Abstract
Introduction
Herein, we present a case of IFI due to
Case presentation
Our patient is a 56-year old Caucasian male who suffered from a chronic myelomonocytic leukemia diagnosed 1.5 years prior to the current episode. He developed progressive cytopenias and features of high risk of progression to acute leukemia, being referred to our hospital for allogeneic-HSCT from 9/10 human leukocyte antigen (HLA) matched unrelated donor.
Other comorbidities included previous tobacco use, arterial hypertension, type 2 diabetes mellitus, obesity, obstructive sleep apnea, chronic kidney disease and hepatic steatosis. He was also diagnosed with latent tuberculosis infection and started therapy with isoniazid 1 month before HSCT. He had a previous job as an environmental inspector.
After HSCT, the patient received acyclovir, letermovir, inhaled AmB (25 mg three times a week) and fluconazole (400 mg per day) as antimicrobial prophylaxis. Regarding immunosuppressive therapies, he received reduced intensity conditioning (fludarabine 30 mg/m2 for 4 days and melphalan 70 mg/m2 for 2 days). As prophylaxis of graft-versus-host disease he received cyclophosphamide 50 mg/kg day +3 and +4, cyclosporine adjusted by serum levels and mycophenolate mofetil 1 g/8 h from +5. Cyclosporine was subsequently withheld on day +7 due to deterioration of renal function. It was switched to sirolimus which was also interrupted on day +21 due to interference in healing. Tacrolimus was started on day +22. He also received abatacept 10 mg/kg on days +1, +10, +24 and +52.
He had an episode of neutropenic fever the day before the infusion of stem cells (day −1), receiving broad-spectrum antibiotic therapy (meropenem) with no microbial isolations (negative blood and urine cultures). On day +3 he presented again with spiking fever. Colistin and vancomycin were added to the antimicrobial regimen. On day +6 he developed mild facial edema and gingival pain without evident lesions on examination. Due to the lack of microbial isolation in previous blood cultures, he was de-escalated to amoxicillin/clavulanate. On day +8 he was diagnosed with sinusoidal obstruction syndrome, and prophylactic fluconazole was stopped due to concerns for hepatotoxicity (as well as isoniazid) and changed to isavuconazole (200 mg three times a day for 2 days followed by 200 mg per day) due to its better adverse-event profile. By then, serum fungal biomarkers (galactomannan and beta-D-glucan assays, extracted on day +6) were negative and a chest computerized tomography (CT) showed pulmonary edema but no signs of fungal infection. Isavuconazole was continued as antifungal prophylaxis. Galactomannan and beta-D-glucan were subsequently determined on a weekly basis during admission and always resulted negative.
On day +14 he remained neutropenic, his mouth discomfort worsened, and edema and mild tumefaction of the facial area over the right maxillary zone was noticed. A facial CT scan was performed showing phlegmonous changes in cervicofacial area without organized collections. Maxillofacial surgeons were consulted and established a diagnosis of odontogenic infection. On day +20, blackish necrotic ulcers appeared overnight on the gingival mucosa around teeth 11, 12, 15 and 16, raising the suspicion of necrotizing fungal infection (Figure 1). A surgical biopsy was performed showing septate and branched hyphae with tissular invasion on direct examination (Figure 2). With the suspicion of breakthrough IFI, antifungal therapy with intravenous liposomal AmB at a dose of 5 mg/kg/day was initiated and isavuconazole was switched to posaconazole at a dosage of 300 mg twice daily on the initial day, followed by 300 mg per day. Waiting for the mycological testing results, micafungin therapy was also added at a daily dose of 100 mg as a third antifungal drug on day +21.

Evolution of oral lesions. Panel (a) and (b) shows initial appearance of necrotic ulcers affecting gingival mucosa in the right superior maxilar zone. Panel (c) shows the appearance after hemimaxillectomy and second debridement. Residual blackish lesions can be noticed.

Microbiologic findings. Panel (a) shows direct microscopy examination with septate and branched hyphae. Panel (b) shows initial “cotton-like” appearance of fungal culture. Panel (c) shows late dark-coloured appearance of fungal culture.
