Abstract
Ankaferd hemostat (Ankaferd blood stopper [ABS], Istanbul, Turkey) is a hemostatic agent affecting red blood cell-fibrinogen interactions. ABS has been traditionally used in Anatolia as a hemostatic agent for centuries. ABS contains a standardized combination of the plants namely Glycyrrhiza glabra, Thymus vulgaris, Alpinia officinarum, Vitis vinifera, and Urtica dioica. The hemostatic effect of ABS depends upon the quick promotion of a protein network, particularly fibrinogen gamma, in relation to the erythrocyte aggregation. The aim of this review is to indicate pharmacobiological basis and clinical backgrounds of ABS. Current perspective for using ABS is to provide hemostasis and accelerating wound healing particularly in cases which are difficult to manage. Future controlled trials are needed to elucidate the actions of ABS with in hemostasis, antithrombotic, anti-inflammatory, anti-infective, antifungal, and anti-oxidative effects.
Introduction
Ankaferd hemostat (Ankaferd blood stopper (ABS), Istanbul, Turkey) is a hemostatic agent affecting red blood cell–fibrinogen interactions. ABS had been traditionally used in Anatolia as a hemostatic agent for centuries. 1 ABS shows the hemostatic effect via affecting the physiology of the red blood cells. 1 ABS contains a standardized combination of the plants namely Glycyrrhiza glabra, Thymus vulgaris, Alpinia officinarum, Vitis vinifera, and Urtica dioica.2,3 Recently, ABS has been established as a novel topical hemostatic agent for the management of clinical hemorrhages if the conventional methods were ineffective to control bleeding.4–6 ABS is as effective in the management of the patients with primary or secondary hemostatic deficiencies as in the bleeding individuals with normal hemostatic parameters.7,8 ABS has an expanding spectrum of clinical indications. Current state of ABS development as a drug is depicted in Table 1. The aim of this article is to indicate pharmacobiological basis and clinical background of ABS.
Ankaferd blood stopper (ABS) as a drug (current state of drug development in distinct pathobiological states).

Basic Ankaferd hemostat (ABS) effects on hemostasis
The hemostatic efficacy of ABS depends upon the quick promotion of a protein network, particularly fibrinogen gamma, in relation to the vital erythrocyte aggregation. 1 ABS induces the constitution of a complex protein network with vital erythroid aggregation covering the whole physiological hemostatic process. 9 The essential effects of ABS are ascribed to the formation of protein network and polymerization modulating the erythroid aggregation and vascular endothelium. Moreover, there are distinct mechanisms affecting hemostatic plug formation. In the presence of ABS, vital erythroid aggregation occurs in conjunction with the spectrin and ankyrin receptors on the red blood cell membranes. 9 ABS contains essential erythroid proteins such as spectrin-alpha, actin depolymerization factor, NADH dehydrogenase (ubiquinone) 1-alpha subcomplex, mitochondrial NADP[+] dependent malic enzyme, and the required adenosine triphosphate (ATP) bioenergy source. 1 ABS up-regulates the GATA/FOG transcription system via affecting erythroid functions and urotensin-II. 3 Urotensin-II is an important component of ABS. Urotensin-II links between the injured vascular endothelium, adhesive proteins, and activated erythroid cells. 1 ABS also affects different regulator molecules such as protein-2 (AP2), androgen receptor (AR), cyclic AMP response element or activating transcription factor-1 (CRE-ATF1), cyclic AMP response element binding protein (CREB), E2F1-5, E2F6, interferon (IFN)-stimulated response element (ISRE), Myc-Max, nuclear factor-1 (NF-1), protein-53 or tumor protein-53 (p53), peroxisome proliferator-activated receptor (PPAR), and Yin Yang-1 (YY1) transcription factors. Those regulatory molecules affect cellular vascular hemostasis, angiogenesis, signal transduction, apoptosis, inflammation, acute-phase reaction, immunity, and several metabolic pathways. 10 Coagulation proteins (Factors II, V, VII, VIII, IX, X, XI, and XIII) were not directly affected by ABS in vitro. Likewise, prothrombin time (PT) and activated partial thromboplastin time (aPTT) were normal during the application of ABS. However, thrombin time (TT) was prolonged in relation to the fibrinogen gamma. 1 On the contrary, a recent hemorheological study showed that ABS has anti-erythroid aggregation effect. ABS inhibits pathological aggregation of red blood cells. Antithrombotic clinical effects of ABS could be ascribed to the paradoxal anti-erythroid aggregation actions of the drug. 11
