Abstract
Objectives:
Studies of palliative care are often performed using single-arm pre–post study designs that lack causal inference. Thus, in this study, we propose a novel data analysis approach that incorporates risk factors from single-arm studies instead of using paired t-tests to assess intervention effects.
Methods:
Physical, psychological and social evaluations of eligible cancer inpatients were conducted by a hospital-based palliative care team. Quality of life was assessed at baseline and after 7 days of symptomatic treatment using the European Organization for Research and Treatment of Cancer QLQ-C15-PAL. Among 35 patients, 9 were discharged within 1 week and 26 were included in analyses. Structural equation models with observed measurements were applied to estimate direct and indirect intervention effects and simultaneously consider risk factors.
Results:
Parameters were estimated using full models that included associations among covariates and reduced models that excluded covariates with small effects. The total effect was calculated as the sum of intervention and covariate effects and was equal to the mean of the difference (0.513) between pre- and post-intervention quality of life (reduced model intervention effect, 14.749; 95% confidence intervals, −4.407 and 33.905; p = 0.131; covariate effect, −14.236; 95% confidence interval, −33.708 and 5.236; p = 0.152).
Conclusion:
Using the present analytical method for single-arm pre–post study designs, factors that modulate effects of interventions were modelled, and intervention and covariate effects were distinguished based on structural equation model.
Introduction
Palliative care is defined by the World Health Organization as an approach that improves the quality of life of patients and their families facing the problems associated with life-threatening illness, through the prevention and relief of suffering by means of early identification and comprehensive assessment and treatment of pain and other problems, physical, psychosocial and spiritual.
1
Palliative care is provided through the combined expertise, knowledge and skills of various healthcare professionals and plays an important role in improving quality of life (QOL) by alleviating pain in patients and their family members. 2
The demand for evidence-based palliative care has been growing in recent years, and randomized controlled trials (RCTs) are indispensable for scientific evaluations of the effectiveness of palliative care. 3 However, because patients requiring palliative care are physically or mentally vulnerable and have diverse cultural and religious backgrounds, ethical considerations often preclude rigorous study designs. Accordingly, randomization of palliative care patients to treatment and reference arms is often difficult,4–8 and most studies in this field still rely on patient and epidemiological surveys.9–11
Although attempts to evaluate the effects of interventions using only a single treatment arm fail to reveal causal relationships due to the absence of rigorous controls, observations from single-arm studies are often critical and may be used to inform planning for subsequent study phases. Paired t-tests are predominantly used to examine intervention effects. However, we propose a novel data analysis approach that can be utilized with all available data from single-arm intervention studies. Specifically, we employ structural equation models (SEMs) with only observed measurements to evaluate intervention effects and simultaneously investigate modelling associations between intervention outcomes and risk factors and among risk factors. Subsequently, we applied the proposed method to a single-arm hospital-based palliative care team (HPCT) intervention study.
Methods
Patients and interventions
Eligible hospitalized patients with malignant tumours were enrolled between 1 November 2009 and 30 March 2010. Inclusion criteria were as follows: (1) age 18 years and over, (2) pathological diagnosis with a malignant tumour, (3) ability to respond to the questionnaire and (4) written consent. Exclusion criteria were as follows: (1) inability to respond to the questions because of disturbed consciousness or cognitive disorder and (2) inappropriateness for the study as judged by the physician in charge.
Decisions for interventions were reached by the HPCT using the screening sheets and criteria described by Morita et al. 12 and Akizuki et al., 13 with minor modifications. Patients requiring intervention were asked to confirm their intention to receive HPCT physical, psychological and social interventions in accordance with pain levels and individual needs. Before and 1 week after interventions, QOL was assessed using the European Organization for Research and Treatment of Cancer QLQ-C15-PAL Questionnaire. 14 Because 9 of 35 patients who satisfied the eligibility criteria for HPCT intervention were discharged from the hospital within 1 week of intervention, the present analysis included only 26 patients for whom QOL assessments were completed at 1 week after intervention. Background variables of the patients are presented in Table 1, and written informed consent was obtained from all participants. The study was approved by the Ethics Committee of Kobe University Hospital and was performed in accordance with the Declaration of Helsinki and the Ethical Guidelines for Clinical Research in Japan.
Demographic and clinical characteristics of subjects.

