Abstract
Background:
Pharmacotherapy is important in reducing morbidity and mortality related to cardiovascular diseases. However, these advantages are limited by drug therapy problems that can impact on a patient’s quality of life, prolong hospital stays, and increase the overall burden of healthcare expenditures. Therefore, this study was aimed to assess drug therapy problems among patients with cardiovascular diseases who were hospitalized and received follow-up at the ambulatory clinic of Hiwot Fana Specialized University Hospital.
Methods:
An institution-based cross-sectional study design was used to collect data from patients with cardiovascular diseases who were admitted to the medical ward and those who had received follow-up at the ambulatory clinic of Hiwot Fana Specialized University Hospital. The collected data were coded, entered, and analyzed using SPSS version 16. The associations of selected categorical variables were done using binary logistic and multivariate logistic regression analyses.
Results:
Out of 216 study participants, females accounted for 123 (57%), whereas 93 (43%) of them were males. Among cardiovascular diseases identified in the medical ward and ambulatory clinics of Hiwot Fana Specialized University Hospital, congestive heart failure 96 (44.4%) and hypertension 93 (43.1%) were the two most commonly diagnosed disorders. Of the total participants involved in the study, 131 (60.65%) had drug therapy problems. Among the seven classes of drug therapy problems assessed, the most commonly observed was the need for additional drug therapy 76 (58%); followed by cases related to unnecessary drug therapy and noncompliance both of which were estimated to be 16 (12.2%). In addition, of independent variables, only the use of more than three drugs was significantly associated in both binary logistic (crude odds ratio = 0.41, 95% confidence interval = 0.234–0.719, p = 0.002) and multivariate logistic regressions (adjusted odds ratio = 4.86, 95% confidence interval = 1.625–14.536, p = 0.005) as compared with those patients who were using less than three drugs.
Conclusion:
The findings of the study indicated that more than half of the study participants experienced drug therapy problems, for which 58% required additional drug therapy. The risk of drug therapy problem is found to increase with the use of more than three drugs. Since these problems are adversely affecting the treatment outcome of patients, this is an area which requires special attention and the cooperation of healthcare professionals to tackle it.
Keywords
Introduction
Cardiovascular diseases (CVDs) remain the biggest cause of death worldwide. According to estimates in a report by the World Health Organization in 2011, approximately 17.1 million people die of CVDs each year, representing 30% of all deaths. Of these deaths, approximately 7.3 million people die of coronary heart disease and 6.2 million people die mainly from heart disease and stroke, which are projected to remain the single leading cause of death. 1 CVDs account for 7%–10% of all adult medical admissions in African hospitals, of which heart failure constitutes approximately 3%–7%. 2 According to a study conducted in Ethiopia in 2015, ischemic heart disease, hemorrhagic stroke, and ischemic stroke were among the top causes of premature mortality and age-standardized deaths. 3 In addition, studies conducted in Addis Ababa, the capital of Ethiopia, also indicated that 75% of CVD deaths were due to myocardial infarction, stroke, and hypertensive heart disease.4,5
Even though a drug may have a significant role for curative, preventive, or diagnostic purposes, it may also have debilitating, negative effect on the health of patients if it is not used properly.6,7 Moreover, the availability of a wide range of drug products and drug consumers as well as complicated regimens of drugs can contribute to more adverse drug events (ADEs) and drug interactions that may create difficulty in the follow-up of the patients. 8
Drug therapy problems (DTPs) are defined as any unintended effects encountered by a patient during treatment. 9 DTP can occur at all steps of the treatment process, mainly during prescribing, transcribing, dispensing, and patient use of medication therapy. 10 These problems significantly influence the treatment outcomes of the patient and the ability to achieve the desired therapeutic goals. 9 Approximately, 20% of patients face DTPs after discharge from the hospital. 11 DTPs increase patients’ healthcare expenditures as well as morbidity and mortality. 12 DTPs are classified based on the possible cause of the problem 11 and may be related to; the choice of formulation, under or overtreatment, inappropriate dosing, drug interactions, noncompliance, and harm caused by ADEs. 13
