Abstract
Background:
Community-acquired pneumonia continues to be a leading cause of morbidity and mortality worldwide. Several validated scoring systems are available for stratifying pneumonia severity, with the CURB-65 being well known. Recently, neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR) have been reported as reliable indicators of disease severity and adverse outcomes in patients with pneumonia, particularly when used alongside established scoring systems.
Objectives:
To evaluate the association of NLR and PLR with higher clinical severity classification (CURB-65 ⩾ 3), and to compare their discriminatory performance with C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) in patients with community-acquired pneumonia.
Design:
Cross-sectional study.
Methods:
In this cross-sectional study conducted at a tertiary care hospital in Peshawar, Pakistan, we enrolled 121 adults with community-acquired pneumonia. Baseline demographic characteristics, comorbid conditions, clinical presentation, and laboratory measures were recorded. Pneumonia severity was assessed using the CURB-65 score. The discriminatory performance of NLR, PLR, CRP, and ESR was evaluated by receiver operating characteristic (ROC) curve analysis, with severe pneumonia defined as a CURB-65 score of 3 or higher. Multivariable logistic regression was performed to assess independent associations after adjustment for age and comorbidities.
Results:
The mean (±SD) age of the patients was 59.2 ± 20.6 years, and 69.4% were male. Common comorbidities included diabetes (31.4%), chronic obstructive pulmonary disease (28.1%), and chronic heart failure (19.8%). The mean CURB-65 score was 2.23 ± 1.20. NLR and PLR were significantly higher in severe pneumonia than in non-severe pneumonia (both
Conclusion:
In this cohort, NLR and PLR demonstrated strong agreement with higher CURB-65 severity classification and superior discriminatory performance compared with CRP and ESR.
Plain language summary
Community-acquired pneumonia (CAP) is a common cause of hospital admission and can quickly become life-threatening. This study evaluated whether two simple measures taken from a routine complete blood count, the neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR), can help identify severe CAP better than traditional inflammation tests (C-reactive protein [CRP] and erythrocyte sedimentation rate [ESR]). We conducted a hospital-based cross-sectional study at Khyber Teaching Hospital, Peshawar (September 2024 to March 2025) including 121 adults with CAP confirmed clinically and by chest X-ray. Severity was classified using the CURB-65 score; severe CAP was CURB-65 ⩾3. NLR and PLR were markedly higher in severe cases than in non-severe cases (NLR 14.5 vs 5.4; PLR 555 vs 230; both p < 0.001), while CRP and ESR did not differ significantly between groups. On ROC analysis, PLR showed excellent ability to distinguish severe CAP (AUC 0.994; cutoff 388.5; sensitivity 95.9%; specificity 98.6%), and NLR also performed strongly (AUC 0.944; cutoff 10.5; sensitivity 79.6%; specificity 97.2%), whereas CRP (AUC 0.603) and ESR (AUC 0.550) performed poorly. Because NLR and PLR require no additional testing beyond a standard CBC, they may provide a rapid, low-cost support tool for triage and early risk stratification, especially in resource-limited settings, alongside clinical assessment and the CURB-65s. As this was a single-center observational study using severity classification rather than hard outcomes, larger multicenter studies are needed to confirm these cutoffs and assess prediction of ICU admission and mortality. These findings suggest that CBC-derived ratios can complement, not replace, clinical judgment, imaging, and severity scores. Vaccination uptake in the cohort was low, reinforcing the importance of influenza and pneumococcal vaccination to reduce CAP burden.
Keywords
Introduction
Community-acquired pneumonia (CAP) remains a major global health concern, leading to significant morbidity and mortality, particularly among the elderly. Accurate assessment of disease severity is essential for guiding treatment and predicting outcomes. Commonly used scoring systems such as the Pneumonia Severity Index (PSI) and CURB-65 stratify patients based on clinical parameters. The CURB-65 score evaluates confusion, urea, respiratory rate, blood pressure, and age ⩾65 years to predict mortality risk. 1 It is widely used due to its simplicity and reliance on readily available bedside variables. However, additional inflammatory biomarkers may further enhance risk stratification and provide complementary information on systemic inflammation.2,3
Hematologic biomarkers have gained attention as adjunctive tools for assessing pneumonia severity. The neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR) are increasingly recognized as useful indicators of systemic inflammation. 4 Both have been reported as potential markers of disease severity in pneumonia.5,6 Derived from routine complete blood count parameters, they are easily accessible and cost-effective. 7 Higher NLR and PLR values have been associated with increased in-hospital mortality and longer hospital stay, 8 and NLR may outperform traditional leukocyte indices in predicting severity. 9
Comparative studies suggest that NLR and PLR may serve as independent markers of severity and improve risk stratification when combined with established tools such as CURB-65 and PSI. 10 For example, combining NLR with CURB-65 has been shown to improve the prediction of in-hospital mortality in elderly CAP patients. 11 Other studies have demonstrated correlations between these ratios and clinical severity scores, 12 while meta-analyses support the role of NLR as a reliable inflammatory marker in CAP. 13 These findings highlight the potential of integrating hematologic biomarkers into clinical assessment frameworks.
