Abstract
We report the case of a 38-year-old woman with human immunodeficiency virus (HIV) infection diagnosed in 2010 and on antiretroviral treatment since 2014, with an undetectable viral load and a CD4 lymphocyte count of 527 cells/µL. The patient presented with weight loss, fever, hyporexia, nausea, vomiting, progressive dyspnea, purpuric abdominal lesions, and lower extremity edema. During hospitalization, she developed hemoptysis and severe respiratory failure and died a few hours later. Parasitological studies of sputum and stool showed abundant Strongyloides stercoralis larvae, confirming disseminated strongyloidiasis. This case highlights that severe S. stercoralis infection can occur even in patients with clinically controlled HIV, underscoring the need for screening in endemic regions.
Plain language summary
We report the case of a 38-year-old woman living with HIV who had a well-controlled immune system and undetectable viral load. Despite her good immune status, she developed a severe, widespread Strongyloides stercoralis infection, leading to rapid respiratory failure and death. The infection was confirmed by stool and sputum tests showing abundant larvae. Her initial symptoms, including fever, abdominal rash, nausea, and shortness of breath, were not recognized as life-threatening. This case highlights that even patients with HIV who appear immunologically healthy can develop life-threatening parasitic infections. Early screening and timely treatment are crucial, especially in endemic areas, to prevent fatal outcomes.
Keywords
Introduction
Strongyloidiasis is an intestinal helminthiasis caused by Strongyloides stercoralis, a nematode capable of perpetuating infection through endogenous autoinfection. In immunocompromised individuals, the infection can progress to a disseminated form or hyperinfection syndrome, characterized by massive larval migration to various organs and associated with high mortality. In people living with HIV, the clinical presentation is often atypical, and the relationship between parasite load and the degree of immunosuppression is not always proportional.1,2 In endemic areas, early diagnosis and prophylactic antiparasitic treatment are essential strategies for preventing serious complications, even in patients with adequate immunological and virological control.3,4
Case presentation
A 38-year-old female patient, originally from Chanchamayo, Junín, was diagnosed with HIV infection in 2010. She began antiretroviral therapy in 2014, with 95% adherence to the tenofovir/lamivudine/dolutegravir regimen. At her last follow-up (August 2025), her CD4 count was 527 cells/µL, her CD4/CD8 ratio was 1.37, and her viral load was undetectable. Serological tests for syphilis, hepatitis B, and hepatitis C were negative. The patient had no history of systemic corticosteroid