Abstract
Background:
Throughout 2007 and January 2008, several glitazones health warnings were published on rosiglitazone myocardial infarction risk. The impact of such warnings on glitazones prevalence of utilization has been extensively studied in the United States but only in one European country (England), which has showed different pattern from US studies. The aim of this study is to evaluate the impact of such safety warnings on glitazones utilization in an area of another European country.
Methods:
We calculated the number of defined daily doses per thousand inhabitants per day of glitazones each month during the period from 2006 to 2008 in a health area of Spain. We analyzed the data graphically and through a segmented regression analysis.
Results:
Rosiglitazone defined daily doses per thousand inhabitants per day were growing before the safety warnings, after the warnings a change in trend occurred and rosiglitazone utilization showed a downturn slope. Pioglitazone defined daily doses per thousand inhabitants per day were stable before the safety warnings, and a linear growth was observed after the safety warnings. Throughout the study period, rosiglitazone defined daily doses per thousand inhabitants per day were higher than pioglitazone defined daily doses per thousand inhabitants per day until near the end of 2008.
Conclusion:
Despite the fact that cardiovascular warnings affected rosiglitazone and not pioglitazone, rosiglitazone was more utilized than pioglitazone until near the end of 2008 which is a pattern similar to the one found in another European studies in England, but very different from studies in the United States, where rosiglitazone was less utilized than pioglitazone from the first month after rosiglitazone cardiovascular safety warnings.
Introduction
Glitazones (Thiazolidinediones) are a group of oral antidiabetic agents that appear to exert their mechanism of action by increasing the sensitivity of insulin target cells. The first representative of this group (troglitazone) was withdrawn from the market due to hepatotoxicity. 1 The two glitazones available in Spain (pioglitazone and rosiglitazone) were granted marketing authorization in the year 2000 (rosiglitazone) and 2001 (pioglitazone).
Even though the mechanism of action of glitazones was different from that of the other oral antidiabetic medicines available at the moment of their commercialization (sulfonylureas, metformin, alpha-glucosidase inhibitors), glitazones efficacy in reducing blood glucose is modest compared to the well-established therapies such as metformin and sulfonylureas 2 and besides glitazones, unlike metformin, failed to demonstrate a reduction in cardiovascular complications.3,4
At the moment when rosiglitazone and pioglitazone were first commercialized, their more worrisome known side effects were a risk of hepatotoxicity and fluid retention; however, subsequent studies showed new risks, such as an increased risk of fractures in women3,5 and in the case of rosiglitazone an increase in myocardial infarction as well, 6 which altered their risk and benefit ratio and has led to warnings from drug regulating agencies. Thus, the Spanish Agency of Medicines and Medical Devices issued the following four safety warnings: in April 2007, a warning about a risk of bone fractures due to rosiglitazone and pioglitazone; in May 2007, a warning about the risk of myocardial infarction with rosiglitazone; in October 2007, a warning stating that a precaution in patients with ischemic heart disease was included in rosiglitazone summary of product characteristics and in January 2008, a warning informing on the introduction into rosiglitazone summary of product characteristics of a contraindication in patients with acute coronary syndrome.
Rosiglitazone cardiovascular safety warnings issued by the Spanish Agency of Medicines and Medical Devices were coincidental in time and content with those of the safety warnings issued by the European Medicines Agency (EMA). The EMA decided in September 2010 that rosiglitazone commercialization should be suspended due to its cardiovascular risks, and the commercialization of rosiglitazone in Spain was suspended at the end of December 2010.
The aim of this article is to evaluate the quantitative changes in the use of pioglitazone and rosiglitazone in Spanish health area following the publication of several health warnings on glitazones.
Methods
The setting of this study was a health area of the Spanish national health system. Being a national health system means that it covers the entire population and its main source of funding is via taxes. Rosiglitazone or pioglitazone treatment was publicly funded by the Spanish national health system, from their first commercialization, to any patient in accordance with EMA rosiglitazone or pioglitazone summary of product characteristics. This criterion did not change after glitazones safety warnings. There was not any kind of change with regard to the criteria for public funding to any other treatment for type 2 diabetes during the period of this study.
