Abstract
Objective:
Our main objective was to evaluate the short- and long-term efficacy of Celon radiofrequency-induced thermal therapy for endovenous treatment of incompetent great saphenous vein. The secondary objectives were to report on possible short-term side effects and complications.
Methods:
This was a retrospective study of 112 consecutive patients included between 2013 and June 2015. These patients were treated (146 great saphenous vein, C2–C6) either at the hemodynamic room using local anesthesia or at the operating theater under general anesthesia with or without phlebectomy. All patients received radiofrequency-induced thermal therapy at 18 W power, 1 s/cm pullback rate and 5–7 pullbacks per segment of 10 cm (i.e. maximum 10 pullbacks). A clinical follow-up via ultrasound monitoring was done at 1 week, 1 month, 6 months, 1 year, 2 years and 3 years.
Results:
The 3-year survival occlusion rate was 96.71% and 98% for overall and radiofrequency-induced thermal therapy patients, respectively. No major side effects were observed. A case of endovenous heat-induced thrombosis was reported. Slight neurological disorders were reported (0.88%).
Conclusion:
Our unit’s endovenous Celon radiofrequency-induced thermal therapy of incompetent great saphenous vein was efficient, well tolerated, without major side effects. Moreover, in order to reduce possible neurological disorders, we advise multiple pullbacks at 1 s/cm and using tumescence anesthesia.
Introduction
Varicose vein disease (VVD) prevalence in industrial world is 30%–60% of the general population. 1 VVD is evolving in the absence of optimal treatment and in the presence of patient’s trophic complications such as eczema, hypodermitis, venous ulcers or superficial venous thrombosis (SVT), as well as sick leaves and high cost of paramedical care. What is at stake for the therapeutic management of patients with varicose veins is the prevention of clinical complications with an estimated prevalence of 3%–11%. 2 Even though there have already been recognized risk factors (e.g. obesity, family history, stationary body positions, heat exposure, pregnancy) which can provoke varicose veins, age remains the major risk factor. 3 Consequently, customized treatments according to age could lead to less aggressive therapies to avoid treatment-related comorbidities. Up to the past few years, the first-line treatment for varicose veins has been conventional surgery via vein stripping resection using general anesthesia (GA). This therapy leads to an extended sick leave longer than 1 month.
The Guidelines (American College of Phlebology, National Institute for Health and Care Excellence (NICE), European Venous Forum, American Venous Forum) recommend endovenous treatment of saphenous vein.4–8 Such treatment has similar medium-term clinical efficacy as surgery and with better patient outcomes. 5
There are different modes of minimum invasive treatments of saphenous vein: thermal endovenous by radiofrequency, laser or steam; sclerotherapy by foam. All these treatments aim at destroying the membrane collagen of the affected vein segment. Vein collagen is the origin of primary sclerosis and secondary fibrosis. Celon radiofrequency-induced thermal therapy (RFiTT®; Olympus Surgical Technologies Europe, Germany) uses a bipolar transducer which generates high-frequency heat waves onto the vein membrane. The paucity of literature on Celon RFiTT has not allowed conclusive results on its short- and long-term efficacy and its feasibility using local anesthesia (LA). We aimed to address these main objectives as well as to identify RFiTT possible secondary side effects and 3-year patient outcomes, that is, complication versus recovery.
Materials and methods
Patient selection and materials
This is a retrospective study on our unit’s management of patients with varicose veins of the lower limbs using endovenous Celon RFiTT between 2013 and June 2015. All 112 patients, that is, 146 incompetent great saphenous veins (GSVs), who underwent RFiTT were included in the study. The sample size estimation was based on previous clinical experience, that is, 2-year learning curves, at least 30 patients treated per year. The diagnosis of superficial venous insufficiency was done on a Toshiba Viamo Doppler ultrasound using a superficial linear transducer at 7.5 MHz. The varicose condition was defined as a truncal reflux (TR) while standing >0.5 s on color and/or pulse mode Doppler ultrasound with a venous diameter of 3 mm on its transversal axis. In accordance with the French High Authority of Health (HAS) recommendations, we only included the TR of the GSV. 9 Inclusion criterion was GSV’s crural intrafascial truncal reflux (IF-TR) of at least 15 cm in length and 5 mm in diameter while standing. Exclusion criteria were as follows: recent (<1 month) SVT, SVT sequelae hindering the probe passage, pace maker, post-thrombotic syndrome, pregnancy and child birth of <6 months, severe systematic disease, active neoplasia, short segment intrafascial saphenous vein (<15–20 cm) or a truncal diameter >10 mm, GSV sus-fascial TR, and critical arterial ischemia.
