Abstract
Objectives:
Combining vorinostat, L-asparaginase, and doxorubicin (Dox) led to improved response rates in the treatment of lymphoid tumors. However, deep-vein thrombosis has been noted as one of the most serious side effects with these drugs, and how these regimens cause deep-vein thrombosis is unclear.
Methods:
We investigated the procoagulant effects of vorinostat, L-asparaginase, and doxorubicin in lymphoid tumors, focusing on tissue factor, phosphatidylserine, and antithrombin. The human vascular endothelial cell line EAhy926 as well as the lymphoid neoplastic cell lines HUT78 (cutaneous T-cell lymphoma), Molt4 (acute T-lymphoblastic leukemia), and Ramos (Burkitt lymphoma) were employed to investigate these procoagulant effects.
Results:
Vorinostat, L-asparaginase, and doxorubicin induced exposure of phosphatidylserine and procoagulant activity on the surface of lymphoid tumor cells. Vorinostat and doxorubicin also induced phosphatidylserine exposure and increased procoagulant activity on EAhy926 cells. Expression of tissue factor antigen was induced by doxorubicin on the surface of each type of cells, whereas expression of tissue factor mRNA was unchanged. Secretion of antithrombin from HepG2 cells was reduced only by L-asparaginase.
Conclusion:
These data suggest that vorinostat and doxorubicin may induce procoagulant activity in vessels through apoptosis of tumor cells and through phosphatidylserine exposure and/or tissue factor expression on vascular endothelial cells. L-asparaginase may induce a thrombophilic state by reducing the secretion of anticoagulant proteins such as antithrombin. The laboratory methods described here could be useful to evaluate the procoagulant effects of antineoplastic drugs.
Introduction
Lymphoid tumors comprise primarily acute lymphoblastic leukemia (ALL), malignant lymphoma, and multiple myeloma. Epigenetic regulation of gene transcription by small-molecule inhibitors of histone deacetylases (HDACs) is a novel type of cancer therapy. Vorinostat (Vor) is the first HDAC inhibitor approved by US Food and Drug Administration for treatment of patients with cutaneous T-cell lymphoma (CTCL) who have progressive, persistent, or recurrent disease during or after two types of systemic therapy. CTCL is a heterogeneous group of extranodal non-Hodgkin’s lymphomas characterized by clonal proliferation of mature T cells localizing to the skin. HDAC inhibitors are a new class of antineoplastic agents and are being evaluated in several clinical trials for various cancer types, as monotherapy, and in combination with other effective therapies.
Healthy cells maintain a balance between acetylation and deacetylation of histones that is mediated by the activity of histone acetyltransferases (HATs) and HDACs. Cancer cells may also have reduced activity of HAT thorough mutations, which results in silencing of genes such as suppressors. HDAC inhibitors bind to the active sites in class-I and class-II zinc-containing HDAC enzymes. Suppression of gene transcription by HDAC inhibitors blocking removal of acetyl-groups permits DNA transcription.1,2
Deep-vein thrombosis (DVT) has been reported to be one of the most serious side effects of HDAC inhibitors. Vor can increase the risk of DVT by ≈5%. DVT is usually caused by stasis, damage to blood vessels, and abnormal activation of coagulation. If DVT is aggravated, it may lead to pulmonary thromboembolism (PTE), which can be lethal.
L-asparaginase (L-asp) is effective against lymphoid tumors, especially ALL and certain types of lymphoma. 3 L-asp can increase the risk of DVT by ≈8%. 4 Most of the important events occur during the induction phase of therapy. 5 L-asp is an enzyme that converts L-asparagine to L-aspartic acid and ammonia. Hence, extracellular pools of L-asparagine in the body are exhausted rapidly. L-asparagine is a type of amino acid necessary for protein synthesis and an essential nutrient to leukemic blasts or lymphoma cells because they lack the enzyme L-asparaginase synthetase, thereby making L-asp highly cytotoxic.6,7
Doxorubicin (Dox) is a chemotherapeutic agent developed in 1967 in Italy. Dox can enter the base pairs of DNA of neoplastic cells and inhibit the reactions of DNA, RNA polymerases, and topoisomerase II, and disrupts the synthesis of DNA and RNA. Dox has particular sensitivity for the S period of the cell cycle. 8 Unfortunately, DVT has been reported to be a side effect of Dox use. Our research team has reported that Dox increases procoagulant activity (PCA) as well as expression of tissue factor (TF) and phosphatidylserine (PS) on the surface of endothelial cells and monocytes. 9 The influence of Dox upon hepatocytes and lymphoid neoplastic cells has not been investigated. Several clinical studies have shown similar thrombotic side effects, but the mechanism of action of thrombosis by Vor, L-asp, or Dox is not clear.