We carried out a retrospective descriptive study on the amount of glitazones (pioglitazone and rosiglitazone) utilized by the inhabitants of Santiago de Compostela health area. This health area is located in northwestern Spain and covers a population of 386,484 inhabitants. The study period extends from January 2006 to December 2008, so we could analyze trends in the use of pioglitazone or rosiglitazone after the publication of health warnings throughout 2007 and January 2008 compared with the previous trend.
Data on pioglitazone and rosiglitazone consumption were obtained from the national health system database as an aggregate number of glitazones-containing packages that had been dispensed per month in the health area under study and billed to the Spanish national health system throughout the study period. Subsequently, these data were transformed into number of defined daily doses (DDD) of pioglitazone or rosiglitazone per month considering that one DDD was 30 mg of pioglitazone or 6 mg of rosiglitazone, and finally, these data were transformed into number of DDD per thousand inhabitants per day (DDD/TID), which is the unit of measure recommended by the World Health Organization for studying drugs utilization. 7
As a first approximation, a graphical representation that contrasts time with the number of glitazones DDD/TID was created. In the statistical analysis, we considered the time between the first and the last safety warnings as a transition period, as they are very close in time and a similar subject is treated, so the two events are considered together as one event (safety warnings).
For statistical analysis purposes, we constructed a segmented regression analysis model, which allows to check whether or not there has been a statistically significantly both an immediate jump in the use of glitazones after the issuing of glitazones health safety warnings, as and any change in the long-term trend in the use of glitazones after the safety warnings compared to the long-term trend before the issuing of such safety warnings. In the regression analysis model, the dependent variable was DDD/TIDs of rosiglitazone, pioglitazone or the sum of glitazones per month and the independent variables were a time since baseline variable (t: 1, 2, 3 …), which would reflect the trend previous to the safety warnings, one binary variable that took a value of 0 before and a value of 1 after the safety warnings, which would show the immediate jump after the warning, and a time since the warning variable that took the value of 0 before the warnings and afterwards took the value of 1, 2,3 …, which would reflect the change in slope after the warning. For controlling autocorrelation, it was necessary to introduce a fifth-order autoregressive term for rosiglitazone and sum of glitazones. As goodness of fit of the model, we measured the R2 value.
Results
Figure 1 shows the pattern of glitazones utilization throughout the study period, where it is observed that rosiglitazone utilization increases previous to the safety warnings and decreases afterwards. In the case of pioglitazone, its utilization was stable previous to the alerts, and afterwards, there was an increase in its utilization. It should be noted that rosiglitazone was more utilized than pioglitazone until near the end of 2008. The sum of glitazones did not diminish after the safety warnings because even though rosiglitazone diminished, the utilization of pioglitazone augmented.
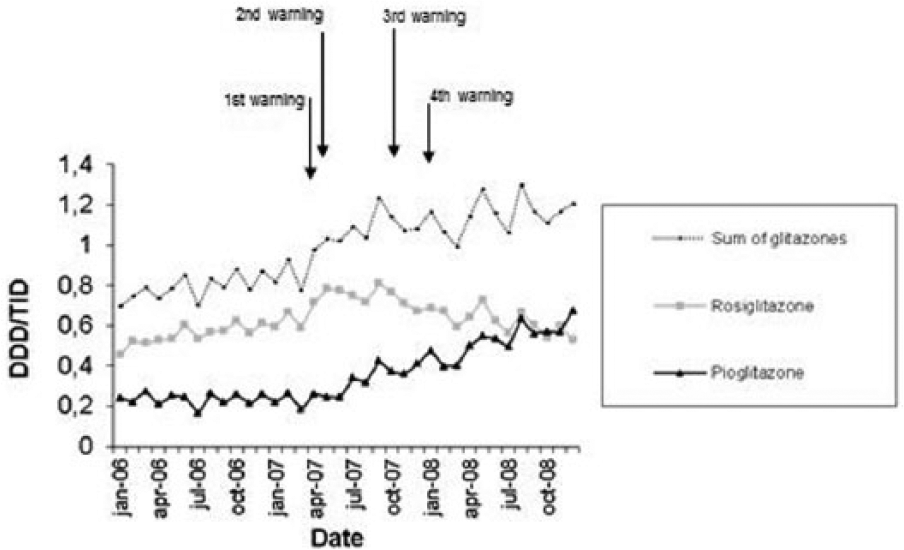
Number of glitazones defined daily doses per thousand inhabitants per day (DDD/TID) from 2006 to 2008.