The presence of tributary veins was not an exclusion criterion. When present in cluster and dilated enough requiring plastic care, phlebectomy could be performed by the vascular surgeon. Other tributary veins, that is, residual or not dilated enough, or persistent after phlebectomy, were treated with foam or liquid sclerotherapy by the vascular physician.
The presence of arteriopathy (systolic pulse index: SPI < 0.9) was regarded as a relevant contraindication, in particular in case of trophic complications induced by superficial venous insufficiency. However, patients with arteriopathy were eligible for endovenous RFiTT only in the absence of anatomical anomalies prone to vein rupture on ultrasound. An anticoagulation or antiplatelet treatment was regarded as a relevant contraindication to phlebectomy and stripping. However, patients on anticoagulation or antiplatelet treatment were eligible for RFiTT. Patients with CEAP (clinical etiology anatomy physiopathology) C1 classification were excluded. Verbal and written information on the treatment by the French Vascular Medicine Society had been given to all patients who agreed to be treated.
Endovenous RFiTT combined with phlebectomy was performed by vascular physician and vascular surgeon at the operating theater using GA (i.e. inpatient). Endovenous RFiTT requiring only LA was performed by the vascular physician at the hemodynamic room (i.e. outpatient) of our cardiology unit under strict asepsis.
Celon RFiTT technique protocol
Celon RFiTT probe’s tip comprises two metallic cylinders separated by a heat insulator (coating). The delivered heat ranged between 60°C and 100°C. This probe was placed into the GSV after performing a venous puncture monitored by longitudinal (either of the upper limb or of the lower crural) ultrasound, using LA (lidocaine, 2 cm3) and an 18-g (BD microlance) BD micropuncture needle. Then, a wireguide was inserted into the vein followed by a 5F sheath (Terumo Radifocus). RF probe was positioned according to the sapheno-femoral junction (SFJ) immediately below the superficial epigastric vein. Tumescence infusion was made using 250 mL saline solution (NaCl 0.9%) and 18 mL lidocaine without adrenaline per treated limb. This infusion was delivered by a tumescence pompe (Nouvag Dispenser DP 30®, Switzerland) and a 23-g 0.6 mm × 25 mm or 21-g 0.8 mm × 40 mm BD micropuncture needle depending on the depth. The tumescence infusion was aimed at obtaining a floppy sapheno and muscular fascia through injection of the tumescence sea as close to the GSV as possible to push the little sub-cutaneous nervous branches away from the vein. This should ease the probe descending the vein >1 cm sub-cutaneously while avoiding burn lesions.
We chose not to treat the tributary veins of GSV by RFiTT. One study suggested postponing the treatment of tributary veins due to their known partial and even complete obliteration failure, after treatment of the main trunk. 10 Per manufacturer’s advise 18 W power was set. An average of 5–7 probe passages (maximum 10 pullbacks) were performed per treated zone at 1 s/cm. In the event of venous spasm (i.e. a probe pullback resistance), initially, a lower venous zone was targeted for descending (please see the pictogram of the protocol in Figure 1). At the end of the therapy, RFiTT probe and wireguide were withdrawn and a long compression band was placed (Biflex® 16+, Thuasne, France) on the treated limb by the physician. Immediately after, an antithrombotic prophylaxis (a Fondaparinux 2.5 mg subcutaneous injection) was administered. The prophylaxis was continued for a week only in case of a history of thrombosis or in case of phlebectomy. The patients remained under surveillance at the outpatient surgery for 1 and 6 h post-intervention in case of LA and GA, respectively. All patients were prescribed low thigh autofitting compression stockings (French class 2; 15–20 mmHg) for 1 month.

For those patients receiving LA, Versatis® was applied at least 6 h before LA in order to reduce tumescence-induced sensitivity. When needed (i.e. in rare cases), a paracetamol IV or even Nitrogen Protoxyde (Kalinox 50®; allergic, tense patients) was administered. The LA patients also received a chloral hydrate hydroxyzine tablet for premedication (Atarax® 25 mg).
All RFiTT and phlebectomy interventions were performed by the same vascular physician and the same vascular surgeon, respectively. The working sick leaves delivered by the physician for patients who underwent RFiTT alone and concomitant with phlebectomy were 1 day, and 1 week, respectively. Contraindication to sports in standing position was given for at least 15 days. A drug prescription for level 1 (paracetamol) analgesic combined with a topical anti-inflammatory was given.
