effects of an exercise programme in HNC patients during RT with or without chemotherapy.85–92 The characteristics of these studies are summarized in Table 1. The delivered total RT dose varies from 60 to 70 Gy.86,87,90–92 One study was a single group, 90 while seven studies were two-armed, comparing an IG to a CG during RT in five studies85–87,91,92 and comparing IGs exercising either during or after RT in two studies.88,89 Exercise training was delivered in hospital and supervised in four studies;86,89–91 in one study, half of the training sessions were in hospital and the other half home-based; 88 and in three studies, exercise training was in hospital during RT and home-based during follow-up.85,87,92 The exercise intervention consisted of combined aerobic and resistance training in three studies,86,87,92 and resistance training alone in five studies.85,88–91 In addition to the resistance training, a nutritional intervention was added in two studies,85,89 and the intervention in Capozzi et al. was a lifestyle intervention consisting of four other components, including physician referral and clinic support, health education, behaviour change support and social support. 88 The frequency of exercise training ranged from two to five times over 6–14 weeks. Aerobic training consisted of walking between 15 and 30 min at a moderate intensity at 11–13 RPE or 3–5 RPE, as measured by the original and modified Borg scales, respectively.86,87,92 Resistance training targeted major muscle groups in five studies85,87,88,90,91 and upper and lower limb muscle groups in three studies.86,89,92 Patients performed between two and four sets of 8–15 repetitions per exercise. Four trials utilized RPE,86,87,91,92 three studies used the RM, ranging from 6 to 15,88–90 and one study used resistance bands to prescribe intensity. 85
Effects of intervention
Exercise training during RT with or without chemotherapy in HNC patients is safe, well tolerated and feasible,85,89,93 even in cachectic HNC patients. 91 No exercise-related adverse events were reported.85,86,90–92 Adherence to exercise sessions ranged from 45% to 83% during RT and from 49% to 62% after RT. Results of the different studies are described below,85–87,90–92 except for the two studies88,89 comparing groups practising exercise during (intervention during treatment (IDT)) and after radiation therapy (intervention after treatment (IAT)), which are described later in the review.
LM was one of the most evaluated variables.85,87,90,91 A pilot study, in which 12 patients carried out a 12-week resistance programme, demonstrated a significant decrease of 5.1 kg in LM after 6 weeks of RT, as well as a decrease in body weight and fat mass. Patients then started to regain LM while they continued to lose total body weight and fat mass, but despite this LM gain, change in LM from baseline to 13 weeks was still significant. 90 Unfortunately, in the absence of a CG, it is difficult to know if this increase in LM was due to the resistance training. The three remaining studies did not find a significant difference in LM at RT completion or at the end of the intervention in either group.85,87,91
Regarding the six studies that assessed functional exercise performance, the 6-min walk test was used in four studies.86,87,91,92 A 6-week combined aerobic and resistance training during RT significantly improved the walking distance by 42 m in the IG after intervention, while a significant decrease of 96 m was observed in the CG. 86 The authors of this pilot study recently confirmed these results by performing a similar intervention over 11 weeks. The IG reported a significant improvement in functional exercise capacity compared to the CG (+37 m vs –73 m, respectively). 92 In addition to the 6-min walk test, functional performance was assessed using other tests (see Supplementary Appendix 1), but with no significant results.85,87,88,90
Muscle strength and endurance were evaluated using a dynamometer,85,87,91 the 1-RM test, 90 and the sit-to-stand test.85,90 A 12-week resistance training showed that upper muscle strength measured by 1-RM decreased significantly after intervention, while lower muscle strength remained almost the same. 90 Similarly, knee extension strength was maintained in the IG for all 14 weeks of intervention, while the CG declined significantly at 7 and 14 weeks. 87 No group difference was observed in upper limb strength. 87
Fatigue was evaluated in three studies.85,91,92 Aerobic and resistance training prevented increased fatigue in the IG compared to the CG. 92 Likewise, after 6 weeks of resistance training, an increase in fatigue was observed in the CG, 85 and the difference between groups (Total Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F) score: CG: –8.0 and IG: –3.4 at 6 and 12 weeks) exceeds the minimal clinical importance difference of 3. 94 Conversely, one study with cachectic HNC patients found no difference after 7 and 15 weeks of resistance training. 91
QoL was assessed in six studies.85–87,90–92 Among them, four studies showed some significant improvements in QoL with exercise intervention in HNC patients undergoing RT.85–87,92 Using the Short-Form (SF)-36 scale, the mental component score demonstrated an improvement (+12%) in the IG, while the CG showed a decrease (–75%). The physical component score remained unchanged in the IG and decreased non-significantly in the CG. 86 A few years later, the same authors showed that changes in physical and mental components were statistically significant between groups in favour of the IG after 11 weeks of aerobic and resistance training. 92 In addition, a difference between groups favoured the IG in the mental health subscale and vitality. 87 Using the Functional Assessment of Cancer Therapy scale, a small to medium effect size improvement was found, favouring the IG over the CG, for overall QoL, physical wellbeing, emotional wellbeing and functional wellbeing. 85 Surprisingly, the IG reported greater HNC-specific symptoms compared to the CG. This result may be explained by the fact that three of the four patients receiving concurrent chemotherapy were assigned to the IG. 85
Muscle biopsies and proteomic evaluation were performed in a pilot study. 90 Along with LM and muscle strength, sarcomeric protein levels declined during the 6-week CRT and thereafter increased during the remaining 6-week intervention, suggesting that structural changes may cause changes in LM and muscle strength. 90 Better quality studies are needed to confirm these results.