The initial antifungal susceptibility test performed at our local mycology lab showed posaconazole resistance and caspofungin sensitivity (no other equinocandins were initially tested). Thus, posaconazole was stopped and micafungin was switched to high dose caspofungin (100 mg per day) on day +26. Final antifungal susceptibility testing results from the mycology reference lab are depicted in Table 1. An aggressive surgical debridement was urgently performed, with hemi-maxillectomy on day +22. Histopathology examination revealed fungal structures with septate hyphae with vascular invasion (Figure 3). CT scan of sinuses and lungs and brain magnetic resonance imaging (MRI) did not show further fungal involvement. The patient remained severely neutropenic at that moment, and the initial evolution was unfavourable, requiring several surgical debridements with biweekly cures and three interventions for extending surgical borders due to disease progression on days +34, +38 and +41 (fungal and bacterial cultures were obtained in all procedures). The extension of the infection to the maxillary sinus and infraorbital groove required further mucosal and bone resection. Due to the profound neutropenia, the patient also received two granulocyte transfusions which were well tolerated, until neutrophil count recovered on day +28. Once the liver function tests improved, and taking into account the difficulties to control the infection with the presence of
Antifungal susceptibility test results from the mycology reference Lab.

MIC, minimal inhibitory concentration.
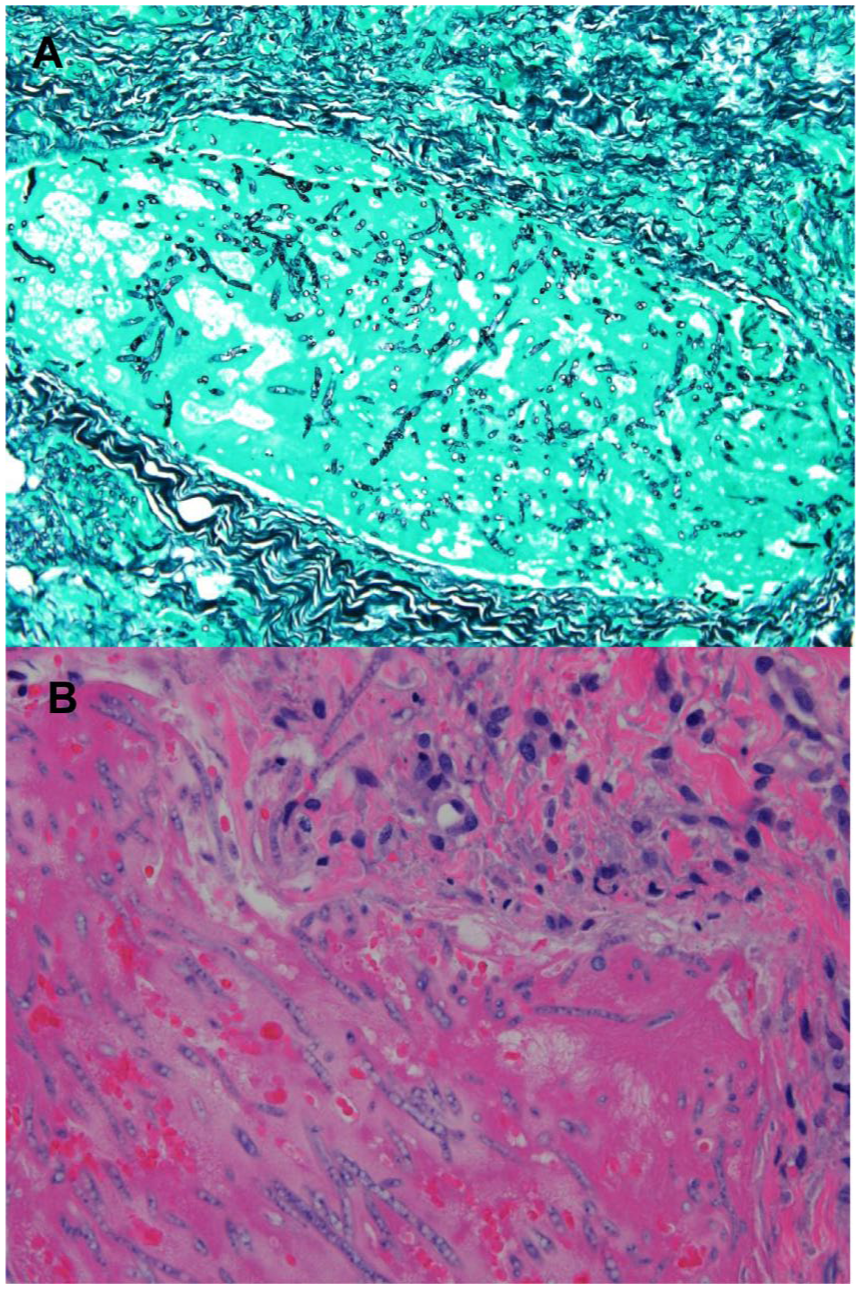
Histopathology examination from surgical samples. Panel (a) (Grocott stain, 20×), Panel (b) (Hematoxylin-eosin stain, 40×). Squamous mucosa fragment with reactive epithelial changes, filamentous fungal structures with narrow septate hyphae invading lamina propria and blood vessels.