Data collection
All collected data were filled into an excel spreadsheet. The collected data were as follows: patient’s demographics (age, gender, treated limb and limb side) and clinical CEAP classification, 11 pain score, type of anesthesia, surgery or hemodynamic treatment, causes of intervention failure, post-intervention complications and ultrasound follow-up data at 1 week, 1 month, 6 months, 1 year, 2 years and 3 years.
Follow-up
All patients underwent an ultrasound at week 1, month 1, month 6, year 1 and once per year (3-year follow-up). CIVIQ-14 (chronic venous insufficiency quality of life questionnaire) 12 was obtained at month 1 follow-up visit (i.e. score 14: highest quality of life to 70: lowest quality of life; please see the supplementary file). Those patients who experienced intervention failure due to technical problems (e.g. venous puncture failure and probe withdrawal), and in the absence of contraindication, underwent an intrafascial foam sclerotherapy assisted by ultrasound (i.e. within 30 min after failure of initial intervention). During the follow-up, for all these patients, in the presence of tributary veins and/or narrow varicose veins, an additional sclerotherapy was proposed.
Clinical outcome
A clinical outcome was regarded as “success” in the absence of functional signs, total occlusion and absence of venous reflux; “satisfactory” in case of disappearance of functional signs and presence of partial occlusion; and “failure” in the presence of functional signs, in the absence of sclerosis and in case of >1 s lasting reflux.
Ultrasound outcome
In accordance with united international phlebology (UIP) consensus 13 for the follow-up of ultrasound-guided endovenous thermal and chemical treatment of varicose veins of the lower limbs, our patient outcomes were as follows: total occlusion, partial occlusion and total permeability. Total occlusion was defined as incompressibility and absence of reflux on color ultrasound. Partial occlusion was defined as partial compressibility and partial reflux on color ultrasound. Total permeability was defined as total compressibility and the presence of total reflux on color ultrasound.
Complications
All post-intervention complications were recorded. Patients’ LA-related pain was evaluated promptly after intervention using the visual analogue scale (VAS: 0 (no pain) to 10 (severe pain)).
Statistics
All quantitative variables were analyzed using Microsoft VBA Excel program and presented in numbers and percentages. In line with our clinical experience (2-year learning curves), it was determined a priori that 38 patients would be needed in each group for a 10% difference in occlusion rate with an 80% power and 0.05 risk of α. Taking into account an expected 30% lost-to-follow-up, 50 patients per group were required (using Biosta TGV: https://marne.u707.jussieu.fr/biostatgv/?module=etudes/sujets#). Vein occlusion rate (%) was computed for each follow-up time period and intervention. Fisher’s exact test was used to compare occlusion rate between two groups, a p value < 0.05 was regarded significant. Student’s t-test was performed to compare quality of life results between two groups. Survival occlusion rate was computed using Kaplan–Meier analysis after censoring for occlusion failure and lost-to-follow-up.
Results
A total of 112 patients (146 GSV) were treated: 34 bilateral treatments, 64 underwent GA and 48 underwent LA. Most of the patients, who underwent GA, were treated by phlebectomy (42/64).
Patients’ demographics and clinical characteristics are displayed in Table 1. Patients’ median age was 48 (23–85), 53.5 (25–78) and 50.7 (23–85) for operative patients, hemodynamic patients and overall patients, respectively.
Characteristics of patients.

RF: radiofrequency; CEAP: clinical etiology anatomy physiopathology.
The mean diameter of truncal segments treated was 5.35 mm (±1.15 SD).
Of note, the orthostatic vein diameters were reported to be greater than the decubitus vein diameters (2–3 mm).
RFiTT failure rates were reported as puncture failure in five cases (4.39%) and as probe ascending failure in three cases (2.63%). The RFiTT success rate was 92.98%. Most of RFiTT failures were due to occurrence of significant venous spasm during interventions performed in the hemodynamic (outpatient) room. This could be explained by the low temperature of hemodynamic room and the cumulative (i.e. after two rounds) puncture-induced venous spasm.
