Abstract
Background:
With implant dentistry growing in popularity as a means of replacing missing teeth, there is an increasing need for guidance to support regular practice and to ensure that implantology is reinforced with an appropriate evidence base for developing safe prescribing habits. This is particularly important as the prescribing of antibiotics in implant dentistry is becoming increasingly controversial owing to the fear of any failure due to post-operative infection; the possible litigation consequences that might emerge can be a major reason for justifying such prescription protocols.
Methodology:
A questionnaire-based pilot study was carried out, which assessed prescribing behaviours by UK-based dentists placing dental implants. The 11-item questionnaire sought information on implant procedures categorised as straightforward and complex implant procedures. The survey was distributed electronically through local implant study clubs and via dental social media forums.
Results:
A total of 54 dentists responded, with almost 89% of respondents prescribing antibiotics pre-operatively and 85% prescribing antibiotics post-operatively, with amoxicillin always the primary choice. The recommended course of treatment varied between one and seven or more days. Almost half of the respondents prescribed analgesics pre- and/or post-operatively with ibuprofen and paracetamol as the first and second choices, respectively. Dexamethasone was prescribed pre- and post-operatively by 7% of the dentists.
Conclusion:
This study highlighted a lack of national antibiotics prescription guidelines and a lack of consistency in implant dentistry practice in the UK. It also indicated the need for multi-centre studies with large sample sizes that can support the foundation for appropriate guidelines on antibiotic prescribing in implant dentistry.
Learning Objectives
To identify antibiotic and analgesic prescribing patterns among GDPs placing dental implants in the UK, and explore the factors influencing prescribing decisions
To highlight the impact of inconsistencies in prescribing habits
To compare clinical guidelines on antibiotic use in implant dentistry with real-world practice and encourage evidence-based prescribing by GDPs to improve patient care and support antimicrobial stewardship
Introduction
In recent years, the threat of antibiotic resistance, which centres largely around the development of bacterial resistance following exposure to a prescribed antibiotics dose, has grown to become a substantial problem facing the world’s population, with senior members of the medical community openly voicing their concerns.1-3
Dental prescribing in the UK contributes to nearly 10% of antibiotics dispensed.4,5 Criticism has been raised about whether dentists appropriately prescribe dependant on the clinical situation, 6 with one study suggesting that up to 80% of prescriptions are unnecessary. 7 This is despite the availability of rigorous national guidelines and the statutory interventions expected of clinicians.8,9 Some have suggested that the adopted mindset and behaviours in dentists is partially responsible for the overprescribing of antibiotics.10,11 Nevertheless, it is crucial to try to understand where some of these behaviours may be developed, and to begin to influence the collective mindset of practicing clinicians to curtail overprescribing where possible.