Statistical models for one-group pre–post design
In pre–post RCTs, endpoints are measured before and after the intervention, and although two-sample t-tests are often employed, analysis of covariance (ANCOVA) models using pre-data as covariates is considered the optimal statistical design.15–18
Because no data from a control arm are available, this study design is referred to as ‘one-group pre–post testing’, and the following statistical model is analogous to ANCOVA for RCTs, although the group effect was omitted as follows

where Di denotes the difference in efficacy between pre- and post-intervention measurements in each patient, α indicates the effects of the intervention, β refers to the influence of the pre-intervention value
Intervention effect models with covariates
The objective of the one-group pre–post design is often limited to hypothesis generation with exploratory data analysis rather than hypothesis testing with pre-specified statistical models. Moreover, the significance of covariates in multiple regression models is strongly influenced by the magnitude of associations between them. Thus, because multiple covariates are often strongly associated with each other, clinical interpretations of covariate effects are often difficult during comparisons of results from several regression models. To resolve this problem, hypothesized models that allow simple clinical interpretations can be created using SEM, which enables simultaneous modelling of associations among covariates and their influences on efficacy indicators. To illustrate the proposed data analysis approach, the path diagram shown in Figure 1(a) is modelled using pre–post outcome change scores as endpoints and age, sex, performance status (PS) and pre-intervention outcome values as covariates. The depicted associations among measurements are only one of many possibly clinically interpretable model structures, and structural equations corresponding to the Figure 1(a) are defined in equation (2)

where Dif (the pre–post difference) in Figure 1(a) corresponds to
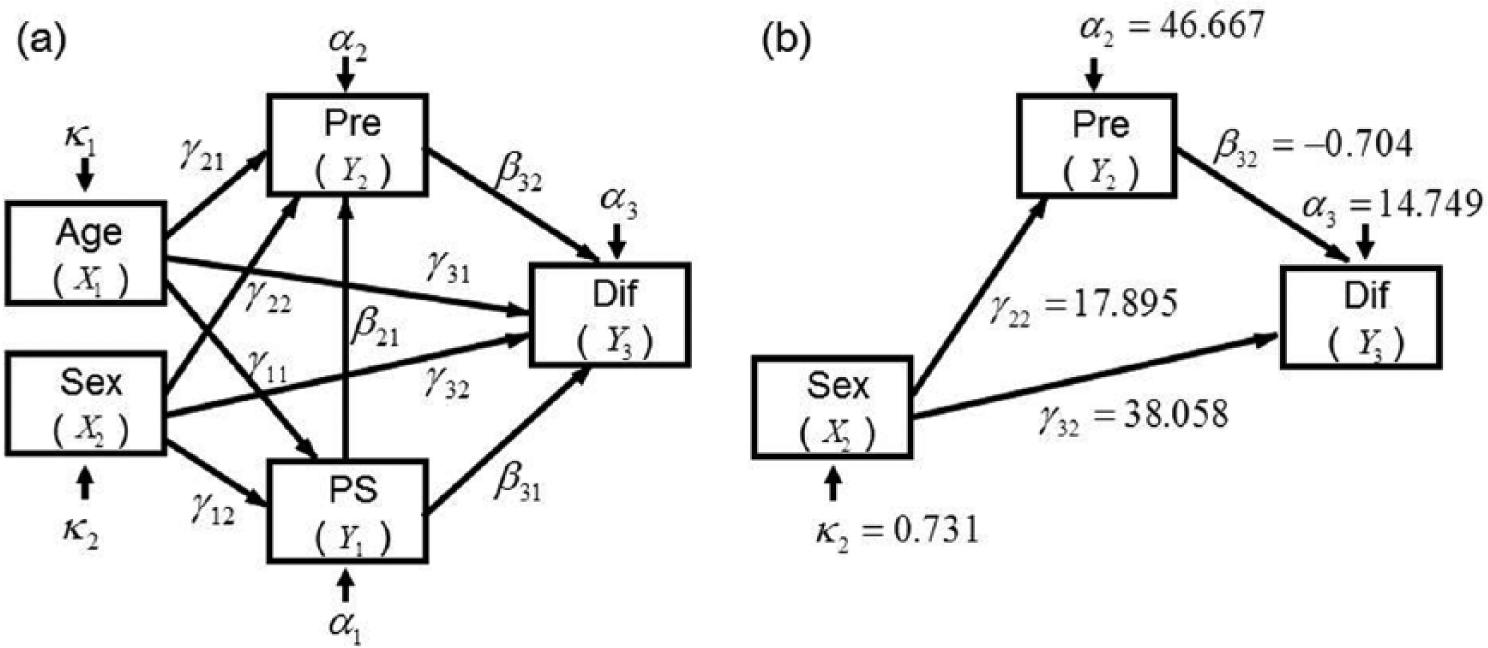
Path diagrams of (a) the full model and (b) the reduced model.
Square box indicates observed measurements, and Greek letter indicates model parameter. Single-headed arrow depicts direction of association between two measurements.
Statistics
Statistical analyses were performed using STATA version 12.0 (Stata Corporation, College Station, TX). All p-values were generated from two-sided tests and were considered significant when p < 0.05. All CIs had a two-sided probability coverage of 95%.
Results
SEMs
The results of analyses using the present method were generated using physical function data. The full model shown in the path diagram (Figure 1(a)) was applied, and estimates, standard errors and CI were calculated for each parameter (Table 2). The total effect comprised the sum of intervention and covariate effects and was consistent with the mean pre–post difference
Intervention effect models with covariates (full model).