Side effects and complications
No burns or pulmonary embolism were reported among the side effects. Table 2 displays patients’ complications and quality-of-life results. One woman presented with edema of one of the treated limbs at 3 weeks. This was due to the sclerosis extension (i.e. endovenous heat-induced thrombosis (EHIT) of common femoral vein with total occlusion; Figure 2). One woman presented with temporary painful paresthesia along the saphenous vein, without other side effects, during 2 weeks. One woman recovered spontaneously from hematoma (>10 cm) at the upper third crural. Other reported hematomas were secondary to phlebectomy. Post-intervention pain score evaluation was very satisfactory for LA (mean VAS: 2.08 over 10). CIVIQ-14 quality-of-life survey at 1-month post-RFiTT results were better for hemodynamic room compared with operative intervention (15.29 ± 1.32 vs 21.48 ± 2.22, p < 0.01).
Patients’ complications and quality-of-life results.

EHIT: endovenous heat-induced thrombosis; CIVIQ-14: quality of life scoring including pain, physical activity and psychological dimensions (highest quality of life to lowest quality of life score: 14–70).
p-value according to Student’s t test.

B-mode ultrasound, sagittal cup: sclerosis extension in common femoral vein (endovenous heat-induced thrombosis).
Follow-up
Inpatients
Patients who underwent a surgical intervention and GA (inpatients) were followed up by their referring physician. Of these patients, 7 (6.14%) and 17 (14.9%) were lost to follow-up at 1 month and 1 year, respectively.
Outpatients
Most RFiTT patients reported a rapid clinical recovery within 2–3 days post-intervention with regard to heavy limbs, night cramps or vesperal edema. This was demonstrated by the results of CIVIQ-14 obtained after an ultrasound monitoring a month after RF treatment.
Occlusion rate
Table 3 displays occlusion results for each treatment (i.e. hemodynamic-outpatient and inpatient-operative and overall patients) and each follow-up period. Occlusion rates between RFiTT (hemodynamic-outpatient) and RFiTT + phlebectomy (operative-inpatient) were not statistically significant for all follow-up periods (p > 0.05).
Occlusion results according to each setting and at each follow-up period.

N: total number of veins; KM: Kaplan–Meier, survival occlusion.
Fisher’s exact test: hemodynamic-outpatient occlusion rates versus operative-inpatient occlusion rates: p = 1, 0.519, 1, and 1 for 1 month, 1 year, 2 years, and 3 years, respectively.
The overall survival occlusion rate was 98.43%, 96.71% and 96.71% at 1 month, 2 years and 3 years, respectively. Most patients presented with fibrosis (Figure 3). Moreover, the 98% and 96.71% 3 year occlusion rates for hemodynamic and overall patients, respectively demonstrated the long-term efficacy of our RFiTT protocol in outpatient hemodynamic using LA.
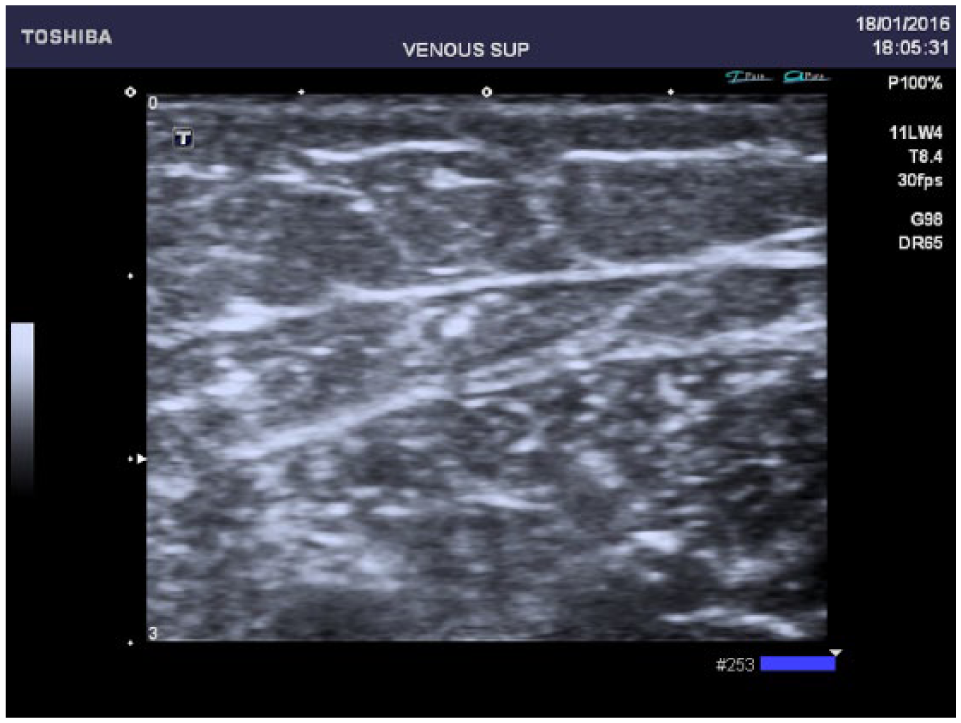
B-mode ultrasound of great saphenous vein’s sagittal cup after 1 year of Celon RFITT application: obliteration and fibrosis.
