Abstract
Background:
Treatment of methicillin-susceptible Staphylococcus aureus (MSSA) bacteremia with antipseudomonal beta-lactams (APBLs) is a common clinical practice in confirmed or suspected polymicrobial infections. Limited data exist to evaluate the impact of APBL treatment on outcomes in MSSA bacteremia.
Objectives:
To determine whether differences in outcomes exist between patients with MSSA bacteremia treated with cefazolin/oxacillin (standard of care; SOC) versus APBLs.
Design:
Retrospective cohort study.
Methods:
Adult and pediatric patients hospitalized between June 2016 and June 2023 with at least one positive blood culture for MSSA who received at least 14 consecutive days of inpatient therapy with cefazolin, oxacillin, cefepime, piperacillin–tazobactam, or meropenem were included. Patients who received APBLs were compared to those who received SOC. The primary outcome was composite clinical failure (i.e., 30-day all-cause mortality and/or bacteremia recurrence within 30 days). Secondary outcomes included 60-day all-cause mortality, intensive care unit and hospital length of stay, 30-day readmission, time to bacteremia clearance, time to mortality, and adverse events (i.e., Clostridioides difficile infection, hepatotoxicity, and acute kidney injury).
Results:
One hundred patients were included, with 50 patients in each group. The most common source of bacteremia was catheter-related (28% total), and twice as many patients met criteria for complicated MSSA bacteremia in the SOC versus APBL group (80% vs 40%, p < 0.0001). Three patients (6%) in the APBL group met the composite primary outcome compared to 1 (2%) in the SOC group (p = 0.62). There were no significant differences in secondary outcomes.
Conclusion:
No differences in mortality or bacteremia recurrence were identified among patients with MSSA bacteremia treated with APBLs compared to SOC. Larger studies should be performed to confirm these findings.
Introduction
Staphylococcus aureus bacteremia is a serious bacterial infection associated with mortality rates greater than 25% at 3 months and prolonged hospital length of stay. 1 The preferred treatment options for methicillin-susceptible S. aureus (MSSA) bacteremia are antistaphylococcal penicillins (e.g., oxacillin or nafcillin) or cefazolin. In clinical practice, antipseudomonal beta-lactams (such as cefepime, piperacillin-tazobactam, or meropenem) may be utilized for treatment of MSSA bacteremia despite a lack of supporting data. This may occur in situations such as polymicrobial bacteremia or severe infections in which there is suspicion for involvement of other organisms beyond MSSA (e.g., diabetes-related foot infection, intra-abdominal infection, or osteomyelitis involving sacral decubitus ulcers). Studies comparing cefazolin and antistaphylococcal penicillins to antipseudomonal beta-lactams for treatment of MSSA bacteremia are limited, and most have found increased mortality with the use of antipseudomonal beta-lactams.2–4 In addition, previous studies have been conducted primarily in adult patients. There is limited data on the optimal approach in pediatric patients with MSSA bacteremia who require antipseudomonal therapy, and treatment decisions are guided by clinical experience. 5
The purpose of this study was to determine whether differences in outcomes exist between patients with MSSA bacteremia treated with cefazolin or oxacillin (standard of care; SOC) vs antipseudomonal beta-lactams (APBLs) (i.e., cefepime, piperacillin-tazobactam, or meropenem).
Materials and methods
Study design
This was a single-center, retrospective cohort study conducted at UF Health Shands Hospital, a 1162-bed tertiary academic medical center in Gainesville, FL. The study included pediatric and adult patients aged 0–100 years with at least one positive blood culture for MSSA between June 1, 2016 and June 30, 2023, who received at least 14 consecutive days of inpatient therapy with cefazolin, oxacillin, cefepime, piperacillin-tazobactam, and/or meropenem. Patients were included in the APBL group if they received at least 14 consecutive days of inpatient therapy with cefepime, meropenem, piperacillin-tazobactam, or a combination of these agents. Those with polymicrobial bacteremia were included only if appropriate antibiotics were administered to target all blood pathogens. Patients were excluded from the study if they received oritavancin or dalbavancin, dual-targeted therapy against MSSA (defined as cefazolin and ertapenem, oxacillin and ertapenem, or daptomycin and ceftaroline; rifampin and gentamicin synergy were allowed), or oral antibiotics for MSSA bacteremia for greater than 48 h. Patients who had a self-directed discharge, transitioned to palliative care, discontinued antibiotics, or died within 72 h following blood culture collection were also excluded from the study. To ensure that factors expected to account for variability in treatment outcomes were represented equally in both study groups, patients were matched 1:1 using three variables expected to contribute to the most confounding: age (adult, ⩾18 years or pediatric, <18 years), presence of an endovascular infection (defined as endocarditis, septic thrombophlebitis, or mycotic aneurysm), and receipt of vasopressors within 24 h of the first positive blood culture with MSSA. 6 The study protocol was approved by the University of Florida Institutional Review Board. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 7
Screening and data collection
Data were collected from the electronic health record with assistance from the University of Florida Integrated Data Repository Research Services.