TF is a key coagulant factor that triggers the extrinsic clotting cascade. TF catalyzes the changeover of the inactive protease factor X/IX into the activated factor X (Xa)/IXa. TF is expressed in a homeostatic manner in several types of extravascular cell but is not, in general, expressed in cells that come into contact with blood. It is known that monocytes and vascular endothelial cells can express TF in response to pathologic stimuli. PS is isolated from the inner leaflet of the phospholipid bilayer, but becomes exposed if the membrane structure collapses by apoptosis, and is thought to be associated with TF “decryption.” 10 PS activates the coagulation cascade. 11 Therefore, increased PS exposure can cause accretion of PCA.
Antithrombin (AT) is an important inhibitor of blood coagulation. Antithrombin or anti-Xa activity of AT is potentiated after its binding to heparan sulfate on the vessel endothelium, or to heparin or heparinoid medicinal agents. In this study, we investigated the procoagulant effects of Vor compared with L-asp or Dox in vascular endothelial cells, hepatocytes, and lymphoid neoplastic cells, and focused particularly on TF, PS, and AT.
Materials and methods
The study protocol was approved by the Ethics Committee of the Faculty of Medicine, Tokyo Medical and Dental University, Tokyo, Japan (no. 1730).
Reagents
Vor (Taihoyakuhinkougyou, Tokyo, Japan) was dissolved in dimethyl sulfoxide, and then added to media to a final concentration of 0.5, 1.0, and 2.0 µM. L-asp (±) (Kyowahakkoukirin, Tokyo, Japan) was dissolved in water (Otsuka Pharmaceuticals, Tokyo, Japan) and normal saline solution (NSS), and then added to media to a final concentration of 0.1, 1.0, and 3.0 U/mL. Dox (Sigma–Aldrich Japan, Tokyo, Japan) was dissolved in ethanol and then added to media to a final concentration of 0.1 and 1.0 µM. As an untreated control, the same amounts of dimethyl sulfoxide, NSS, and ethanol were added to the culture medium, respectively. The pharmacologic concentrations of each agent were added to media and incubated for the times indicated.
Cell culture
As a model of endothelial cells within blood vessels, we used the representative human umbilical vein endothelial cell line EAhy926. The latter was kindly provided by Dr Edgell (University of North Carolina, Chapel Hill, NC, USA). As model of hepatocytes, we used HepG2 cells (Riken BioResource Center (BRC), Ibaraki, Japan). EAhy926 and HepG2 cells were cultured in Dulbecco’s modified Eagle’s medium with low-glucose media (Wako Pure Chemicals, Osaka, Japan) supplemented with 10% fetal bovine serum (Sigma–Aldrich Japan) and 50 U/mL penicillin-streptomycin. HUT78 (CTCL), Molt4 (acute T-lymphoblastic leukemia), and Ramos (Burkitt lymphoma) lymphoid neoplastic cell lines were obtained from Riken BRC and the American Type Culture Collection (Manassas, VA, USA) and cultured in RPMI1640 medium (Wako Pure Chemicals) supplemented with 10% fetal bovine serum and 50 U/mL penicillin-streptomycin. We could not obtain sufficient numbers of vascular endothelial cells and lymphoma cells for assays, so we used these cell lines. To assess the various effects of antineoplastic drugs, those cell lines with each malignant cell type are most popularly used in the world.