The regression model showed as goodness of fit a R2 value of 0.932 for sum of glitazones, 0.730 for rosiglitazone and 0.974 for pioglitazone. The significance of regression coefficient results is given in Table 1, which shows that pioglitazone underwent a statistically significant jump after the safety warning as well as a statistically significant change in the rate of growth. In the case of rosiglitazone, the growth rate previous to the safety warnings was positive and the growth rate after the safety warnings was negative and statistically significant.
Segmented regression analysis.

Discussion
The results of our study indicate that the sum of glitazones did not diminish after the safety warnings because even though rosiglitazone utilization diminished, the utilization of pioglitazone augmented. Rosiglitazone utilization increased previous to the safety warnings and decreased afterwards. In the case of pioglitazone, its utilization was stable previous to the alerts, and then, there was an increase in its utilization. Rosiglitazone was more utilized than pioglitazone until near the end of 2008.
The temporal trends of the sum of glitazones utilization observed in our study are in line with studies in England8,9 and Autralia 10 where the sum of glitazones utilization did not diminished immediately after rosiglitazone cardiovascular warnings, but in contrast with studies in United States,11–13 where a decrease in global glitazones utilization was observed.
As each glitazone in particular, the trend in the use of rosiglitazone in our study is similar to that found in studies in England and Australia, where a slow decline in rosiglitazone use was observed, but rosiglitazone was used more than pioglitazone at the end of the study period in Hall et al.’s 8 study in England (this study ended in January 2008) or rosiglitazone was used more than pioglitazone until the end of 2008 in studies in England 9 or Australia. 10 This is in contrast with the studies in the United States,11–14 which showed a sharp decline in rosiglitazone utilization, rosiglitazone being used less than pioglitazone from the next month after the first rosiglitazone safety warning.
Pioglitazone in our study experienced a steady increase after the warnings, similar to studies in England8,9 or Australia, 10 while in studies in the United States, pioglitazone either remained stable or experienced a small initial increase to remain stable thereafter.11–14
Before the publication of our study, the impact of glitazones warnings on their prevalence of utilization had been extensively studied in the United States but only in one European country (England), which had showed different pattern from US studies. The results of our study, which are similar to those of England, point to that the response in Europe to the glitazones safety warnings was worse than in the United States. It has to be noted that even though a study in the Netherlands has been published, 15 it has two main limitations: first, the study does not examine the prevalence of glitazones utilization, unlike the rest of glitazones studies, it only analyzes the incidence of glitazones utilization in a cohort of incident users since participants with a dispensing during the first 6 months of follow-up where excluded, and we consider that those patients should be studied because if they continue taking rosiglitazone, they continue to be exposed to a higher risk of the diseases included in the safety warnings. The second limitation is that in the Netherlands, prior authorization for glitazones was implanted in July 2007, 16 a time that is coincident with the safety warnings, and it is considered that prior authorization can cause a greater decrease in medicines utilization after its implantation than a safety warning;17,18 since prior authorization has an impact on glitazones utilization, 19 it would be difficult to disentangle whether a decrease in glitazones utilization is due to a safety warning or to prior authorization.