Discussion
Thermal or chemical endovenous venous treatments are among the first-line treatments of varicose veins (e.g. venous compression, lifestyle rules and veno-active drugs). The endovenous technique, although known for several decades, has only been used for the past 15 years. 14
Celon RFiTT uses a bipolar transducer which generates increasing power heat waves (60°C to 100°C) onto the venous membrane, that is, collagen and elastin, for optimal vein occlusion. This leads to an initial venous spasm and a secondary obstruction. An acoustic feedback monitors closely the RFiTT, that is, the higher the venous occlusion rate, the higher would be the acoustic signal alarming the end of the therapy. Gradual increase in the power of heat waves reduces the risk of venous membrane lesion and perforation.
The disadvantage of RFiTT is the coagulated blood build-up at the probe’s end (4 mm). This coagulum is a constraint to treatment continuation and leads to probe’s withdrawal, cleaning and replacement. An acoustic signal warns such coagulum formation. Moreover, the presence of a high volume of remaining venous blood and the absence of peripheral, intrafascial tumescence can increase the coagulum formation, thus leading to intervention discontinuation. In case of narrow and winding veins or ecstatic blisters, the probe ascending becomes a challenge.
The manufacturer’s recommendations in 2007 and 2012 were 25 W power, 1 cm/s withdrawal speed and 18–20 W power ⩾ 1.5 s/cm, respectively. Multiple passages of the probe were recommended without specific details or methodology.
Some authors have reported 97.6% obliteration using 18 W power and ⩾3.4 s/cm withdrawal speed. 15 Others have reported 98.4% obliteration using between 18 and 20 W power and emphasizing on RFiTT operator’s experience. 16
Our results of GSV truncal obliteration (RFiTT: 98%, 98%; Overall: 98.43% and 96.71% at 1 month and 1 year, respectively) were better than those of the literature. Braithwaite et al., 16 Goode et al. 17 and Doerler et al. 18 reported 98.5% at 180 days and 88.2% at 360 days, 95% at 10 days and 74% at 1 year, and 90% at 22 months, respectively. Braithwaite et al. followed the manufacturer’s guidelines using a power of 18–20 W during 1.5 cm/s in average. Their obliteration rate was improved from 90.2% to 98.4% by increasing the passage time to 3.5 cm/s. Goode et al. obtained a 98% obliteration rate by increasing the passage time and reducing the power (10–18 W vs 20–25 W). We believe that 60–70 J/cm energy is required for a sustainable occlusion. One pullback passage of 1 cm/s delivers only 25 J/cm. We recommend performing multiple pullbacks, thus increasing the passage time for most optimal obliteration rate.
Our study’s clinical follow-up via ultrasound monitoring was done at 1 week, 1 month, 6 months, 1 year, 2 years and 3 years. This follow-up monitoring and duration further strengthened our described RFiTT protocol (e.g. multiple pullbacks thus increasing the probe’s passage time) and findings. Our study’s 3-year survival occlusion rate of 96.71%, 98% for overall and RFiTT patients, respectively, demonstrated the long-term efficacy of our RFiTT protocol.
According to the literature, RFiTT can be cost-effective for the society.19,20 We believe that RFiTT can reduce procedure time and hospitalization rate and increase treatment turn-over. All of which contribute to the feasibility of RFiTT in a hospital outpatient setting. The clinical effectiveness and cost-effectiveness of RFiTT versus operative intervention are reported to be based on rates of post-operative technical recurrence rather than symptomatic recurrence. 20 Our 3-year post-intervention obliteration rate results clearly demonstrated dominant effectiveness of RFiTT compared with operative technique.