PCA assay
Each cell line was incubated with Vor, L-asp, or Dox for 8 or 24 h. EAhy926 cells and HepG2 cells were confluent and the other tumor cell lines proliferated in suspension. EAhy926 cells and HepG2 cells were taken off from plates by 0.25% trypsin and washed with phosphate-buffered saline (PBS) twice. A portion of cells (2 × 106) was suspended in 50 µL of PBS, and added to 50 µL of pooled normal human plasma. After incubation at 37°C for 3 min, 50 µL of 25 mM calcium chloride was added and the plasma recalcification time measured using a semi-automatic coagulator (CA-50; Sysmex, Kobe, Japan).9,12
Cell-surface TF antigens (flow cytometric analyses)
Each cell line was incubated with Vor, L-asp, or Dox for 24 h. Cell suspensions in PBS were incubated first with a monoclonal anti-TF antibody (American Diagnostics, Greenwich, CT, USA) and then with a fluorescein isothiocyanate (FITC)-labeled anti-mouse IgG antibody (Beckman Coulter, Fullerton, CA, USA) for 60 min on ice. After washing with PBS, cell suspensions were run on a Flow Cytometer (FACScan; Becton Dickinson, San Diego, CA, USA) using acquisition and analysis software (CellQuest; Becton Dickinson).
Quantitative reverse transcription polymerase chain reaction and densitometric analyses
EAhy926 and Molt4 lines were incubated with Dox for 4 h. Total cellular RNA was isolated using a high-purity RNA isolation kit (Roche Diagnostics, Mannheim, Germany). Reverse transcription polymerase chain reaction (RT-PCR) was carried out using a Titan One Tube RT-PCR kit (Roche Diagnostics) according to manufacturer instructions. cDNA derived from each cell line was amplified by 28 PCR cycles. The relative signal intensity of bands was determined and standardized using imaging software (Scion, Frederick, MD, USA), as described previously.9,12
Exposure of cell-surface PS (flow cytometric analyses)
Suspensions of each cell line in 1 × annexin V binding buffer including 1.8 mM CaCl2 (Beckman Coulter) were incubated with FITC-labeled annexin V (Beckman Coulter) for 15 min on ice and with propidium iodide (Beckman Coulter). Immediately after the antibody reaction, aliquots were run on a Flow Cytometer (FACScan) using acquisition and analysis software (CellQuest).
Cell-surface PCA after incubation with anti-TF antibody
To investigate the effect of cell-surface TF on PCA induced by Dox, all cell lines were incubated with 1.0 µM Dox. After 8 h, samples were preincubated with 10 µg/mL monoclonal anti-TF antibody (ADG4509; American Diagnostics), monoclonal mouse anti-human TF IgG, or the same amount of “irrelevant” IgG, in PBS for 60 min on ice. After washing with PBS, cell-surface PCA was assessed as described above.
Cell-surface PCA after incubation with annexin V
To investigate the effect of exposure of cell-surface PS to 2.0 µM Vor, 1.0 µM Dox, or 3.0 U/mL L-asp upon PCA stimulation, samples were post-incubated with 1 µg/mL annexin V (AnaSpec, San Jose, CA, USA) in 300 µL annexin V binding buffer at 37°C for 30 min. After incubation, cell-surface PCA was assessed as described above.
Anti-FXa activity of AT
HepG2 cells were incubated with Vor, L-asp, or Dox for 48 h. An aliquot of cells (1 × 106) was cultured in serum-free Dulbecco’s modified Eagle’s medium with 50 U/mL penicillin-streptomycin. We examined the anti-FXa activity of AT in 2-mL cell culture media using an Antithrombin III Kit Testzym S (Sekisui Medical, Tokyo, Japan). Finally, activity levels were measured at an absorbance of 405 nm. We used an analytical curve in which the reagent blank was NSS (0%) and the calibrator was normal human plasma (100%).
Statistical analyses
Data are the mean ± standard deviation (SD). Statistical analyses were carried out using the non-parametric Mann–Whitney U-test for paired data using GraphPad Prism 5 (GraphPad, La Jolla, CA, USA). Significant differences are indicated (*) with (p < 0.05) compared with control, respectively, whereas (**) indicates a significant difference with (p < 0.01), and (***) with (p < 0.001) compared with control, respectively.
Results
Effects of Vor, L-asp, and Dox on cell-surface PCA
We ascertained whether or not Vor, L-asp, or Dox could increase cell-surface PCA in HUT78, EAhy926, Molt4, and Ramos lines (Figure 1).