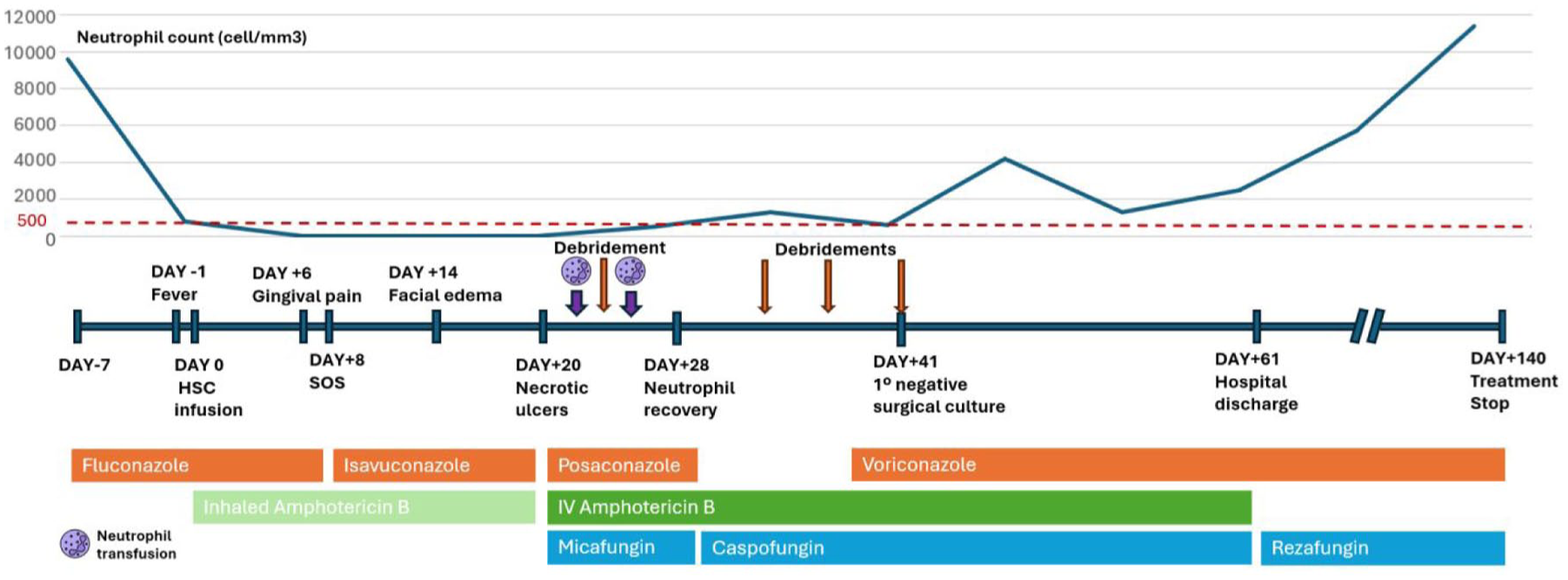
Temporal line showing clinical evolution and antifungal therapy.
Discussion
Our case shows the potential of
A small proportion of patients with lung involvement have demonstrated positive results for serum beta-D-glucan and galactomannan in bronchoalveolar lavage. 4 In our patient, both fungal biomarkers were repeatedly negative in serum.