In a study conducted in Singapore, which involved 347 patients, DTPs were found to be one of the major reasons for admission that resulting in 71.9% of patient admissions. 14 Similarly, an observational study conducted in hospitals in the Netherlands indicated that out of 13,000 unintended admissions, 714 (5.6%) were most likely due to issues related to drug therapy. Of these drug-related admissions, 46.5% were potentially preventable. 15 Furthermore, in the United States, around 44,000 people reportedly died each year in the hospitals as a result of medical problems that were preventable as indicated by different studies.16,17 The literature review conducted on DTPs has demonstrated that about 28% of cases encountered at the emergency ward were drug-related problems such as adverse drug reactions (ADRs), of which 70%–90% were potentially preventable. 18 A cross-sectional study conducted in Lahore, Pakistan also indicated that more than half (58.4%) of the ADEs were preventable. Accordingly, 43.6% of the ADEs were definitely preventable, whereas 14.8% were probably preventable and less than half (41.6%) were non-preventable ADEs or ADRs. 19
Coming to our country Ethiopia, a study conducted in Felege Hiwot Referral Hospital among cardiovascular patients identified a total of 105 DTPs. 20 An additional study conducted in Jimma Referral Hospital showed that of 257 participants, 189 (73.5%) had developed DTPs. 8 Similarly, findings from a medical ward of Gondar University Referral Hospital indicated that of 256 participants engaged in the study, 169 (66.0%) of patients had drug-related problems. 7 This could imply that DTPs are common in our settings. Therefore, this study was targeted to assess DTPs among patients with CVD who were admitted to the medical ward and those who had received follow-up at the ambulatory clinic of Hiwot Fana Specialized University Hospital (HFSUH).
Methods
Study design and study period
The study was conducted using institutional-based cross-sectional study design among patients with CVDs who were admitted to the medical ward and those who had received follow-up at the ambulatory clinic of HFSUH that is located in Harar town, which is 526 km away from Addis Ababa, the capital of Ethiopia. The hospital provides internal medicine, surgery, pediatrics, emergency, maternity, family planning, gynecologic/obstetric, ambulatory clinic, dental, psychiatric, physiotherapy, tuberculosis (TB), and human immunodeficiency virus (HIV) services to the people of eastern Ethiopia, especially those from both Hararghe zone and Harari region. The data were collected from March 1 to 30, 2017.
Source population
All patients who were admitted to the medical ward and those who had a follow-up at the ambulatory clinic of HFSUH in the study period.
Study population
All patients with CVDs who were admitted to the medical ward and those who had a follow-up at the ambulatory clinic of HFSUH during the study period.
Eligibility criteria
Inclusion criteria
Patients who were admitted with CVD to the medical ward and who had taken cardiovascular drug therapy;
Patients with CVD who received follow-up at the ambulatory clinic of HFSUH;
Patients with age ⩾18 years old.
Exclusion criteria
Patients with incomplete medical records;
Patients who were seriously sick and unable to respond for questions;
Patients with age < 18 years old.
Sample size and sampling technique
The sample size was calculated using a single proportion formula with the assumption of 50% prevalence of DTPs among patients with CVDs, 95% confidence interval (CI), 5% margin of error, and 10% of contingency for non-response rate. Based on this assumption, the calculated sample size was found to be 216.
Convenience sampling technique was used as per inclusion criteria and data collection period. Data abstraction questionnaires assessing sociodemographic characteristics, vital signs, relevant laboratory test, diagnosis, current medical conditions, medication prescribed, the effectiveness of drugs, regimens, duration of treatment, comorbid illness, and categories of DTPs were used. These questionnaires are not validated; however, it was pretested in separate similar hospital in Harar town to check whether there is any ambiguity with the questions and to modify it accordingly. The data were collected by reviewing patients’ medical records and asking the patients’ pertinent information related to adherence and compliance, and other issues based on the objective of the study. Two clinical pharmacists were involved in the data collection.