The provision of dental implants, largely considered an elective procedure, has often seen the prescription of antibiotics in a bid to reduce potential failures because of bacterial contamination, but the question as to their true effectiveness has been posed.12,13 Furthermore, irrespective of historic high-level evidence, a broad range of behaviours are still exhibited in prescribing habits with respect to drug choice and regimen.14-16 Two recent cross-sectional studies examining behaviours in Spain showed regular prescribing of antibiotics for implant procedures outside of published guidance, with limited consensus.17,18 When considering the concept of antibiotics misuse and dental implants, it appears that this is indeed a global problem and one that has been growing for some time.19-21 This pilot study aims to explore the prescribing habits of medications in implant dentistry in the UK, focusing on antibiotic prescription and any particular protocols.
Methodology
The medications and questions outlined in the questionnaire were initially based on a pilot survey to 15 implant dentists – members of the British Society for Advanced Dentistry (BSAD) – and the feedback gained from this. Further input was fielded from a senior academic colleague to help finalise the definitive questions. Utilising different questions (Figure 1), the survey asked participants to categorise their training in implant dentistry in terms of the nature of training (i.e. formal university programmes, NHS specialty training, short courses or company led product training), and their years of experience post-graduation. They were also asked to classify their clinical treatments into straightforward and complex implant procedures, and their prescribing habits and regimen for both analgesics and antibiotics pre- and postoperatively. The survey was circulated electronically on a social media implant and restorative forum (British Association of Restorative Dentistry [BARD]) and passed, via the BSAD email lists, to a regional implant study club (Association of Dental Implantology [ADI] Hampshire). Thus, overall reach was estimated to be to approximately 600 practising dentists. The survey was circulated for two months with an initial reminder circulated two weeks later and again every two weeks, following-up until eight weeks.

Results
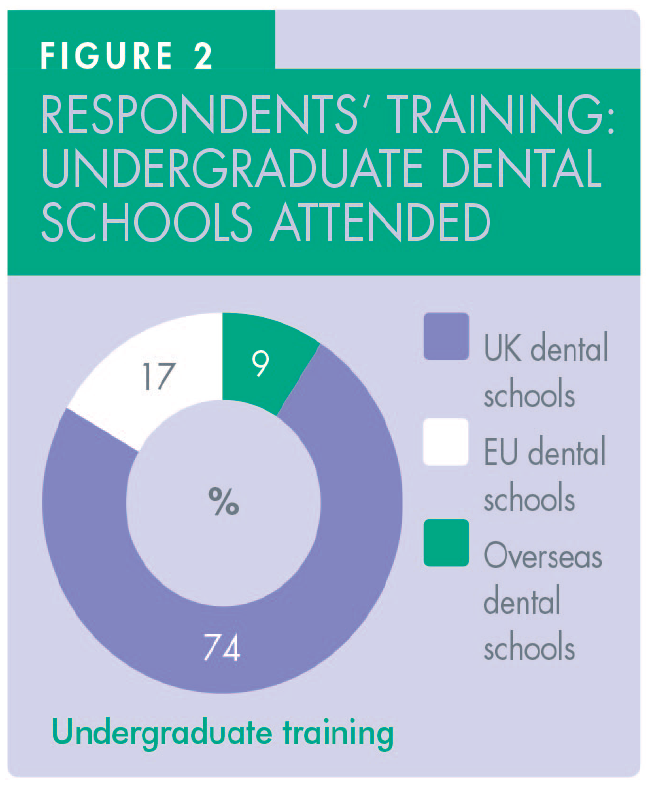
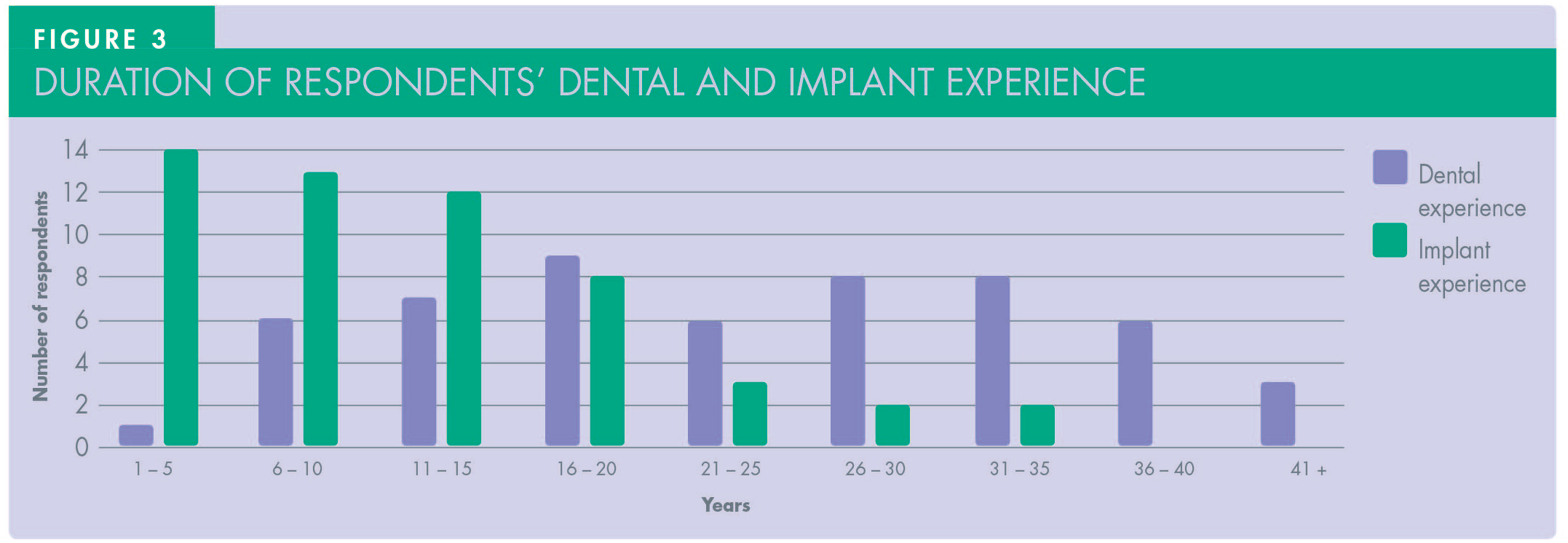
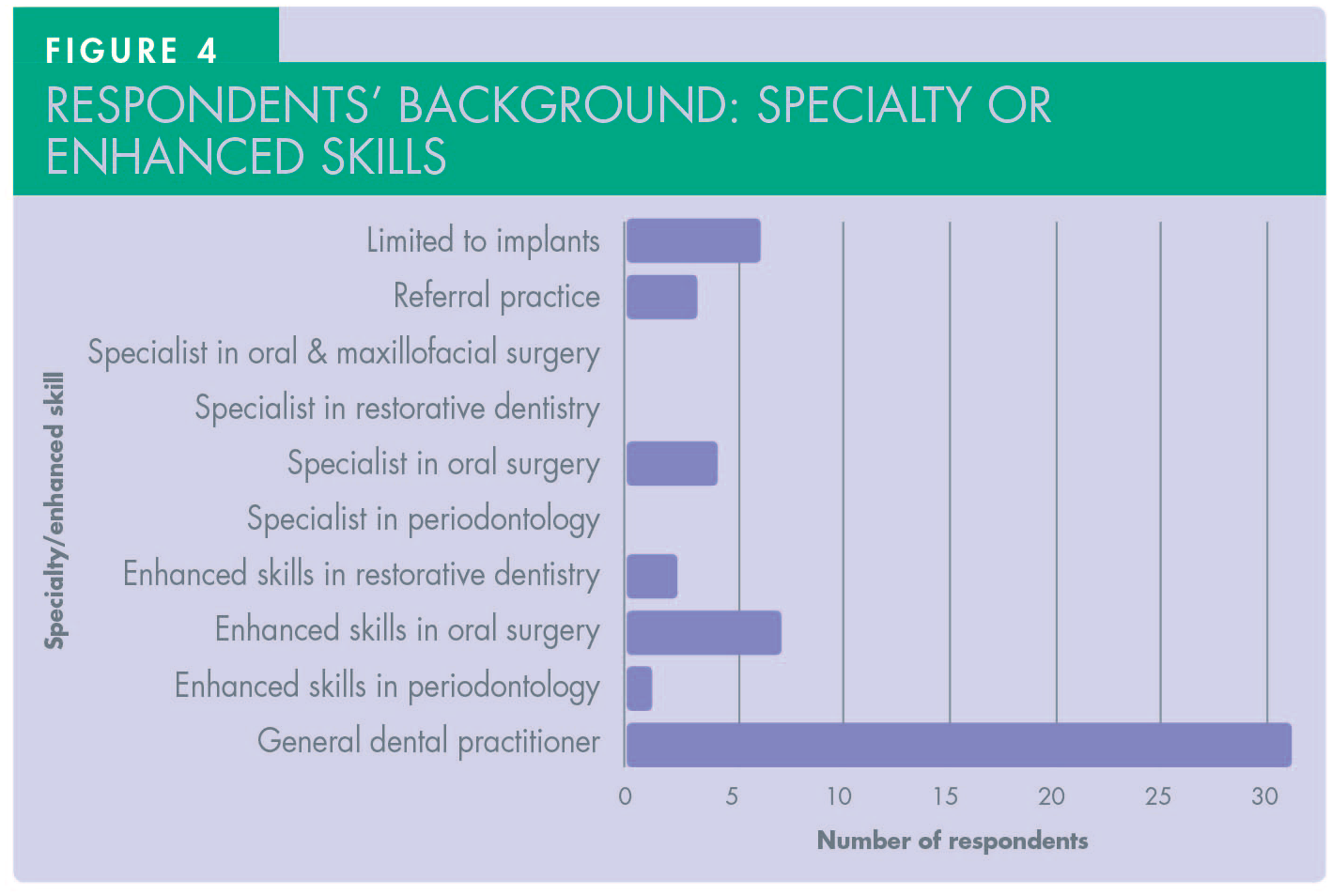
A total of 54 dentists responded to the questionnaire with 74% of them having graduated from UK dental schools (Figure 2). Duration of postgraduate experience varied between one and 35 years (mean = 17.5 years) (Figure 3). The majority constituted general dental practitioners (57.5%) with both specialists (7.5%) and clinicians with enhanced skills in periodontology, oral surgery and restorative (35%) accounting for the rest (Figure 4).
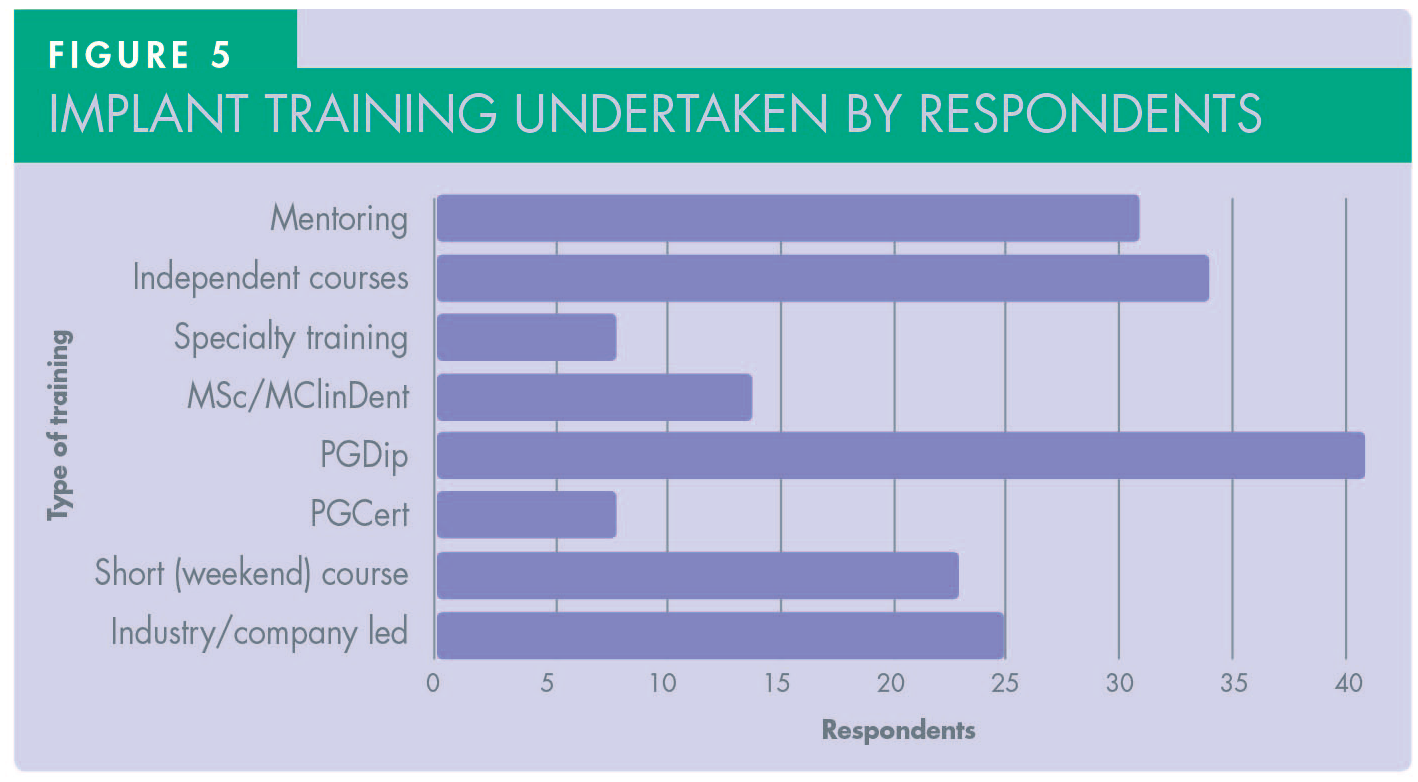
Most respondents had formal postgraduate training, including postgraduate certificates, diplomas and/or Master’s degrees. However, many also attended independent and/or industry-led courses. More than half of the respondents (57%) were clinically mentored by other experienced colleagues during their training. (See Figure 5.)