Regarding the antifungal therapy, the complexity of the case and the initial poor response with persistence of fungal structures in the surgical samples, prompted the utilization of a combination of different antifungal drugs. Prior small-size studies, subject to a high risk of bias (confounding by indication), demonstrated better outcomes with the combination of voriconazole and caspofungin than with other treatments. Sal et al described in their review a lower mortality in patients treated with caspofungin (28%) and voriconazole (46%) monotherapies than in patients treated with AmB (55%) or other triazoles (73%) in monotherapy. The mortality rate was also lower with the combination of caspofungin and voriconazole (1/4, 25%) than with the azole and AmB combination (4/6, 66%).
4
Despite the described limitations and due to the scarcity of previous data, we decided to maintain combination therapy with at least two active antifungal drugs, and eventually, up to three simultaneous drugs (AmB, voriconazole and caspofungin), until the confirmation of clinical and microbiological response in surgical samples, with negativization of fungal cultures. With this rationale, and due to the extreme frailty of the patient and the immunosuppression derived from HSCT, outpatient therapy was continued with a combination of oral voriconazole and weekly parenteral rezafungin. The selection of antifungal agents in our case was determined by the availability of preliminary and final susceptibility testing, the clinical course and concomitant toxicities. Voriconazole was not added immediately after receiving susceptibility testing report as the patient was receiving two other active agents, due the concerns of excessive toxicity. However, due to the lack of clinical improvement it was finally included in the therapeutic scheme. Despite the absence of established minimal inhibitory concentration (MIC) breakpoints,
Rezafungin is a novel echinocandin with a prolonged elimination half-life, enabling its administration on a weekly basis. It disposes an improved activity against filamentous fungi compared to other echinocandins.7,8 However, there are no reports of its activity against
The other cornerstone of the management of aggressive soft tissue IFIs is surgical intervention, aimed at reducing the fungal burden. Surgery also facilitates penetration of the antifungal drug by removing necrotic tissue, and prevents further dissemination of the infection.
10
The initial clinical presentation of the patient suggested mucormycosis, and the finding of septate hyphae in the initial microscopy examination extended the differential diagnosis to
Lastly, the other important therapeutic measure was to achieve a correction of the predisposing factor. In this particular instance, the objective was to enhance the patient’s immunological status through granulocyte infusions, in the context of persistent neutropenia. The role of granulocyte transfusions in invasive fungal infections is controversial, due to the potential for severe side effects and contradictory reports regarding their efficacy. However, they can be useful in selected cases.11,12 In our case the transfusion was performed as a provisional intervention to stabilize the infection until the subsequent occurrence of engraftment and neutrophil normalization. The patient did not experience any adverse events related to the granulocyte transfusion.
Conclusion
In conclusion,
Supplemental Material
sj-docx-2-tai-10.1177_20499361251403060 – Supplemental material for Necrotizing gingival infection due to Trichoderma longibrachiatum in an hematopoietic stem cell transplant recipient: case report
Supplemental material, sj-docx-2-tai-10.1177_20499361251403060 for Necrotizing gingival infection due to Trichoderma longibrachiatum in an hematopoietic stem cell transplant recipient: case report by Eduardo Aparicio-Minguijón, Nieves López-Muñoz, Pilar Martínez-Sánchez, María Calbacho, Ana Pérez-Ayala, Justino Jiménez-Almonacid, María Asunción Pérez-Jacoiste Asín and Isabel Rodríguez-Goncer in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-pdf-1-tai-10.1177_20499361251403060 – Supplemental material for Necrotizing gingival infection due to Trichoderma longibrachiatum in an hematopoietic stem cell transplant recipient: case report
Supplemental material, sj-pdf-1-tai-10.1177_20499361251403060 for Necrotizing gingival infection due to Trichoderma longibrachiatum in an hematopoietic stem cell transplant recipient: case report by Eduardo Aparicio-Minguijón, Nieves López-Muñoz, Pilar Martínez-Sánchez, María Calbacho, Ana Pérez-Ayala, Justino Jiménez-Almonacid, María Asunción Pérez-Jacoiste Asín and Isabel Rodríguez-Goncer in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
We acknowledge the hematology, maxillofacial surgery, microbiology and infectious disease practitioners who provided care of the patient and have not been able to participate as authors of this manuscript. We specially acknowledge Laura Alcazar-Fuoli from Mycology Reference Laboratory for her contributions.
Author note
The case was conducted and reported in accordance with CARE guidelines.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