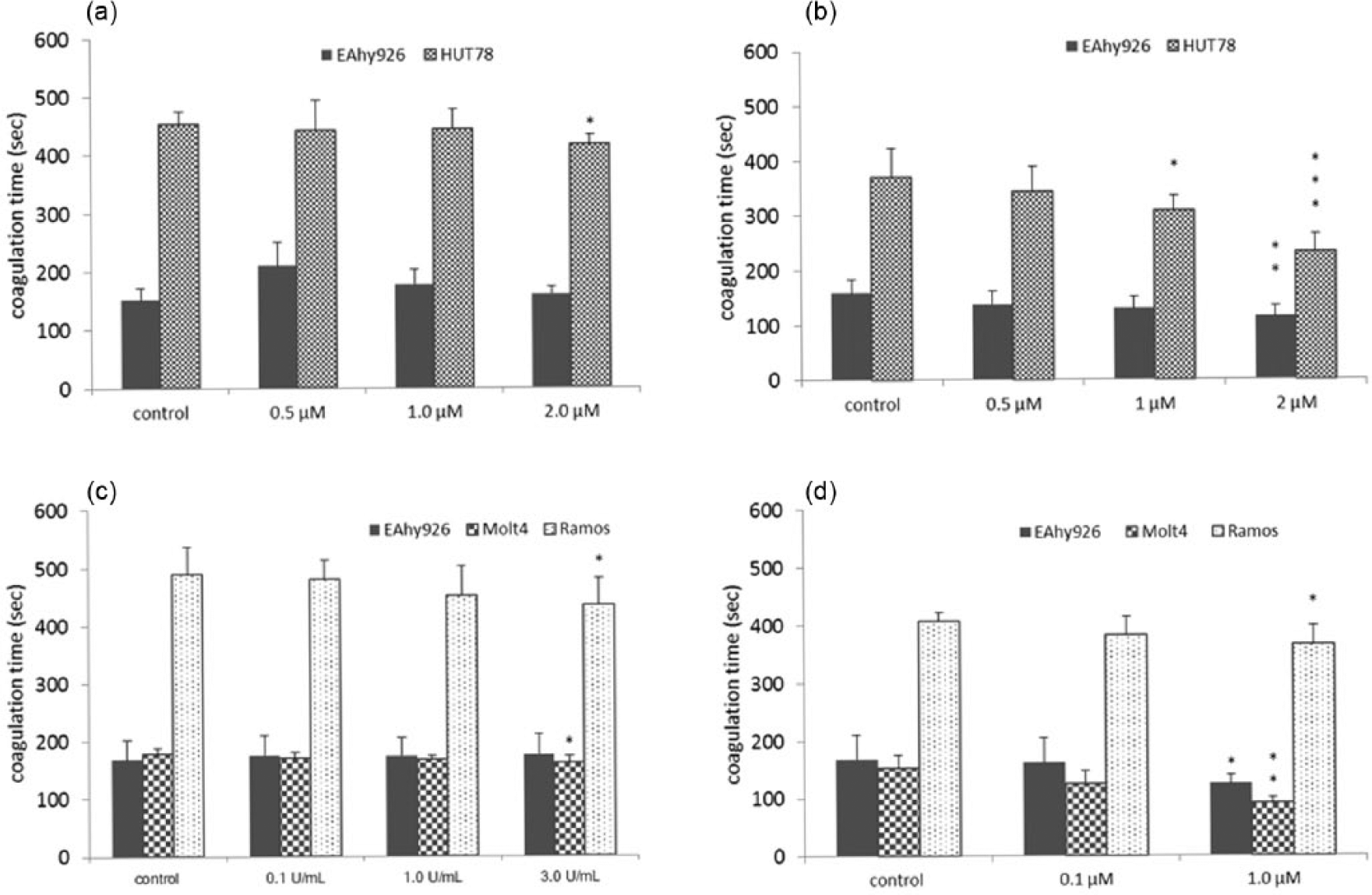
Effects of Vor, L-asp, and Dox on cell-surface PCA: (a) Vor (8 h), (b) Vor (24 h), (c) L-asp (8 h), and (d) Dox (8 h).
After 8 h of treatment with 2.0 M Vor, PCA was increased more in HUT78 cells than in control cells (Figure 1(a)). In EAhy926 cells, PCA was not significantly different compared with control cells, so we incubated EAhy926 cells for 24 h with Vor. Vor increased PCA of HUT78 cells and EAhy926 cells clearly after 24 h of treatment (Figure 1(b)).
After 8 h of treatment with 3.0 U/mL L-asp, PCA was increased to a greater extent in Molt4 cells and Ramos cells than in control cells (Figure 1(c)). These results suggested that L-asp induced PCA on the surface of lymphoid tumor cells.