use, immunosuppressive therapy, chemotherapy, organ transplantation, alcohol abuse, substance use, malnutrition, or chronic liver disease, according to clinical records and family report. No comorbidities or recent travel were reported. She had no relevant family history or known hereditary conditions. The patient lived in a rural area of the central jungle, with frequent exposure to damp soil and agricultural areas. No significant psychosocial factors were identified.
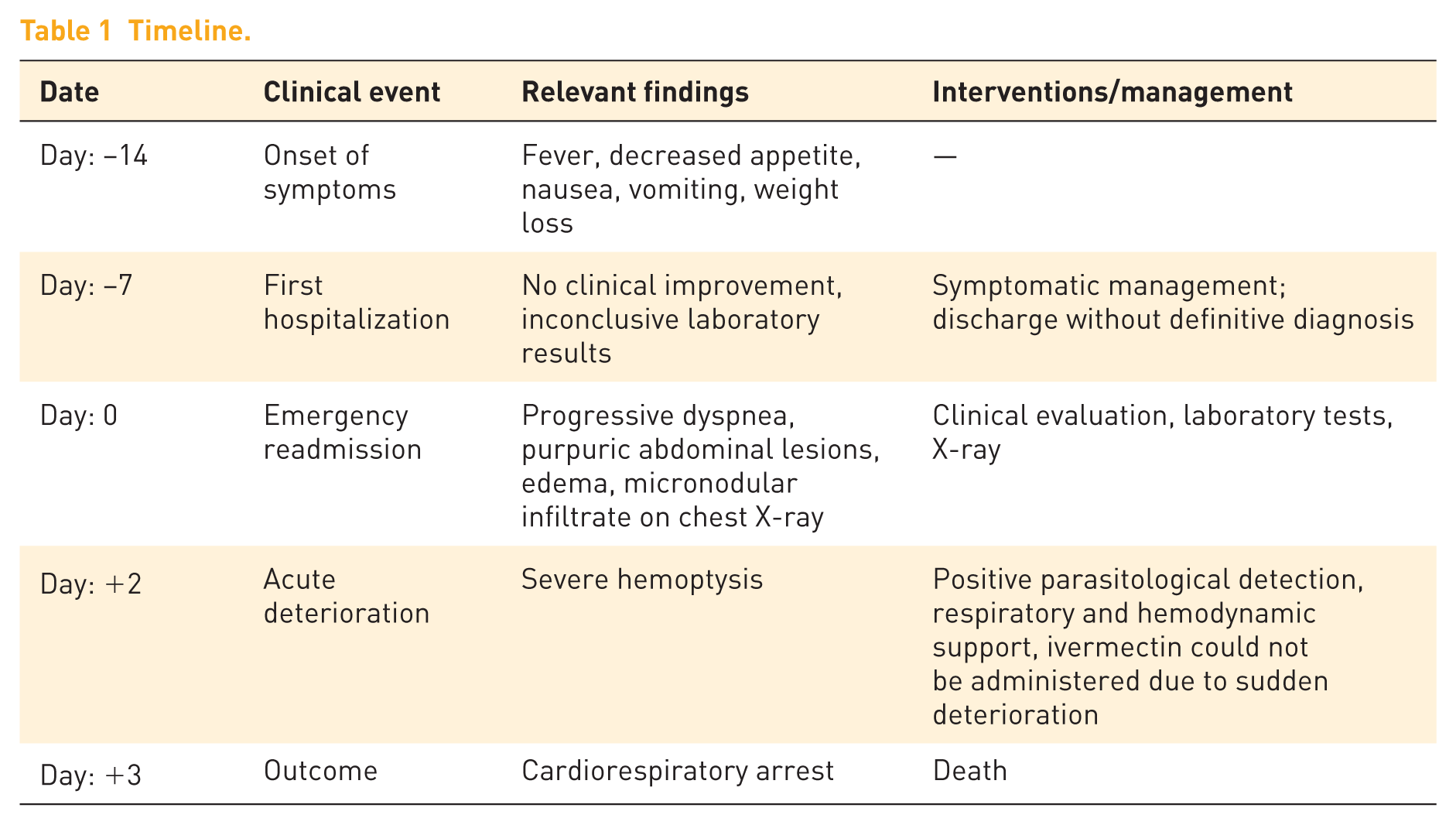
Two weeks prior to admission, she developed fever, decreased appetite, weight loss, nausea, and vomiting. She was hospitalized without improvement and subsequently discharged. Days later, she was admitted to the emergency department with progressive dyspnea that worsened to the point of requiring oxygen support, diffuse abdominal pain without peritoneal signs, purpuric lesions on the abdomen and thighs (Figure 1(a)), and edema in the lower extremities. Laboratory tests showed: leukocytes 7100/µL (segmented neutrophils 87%, eosinophils 1%), hemoglobin 7.7 g/dL, hematocrit 23.3%, and platelets 262,000/µL. The chest X-ray showed bilateral micronodular infiltrate (Figure 1(b)). Forty-eight hours later, sputum and stool smears were obtained and were negative for tuberculosis; however, abundant S. stercoralis larvae were found (Figure 1(c)). Hours after this finding, the patient presented with massive hemoptysis and severe respiratory deterioration, which is why respiratory and hemodynamic support were prioritized, and therefore, antiparasitic treatment with ivermectin could not be initiated. However, the patient died a few hours later without receiving specific treatment for the parasite. The final diagnosis was disseminated strongyloidiasis in a patient with HIV infection and an undetectable viral load.

(a) Purpuric abdominal lesions (“Thumbprint purpura”). (b) Chest X-ray showing bilateral micronodular infiltrates. (c) Sputum smear showing S. stercoralis larvae.
Due to the patient’s death, the perspective of her family members is included. They commented that the progressive symptoms were not initially recognized as serious. Informed consent was obtained from the family members for the publication of the case and clinical images. The clinical course of the patient from symptom onset to outcome is summarized in Table 1.
Timeline.
Discussion
The case presented demonstrates a disseminated form of S. stercoralis in a patient with clinically controlled HIV infection, with CD4 counts above 500 cells/µL and undetectable viral load. This finding is uncommon, as the literature indicates that severe strongyloidiasis is usually associated with profound immunosuppression or immunosuppressive therapy1,3,5; In contrast, this patient had preserved CD4 counts and sustained virological suppression.