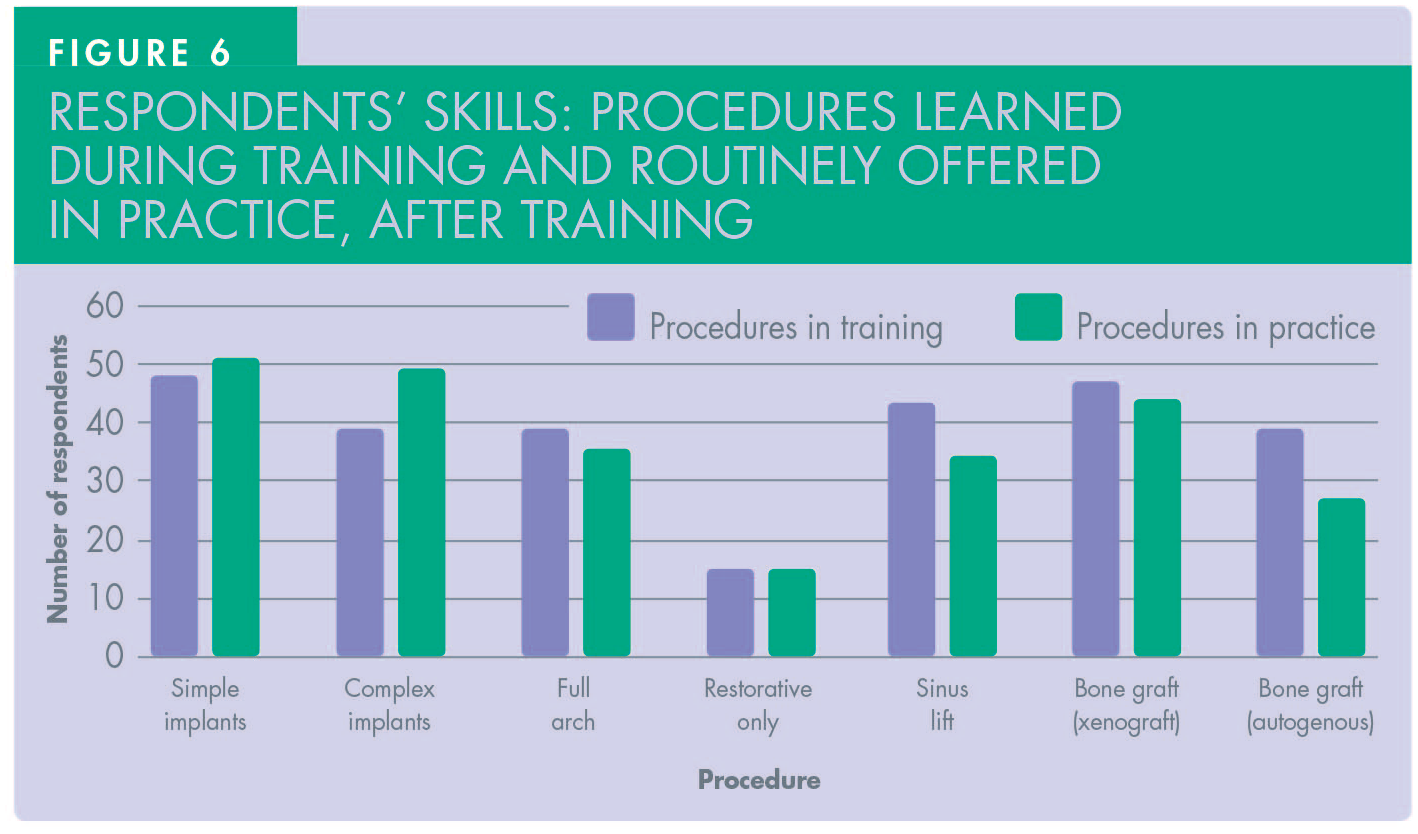
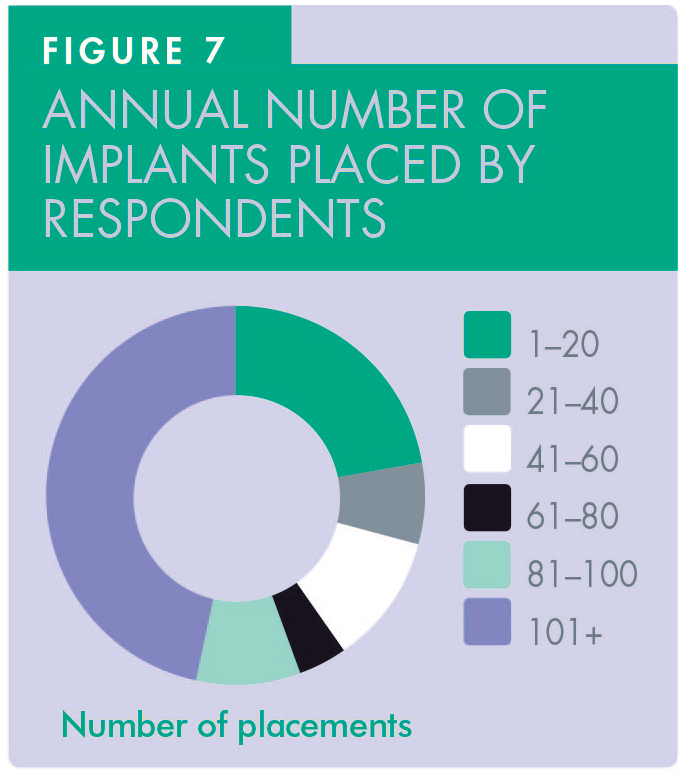
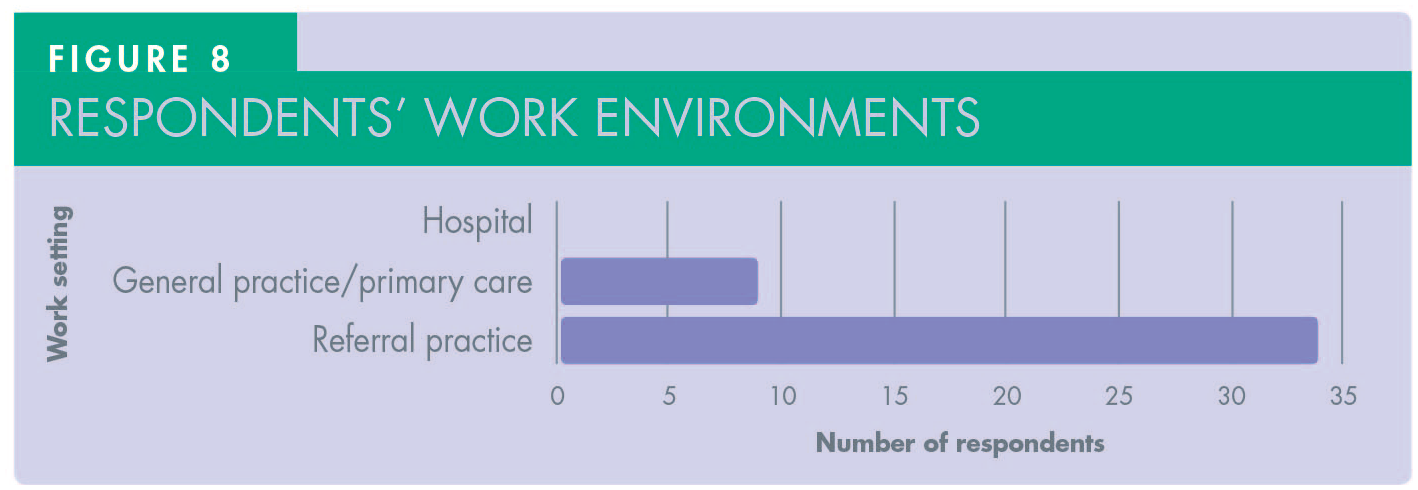
In terms of training, the portfolio of cases that respondents were exposed to was extensive, covering simple and complex cases, the latter including full arch, sinus lift and bone grafting. This was reflected in the treatments offered to cover all aspects of implant dentistry including the restorative stage (Figure 6). More than 46% of participants have placed and restored 25+ implants (Figure 7). The majority of respondents (90%) were mainly based in primary care, with some (16.6%) also working in referral centres (Figure 8).
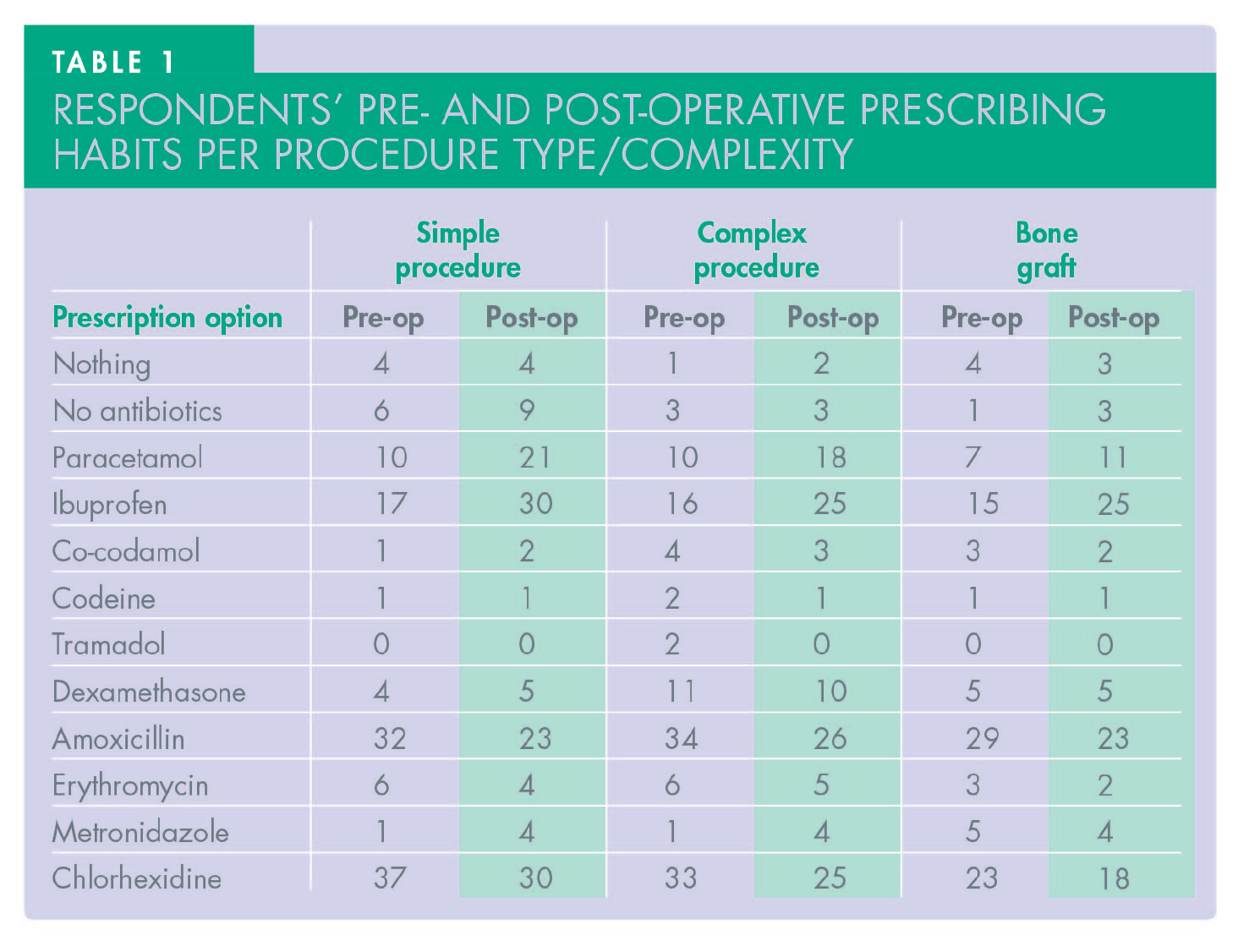
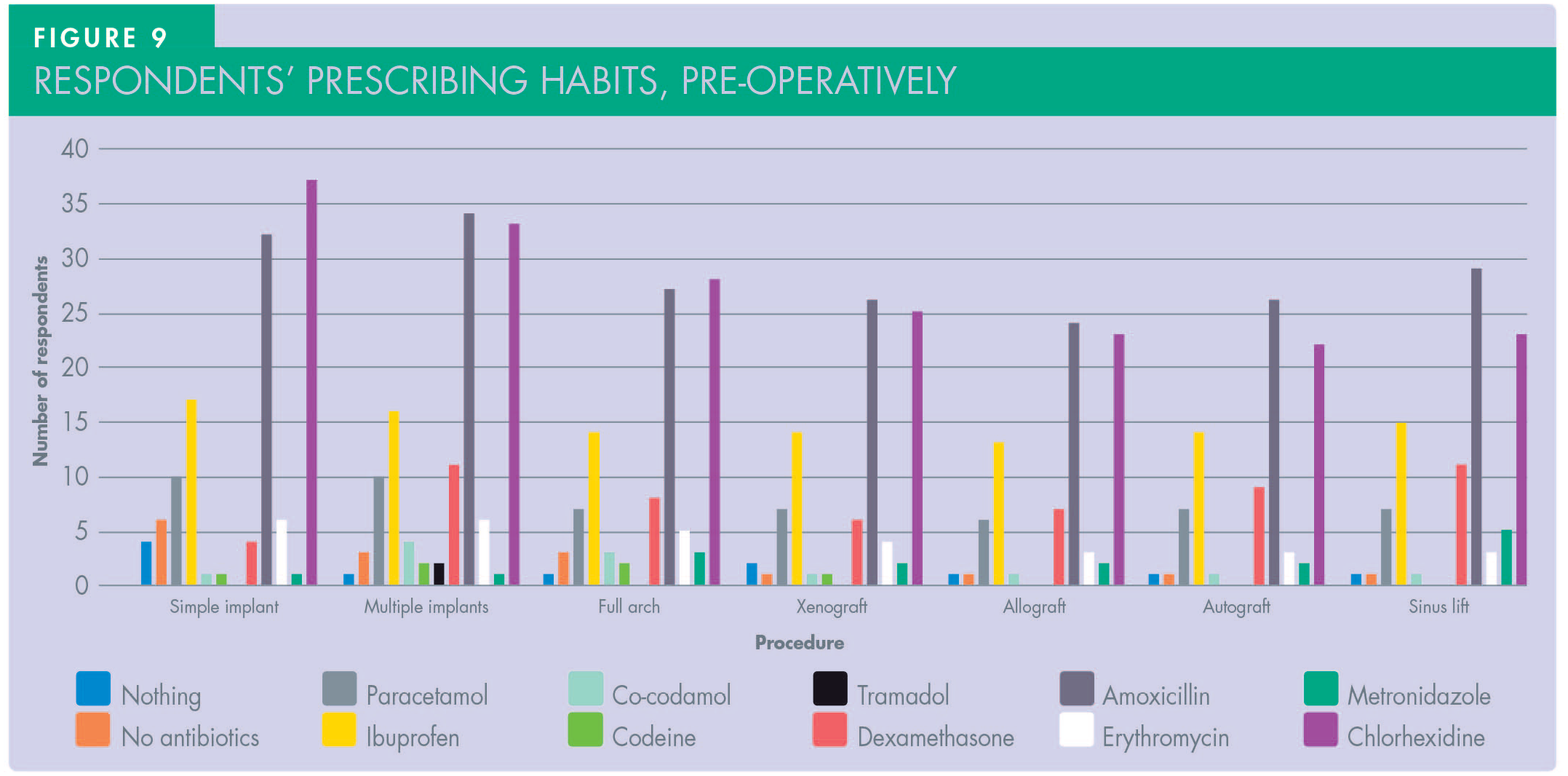
The questionnaire categorised prescribing habits into two groups: pre-operative and post-operative. Table 1 and Figures 9 and 10 show the pre- and post-operative prescribing habits for a range of procedures, based on survey responses. The pre-operative responses were extremely inconsistent: for simple implant placement, 11.1% of respondents said they do not prescribe any antibiotics; 5.5% do not prescribe for complex placement (full arch and sinus lift); and only one respondent did not cover with antibiotics for bone grafting. Almost 70% of respondents prescribed a course of antibiotics for all placements, with amoxicillin being the primary choice (almost 89%) followed by erythromycin and metronidazole. In parallel, analgesics were also prescribed by 53.7% of respondents, with ibuprofen and paracetamol as the first and second choices respectively; only