Two studies investigated the ideal time to perform an exercise programme by comparing groups practising during or after RT.88,89 Capozzi et al. compared a 12-week lifestyle intervention including resistance training either IDT or IAT. Greater adherence was reported for the IAT (IDT = 45% vs IAT = 62%). The authors showed a significant decline in LM after RT in both groups (–4.9 cm²/m² for IDT vs –5.4 cm²/m² for IAT) that remained at 24 weeks (–4.5 cm²/m² for IDT vs –4.4 cm²/m² for IAT). 88 They also observed a decline in the distance walked in both groups during the first 12 weeks (–13 m for IDT and –35 m for ADT) that increased by 43 and 18 m, respectively, compared to baseline scores at 24 weeks. It is important to note that this increase from baseline to 24 weeks was not statistically significant, but it was clinically important for the IDT. 88 Concerning muscle strength, the authors showed a significant decline in handgrip strength in both groups through the first 12 weeks (–3 kg for IDT vs –6.7 kg for IAT) with a return to baseline at 24 weeks (+0.2 for IDT and –1.3 for IAT). 88 Both groups maintained functional lower body strength between 0 and 12 weeks, which increased significantly by 24 weeks. 88 Finally, they demonstrated a decrease in the cancer-specific QoL score across the first 12 weeks in both groups that returned to baseline by 24 weeks. 88 The authors suggest that patients should receive education before RT treatment and start the IAT completion. However, this study is limited by the lack of a CG, a resistance programme not physiologically demanding enough, and a small sample size. In addition, another study compared a group performing IDT (2×/week for 6 weeks) versus IAT (3×/week for 3 weeks). Adherence to the resistance programme was higher when exercise was performed during RT (IDT = 74% vs IAT = 49%). The authors explain this difference by the fact that the exercise training probably started too quickly after the end of the treatment (2–4 weeks after RT completion), not allowing the patient time to recover; furthermore, patients took at least 2 h to get to the rehabilitation centre post-treatment and spent the weekdays there. 89 After RT, both groups showed a significant decline of LM, but the decrease was less pronounced in the IDT (–1.7 cm²/m² vs –4.0 cm²/m²; ES = 0.79 in favour of IDT). These results suggest that resistance training during RT may be an effective way to limit muscle mass loss. However, the change in LM from week 6 to week 14 showed a trend towards mitigating LM loss, with a medium effect size in favour of IAT. Therefore, the authors suggested that to minimize the muscle mass loss in HNC patients undergoing RT, exercise training should be initiated at the start of treatment and continue after treatment. Two studies failed to demonstrate such a difference in LM in the IG and the CG either at RT completion or at the end of the intervention.85,87
Summary
Exercise training during CRT in HNC patients is feasible and well tolerated, and no exercise-related adverse events were reported, even in cachectic HNC patients.85,86,90–92 Improvement was shown for functional exercise capacity86,92 and some domains of QoL.85–87,92 Exercise training may also prevent a decline in lower muscle strength 87 and an increase in fatigue.85,92 Concerning the preservation of LM, which is a major challenge for these patients during and after RT, the current findings are not strong enough to affirm the benefit of resistance training to maintain or increase LM. Futures RCTs should be conducted to determine the optimal type and intensity of exercise to counteract the side effects of RT, especially in relation to LM, in HNC cancer patients.