Upon incubation with 1.0 µM Dox, PCA was increased in EAhy926, Molt4, and Ramos cells compared with control cells (Figure 1(d)). In particular, the increase in Molt4 cells was considerable. Moreover, Dox induced a shorter coagulation time than that seen for L-asp.
Effects of Vor, L-asp, and Dox on expression of TF antigen on cell surfaces
Quantification of expression of TF antigen on the cell surface was carried out by flow cytometry. We measured this parameter for all cell lines upon treatment with Vor, L-asp, or Dox for 24 h (Figure 2).

Effects of Vor, L-asp, and Dox on expression of surface TF antigen: (a) Vor, (b) L-asp, and (c) Dox.
After treatment with Vor, expression of TF antigen on the cell surface was barely greater than that of control (Figure 2(a)). Upon stimulation with 3.0 U/mL L-asp, the level of TF antigen on Ramos cells was increased to ≈0.3% (Figure 2(b)).
When all cell lines had been stimulated with Dox, expression of TF antigen on the cell surface was higher than that seen in control cells (Figure 2(c)). Expression of TF antigen on the surface of all cell types after Dox treatment was induced further compared with that observed for Vor or L-asp.
Effects of Dox on expression of TF mRNA
To reveal the transcriptional stimulus for TF production in EAhy926 cells and Molt4 cells, levels of TF mRNA were quantified by RT-PCR. Expression of TF mRNA in Ramos cells was not observed. After Dox treatment, levels of TF mRNA were unchanged in all cell types (Figure 3).

Effects of Dox on TF mRNA.
Effects of Vor, L-asp, and Dox upon cell-surface PS exposure
Annexin V associates with PS with high compatibility in the phospholipid bilayer (which contains PS). Therefore, an increase in levels of annexin V suggests that alterations in the plasma membrane have occurred because of the medication administered, and coagulation may be promoted.13,14 Flow cytometry was used to distinguish between living and apoptotic cells after treatment for 24 h. PS exposure on the surface of each cell line was increased significantly compared with that observed with control cells (Figure 4). Increasing concentrations of Vor, L-asp, and Dox were associated with increasing PS exposure.

Effects of Vor, L-asp, and Dox on expression of cell-surface PS exposure: (a) Vor, (b) L-asp, and (c) Dox.
Effects of Dox on cell-surface PCA blocked by anti-TF antibody
Dox (1.0 µM) increased expression of TF antigen on the surfaces of all cell types markedly. We examined if anti-TF antibody could block cell-surface PCA.
In all cell lines, upregulated cell-surface PCA was blocked significantly by anti-TF antibody. These results suggested that increases in PCA by Dox regimens were induced by upregulation of the expression and activity of TF on the cell surface (Figure 5).

Effects of Dox on cell-surface PCA blocked by anti-TF antibody.
Annexin V blocks the procoagulant effects of PS
We examined whether or not exposure of cell-surface PS to 2.0 µM Vor, 3.0 U/mL L-asp, or 1.0 µM Dox could increase cell-surface PCA. We used annexin V as a PS inhibitor. In all cell lines, post-incubation of samples with annexin V suppressed the increase in PCA by each drug regimen (Figure 6).