two dentists preferred co-codamol or codeine. Tramadol was preferable in complex cases by only two dentists. The majority (68%) also favoured a 30-second chlorhexidine pre-operative rinse as part of their protocol. Dexamethasone was prescribed by 7% of the dentists post-operatively.
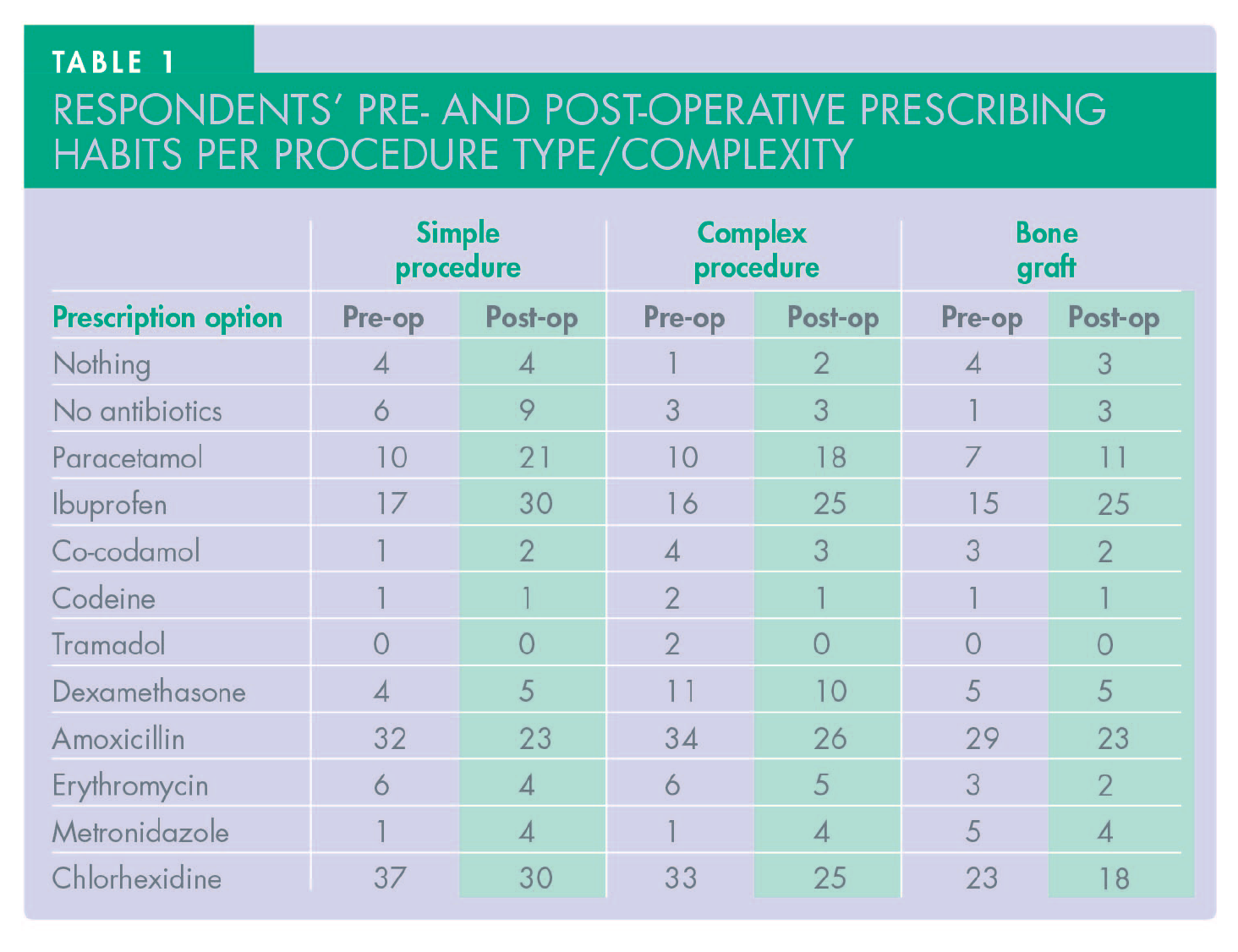
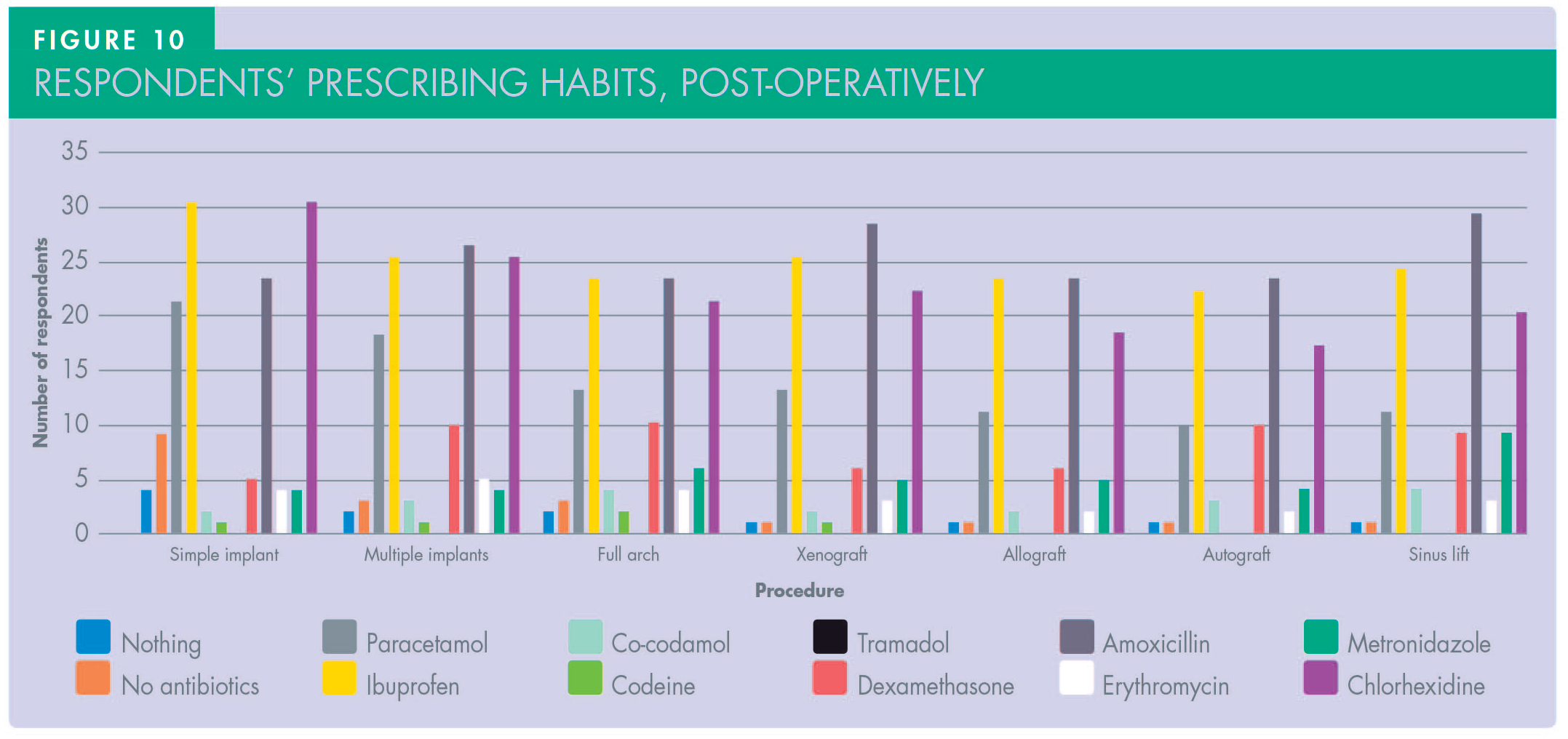
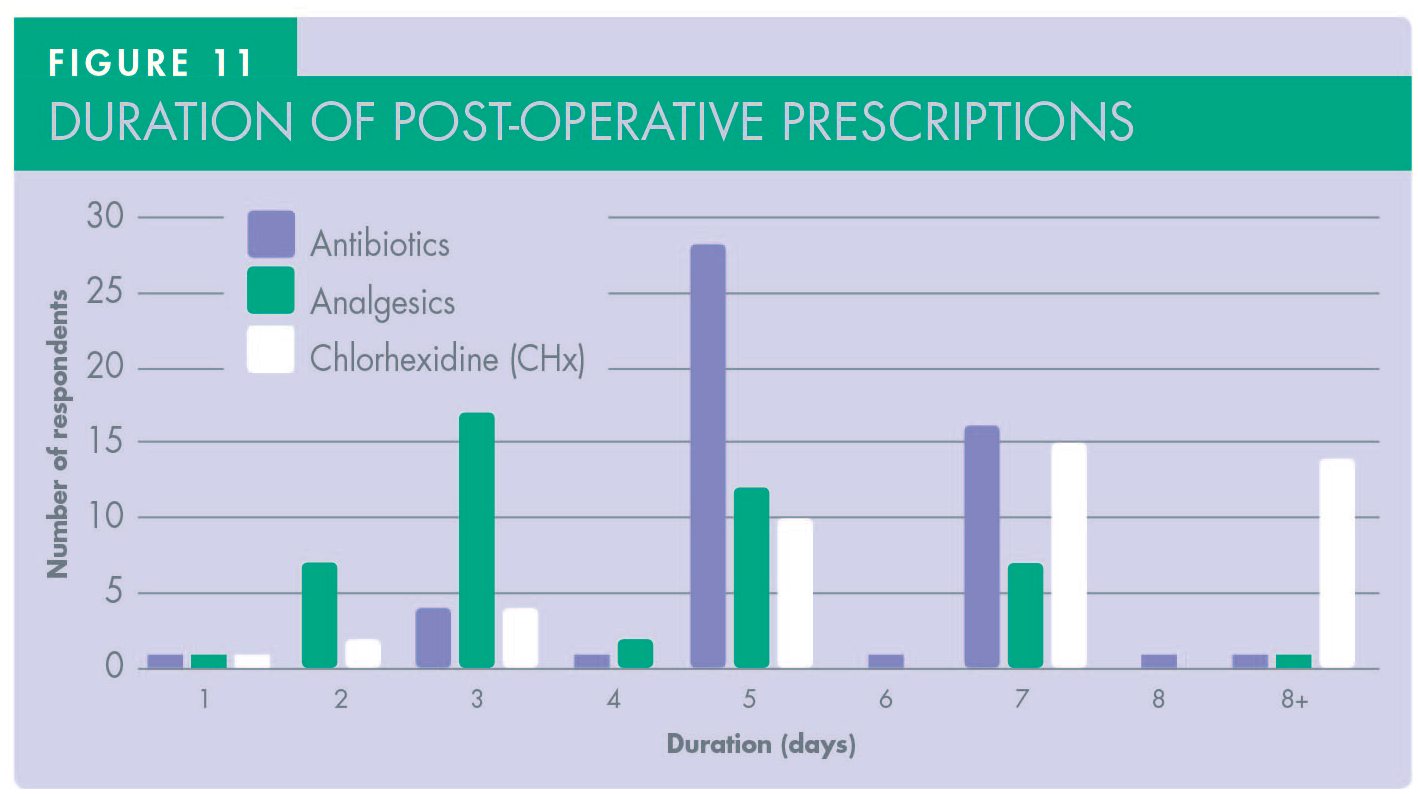
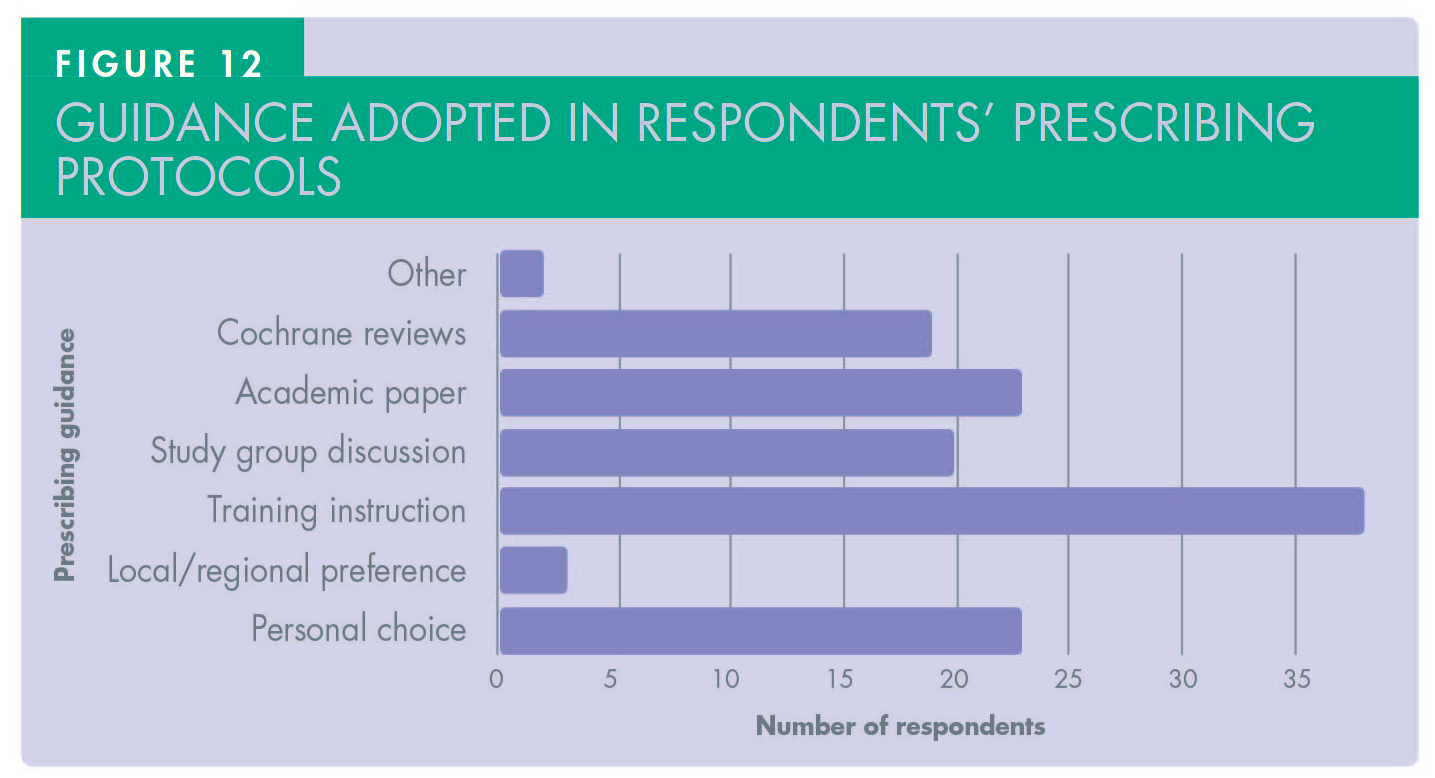
Regarding post-operative results (Figure 10 and Table 1), the number of dentists who did not prescribe antibiotics was slightly higher at 16.6% in simple cases but the same (5.5%) in complex cases. Analgesic prescriptions were slightly lower, but ibuprofen and paracetamol were still the first two choices respectively. None of the respondents prescribed tramadol regardless of the complexity of implant surgery performed. Dexamethasone was prescribed by 9% of respondents. Assessing the duration of post-operative medications, there was extreme difference in both antibiotics and analgesic prescriptions among participants (Figure 11). In terms of antibiotics, a five-day course was the choice for 51.8% of respondents, a seven-day course was the choice for 29.6% of respondents, and three-day course was the choice for 7.4%, with the rest varying between one to seven or more days. Regarding analgesics, prescription for three days was reported by 31.4%, five days by 22.2%, and two days was the choice of 12.9%, with the rest also varying between one to eight or more days. The majority of respondents’ protocols were adopted from their training (70.3%), with personal choice (42%), academic papers (42%), and Cochrane reviews (35%) also influencing decision making (Figure 12).