Ankaferd hemostat (ABS) effects on cellular biology
Ankaferd hemostat in cancer
The anti-neoplastic effect of ABS was preliminary defined by Goker et al. 12 They investigated the anti-neoplastic effects of ABS on SAOS-2 osteosarcoma cell survival and growth. A dose-dependent inhibition in cell proliferation and a marked decrease were observed in the survival of SAOS-2 cells. In another study, the anti-neoplastic effects of ABS on colon cancer cells were defined. 13 Following the addition of ABS to the culture medium, the inhibition of cellular reproduction and loss in the viabilities of human colon CaCo-2 cells were observed. Akalın et al. showed the anti-proliferative effects of ABS on lymphoid neoplastic cells (B-CLL and RAJI tumor cell lines). With the addition of ABS, first, the inflation of the hematopoietic tumor cells was detected. The inflation and proliferation continued on B-CLL cells at day 3 and produced aggregation islands. 14 Mumcuoglu et al. 15 demonstrated that depending on the concentration and duration of the application, ABS could cause apoptosis via regulating PAR1 and p53-independent p21 involvement in apoptosis stimulation within leukemia cells. Turk et al. also demonstrated that the most resistant cell type was SK-MEL-10 and the least resistant neoplastic cell type was A2058 since the effect of 0.19% ABS could be seen within just 15 min. The anticancer effect of ABS was also apparent on the light microscopic images of untreated M307 primary cells. 16 Another study disclosed that ABS induces DNA damage, apoptosis, and cytotoxic activity via generating reactive oxygen species in melanoma cell lines. 17
Animal studies in Ankaferd hemostat (ABS)
Many studies have been conducted on preclinical animal models to demonstrate the efficacy and safety of ABS.18–23 Bilgili et al. 18 reported that the application of ABS (spray, solution, and tampon forms) to superficial and deep abdominal lacerations could control bleeding successfully. In another animal study, partial nephrectomy was applied to 24 Wistar rats. The effectiveness of ABS was compared to several conventional anti-hemorrhagic methods. Hemostasis was successfully achieved by ABS. Moreover, warm ischemia times decreased by ABS. 24 Kuru et al. reported that rectal application of ABS showed favorable effects on bursting pressures, tissue prolidase and hydroxyproline levels, and the histopathological findings of colonic anastomosis. The rectal administration of ABS had positive effects on the improvement of colorectal anastomosis. As a natural product of plant origin, ABS may be used effectively and safely to obtain better healing results after colorectal anastomosis. 25 In another study, the rats underwent femoral vein puncture. One subgroup was treated with ABS tampon or spray, and the other control group was left untreated. After 2 weeks, each group underwent partial tissue excision from the same femoral region as well as from the brain, heart, kidney, and liver. ABS provided hemostasis and was observed to stop bleeding. There were no histopathological changes at the tissue level and no pathological effects in other organ tissues under light microscope. 26
Ankaferd hemostat (ABS) in clinical medicine
ABS has hemostatic, antithrombotic, anti-infective, anti-neoplastic, and wound-healing effects. The specific molecules which have the potential to contribute within ABS to its action are depicted in Table 2. ABS has also been employed in controlled clinical trials as depicted in Table 3.
Hypothesized mechanisms of the molecules within the Ankaferd hemostat (ABS) actions.

ABS: Ankaferd blood stopper; ATP: adenosine triphosphate; TBHQ: tertiary butylhydroquinone; BHT: butylated hydroxytoluene; VEGF-A: vascular endothelial growth factor A; YY1: Yin Yang-1.
Clinical trials that used Ankaferd hemostat.

ABS: Ankaferd blood stopper; HCT: hemostasis by conventional technique; ACB: autogenous cortical bone graft; PCNL: percutaneous nephrolithotomy; INR: international normalized ratio; OAT: Oral anticoagulant therapy; CABG: coronary artery bypass grafting; ASA: acetylsalicylic acid; SS: saline solution.