The current studies do not highlight the optimal timing (during or after RT) to initiate an exercise programme, especially for maintaining or increasing LM, and future RCTs should be conducted to determine the optimal timing.
NSCLC undergoing RT
RT with concurrent chemotherapy is a standard of care in locally advanced unresectable NSCLC. 3 Although RT techniques have improved in recent years, acute and late toxicities still occur, such as fatigue, oesophagitis, radiation pneumonitis, and haematologic and pulmonary toxicities. 22 Exercise training during RT is poorly studied in this type of cancer.
Description of intervention
A recent randomized controlled feasibility study focuses on pre-RT daily exercise training in locally advanced NSCLC. 95 All patients received the same total dose of 66 Gy in 33 fractions. Fifteen patients were randomized either in the CG or in the IG. The exercise programme consisted of 20 min moderate- to high-intensity aerobic interval training on a cycle ergometer over a 7-week period. Each session was supervised and performed daily prior to RT.
Effects of intervention
The study demonstrates that daily moderate- to high-intensity aerobic exercise is feasible, safe and well tolerated in patients with locally advanced NSCLC undergoing concurrent CRT. 95 The recruitment rate was 44% and the overall attendance rate to exercise was 90%, with an adherence rate to full exercise participation of 88%. No adverse event was observed during the exercise sessions. No secondary outcomes, including VO2peak, functional exercise capacity, pulmonary function and psychological parameters, demonstrated statistical differences. These results might be explained by the small sample size or by the fact that although the CG did not receive any exercise programme, it performed more steps per day (7572 ± 2445 steps), monitored by an activity tracker, than the IG (6254 ± 2337 steps).
Summary
Moderate- to high-intensity exercise training is feasible, safe and well tolerated in NSCLC patients undergoing RT. 95 Due to the limited number of studies available, more studies are needed to determine the safety and effectiveness of exercise training during RT.
Discussion and experts’ opinion
This narrative review aims to provide an up-to-date review of the effect of an exercise intervention in cancer patients during RT. In the last few years, progress has been achieved in RT treatment and it has provided many benefits to patients. Unfortunately, however, RT still results in a range of acute and late toxicities. Most of these side effects are specific to the irradiated area. The objectives targeted by performing exercise therefore differ somewhat between the types of cancers due to the tumour-specific side effects and care path (e.g. concurrent chemotherapy or ADT or future surgery).
Current literature recommends being as physically active as possible during cancer treatment. 96 However, patients undergoing RT significantly decrease the number of days spent exercising and the duration of exercise. 51 Reasons given by the participants to explain this decline are various and include lack of energy, tiredness, fatigue, pain, shortness of breath on exertion, decreased motivation or time constraints. Cancer care practitioners should inform patients of the benefits of exercise during cancer treatment to alleviate this decline in physical activity. A survey of 15,524 colorectal cancer patients yielded that only 31% received physical activity advice from the healthcare professionals. 97 Patients with cancer showed an interest in receiving information and advice about exercise. Indeed, a study reported that 71% of patients treated by RT indicated that they were interested to receive information about performing exercise during treatment. 51 In another study, written information on CRF and the capacity of exercise to manage it were provided to cancer patients: results showed that 70%–78% of patients found the information provided helpful or very helpful, 88% reported exercising during treatment, and 89% were still exercising 4–6 weeks after treatment completion. 98 It has previously been shown that if this information about exercise is provided by healthcare professionals, the potential benefits should be greater than if it is provided by an unqualified person. 99
Patients performing exercise training during RT reported high satisfaction, felt better, were more energetic and were able to face the treatment more easily. 76 They would recommend the programme to other patients undergoing RT. 76 An exercise programme for cancer patients during RT is generally safe. Only one study reported a serious exercise-related adverse event during aerobic training for a PCa patient, 62 but caution must be exercised because patients at risk were generally an exclusion criterion in the studies. To deliver exercise safely, medical clearance before exercise is indicated in diverse situations reported by the consensus statement from the international multidisciplinary roundtable based on the National Comprehensive Cancer Network. 29