Discussion
This pilot study aimed to evaluate the prescribing of medications (particularly antibiotics) commonly used in implant dentistry in the UK. Despite aiming to reach 600 dentists, only 54 dentists responded to the questionnaire. This response rate is unfortunately low and may reflect the way in which the survey was circulated. 22 Unlike the paper by El-Kholey et al. 21 which used multiple-choice questions based on scenarios, the questions posed in this study were similar in design to studies performed in Spain and India.18,19 However, it must be noted that when considering all these papers, there was no consistency in methodology and a lack of homogeneity makes the comparison difficult. Despite most respondents having attended formal training in implant dentistry, their prescription habits varied in terms of protocols, medications and duration of prescriptions. All respondents, however, showed broad experience in terms of both training and treatment offered in their surgeries, including both primary care and referral centres settings.
Although the regimes of antibiotic prescription were inconsistent, the main aim is to prevent delayed wound healing and avoid early failure of implants. 23 This is often linked to specific bacterial strains. The most frequently involved bacteria include streptococci, anaerobic Gram-positive cocci, and anaerobic Gram-negative rods. 24 A study by Heydenrijk and colleagues 25 on the bacteria found in peri-implantitis revealed significant colonisation of inflamed implants by anaerobic Gram-positive bacteria, such as fusobacteria spirochetes, Bacteroides forsythus, as well as black-pigmented bacteria like Prevotella intermedia, Prevotella nigrescens, and Porphyromonas gingivalis. Additionally, Actinobacillus actinomycetemcomitans was identified in the collected samples, suggesting that the microbial community in inflammatory lesions surrounding implants closely resembles that seen in adult periodontitis, including refractory periodontitis. 25
The use of corticosteroids in dental implant surgery has not been widely reported in the literature, except in the long term usage of these medications for the management of systemic conditions and the effects of corticosteroids on implants. 26 However, they are typically used in an attempt to reduce post-operative swelling and improve patient comfort. 27 A recent review on general surgical patients, has suggested that it is still unknown whether the typical regimen of a single peri-operative dose affects healing, but it is thought not to impact the risk of infection. 28
Post-operatively, 85% of respondents prescribed antibiotics, with amoxicillin revealed as the primary choice. Duration of prescriptions were found to be very inconsistent. This disparity is in keeping with other studies conducted, such as that by Khalil (2015) which looked at prescribing patterns in Sweden and found similar variations. 