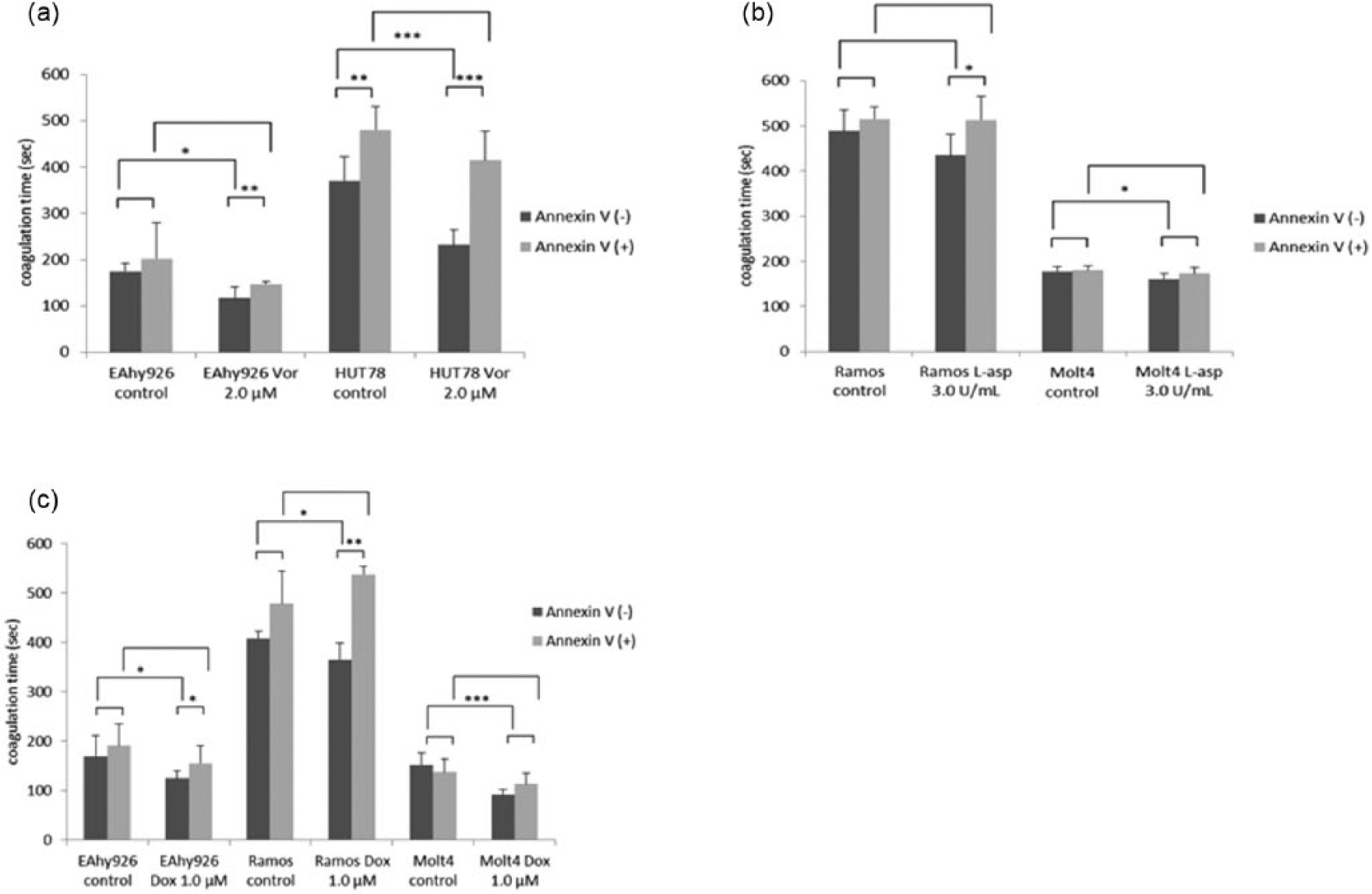
Effects of incubation with annexin V on cell-surface PCA: (a) Vor, (b) L-asp and (c) Dox.
Effects of Vor, L-asp, and Dox upon the anti-FXa activity of AT in culture supernatants
HepG2 cells were used as a model of hepatocytes to produce AT. Anti-FXa activity of AT in cell culture supernatants was decreased significantly by L-asp compared with that observed with control cells (Figure 7(b)). However, when HepG2 cells were stimulated with Vor or Dox, the anti-FXa activity of AT was barely changed (Figure 7(a) and (c)).