This narrative review has shown benefits related to exercise training in breast, prostate, rectal, NSCLC, HNC patients undergoing RT. Exercise may be considered as a concurrent treatment alongside RT to alleviate treatment-related side effects and facilitate effective recovery. Future studies are needed to confirm the potential benefits of exercise observed on treatment-related side effects. In addition, initiating regular physical activity at the start of treatment may be a ‘teachable moment’ to make long-term lifestyle changes and thus provide long-term health benefits. We observed this maintenance in a study including HNC patients: the recommended weekly physical activity was significantly increased during the 12-week intervention, and this change was maintained after the intervention. 88
It is important that adherence to the exercise programme is high to potentiate the effects of exercise. Physical and psychological impairments caused by the cancer and its treatment, as well as patients’ concerns about their future or employment and family issues, may affect the adherence to exercise during RT. 29 Current literature reports that supervised exercise programmes show better results and adherence.100,101 In this review, supervised and non-supervised programmes show similar adherence (74%–96% vs 72%–97%, respectively). This good to high adherence associated with unsupervised training may be explained by the fact that patients received weekly telephone calls during the intervention. Activity levels, medical status, preferences and barriers of the patients must be taken into account in determining the exercise programme to maximize adherence. In addition, patients’ symptoms may change from day to day and from week to week, so a clinical re-evaluation seems strongly advisable in order to adapt the exercise programme depending on symptoms and side effects. Therefore, an exercise professional is needed to personalize exercise training and tailor it throughout the intervention according to progress and the patient’s medical status.
Most of the exercise interventions were provided at moderate intensity. However, studies on high-intensity exercise (HIE) training in patients with cancer have recently arisen and they show promising results. HIE is a safe and feasible method during cancer treatment, 102 and it has been shown to improve cardiorespiratory fitness, strength, body mass and body fat significantly more than usual care and/or moderate-intensity continuous training in cancer survivors.103,104 Furthermore, these improvements in cardiorespiratory fitness are more sustainable compared to low-intensity exercise. 105 In contrast, a systematic review with a meta-analysis including only studies with aerobic training showed significantly larger improvements in cardiorespiratory fitness in HIE compared to the CG, while no additional benefit of HIE was found compared to moderate-intensity continuous training. Nevertheless, while lack of time is a reason reported by patients for decreasing the time spent exercising during radiation, 51 HIE may be a time-efficient strategy to improve health outcomes. 104 During RT, only one study reported the feasibility of a moderate- to high-intensity aerobic interval training in NSCLC, with an excellent retention rate of 100%, an overall attendance rate to exercise of 90.0%, and no adverse exercise-related events. 95 However, no secondary outcomes showed a significant difference from baseline to post-intervention, probably because of the small sample size (n = 15). 95 Therefore, future high-quality research is warranted to investigate the effects of HIE during RT in various types of cancer.
Some studies performed a non-conventional intervention during RT. Yoga led to reported improvement in fatigue 106 and in some aspects of QoL106,107 compared to the CG in BC patients undergoing RT. Qigong programmes improved fatigue and overall QoL in BC patients with high depressive symptoms at RT onset 108 compared to the CG, while qigong/tai chi improved sleep duration at mid-RT compared to light exercise and wait control groups in PCa patients undergoing RT, but this improvement was not sustained over time. 109 Due to the limited data available, more studies exploring the effects of mind-body programmes should be performed to determine the effectiveness of mind-body programmes to improve RT-related side effects.
All studies evaluated the patient before and at the end of the intervention, but only few studies included a follow-up period.41,63,75,77,90,91 Research with a longer follow-up is needed to identify the long-term effects of exercise throughout RT.
Some side effects and symptoms have been reported to influence the tolerance and safety of exercise programmes during RT, in addition to the potential side effects induced by other types of treatment (e.g. chemotherapy, surgery, ADT). 29 These different side effects and symptoms are presented in Figure 3.

Side effects and symptoms influencing the tolerance and safety of exercise during radiotherapy.
It is important that physiotherapists administering exercise programmes have knowledge of the common approaches to treat cancer and the treatment-related side effects and symptoms that are specific to each treatment and cancer type. 29 Exercise professionals should work closely with the radiotherapist and other team members in order to be continuously informed of the patient’s clinical progress and possible therapeutic changes during treatment. Weekly multidisciplinary discussion would be useful. In addition, they must be able to identify signs that require referral to a healthcare provider.