Given the high burden of CAP in low- and middle-income countries such as Pakistan, where access to advanced diagnostics may be limited, there is a need for simple and accessible tools for early severity assessment. Biomarkers such as NLR and PLR could provide a rapid and cost-effective approach to identify high-risk patients and guide management. Therefore, this study evaluated the ability of NLR and PLR, compared with CRP and erythrocyte sedimentation rate (ESR), to discriminate severe CAP (CURB-65 ⩾ 3) from non-severe CAP in our setting.
Methods
Study design and setting
This was a hospital-based cross-sectional observational study, conducted at Khyber Teaching Hospital Peshawar, Pakistan, between September 2024 and March 2025, during the peak pneumonia season. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement for cross-sectional studies. The completed STROBE checklist is provided as Supplemental Table 1.
Objective
The primary objective of the study was to evaluate the discriminatory performance of NLR and PLR, and to compare them with ESR and serum CRP, for identifying severe community-acquired pneumonia, using CURB-65 severity classification (severe defined as CURB-65 ⩾ 3) as the clinical benchmark.
Ethical consideration
Ethical approval for the study was obtained from the Institutional Review Boards (IRB) of the Khyber Medical College (Reference No. 105/DME/KMC) prior to the initiation of data collection. Written informed consent was obtained from all participants, ensuring that they were fully aware of the study’s purpose, procedures and potential implications of the study. Patient confidentiality was strictly maintained, and all collected information was used solely for research purposes.
Selection of participants
Adult patients (aged ⩾18 years) presenting to the emergency department (ED) and/or admitted to inpatient medical wards with suspected CAP were enrolled.
Sampling technique
Patients were selected using a non-probability consecutive sampling technique, ensuring the inclusion of all eligible patients admitted with CAP during the study period. An a priori sample size calculation was performed using the WHO sample size calculator based on expected area under the curve (AUC) values for NLR in predicting CAP severity reported in previous literature, assuming a 95% confidence level and 5% margin of error.
1
The minimum required sample size was estimated to be 121 participants. A total of 121 patients met the eligibility criteria and were included in the final analysis. Additionally, the number of severe cases (
A total of 168 patients were screened during the study period. Of these, 47 were excluded based on predefined criteria, including incomplete medical records (

Participant flow diagram.
Inclusion and exclusion criteria
Suspected cases of CAP were identified based on the presence of lower respiratory tract symptoms, including a new cough, sputum production, shortness of breath, fever (either hypothermia or hyperthermia), and abnormal breath sounds on physical examination, accompanied by a newly detected infiltrate on chest radiography. Initial chest X-rays were assessed by the ED physician and later reviewed by a senior radiologist, who was blinded to clinical and laboratory findings to ensure an objective evaluation.
Patients were excluded if they were under 18 years of age or had active tuberculosis or other chronic lung conditions that could confound diagnosis or inflammatory indices (e.g., bronchiectasis, interstitial lung disease, or lung malignancy). Additional exclusion criteria included known hematologic disorders affecting blood indices, active malignancy, current chemotherapy, chronic systemic corticosteroid therapy prior to admission, other immunosuppressive treatments, pregnancy, and incomplete medical records, to minimize confounding and ensure accurate interpretation of inflammatory markers.