DTPs were identified and classified using the Cipolle’s and Strand’s method9,21 and discussion held by clinical pharmacists on the identification of DTP. Then, these pharmacists further refined DTP identification and classification method to the study setting based on treatment guidelines and literature reviews.22–24 The identification of DTP was based on a review of patients’ medical and medication records, assessment of laboratory investigations, and patient’s interview about medication experience. Specific information about medication therapies, such as the recommended drug of choice, recommended dosages, frequency of administration, duration of therapy, drug interactions, and ADEs were compared based on details from the standard pharmacotherapy textbooks, and guidelines of hypertension and heart failure management.22,25–27
Data entry and analysis
The data were cleaned, coded, entered, and analyzed using SPSS version 16. Categorical variables were described by frequencies and percentages, whereas the associations of selected categorical variables were done using binary logistic and multivariate logistic regressions.
Operational definitions
Adverse drug event is any injury related to the use of a drug, even if the causality of this relationship is not proven. 28
Adherence is drug-taking behavior of a patient measured with the Morisky scale in which when a patient says no at least for one of the questions, he or she is noncompliant. 9
Contraindication is a medical condition in which specific drugs are not going to indicated because it may be associated with severe medical complication. 29
Comorbidity is the presence of one or more additional disease or disorders co-occurring with a primary disease. 30
Adverse drug reaction refers to any noxious, unintended, and undesired effect of a drug, which occurs at doses used in humans for prophylaxis, diagnosis, or therapy. 31
Dosage includes the dose given, frequency of administration, and duration of therapy.
Definition of terms related to DTPs9,21
Unnecessary drug therapy—the drug therapy is unnecessary because the patient does not have a clinical indication at this time.
Need for additional drug therapy—additional drug therapy is required to treat or prevent a medical condition or illness from developing, or the clinical condition required initiation of drug therapy.
Ineffective drug—the drug product is not being effective at producing the desired response or the medical condition is refractory to the drug product.
Inappropriate dosage—refers to dosages both too low and too high.
Dosage too high—indicates that the dose is too high and resulting in undesirable effects. Dosage too low—refers to the dose is too low to produce the desired response.