SE: standard error; CI: confidence interval; PS: performance status.
Age and PS were affected less by covariates and were excluded from the full model to generate the reduced model. To obtain a parsimonious construct and retain potentially significant covariates in the reduced model, p-values of covariates in the full model were set to 0.15. Accordingly, the path diagram (Figure 1(b)) shows that the reduced model encompasses the effect of gender mediated by a pre-value and the direct effect of the pre-value and gender on Dif. Table 3 shows the intervention effect on physical function and the estimated values of covariates. As in the full model,
Intervention effect models with covariates (reduced model).

SE: standard error; CI: confidence interval.
Impact of small sample size on SEM
Only small samples were available to fit full and intervention effect models. However, all models converged after few iterative calculations. Because the normal likelihood method was used to estimate standard errors of parameters, the stability of these estimates was examined by evaluating the variance of the estimator using bootstrapping. Estimates of asymptotic standard errors and bootstrap standard errors, and of bias-corrected CI, are shown in Table 4. Results of bootstrap analyses indicated that SEM with observed measurements was applicable to relatively small sample sizes.
Estimates of standard error using the bootstrap method.

SE: standard error; CI: confidence interval.
Discussion
Studies of palliative care often adopt single-arm study designs to accommodate patient conditions that preclude randomization. However, single-arm studies offer limited estimates of causal relationships between interventions and effects. Hence, study designs that meet the requirement of evidence-based palliative care have recently been proposed.20,21 In this study, we propose a method for effective use of data from single-arm studies by modelling relationships among covariates.
In one-group pre–post studies, intervention effects are usually evaluated using paired-t tests.22,23 In contrast, the present analytical method uses ANCOVA models that exclude group effects and enables clinical interpretation using SEM to evaluate the effects of palliative care interventions on QOL. Critically, this model accommodates associations among covariates and permits extraction of factors that directly or indirectly influence efficacy indicators, thus enabling consideration of randomization designs in which these factors serve as allocation factors. However, future studies are required to assess the advantages and shortcomings of this approach in comparison with conventional ad hoc allocation factor selection methods.
During modelling of associations among covariates using SEM, trial calculations of bias-free sample sizes are possible as in analyses using ANCOVA models, which are used to estimate intervention effects (not affected by covariates) and their dispersions. However, the present method enables estimation of intervention effects by adjusting the influences of covariates on efficacy indicators. Moreover, HPCT interventions and covariate effects were estimated even during modelling of simultaneous influences of multiple covariates. Thus, the proposed method may serve as a novel analytical tool for one-group pre–post studies that are performed under study-limiting conditions. Data analyses using SEM usually require large sample sizes. 24 Thus, application of SEM to small samples may lead to unstable estimates of model parameters. Thus, unusually large parameter estimates require cautious interpretation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethics Approval
Ethical approval for this study was obtained from Ethics Committee of Kobe University Hospital.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Written informed consent was obtained from all subjects before the study.
Trial Registration
UMIN Clinical Trials Registry: UMIN000002138.