Data collection
Data were collected using a structured questionnaire developed for this study to record demographic, clinical, and laboratory variables (Supplemental Table 2). The form captured patient characteristics such as age, gender, occupation, residence (urban or rural), comorbid conditions, CURB-65 components, and relevant laboratory parameters. The data collection form was reviewed by senior faculty prior to implementation to ensure clarity and completeness. Laboratory investigations, including complete blood count, CRP, and ESR, were obtained at the time of initial presentation to the emergency department prior to initiation of antibiotic therapy or other hospital-based treatments. NLR and PLR were calculated from these baseline complete blood count parameters, and these initial values were used for all analyses. Additionally, influenza and pneumococcal vaccination status were documented for each patient. The CURB-65 score was calculated based on five components: confusion status, serum urea level, respiratory rate, blood pressure, and age ⩾65 years.
Vaccination status was recorded for descriptive purposes and was not included in analytical models due to low uptake in the study population.
Statistical analysis
All analyses were conducted using R software (version 4.3.3, R Foundation for Statistical Computing, Vienna, Austria). Continuous variables were summarized as means with their corresponding standard deviations, while categorical variables were described using counts and percentages. Comparisons between groups were made using either the
Receiver operating characteristic (ROC) curve analysis was performed to evaluate how well CRP, ESR, NLR, and PLR could discriminate pneumonia severity according to the CURB-65 score. For each biomarker, the AUC was calculated, and the most effective cutoff value for predicting severity was identified using the Youden Index. A
Multivariable logistic regression models were constructed to evaluate the independent association of NLR and PLR with pneumonia severity, adjusting for age, chronic obstructive pulmonary disease (COPD), congestive heart failure, and diabetes mellitus. Due to collinearity between NLR and PLR, the two biomarkers were analyzed in separate models. Additionally, quasi-complete separation was observed in models including PLR, resulting in unstable adjusted estimates; therefore, multivariable results for PLR were not interpreted.
Results
A total of 121 patients with community-acquired pneumonia were included. The mean age of the cohort was 59.2 ± 20.6 years, and 84 patients (69.4%) were male. Baseline characteristics are shown in Table 1.
Baseline characteristics of participants.

COPD, chronic obstructive pulmonary disease; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.
Diabetes mellitus (38/121, 31.4%), chronic obstructive pulmonary disease (34/121, 28.1%), and chronic heart failure (24/121, 19.8%) were the most common comorbid conditions (Table 1). Sepsis on admission occurred in 29 patients (24.0%). Supplemental oxygen was administered to 72 patients (59.5%). The mean CURB-65 score was 2.23 ± 1.20. The in-hospital mortality was 9/121 (7.4%), and the mean duration of hospitalization was 6.2 ± 3.7 days.
Patients with severe pneumonia had significantly higher NLR and PLR compared with non-severe cases (both
Comparison of age and inflammatory markers between non-severe and severe community-acquired pneumonia, with statistical tests and

CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.

Boxplots of CRP, ESR, NLR, and PLR in severe versus non-severe pneumonia.
ROC analysis showed PLR had the best discriminative performance (AUC 0.9935, 95% CI 0.9847–1.0000; cutoff 388.5; sensitivity 0.9592; specificity 0.9861), followed by NLR (AUC 0.9436, 95% CI 0.8977–0.9895; cutoff 10.5; sensitivity 0.7959; specificity 0.9722). CRP (AUC 0.6028, cutoff 65.5) and ESR (AUC 0.5501, cutoff 78.5) showed poor discrimination (Table 3 and Figure 3).
Diagnostic performance of inflammatory markers for predicting severe community-acquired pneumonia using ROC analysis (AUC, 95% CI, optimal cutoff, sensitivity, specificity).