The occurrence of disseminated strongyloidiasis in individuals with preserved CD4 counts and sustained virological suppression remains incompletely understood.3,5,6 Although antiretroviral therapy restores quantitative CD4 levels, functional immune dysregulation may persist, particularly in helminth-specific Th2 responses that are critical for parasite control.6,9 In endemic settings, continuous exposure to S. stercoralis may lead to chronic autoinfection and progressive accumulation of parasite burden, potentially overwhelming host defenses despite apparently preserved immunity.3,10 These observations suggest that CD4 cell counts alone may not accurately reflect the functional integrity of the immune response required for effective control of helminth infections.6,9
The delayed diagnosis in this case was primarily related to the absence of eosinophilia and the patient’s preserved immune status, with an undetectable HIV viral load and normal CD4 counts, which initially reduced clinical suspicion of parasitic infection. Given the strong epidemiological association between tuberculosis and HIV infection in our setting (regardless of immune status), sputum smear microscopy for Mycobacterium tuberculosis was systematically prioritized (a process requiring serial sample collection over three consecutive days and processing times of up to 24 h due to our laboratory limitations). During the analysis of these tuberculosis smears, S. stercoralis larvae were incidentally identified in sputum samples. This highlights how the reliance on eosinophilia and the burden of other comorbidities can delay the recognition of disseminated strongyloidiasis.
Although HIV infection alters the cellular immune response, disseminated strongyloidiasis is uncommon in these patients. Viney et al. 6 postulated that the relative preservation of Th2 immunity in HIV would explain the lower frequency of hyperinfection, in contrast to patients treated with corticosteroids or transplanted. However, several reports describe cases of dissemination in patients with controlled HIV, suggesting that there are additional factors, such as viral coinfections, genetic variability of the host, or continuous reinfections in endemic areas.7–9
Testing for human T-lymphotropic virus type 1 (HTLV-1) was not performed in this patient, which represents an important limitation of this report. In our setting, HTLV-1 infection is considered uncommon; therefore, routine screening is not included in HIV care programs, and diagnostic tests are not widely available in most healthcare facilities. Despite these limitations, HTLV-1 coinfection has been shown to shift the immune response toward a Th1-dominant profile, suppressing protective Th2 cytokines and eosinophilia, and has been strongly associated with severe and disseminated strongyloidiasis.6,9 Given its clinical relevance and the potential for misleading CD4 counts, routine HTLV-1 screening should be considered in patients with severe Strongyloidiasis and in people living with HIV from endemic or high-risk regions.6,9
The patient came from a jungle region of Peru, where the prevalence of S. stercoralis in people living with HIV can exceed 20%, according to Otero-Rodríguez et al. 10 In such environments, constant exposure to the parasite and chronic autoinfection can maintain a subclinical larval load that reactivates with minimal immunological changes.
The purpuric abdominal lesions observed in the patient represent a characteristic clinical sign of the disseminated phase. They are caused by the invasion of filariform larvae into the dermal microvasculature, causing mechanical damage, extravasation of erythrocytes, and cutaneous hemorrhages in a reticulated or periumbilical pattern, known as “thumbprint purpura.”3,11 This finding, described by Weiser et al., 11 reflects a specific cutaneous manifestation of hyperinfection and is usually associated with severe systemic involvement. In advanced stages, vascular destruction favors secondary bacteremia and sepsis, contributing to the high mortality described in these patients.4,8
The absence of eosinophilia in this patient is consistent with other reports of hyperinfection, where the immune response is suppressed or shifted toward a non-protective profile, allowing uncontrolled larval migration.2,5,9
Timely diagnosis was hampered by the absence of eosinophilia, a finding that can delay clinical suspicion of disseminated strongyloidiasis. The initial hospitalization did not allow for a conclusive diagnosis, and the patient was discharged without improvement, further delaying the identification of the infection. Additionally, the similarity of the respiratory symptoms to tuberculosis or pneumonia, common pathologies in the region, led to clinical overlap that contributed to the diagnostic delay.
The case management presented several limitations; the late recognition of the purpuric abdominal lesions delayed the suspicion of disseminated strongyloidiasis, despite the patient coming from an endemic area. Furthermore, the clinical evolution was fulminant, which prevented the administration of ivermectin and limited the available therapeutic options, contributing to the fatal outcome.