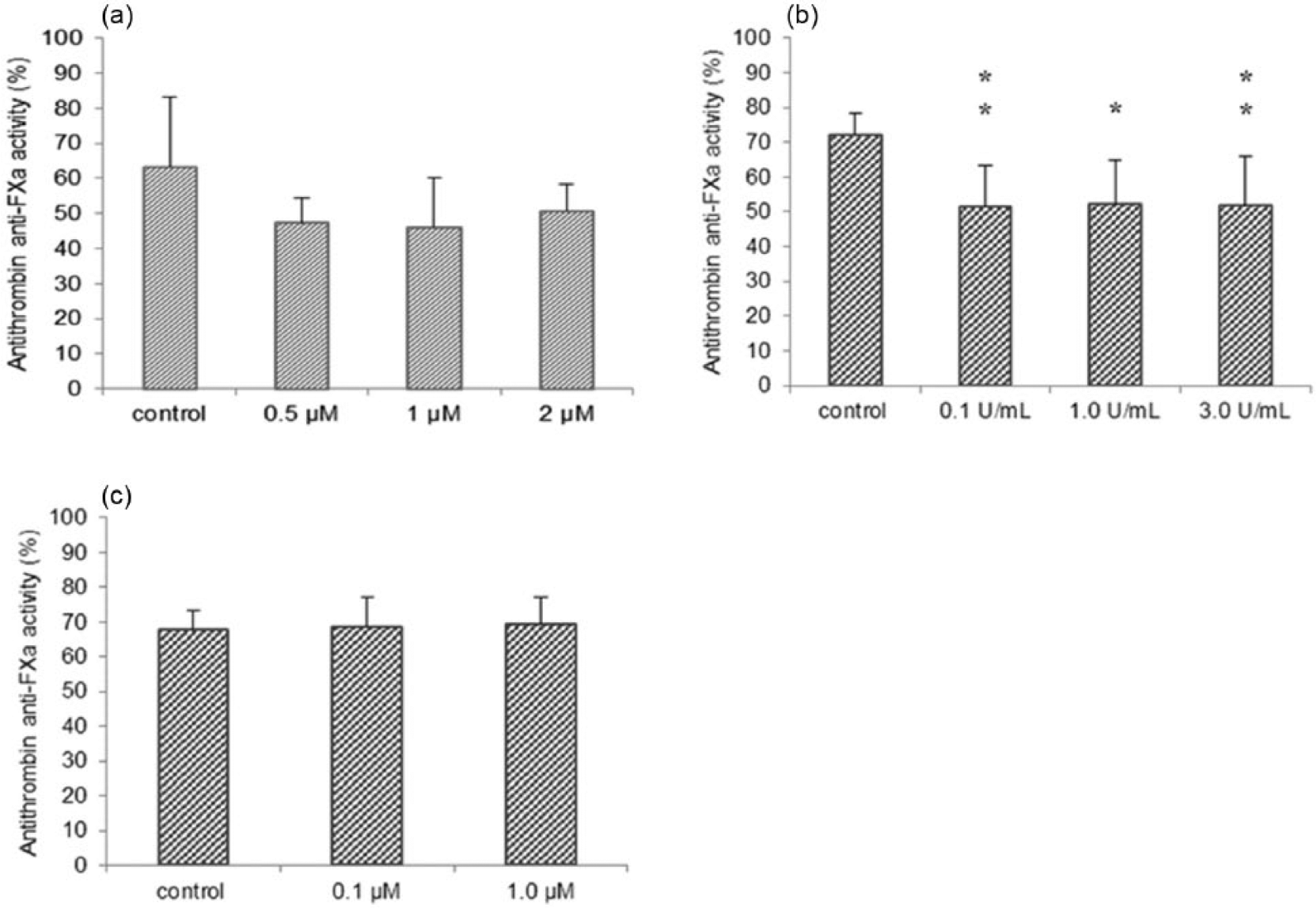
Effects of Vor, L-asp, and Dox on AT anti-FXa activity in culture supernatants: (a) Vor, (b) L-asp, and (c) Dox.
Discussion
We showed that antineoplastic drugs can increase PCA on the surface of vascular endothelial cells and lymphoid tumor cells. To ascertain why PCA was increased by these antineoplastic drugs in the vasculature of patients, we focused on expression of TF and PS on the surface of human cells.
In the recalcification time using the normal human plasma-based cell suspension, TF-expressing cells embody the prothrombin time and reflect the extrinsic coagulation cascade. Conversely, TF-absent cells embody the partial thromboplastin time and reflect the intrinsic clotting system (which is dependent upon PS).
Upon Vor treatment, PCA on the surface of vascular endothelial cells and lymphoid tumor cells was increased. When these cells were incubated with Vor for 24 h, PCA increased more obviously than that after incubation with Vor for 8 h. These data suggest that exposure to Vor regimens for a long period increases the risk of thrombosis. The level of TF antigen on all cell types barely changed. Conversely, PS expression on the surface of lymphoma cells was increased markedly, which suggests that Vor has antitumor activity because PS expression is dependent upon apoptosis. Annexin V suppressed PCA in all samples to that seen for control cells, so cell-surface PS exposure may be correlated with an increase in PCA. However, AT activity was unchanged and did not seem to be associated with hypercoagulability.