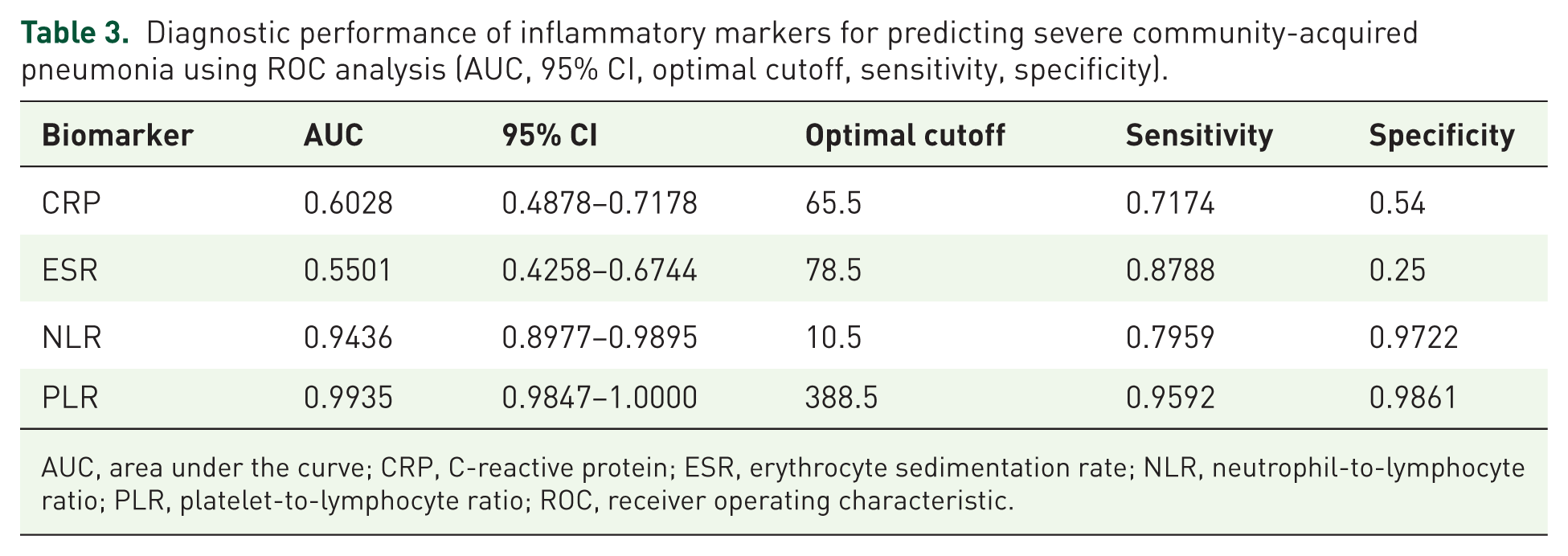
AUC, area under the curve; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; ROC, receiver operating characteristic.

ROC curves of CRP, ESR, NLR, and PLR for predicting severe pneumonia.
Bootstrap internal validation demonstrated negligible optimism for both biomarkers (NLR: −0.00037; PLR: −0.00013). The optimism-corrected AUCs were 0.9440 for NLR and 0.9936 for PLR, confirming the stability of the observed discriminatory performance (Supplemental Table 3).
After adjustment for age and comorbidities, NLR remained independently associated with severe pneumonia (adjusted OR 1.91, 95% CI 1.46–2.51;
Overall, NLR and PLR demonstrated strong discriminatory performance and outperformed traditional inflammatory markers in distinguishing severe from non-severe community-acquired pneumonia.
Discussion
Accurate early assessment of CAP severity is essential for guiding clinical decision-making, hospitalization, and outcome prediction. While CURB-65 is widely used, additional biomarkers may complement clinical assessment, particularly simple and rapidly available measures of systemic inflammation.14,15 Hematologic indices such as the NLR and PLR, derived from routine blood counts, have gained attention as inexpensive markers and may help identify severe CAP (CURB-65 ⩾ 3).16–19 In our study, both NLR and PLR were significantly higher in severe CAP and demonstrated better discrimination than traditional inflammatory markers, with PLR showing the highest AUC, followed by NLR, while CRP and ESR showed poor performance. These ratios are not intended to replace clinical scoring systems but to complement them at presentation.
Patients with severe pneumonia had significantly higher NLR and PLR values, whereas CRP and ESR did not differ significantly. These findings are consistent with studies showing that higher NLR is associated with increased severity and worse outcomes, including ICU need and mortality.20,21 NLR has also been reported to outperform conventional leukocyte indices and complement clinical severity scores such as CURB-65.17,18 Our findings support this evidence, demonstrating strong discriminatory ability even in a cohort with prevalent comorbidities.
PLR, though less extensively studied, also showed strong discriminatory capacity, consistent with prior reports linking it to inflammation and poorer outcomes in respiratory infections.22,23 Elevated NLR and PLR in severe pneumonia may reflect neutrophilia, lymphopenia, cytokine-mediated platelet activation, and heightened inflammatory response.24–27 The near-perfect AUC observed for PLR should be interpreted cautiously. Although bootstrap validation showed minimal optimism, such high performance is uncommon in clinical practice. The clear separation of PLR values between groups likely contributed to this finding, while the modest sample size and single-center design may limit generalizability. External validation is required.