Noncompliance—the patient is not able or willing to take the drug therapy as intended.
Drug–drug interaction—occurs when the effect of one drug is changed due to the presence of other co-administered drugs, food, or drinking substance.
Results
Sociodemographic characteristics of patients
Of the total patients involved in the study, 93 (43%) of them were males, whereas 123 (57%) were females. Approximately, 164 (75.9%) of the study participants were married. Of 216 participants engaged in the study, 83 (38.4%) of them had attended secondary school, whereas 80 (37%) of them were illiterate. Sixty-five (30.1%) of the study participants were aged >58 years. Out of the 123 females participated in the study, 10 (8.1%) were pregnant and 9 (7.3%) were breastfeeding (Table 1).
Sociodemographic characteristics of patients admitted to the medical ward and those who had a follow-up at the ambulatory clinic of HFSUH, Harar town, from March 1 to 30, 2017 (N = 216).
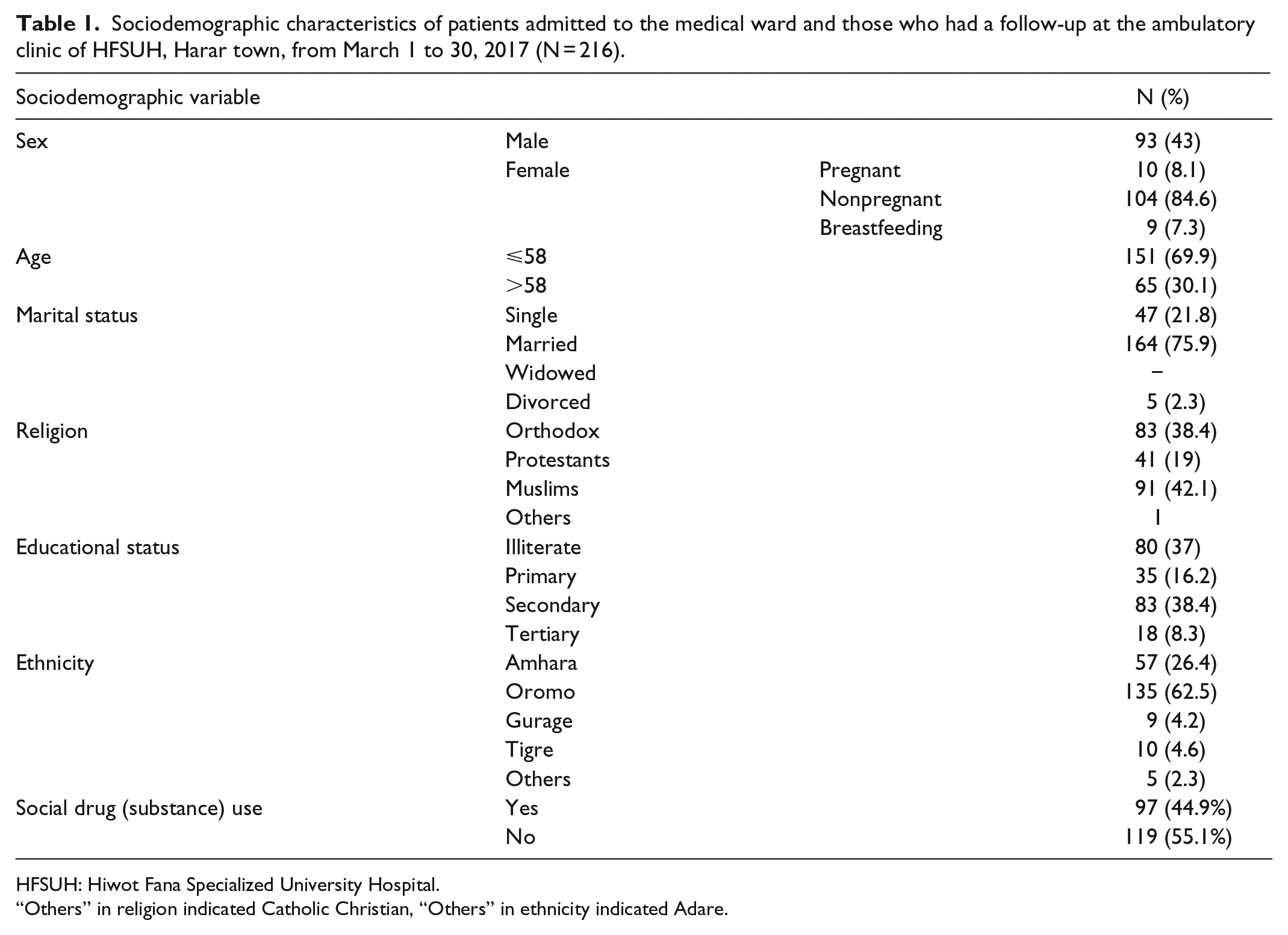
HFSUH: Hiwot Fana Specialized University Hospital.
“Others” in religion indicated Catholic Christian, “Others” in ethnicity indicated Adare.
Common CVDs, family history, laboratory test, and comorbid illness observed among patients with CVD admitted to the medical ward and those who had a follow-up at the ambulatory clinic of HFSUH
Among CVDs identified, the most common were congestive heart failure (CHF; 44.4%) and hypertension (43.1%). Approximately, 10 (4.6%) of participants reported a family history of cardiovascular disorders, whereas few of them also reported the presence of diabetes mellitus. Nearly, more than half of the patients had no relevant laboratory test, whereas most of them had documented vital sign. Out of the patients attending the medical ward and ambulatory clinic of HFSUH, diabetes (17.6%) and pneumonia (15.3%) were the two leading comorbidities. Regarding the number of drugs prescribed per patient, 137 (63.4%) patients were prescribed with more than three drugs (Table 2).
The most common CVDs, number of drugs prescribed, family history, laboratory test, and vital sign of patients admitted to the medical ward and those who had received follow-up at the ambulatory clinic of HFSUH, Harar town, from March 1 to 30, 2017 (N = 216).

CHF: congestive heart failure; HTN: hypertension; IHD: ischemic heart disease; VTE: venous thromboembolism; ACS: acute coronary syndrome; DM: diabetic mellitus; CVD: cardiovascular disease; AF: atrial fibrillation; HFSUH: Hiwot Fana Specialized University Hospital.
Cases not related to cardiovascular disease.