29 A survey by Camps-Font et al. found that 40.5% of their respondents provided antibiotics post-operatively only, and 38.1% both pre- and post-operatively; a wide range of dosages were provided and are different to the UK. 18 Furthermore, the most common medication provided was a mixture of amoxicillin and clavulanic acid. In 2014, Datta et al. showed that Indian prescribers often favoured amoxicillin, with a large subset prescribing co-amoxiclav while quinolones formed the next largest group. 19 These results were found consistent when compared to a cross-sectional study of dentists in Jordan. 30 Datta et al. 19 found the average length of prescription post-operatively was five days, with a broad range extending from one- to ten-days in total, whereas Camps-Font 18 found this to be seven days, and El-Kholey et al. 21 noted three- to five-day prescriptions. The study by El-Kholey et al. highlighted a difference in prescribing habits between those with and without postgraduate training, the former issuing fewer prescriptions pre-operatively but more post-operatively for antibiotics. 21 With regard to the trend in this study, the authors were unable to differentiate between short courses and formal postgraduate education as, although there were those who just confirmed having attended short courses (33.3%), there were many in postgraduate training who attended both (66.6%) making it harder to isolate the data in this manner based on the software used in data collection. Other studies, such as El-Kholey, 21 also examined postgraduate training, and while they did not correlate the type of training exactly to prescribing habits, they did note that 73.4% of respondents based their decision making on lectures, scientific reading or courses.