Ankaferd hemostat (ABS) in gastrointestinal bleeding
Gastrointestinal (GI) bleeding is a significant life-threatening condition and a common cause of hospitalization. Although endoscopic management reduces the rates of re-bleeding, need for surgery, and mortality in active bleeding, early recurrence still occurs in around 20% of cases despite the effective initial hemostasis. Therefore, there is an ongoing intensive research for novel techniques or treatment modalities that are efficient and safe for the managing of GI bleedings. During the research for a supplementary hemostatic agent for the treatment of GI hemorrhages, collected findings suggested that ABS could be efficient for the “difficult-to-manage” subtypes of GI hemorrhage.41–43 In an observational study by Ozaslan et al., 44 five adult patients with bleeding peptic ulcer disease, in which ABS was used as a primary hemostatic agent, were reported to have achieved success in controlling of the bleeding within minutes. Similarly, Purnak et al. reported a successful hemostasis control in a patient with a bleeding peptic ulcer complicated with defective hemostasis. In this reported case, at the time of bleeding, the patient was under-treated with a cytotoxic chemotherapeutic agent leading to thrombocytopenia. 45 There are case reports showing that ABS was used safely in GI bleeding in infants and children. The first pediatric experience with ABS in an infant with bleeding peptic ulcer was recently demonstrated by Yarali et al. They applied 1-mL ABS which was spurt over the ulcer surface, and in a very short period white-gray adherent clot was developed on the ulcer and bleeding ceased. 46
Ankaferd hemostat (ABS) in cardiovascular surgery
The amount of cardiac operations increases in all countries and parallel to this situation, the problems of sternotomy healing and hemorrhage lead surgeons to research for new options and use several new techniques and materials. Ergeneoglu et al. 47 showed that applying ABS was a useful different technique for the control of sternal bleeding during cardiac surgery. Atalay et al. showed that the local use of 10-mL ABS decreases bleeding significantly during the operation. Therefore, transfusion requirements of erythrocyte suspension and platelets decreased in patients who received clopidogrel and acetylsalicylic acid undergoing emergent operations. 36
Ankaferd hemostat (ABS) in ear, nose, and throat bleeding
Active bleeding from the nose is a common ear, nose, and throat emergency. Epistaxis can be serious or even fatal. Local or systemic diseases cause epistaxis. Yasar et al. evaluated the efficacy of ABS tamponade in the control of intra-operative bleeding occurring during adenoidectomy performed in children under the age of 12. The results showed that ABS aids in the control of intra-operative bleeding and reduces the number of packets tamponade required to achieve hemostasis. 37
Ankaferd hemostat (ABS) in surgical bleeding
There are numerous studies in the literature indicating that ABS can be used safely in surgical procedures and dental procedures in patients with normal and abnormal hemostasis. Huri et al. presented the first case report demonstrating the efficacy of ABS in a patient with prostate adenocarcinoma undergoing radical retropubic prostatectomy. They provided hemostasis via applying ABS onto tissues with active hemorrhages during radical prostatectomy and reported the clinical efficacy of ABS during radical prostatectomy. 48 Another report had also demonstrated similar hemostatic effects of local ABS application to a patient who underwent open partial nephrectomy. ABS was successfully applied to control bleeding without suturing the renal parenchyma. That application on the transected kidney area had provided active hemostasis in the partial nephrectomy case. ABS has a significant effect on active hemostasis during urogenital operations. Major bleeding from renal tissue could be controlled with additional doses of ABS solution which were applied to the bleeding area. 49
Ankaferd hemostat (ABS) in wound healing