In order to understand the observed performance differences between European, Australian and US studies with regard to glitazones and its safety warnings, it can be postulated that there are several differences in European or Australian and US scenarios: first in the United States, a new oral antidiabetic drug (sitagliptin) had been marketed shortly before the first safety warning, which experienced a strong growth in its use, 11 while a decrease in the use of rosiglitazone was observed; while in Spain or other European country or Australia, no new oral antidiabetic was marketed neither shortly before nor during our study period, and a possible explanation why studies in Europe and Australia found that the use of pioglitazone increased while in the United States it did not increase is that prescribers the United States after the rosiglitazone cardiovascular alerts might have changed their prescriptions to new medicines such as sitagliptin (although it is difficult to know to what extent those changes from rosiglitazone to new medicines would have occurred anyway even if rosiglitazone health alerts had not happened) while in Europe or Australia, where no new antidiabetic medicines had been marketed, prescribers after rosiglitazone cardiovascular alerts decided to change their prescriptions to the other glitazone that presented no cardiovascular health warnings (pioglitazone). On the other hand, in the United States, these rosiglitazone safety warning was strongly scattered through the mass media because glitazones were popular to the public since prescription drugs advertising is permitted to the lay public; while in Spain, where like in any other European countries or Australia, prescription drugs advertising to the lay public is forbidden and glitazones were unknown to the general public, the mass media did not echo this drugs warnings. A study conducted in the United States found that 45.1% of patients who had discontinued rosiglitazone after the warnings had done so due to their own decision after learning the existence of safety warnings for that particular drug and not by decision of the prescribing healthcare professional. 20
Studies on the influence of safety warnings about other groups of drugs other than glitazones have found mixed results. In Spain, it was found that the use of selective serotonin reuptake inhibitor (SSRI) antidepressants in children and adolescents did not decrease despite a warning about the risk of suicide by these drugs, 21 or that a warning on telithromycin had no influence on the subsequent use of such medicine, 22 or that piroxicam utilization did not diminish after a safety warning regarding its relative toxicity 18 or that the use of strontium ranelate continued to grow despite a warning about its risks; although in the last case, the growth rate was slower after the warning. 23 Studies in other locations have also found mixed results, as the use of SSRI antidepressants in children and adolescents decreased after health alert in the United States 24 but not in Canada, 25 the use of atypical antipsychotics in the elderly continued to grow after health warnings in Canada (although at a slower pace than previously) 26 but decreased in the United States 27 or the use of telithromycin in the United States decreased after a health alert about such medicine. 28
There are several limitations to this study. First, this is an ecological study analyzing time series, which implies that any causal relationship between the issuance of a safety warning and changes in this drug utilization trends should be taken with caution. On the other hand, this study did not analyze from the qualitative point of view the compliance of the safety warnings in terms of fraction of glitazones users presenting cardiovascular contraindications; therefore, more studies would be needed. Our study also has many advantages: for example, as we have noted, the fact of using a national health system perspective, using DDD/TID as a unit of measurement and no any new antidiabetic drug being marketed shortly before the study period nor during the study period.
The two glitazones (rosiglitazone and pioglitazone) available during the study period had a similar hypoglycemic efficacy and a level of adverse effects that were similar except at cardiovascular level, where rosiglitazone was the only one with cardiovascular safety warnings, so it is logical to think that the use of rosiglitazone should be lower than the use of pioglitazone after rosiglitazone cardiovascular safety warnings. The sooner patients stopped being exposed to the cardiovascular risks of rosiglitazone the better. Studies for another group of medicines have found that following a health safety warning for such medicines, pharmaceutical companies’ promotion for such medicines did not cease and also their advertising campaigns continued trying to increase sales, including via medical associations. 29 For example, it has been found that when clinical trials were published with negative results for hormone replacement therapy (HRT), since they presented certain adverse effects, 30 some scientific societies provided information that was trying to counteract the information of such clinical trials and saying that prescribing of HRT should not change by such clinical trials, 31 and has been known that pharmaceutical companies had secretly briefed medical societies on HRT 32 and had used medical associations in order to promote HRT. 33 Studies are needed to ascertain whether something similar happened with rosiglitazone and how promotion evolved after safety warnings.
As a conclusion it can be said that despite the fact that cardiovascular warnings mentioned rosiglitazone and not pioglitazone, rosiglitazone was more utilized than pioglitazone in Spain until near the end of 2008 which is a pattern similar to the one found in studies in other European countries like England but very different from studies in the United States where rosiglitazone was less utilized than pioglitazone from the first month after rosiglitazone cardiovascular safety warnings. More studies are needed to delve into the possible explanation of such differences.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