Although the inability to initiate antiparasitic therapy in this patient was related to abrupt clinical deterioration, alternative therapeutic strategies for severe strongyloidiasis have been described. In patients from endemic areas with clinical suspicion of disseminated infection, early empirical treatment with ivermectin is recommended, even before parasitological confirmation, given the high mortality associated with delayed therapy.3,8 In critically ill patients with impaired gastrointestinal absorption, ivermectin administered via nasogastric tube or subcutaneous route has been successfully used, particularly in cases of hyperinfection syndrome. 8 Importantly, the presence of characteristic abdominal purpuric lesions, as observed in this case, should prompt immediate consideration of empirical treatment for S. stercoralis, as this finding represents an indirect marker of disseminated disease and hyperinfection.3,11 Despite parasitological confirmation, the rapid progression to severe respiratory failure in this patient precluded timely ivermectin administration, highlighting a major challenge in disseminated strongyloidiasis, namely its potential for fulminant progression before specific treatment can be initiated. 8
The clinical manifestations of hyperinfection are diverse. In this case, pulmonary involvement was predominant, consistent with the findings described by Bravo et al. 1 and Le Pogam et al., 3 who reported cases of acute respiratory failure with high mortality. Mortality from disseminated strongyloidiasis can reach up to 70%–90% when the diagnosis is late.5,8
This case underscores the importance of considering strongyloidiasis in the differential diagnosis of purpuric skin lesions and pulmonary manifestations in people living with HIV from endemic regions, regardless of CD4 cell count or virological control. Early recognition of highly suggestive clinical signs, particularly characteristic abdominal or periumbilical purpura, should be interpreted as a critical warning sign of hyperinfection or disseminated disease and should prompt immediate therapeutic decision-making, even in the absence of overt pulmonary involvement or before parasitological confirmation, given the fulminant course and high mortality associated with delayed treatment.3,8,11 In endemic regions, the implementation of systemic screening strategies and preventive antiparasitic treatment within HIV care programs is essential to reduce severe outcomes, as demonstrated in recent studies from the Peruvian Amazon. 10
Conclusion
Disseminated strongyloidiasis can occur in patients with clinically controlled HIV infection. This case demonstrates that a preserved CD4 count does not exclude the risk of severe forms, especially in endemic areas. Routine parasitological screening and prophylactic treatment should be included in comprehensive care programs for HIV-positive patients. Early detection and timely initiation of ivermectin are crucial for reducing the high mortality associated with this parasitic infection.
Recommendations
Systematic screening in endemic areas
In tropical and subtropical regions where S. stercoralis is endemic, routine parasitological screening is recommended for all people living with HIV, regardless of their CD4 count or clinical stage. Active screening through serial stool examinations or immunological tests can identify asymptomatic infections and prevent severe forms. 10
Prophylaxis with preventive treatment in cases of prolonged exposure
In patients with a history of continuous exposure to contaminated soil or with previous parasitic infections, preventive administration of ivermectin should be considered, especially before procedures that modify the immune system, such as the initiation of corticosteroids, chemotherapy, or transplantation.3,8,11
Monitoring for nonspecific cutaneous and pulmonary signs
The appearance of purpuric abdominal or periumbilical lesions should be considered a warning sign, particularly the pattern described as “thumbprint purpura.” This sign may precede hyperinfection, and its early recognition is key to diagnosis. 11
Comprehensive interpretation of immunological parameters
The CD4 count should not be considered an absolute marker of protection against S. stercoralis infections. It is recommended to interpret immunological values in conjunction with the epidemiological and clinical context and parasitic history, given that disseminated infection can occur even with apparently preserved immunity.6,9
Strengthening the training of healthcare personnel
It is essential to train medical and laboratory personnel in the clinical and microscopic recognition of S. stercoralis, as well as in the interpretation of parasitological results. Early diagnosis is crucial to reducing the high mortality associated with hyperinfection.1,4
Integration of parasitic management into HIV programs
Comprehensive care programs for people living with HIV should include screening, treatment, and follow-up protocols for helminthiasis, especially S. stercoralis, within their strategies for controlling opportunistic infections. 10
Supplemental Material
sj-docx-1-tai-10.1177_20499361261425327 – Supplemental material for Fatal dissemination of Strongyloides stercoralis in an HIV-positive patient with preserved immunity: a case report
Supplemental material, sj-docx-1-tai-10.1177_20499361261425327 for Fatal dissemination of Strongyloides stercoralis in an HIV-positive patient with preserved immunity: a case report by Joel Hurtado Dominguez and Silvana Fernanda Loza Mollinedo in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
The authors would like to thank the healthcare and the laboratory staff involved in the care and diagnostic evaluation of the patient.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