Some relative contraindications are important to consider for exercise programmes during RT. Radiation-induced cardiotoxicity, especially in lymphoma, breast and lung cancers, requires greater monitoring during exercise and tailored exercise prescription. As this cardiotoxicity may occur several years after irradiation, 110 prolonged and strict monitoring is necessary. Radiation pneumonitis may occur 1–3 months after RT completion in patients irradiated in the chest area and lungs 111 and may alter lung function. Continuous monitoring is crucial to refer the patient if symptoms appear. Patients with bone metastases should minimize load on the affected sites. 29 The safety and feasibility of an isometric resistance training during RT in patients with spinal bone metastases have been demonstrated, 112 showing enhanced bone density in the metastases after 3 and 6 months compared to a passive physical therapy, with no increase in pathological fracture rate. 113 RT increases the risk of infection by affecting the immune system. 114 Patients have to reduce the risk of developing an infection by washing their hands frequently and having good hygiene, and avoiding public places if their white blood cell count is low. 115 Importantly, there are no related data about infection acquired during an exercise programme. Swimming in a pool is contraindicated due to the increased risk of infection and skin irritation in the irradiated area. 116
This narrative review has some limitations to mention. First, participants in the majority of studies did not receive only RT for cancer treatment. Although these combinations in treatment have probably influenced the results, we decided to extend the review to all studies in which all participants received RT with or without other cancer treatments (chemotherapy, hormone therapy or surgery) because that reflect more closely the reality due to the guidelines for treatment of cancers. Second, almost 40% of studies did not report the total dose received by the body during RT. However, among studies reporting this data, the delivered dose was the same for all study participants, except for one study. 60 We suggest that future studies should report these data. Nevertheless, although there is heterogeneity in the dose delivered between some studies, it has been reported that the total dose of RT did not predict variance in fatigue severity. 117 Furthermore, because of the narrative design of this study and even if we closely followed the PRISMA checklist (see Supplementary Material) in our process, a systematic review is required to evaluate the strength of the evidence reported.
Conclusion
The findings in this narrative review show that exercise training is beneficial during active RT treatment and appears to be an effective and crucial component to counteract the side effects induced by RT. In view of the value of exercise during RT to manage treatment-related side effects, exercise programmes should be incorporated as a routine part of cancer patient care during RT, similar to cardiac and pulmonary rehabilitation. Exercise prescription should be individualized depending on the patient’s characteristics, cancer type, treatment prescribed and related toxicities. A tailored approach is needed throughout intervention, according to progress and to the evolution of the patient’s medical status. Further research with the following aims is needed: (1) to determine the optimal type, frequency and timing of exercise, (2) to compare various intensities of exercise training to ascertain the most suitable, and (3) to identify the long-term effects of exercise performed during RT.
Supplemental Material
Appendix_1 – Supplemental material for Effects of exercise therapy in cancer patients undergoing radiotherapy treatment: a narrative review
Supplemental material, Appendix_1 for Effects of exercise therapy in cancer patients undergoing radiotherapy treatment: a narrative review by Elise Piraux, Gilles Caty, Frank Aboubakar Nana and Gregory Reychler in SAGE Open Medicine
Supplemental Material
Attached_file__PRISMA-2009-Checklist-MS-Word – Supplemental material for Effects of exercise therapy in cancer patients undergoing radiotherapy treatment: a narrative review
Supplemental material, Attached_file__PRISMA-2009-Checklist-MS-Word for Effects of exercise therapy in cancer patients undergoing radiotherapy treatment: a narrative review by Elise Piraux, Gilles Caty, Frank Aboubakar Nana and Gregory Reychler in SAGE Open Medicine
Footnotes
Acknowledgements
The authors thank Dr Benjamin Ledoux for his help in manuscript writing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: E.P. is funded by a grant from the Fonds National de la Recherche Scientifique (FRIA–FNRS). F.A.N. is supported by Télévie (Fonds National de la Recherche Scientifique (FNRS)) (7.4624.15); Fonds Spécial de Recherche (FSR) (Communauté Française de Belgique); Fondation Willy and Marcy De Vooght; and Fonds de Recherche Clinique (FRC) from the Université catholique de Louvain (UCLouvain), Belgium. G.R. received a grant from the Institut de Recherche Expérimentale et Clinique (Université catholique de Louvain, Brussels, Belgium).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