The limited performance of CRP and ESR aligns with prior studies showing that these markers lack specificity for severity.28,29 Although CRP correlates with infection, it does not reliably predict clinical deterioration, particularly early in disease.30,31 CRP and ESR reflect downstream inflammatory processes and may respond more slowly than cellular immune changes. In contrast, NLR and PLR capture early shifts in leukocyte and platelet dynamics, providing a more immediate reflection of disease severity. The lack of significant differences in CRP and ESR may also reflect single time-point measurement, variability in symptom duration, and the high prevalence of comorbidities, which may elevate baseline inflammatory states and reduce group differences. Additionally, the sample size may have been insufficient to detect smaller differences.
Comorbidities such as COPD, CHF, and diabetes are known to worsen pneumonia outcomes and may influence inflammatory markers. Although sepsis was more frequent in severe cases, NLR has been validated as a prognostic marker in both pneumonia-related and primary sepsis,6,32 supporting its biological plausibility.
Our findings are consistent with prior studies reporting AUC values of approximately 0.75–0.88 for NLR in predicting severe pneumonia or mortality.17–21 The higher discriminatory performance observed in our cohort may reflect differences in population characteristics, study design, and the use of CURB-65 as the severity benchmark. Similarly, PLR has been associated with worse outcomes, though it remains less studied. These findings require validation in larger, multicenter cohorts.
Vaccination rates in this cohort were extremely low, reflecting broader immunization gaps and highlighting an important area for prevention. Influenza vaccination has been shown to reduce severe respiratory infections and ICU admissions,20,21 and improving uptake may reduce the burden of CAP.
Study limitations
This study has several limitations. First, its observational design introduces the possibility of residual confounding, as biomarkers may be influenced by comorbid conditions, concurrent infections, or medications. Additionally, the single-center setting may limit generalizability to broader populations or different healthcare systems. Larger multicenter studies are needed to confirm these findings and improve their clinical applicability.33,34
Severity in this study was defined using the CURB-65 clinical scoring system rather than hard clinical outcomes such as mortality or need for intensive care. Therefore, our findings reflect agreement with an established clinical severity classification rather than direct prognostic ability. Prospective studies incorporating clinical endpoints are required to establish independent prognostic value. Although CURB-65 is widely validated and practical, it is not a definitive gold standard and may not fully capture disease complexity or clinical trajectory.
The relatively small number of outcome events limited robust comparisons of whether NLR or PLR outperform CURB-65 in predicting outcomes such as mortality or ICU admission. Future prospective studies with larger cohorts are needed to evaluate the incremental prognostic value of these biomarkers beyond established clinical scoring systems.
An important consideration is the significant age difference between severity groups, which is expected given that age ⩾65 years is a component of the CURB-65 score. This introduces structural confounding, as older patients are more likely to be classified as severe. Age-related immune changes, including relative lymphopenia, may also influence NLR and PLR independently of disease severity. Although we adjusted for age in multivariable analysis, residual confounding cannot be excluded, and part of the observed association may reflect age-related variation rather than severity alone.
Conclusion
In conclusion, while established clinical scoring systems such as CURB-65 remain central to severity assessment in community-acquired pneumonia, NLR and PLR demonstrated strong agreement with higher CURB-65 severity classification and superior discriminatory performance compared with CRP and ESR in our cohort. These readily available hematologic ratios may serve as practical adjuncts to clinical severity assessment, particularly in resource-limited settings, but should not be viewed as replacements for validated scoring systems. Prospective studies incorporating clinical outcomes are warranted to further clarify their independent prognostic role.
Supplemental Material
sj-docx-1-tai-10.1177_20499361261441703 – Supplemental material for Assessing pneumonia severity using neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios: a cross-sectional comparison with CURB-65 score
Supplemental material, sj-docx-1-tai-10.1177_20499361261441703 for Assessing pneumonia severity using neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios: a cross-sectional comparison with CURB-65 score by Muhammad Osama, Ahmad Iftikhar, Mallahat Abdul Baseer, Nazim Khan, Abdul Majid, Akshay Amaraneni, Faiz Anwer and Pawan Kumar Thada in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