HDAC inhibitors are used for the treatment of blood cancers. HDAC inhibition also results in beneficial outcomes in various type of neurodegenerative diseases, inflammatory disorders, and cardiovascular diseases. 15 Clinical results with Vor used as a single agent have not been successful in the treatment of solid malignancies, but preclinical data strongly suggest that combination with conventional cancer therapies would be beneficial. 16 However, owing to the reported side effects of PTE when Vor has been used as a single agent, combination therapy could cause severe thrombosis.
L-asp showed similar results to that seen for Vor. In contrast with Vor, AT activity from hepatocytes was decreased markedly. This decrease in AT activity may appear to induce hypercoagulation. Hunault-Berger et al. 17 showed that AT levels were <60% in 73% of patients. Thrombosis occurred in 9.3% of those patients, who had a median AT level of 53% at the time of the event. Our results are in accordance with those observed in clinical studies. AT deficiency could promote the procoagulatory effects of L-asp. Most studies have shown that infusion of fresh frozen plasma or AT concentrates is very effective for lowering the risk of DVT.17,18 Anticoagulant therapy may be necessary if patients with lymphoid tumors are treated with Vor or L-asp.
Upon stimulation with Dox, PCA was increased in endothelial cells and lymphoid tumor cells compared with that observed in control cells. Expression of cell-surface TF antigen and PS exposure were increased markedly on all cell types but was not accompanied by upregulation of expression of TF mRNA. In each cell line, increased PCA was blocked by anti-TF antibody and a PS inhibitor. In accordance with other reports,9,19,20 expression of TF mRNA remained unchanged, so increased expression of TF antigen may originate from “encrypted” TF. Previously, we reported that Dox increases PCA as well as expression of TF and PS on the surface of endothelial cells and monocytes. 9 This study has revealed similar effects on lymphoid neoplastic cells. AT activity does not seem to be associated with hypercoagulability.
TF in the vasculature can contribute to thrombin generation and fibrin formation after an activation event. Most of the TF on the cell surface is encrypted or “coagulation-inactive.” 21 The “decryption” step involves PS exposure, but PS exposure alone is not sufficient for the full activity of TF. Externalization of PS is a component of apoptosis. Apoptotic cells release microparticles (MPs), which are derived from various cell types (e.g. platelets, leukocytes, and endothelial cells). Endothelial MPs are directly indicative of stress or damage to endothelial cells, and may reflect endothelial inflammation. Increased circulating levels of MPs are encountered in diseases associated with vascular involvement and hypercoagulability.22–24 Fu et al. reported that another anthracycline, daunorubicin, induces PCA on endothelial cells and human umbilical vein endothelial cells through PS exposure and MP release. 20 Therefore, in the blood vessels of patients, the cell membrane may be damaged by anthracycline antineoplastic medications. Cells exposed to increased PS or TF as well as MPs may circulate through the whole body and induce blood coagulation. We intend to further investigate changes in the secretion of the anticoagulant proteins, protein C and protein S, from hepatocytes and thrombomodulin expression in vascular endothelial cells due to treatment by antineoplastic drugs. D-dimer is a marker used in the diagnosis of DVT. For DVT prevention in the treatment of patients with lymphoid tumors, monitoring of D-dimer levels and consideration of anticoagulant therapy may be necessary.
Limitations of this study may be as follows. Our laboratory methods presented in this study do not include whole blood assay. Intravascular PCA is mainly dependent upon coagulation activity in plasma but may be also influenced in part by blood cells such as platelets, monocytes, neutrophils, and erythrocytes. We are now preparing inclusion of a whole blood assay, rotational thromoboelastometry into our laboratory assessment of PCA by antineoplastic drugs. Furthermore, in vivo intravascular PCA reflect patients’ performance status and vascular integrity. While we always have to take patients’ clinical status into consideration, our laboratory methods presented here are well compatible with procoagulant effects in real clinical scenes. Therefore, in conclusion, our cell line and plasma-based laboratory methods may be simple and helpful to evaluate procoagulant effects of antineoplastic drugs.
Footnotes
Acknowledgements
Misae Tsunaka and Reina Arai contributed equally to this study. This study was presented in part at the 24th and 25th Congress of the International Society of Thrombosis and Haemostasis, Amsterdam, The Netherlands, July 2013 and Toronto, ON, Canada, June 2015.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study protocol was approved by the Ethics Committee of the Faculty of Medicine, Tokyo Medical and Dental University, Tokyo, Japan (no. 1730).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects before the study.