Very few RFiTT-induced complications were reported at the end of the intervention. No pulmonary embolism was reported. One RFiTT patient who received thromboprophylaxis (single injection of 2.5 mg Fondaparinux) presented with common femoral vein sclerosis expansion of only one limb at 3 weeks post-treatment. She wore class 2 low thigh venous compression stockings post-intervention. The limb’s EHIT 21 and edema recovered completely. No lesions were observed on ultrasound Doppler after 3 weeks of Rivaroxaban (Xarelto®, Bayer, Germany) at curative dose. In line with the literature, this patient did not practice any sports such as running for 15 days to avoid EHIT post-intervention. 21 Sutton et al. 22 showed an increased thrombotic risk after GA and phlebectomy. Newman et al. 23 reported three deep venous thrombosis (DVTs) in each treatment group for a total of 534 patients. Braithwaite et al. 16 reported four DVTs in 462 patients. Hamel-Desnos and Desnos 24 reported no post-intervention thrombotic complications.
No neurological disorders were reported in our study, that is, one case of painful paresthesia of medial thigh for 15 days was recovered after treatment with analgesics and oral nonsteroidal anti-inflammatory drugs (NSAIDS). These results were better than those of the literature. Hamel-Desnos and Desnos 24 reported 13 over 119 cases of dysesthesia with spontaneous recovery and no sequelae in less than 6 months. For these cases, the intervention time has been reported between 9 and 14 s per cm for a power of 19 W (i.e. a single pullback passage).
Braithwaite et al. reported 39 dysesthesia of 672 treated veins, in 462 patients. These dysesthesia recovered in less than 9 weeks. 16 In comparison with our work, Braithwaite et al. showed that higher rate of neurological disorders could be explained by their higher use of GA compared with tumescence infusion (GA: 81.5% vs tumescence: 73.1%).
We strongly suggest the use of tumescence infusion in prevention of neural lesions (i.e. neurological disorders). The tumescence infusion was aimed at obtaining a floppy sapheno- and muscular fascia. This was achieved by injection of the tumescence sea very close to GSV to push the little sub-cutaneous nervous branches away from the vein. In this manner, patients who undergo LA and tumescence infusion can describe the treatment-induced pain, thus hinder possible neural lesions. The 18 W power setting on Celon RFiTT used in our study most likely hindered the potential side effects. The mean VAS was 2.08, immediately post-intervention. This was consistent with the results reported by Hamel-Desnos et al. (VAS = 2.1 and 1.5). In two cases of hypersensitivity and anxiety regardless of premedication, Kalinox 50® was used before tumescence injection. This additional anesthesia was well tolerated by the two patients.
Given our unit’s experience, the only disadvantage of Celon RFiTT is the coagulum build-up at the tip of the probe. There does not seem to be a significant difference in procedure time between RFiTT and Venefit® (“Closure Fast”). According to our clinical experience, treatment of a 28-cm segment takes around 1.40 min for “Closure Fast” versus 2.20 min for RFiTT after five pullbacks.
There are several limitations to our study. First, this was a retrospective study thus not designed to use a comparator control group. It is worth mentioning that given the small number of cases according to our clinical experience, using an historical comparator group did not seem justified. Other limitation was that we did not carry a Clinical Satisfaction Survey Score among patients. Nevertheless, given the fact that patients could return to work or take up a physical activity within 2–3 days post-intervention, we do believe that the quality of life after RFiTT is good. This was demonstrated by the results of CIVIQ-14 obtained after an ultrasound monitoring a month after RF treatment. According to these results, patients recovered within a few days from post-RF complications.
Conclusion
Our study demonstrates that thermal endovenous treatment of varicose veins using Celon RFiTT to be efficient at short, medium and long term. Celon RFiTT is safe, easy to be deployed in outpatient unit and under LA. We strongly recommend an 18 W power setting with a withdrawal speed of 1 s/cm and 5–10 passages on the treated segment. Our Celon RFiTT protocol avoided adverse and lasting side effects. This protocol seems promising in regard to patients’ outcomes as well as patients’ preferences (e.g. quality of life) for more rapid recovery after surgery. The reported dominant effectiveness of RFiTT versus operative technique can assist healthcare decision-makers in comparing the impact of various alternative interventions on the entire target population, under a pre-specified budget constraint. Moreover, Celon RFiTT technique requires training of healthcare providers, such as vascular physicians. This should be done in combination with expert use of vascular ultrasound for the best targeting and management of the varicose indications and in prevention of adverse side effects.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was waived by Brest CHRU Institutional Review Board.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects before the study (an information sheet on the venous treatment technique provided by the French Vascular Medicine Society was given to the patients who signed/agreed to be treated in accordance with the French guidelines). Verbal informed consent was obtained from all subjects before the study.