Camps-Font et al. noted that maxillofacial surgeons (although a small number of respondents) tended to operate a very different prescription criteria to other dentists, using broad spectrum antibiotics in both pre- and post-operative periods. 18 They found that these results were similar to an American study of oral and maxillofacial surgeons, which observed no consensus in prescription habits in this group and showed similar results subsequently. 16 These variations in professionals’ behaviours can be due to multiple factors. Flottorp et al. identified seven determinants in healthcare professionals’ behaviours that typically influence antibiotic prescribing practices: 31
Guideline factors (e.g. guideline characteristics or presence of contradictory guidelines);
Individual prescriber preferences (which could be influenced by many factors, such as lack of agreement with specific guidelines, lack of motivation or inertia of previous practices etc.);
Patient factors (e.g. patient’s expectations, inability to reconcile patient’s preferences with guideline recommendations);
Professional interactions (e.g. leadership, key individuals, team processes);
Incentives and resources (e.g. economic incentives, technical knowledge, organisational size);
Capacity for organisational change (e.g. planning, engaging, executing and evaluating); and
Social, political and legal medical norms (e.g. legislation or regulations, priority on societal agenda, corruption, political stability, etc.)
All these determinants have been explored widely in the literature and found to influence inpatient and outpatient settings32-34 however there are still factors that are likely to influence clinicians in primary care.
This pilot study highlights only a very small snapshot of habits within what is largely limited to private practice. Longitudinal studies that correlate with primary care and secondary care behaviours would be useful, from a governmental perspective, in assessing true trends and supporting recommendations and targeted interventions with practitioners. 35
When considering the results of all the studies in general, they highlight behaviours and patterns beyond those indicated for typical scenario management as noted in national guidelines such as those by the College of General Dentistry. 36 It is noteworthy to mention that some studies recommended a course of antibiotics, such as the meta-analysis of Rodríguez Sánchez et al. (2018) and the Cochrane review by Esposito et al. (2013).37,38 The meta-analysis suggested that a single dose (1g, 2g or 3g) of oral amoxicillin pre-operatively prior to implant surgery was found to significantly prevent patients from developing dental implant failures. Other reviews reported similar effects, such as the Cochrane review which specified that 2g or 3g of oral amoxicillin, as a single administration one hour pre-operatively, significantly reduces failure of dental implants. Both reports suggested that post-operative use of oral amoxicillin did not significantly achieve a prophylactic effect, and it is still unknown whether post-operative antibiotics are beneficial; equally, recommendations towards studies exploring varying dosages and regimes is required for further clarity on clinical standards.
Exploring the literature further, a 2014 study by Chrcanovic et al. stated in their systematic review that there is evidence suggesting that a prophylactic antibiotic regimen significantly reduces failures of dental implants placed within ordinary conditions, though, there were no apparent significant effects of prophylactic antibiotics on the occurrence of post-operative infections in healthy patients receiving implants. 39 Lund et al. also specified that the use of antibiotic prophylaxis in conjunction with implant placement reduced the risk of implant loss by 2%. 40
Conversely, however, there is emerging evidence supporting an alternative view. Park et al. stated that antibiotic use in healthy patients for the prophylaxis of surgical infection associated with dental implant placement does not appear to improve clinical outcomes; this includes both pre- and post-operative doses. 20 This evidence is supported elsewhere in studies, initially by Tan et al. and subsequently by Payer et al. who found that there was no statistically significant difference in impact of patient related outcomes and post-operative complications during simple implant placement or implant placement with simultaneous bone grafting respectively.13,41
Regarding analgesics, 90% of dentists prescribed them, with ibuprofen being the most favoured. Paracetamol, co-codamol and codeine were also mentioned. Tramadol however was not prescribed at all, and 7.5% prescribed dexamethasone. Prescription length varied from one to eight days. Reassuringly, analgesic usage was largely conservative and in keeping with documented and recognised management which considers the use of non-steroidal anti-inflammatory drugs (NSAIDs) and acetaminophen (paracetamol) as primary interventions to manage pain following minor surgery, and adjunctive use of opioids as necessary;42,43 this is comparative to prescribing habits elsewhere in the world. 44
The protocols described in this pilot study were adopted from previous training as stated by 70% of the respondents; however, there is lack of evidence to prove which courses are providing their trainees with evidence-based antibiotic prescription protocols. It is worth mentioning that prophylactic antibiotics in implant dentistry are becoming more controversial as the fear of any failure due to post-operative infection and the possible litigation consequences that might emerge can be a major reason for justifying such prescription protocols. In order to reduce antibiotic prescriptions, varying measures may have to be considered. A study by Kim et al. showed that, in conjunction with circulation of guidelines, there was a behaviour change when prescriptions were recorded automatically within medical records which might be easier to scrutinise. 45 Furthermore, the review by Thompson et al. showed that there are multiple factors associated with decision making in prescribing antibiotics which has made stewardship and information sharing rather challenging. 46
This pilot study has several limitations that should be acknowledged, particularly in considering developments for further studies. The small sample size limits the generalisability of the findings to the broader population of implant dentists in the UK. Additionally, the distribution of the survey through specific channels, such as implant study clubs, may have introduced selection bias, capturing responses predominantly from dentists actively engaged in professional networks. The reliance on self-reported data introduces potential reporting bias, as participants’ responses may have been influenced by social desirability or their interpretation of best practices. The study predominantly included respondents based in primary care, limiting insights into prescribing habits across other practice settings, such as secondary care or academic institutions, but remains a realistic interpretation of where the majority of implants are placed in the UK.
Furthermore, the pilot questionnaire format, with closed-ended questions, may not have fully captured the complexity of prescribing behaviours or nuanced decision-making processes. Attempts to correlate training types with prescribing habits were hindered by overlapping responses, making it difficult to isolate the specific impacts of formal versus informal training. The two-month survey period might have further limited broader participation and engagement, and the cross-sectional design only provides a snapshot of prescribing behaviours at a single point in time, without capturing temporal trends or changes influenced by emerging evidence. Lastly, the study did not sufficiently explore patient-specific factors, such as comorbidities or preferences, that could impact prescribing decisions. These limitations highlight the need for larger, multi-centre, longitudinal studies with more comprehensive data collection methods to address these gaps and provide a more robust evidence base.
Conclusion
The growing adoption of implant dentistry as a solution for missing teeth highlights the importance of evidence-based guidance to promote safe and effective prescribing practices in clinical settings. Despite the small sample size, this pilot study highlighted a lack of consistent antibiotic prescription regimens in implant dentistry practice in the UK. It also emphasised the inconsistency within the UK and this appears in line with findings elsewhere. Furthermore, it algins with the growing demand to consider multi-centre studies with large sample sizes that can support the foundation for appropriate guidelines in an effort to mitigate antimicrobial resistance.
Footnotes
Acknowledgements
The authors wish to extend their thanks to the local ADI section, BARD and BSAD for supporting them in the development and distribution of the questionnaire. We also wish to thank Professor Nikos Donos for his input on question design in the early phases of the study design.