DTPs among patients with CVD who were admitted to the medical ward and those who had a follow-up at the ambulatory clinic of HFSUH
Out of 216 participants involved in the study, DTPs were observed in approximately 131 (60.65%) patients, in whom the ratio of DTPs per patient was 0.6. Of DTPs identified in this study, the need for additional drug therapy accounted for 76 (58%), followed by unnecessary drug therapy and noncompliance—both of which accounted for 16 (12.2%) (Table 3).
DTPs among patients with CVD who were admitted to the medical ward and those who had received follow-up at the ambulatory clinic of HFSUH, Harar town, from March 1 to 30, 2017 (N = 131).

HFSUH: Hiwot Fana Specialized University Hospital; DTP: drug therapy problems; ADR: adverse drug reaction; DI: drug interaction; CVD: cardiovascular disease.
Factors associated with DTPs among patients with CVD who were admitted to the medical ward and those who had received follow-up at the ambulatory clinic of HFSUH
To identify independent factors predicting the likelihood of DTPs, binary logistic and multivariate logistic regressions were conducted. In binary logistic regression, the risk of DTP among patients using more than three drugs was decreased by 0.410 (crude odds ratio (COR); 95% CI = 0.234–0.719, p = 0.002) as compared with those who were received less than three drugs. However, in multivariate logistic regression, patients who were received greater than three drugs were 4.86 times more likely to develop DTPs than those who were using three or fewer drugs (Adjusted odds ratio [AOR]; 95% CI = 1.625–14.536, p = 0.005) (Table 4).
Factors associated with DTPs among patients with CVDs who were admitted to the medical ward and those who had received follow-up at the ambulatory clinic of HFSUH, Harar town, from March 1 to 30, 2017.

HFSUH: Hiwot Fana Specialized University Hospital; COR: crude odds ratio; AOR: adjusted odds ratio; CI: confidence interval; DTPs: drug therapy problems; CVDs: cardiovascular diseases.
p < 0.05 is considered to be statistically significant.
Discussion
Drug-related problems are a major healthcare challenge and most of them are preventable.32–35 A large number of reports illustrated the incidence, prevalence, and preventability of drug therapy-related losses of life.36,37 Inappropriate medication use has been associated with hospitalization35,38,39 and untoward drug effects that have adversely affected the quality of life of patients. This has been shown to cause considerable morbidity and mortality in the inpatient and ambulatory clinics of health facilities.28,40–42 Taking this into account, this study was intended to evaluate the occurrence of DTPs among patients with CVD who were hospitalized as well as those who had a follow-up at the ambulatory clinic of HFSUH. In addition, the study identifies factors related to the development of DTP such as age, sex, pregnancy, breastfeeding, comorbidity, and the number of medications prescribed per patient.
The study identified that a large number of the study participants were presented with CHF and hypertension. This finding is higher than the results reported from other studies of which the prevalence of CHF and hypertension were less than 30%.43,44 However, it is consistent with a study conducted in Jordan. 45 In addition, our study also indicated that the most prevalent comorbidities among patients with CVDs were diabetes and pneumonia. These conditions put the patients at high risk of DTPs due to the need for multiple drugs to manage these health problems parallel to other CVDs. This could explain why most of the study participants in this study received three or more drugs. These results are in agreement with a study conducted in the northwest of Ethiopia in which most patients experienced more than one comorbid illness and received more than five drugs. 7
Coming to DTP, the study identified different types of DTPs among patients who were admitted to the medical ward and those who had received follow-up at the ambulatory clinic of HFSUH. These were unnecessary drug therapy, the need for additional drug therapy, ineffective drug therapy, inappropriate dose, and noncompliance with the treatment. These all have detrimental effects on the treatment outcome of the patients in addition to forcing them to make unnecessary healthcare expenditures. According to this study, more than half of the study participants experienced DTPs in which the ratio of DTPs per patient was 0.6. However, this finding is lower than the results reported from studies conducted in the southwest 8 and the northwest of Ethiopia. 7 Moreover, our finding is also lower than a report from another study which indicated that the number of DTPs per patients was 2.5. 46 This difference is probably related to the small sample size that was used in our study, which attributed to relatively lower DTPs compared with studies conducted in other areas.