Wound healing, hemostasis, and infection have closed pathobiological conditions. 50 Functional proteomic analyses had defined that antithrombin and pro-hemostatic activities of ABS are related to fibrinogen gamma chain and prothrombin. 51 ABS accelerated wound-healing in animal studies and case reports as depicted in Figure 1. Topal et al. investigated the effects of ABS on the deep second-degree burn wound healing and compared its efficiency with silver sulphadiazine (SSD). They showed that the mean percentage of wound contraction in the ABS and SSD groups was significantly higher than in the control group on days 14, 21, and 28 and suggested that ABS could be successfully used for burn wound healing besides (SSD). 52 ABS can improve the wound-healing process via providing inhibition of extra cellular matrix-degrading enzymes during wound repair. Moreover, ABS enhanced the stimulated migration of 3T3 fibroblasts to an artificial wounded area. 53 ABS may be useful in the treatment of burn lesions. It was observed that the wounds healed rapidly with the topical application of ABS onto the burn lesions. 54 The antioxidant components of ABS regulate the cellular proliferation, vascular dynamics, and hemostatic hemodynamic activity. 55 Therefore, ABS may be useful by protecting the gastric mucosa from oxidative injury and by accelerating the healing of gastric ulcers. 56 One experimental study disclosed that ABS was associated with significantly improved gastric mucosal structure. Therefore, oral administration of ABS could not only be useful in GI hemorrhage but also in infections and wound-related pathologies. 57
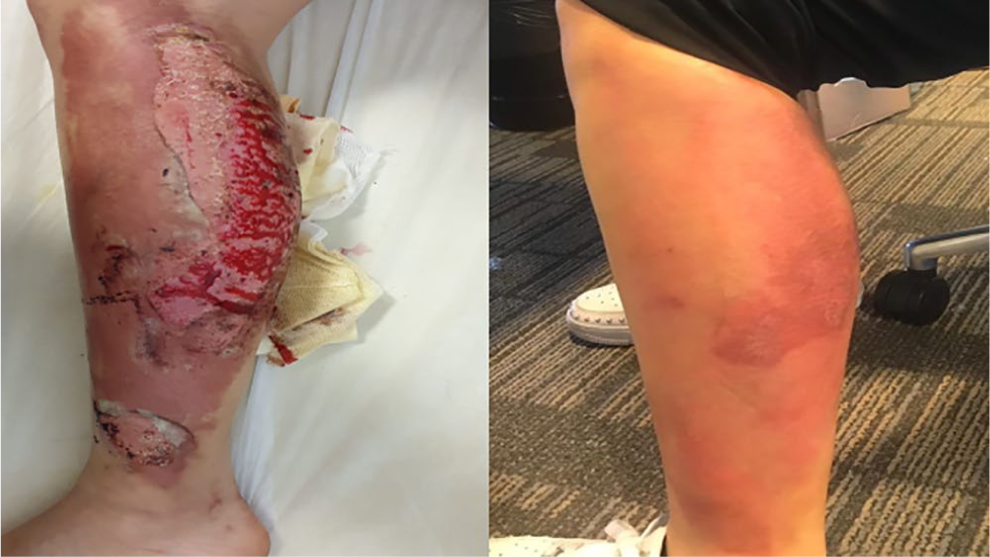
Rapid healing of burn lesions Ankaferd blood stopper (ABS) following the topical treatment (photographs from one of our burn patients before and after topical ABS therapy).
Ankaferd hemostat (ABS) in infections
ABS has an anti-microbial activity against many microorganisms such as gram-positive and gram-negative bacteria (Figure 2) and several fungal infections. ABS was assessed on 102 clinical isolates from both gram-negative and gram-positive bacteria and four standard strains, including MRSA ATCC 43300, MSSA ATCC 25923, P. aeruginosa ATCC 27853, and E. coli ATCC 35218. Fisgin et al. 58 showed that ABS was significantly active against all of the bacteria investigated. Another study disclosed that ABS is highly effective against several gram-negative and gram-positive bacteria including frequent foodborne microorganisms. 59 Akkoc et al. assessed the antifungal effect of ABS using an agar well diffusion test. They found that ABS has a high antifungal effect against Zygosaccharomyces bailii, Candida albicans, Aspergillus flavus, and Aspergillus parasiticus. However, Ciftci et al. did not observe any effect of ABS on Candida. They also reported that when ABS was directly applied onto Candida species, it resulted in changes in the growth conditions. 60 Another study indicated that ABS could act against Helicobacter pylori. ABS is clinically used for the management of GI bleeding due to benign and malignant lesions. Thus, the possible anti–H. pylori effect of ABS shall expand the therapeutic spectrum of the drug in GI lesions in relation to H. pylori infection such as peptic ulcer disease (PUD) and lymphoid tissue (MALT) lymphomagenesis. 61

Ankaferd hemostat anti-microbial effects on Salmonella species.
Future perspectives in Ankaferd hemostat
ABS could be an alternative treatment modality for different types of bleeding that are refractory to treatment with conventional procedures. ABS also provides effective anti-infective, anti-neoplastic, and curative modulator properties. Current perspective for using ABS is to provide hemostasis and accelerating wound healing. Future controlled trials are needed to show the effects of ABS in pleiotropic actions such as anti-neoplastic, antithrombotic, anti-inflammatory, anti-infective, antifungal, and anti-oxidative effects.
Conclusion
ABS serves as a hemostatic agent in a wide variety of clinical hemorrhages. Moreover, ABS also has numerous pleiotropic effects. The expanding spectrum of ABS include anti-infective, anti-neoplastic, and wound-healing properties. Randomized controlled clinical trials of ABS had established its anti-hemorrhagic clinical profile. However, further investigations are needed to elucidate the other pleiotropic activities in clinical backgrounds.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and informed consent
Because the article does not contain patients or animals, there are no ethics committee approval and informed consent.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