Of the DTPs identified in this study, the most prevalent one was the need for additional drug therapy; followed by unnecessary drug therapy and noncompliance. This finding is concordant with a study conducted in the Southwest of Ethiopia at Jimma University Specialized Hospital8,47 as well as a study conducted in Jordan among patients with chronic disease 48 in which the need for additional drug therapy was higher than other categories of DTPs. However, in terms of the need for additional drug therapy, our finding is higher than what is reported in a study conducted in the northwest of Ethiopia. 7 This study also reported a higher prevalence of inappropriate dosage and noncompliance 7 than the findings of our study. Similarly, a study carried out at Ambo General Hospital showed that a higher percentage of unnecessary drug therapy and a lower prevalence of the need for additional drug therapy 49 compared with the results of our study. This difference might be ascribed to factors such as the level of knowledge of health professionals among these health facilities regarding the appropriate indication of drugs, the proper selection of drugs as per cases observed, and issues related to comorbidities as well as the patient’s condition.
With regard to variables influencing the occurrence of DTPs among patients with CVD; only the use of multiple drugs was significantly associated in both binary logistic and multivariate logistic regressions. In binary logistic regression analysis, the likelihood of developing DTPs among patients using more than three drugs is decreased by 0.410 (95% CI = 0.234–0.719, p = 0.002), whereas in multivariate logistic regression analysis, those patients who received greater than three drugs were 4.86 times more likely to experience DTPs than those who were using three or fewer drugs (95% CI = 1.625–14.536, p = 0.005). The most probable explanation for the difference between unadjusted and AOR is due to the presence of confounding factors which masked the true effect of a risk factor of multiple drugs used as a predictor of DTPs in binary logistic regression. However, when multiple variables are run at a time using multivariate logistic regression, possibly the confounding factors were controlled and changed the direction of the association between multiple drugs used and DTPs. This finding is in agreement with the study done in Jordan where the number of medications used were strongly linked to treatment-related problems 12 as well as the study conducted in the northwest of Ethiopia in which the number of medications per patient is associated with the risk of DTPs (AOR = 9.088; 95% CI: 1.012–81.623; p = 0.033). 7 In addition, the finding of our study is also in agreement with another study conducted in the southwest of Ethiopia in which the use of more than five drugs per day per patient was found to be a predictor of the occurrence of DTPs (AOR = 5.23; 95% CI = 1.15–23.75). 8
Limitation of the study
Limitations of this study include the following: first, the study only covers patients who were admitted to the medical ward and those who had received follow-up at the ambulatory clinic, and it only involves a single health facility. Second, except questionnaires used to assess categories of DTPs those that are adapted from pharmaceutical care practice, 9 the remaining questionnaires are not validated and prepared based on the objective of the study. Due to this reason, the tools may have their own limitation. Therefore, further studies are recommended to involve different facilities using standardized tools to identify the most important factors likely to be associated with the occurrence of DTPs among hospitalized patients and patients attending ambulatory clinics in order to generate sufficient evidence that can be generalized to a larger population.
Conclusion
The prevalence of DTPs was common among patients who were admitted to the medical ward and those who had received follow-up at the ambulatory clinic, in which approximately 60.65% of patients developed DTPs. Of all the DTPs which occurred, the need for additional drug therapy was the most prevalent and accounted for 58%, whereas the proportion of almost all other DTPs was lower. Since these problems are adversely affecting the treatment outcome of patients, this is an area which requires special attention and the cooperation of healthcare professionals to tackle it.
Footnotes
Acknowledgements
The authors would like to thank the administrative office of HFSUH, head of the medical ward and the ambulatory clinic, and data collectors without which this study cannot be realized. Finally, our gratitude also goes to Ms. Kelly Alliston for her cooperation in the English language editing of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Institutional Health Research Ethics Review Committee of College of Health and Medical Sciences, Haramaya University. An official letter was obtained from the School of Pharmacy (ref. No. SOP 980/02/2017) and given to HFSUH administration to secure permission.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Verbal informed consent was obtained from all subjects before the study. However, written informed consent was not obtained because it is expected that the study involved participants who are illiterate and cannot understand written informed consent. Therefore, by considering this expectation, the Institutional Health Research Ethics Review Committee waived verbal informed consent proposed during proposal submission.