Outcomes and definitions
The primary outcome was a composite of clinical failure consisting of all-cause mortality within 30 days from the index date (collection date of first positive blood culture with MSSA) and/or bacteremia recurrence within 30 days following blood culture clearance. Secondary outcomes included all-cause mortality within 60 days from the index date, intensive care unit (ICU) length of stay, hospital length of stay, readmission within 30 days following the discharge date, time to blood culture clearance from the index date, time to mortality from the index date, and incidence of C. difficile infection, acute kidney injury (AKI), and hepatotoxicity. This study did not evaluate whether adverse effects were specifically caused by the study antibiotic(s). The decision to evaluate mortality at 30 and 60 days was based on a study that found a declining proportion of deaths in patients with S. aureus bacteremia attributable to the bacteremia episode as follow-up extended beyond 30 days. 8 Complicated MSSA bacteremia was defined according to the Infectious Diseases Society of America (IDSA) criteria in patients who met one of the following: the presence of endocarditis, implanted prostheses, repeat positive blood cultures obtained 2–4 days following the initial set, lack of defervescence within 72 h of initiation of effective therapy, or evidence of metastatic sites of infection. 9 ICU length of stay was calculated as the total number of patient days in any ICU from the index date to the end of treatment for the MSSA bacteremia episode. Incidence of C. difficile infection was defined as both a positive C. difficile polymerase chain reaction result and administration of treatment directed toward active C. difficile infection for ⩾48 h (i.e., oral vancomycin or fidaxomicin) from the index date to 30 days after the end of treatment for MSSA bacteremia. AKI was determined using the Kidney Disease: Improving Global Outcomes (KDIGO) definition of serum creatinine (SCr) increase of at least 0.3 mg/dL within 48 h or a 50% increase in SCr from baseline within 7 days. 10 Hepatotoxicity was defined as an alanine aminotransferase (ALT) and/or aspartate aminotransferase (AST) greater than or equal to three times the upper limit of normal (ULN).
Statistical analyses
Continuous variables were described as means and standard deviations for parametric data; medians and interquartile ranges were reported for nonparametric data. Categorical variables were described as frequencies and proportions. Inferential statistics were performed using the Wilcoxon rank-sum test for continuous variables and the χ2 or Fisher’s exact test for categorical variables, as appropriate. Statistical significance was defined by a threshold of α < 0.05. All statistical analyses were performed using Microsoft Excel and JMP® Pro version 17.0.0 software (SAS Institute, Cary, NC).
Results
A total of 1011 patients with MSSA bacteremia who received at least one dose of the study antibiotics (i.e., cefazolin, oxacillin, cefepime, piperacillin-tazobactam, and/or meropenem) were identified during the study period (Figure 1). Four patients were excluded for receiving oritavancin or dalbavancin, and 728 were excluded due to not receiving 14 total days of study antibiotics in the inpatient setting. Of the 279 remaining patients, 84 received at least 14 total days of APBLs. Out of these 84 patients, 11 were moved to the SOC group as they did not receive 14 consecutive days of APBLs, but did receive at least 14 consecutive days of cefazolin and/or oxacillin, and 23 were excluded from the study because they did not receive 14 consecutive days of either APBLs or SOC. A total of 50 patients were included in the APBL group, and these patients were matched 1:1 to identify a total of 50 patients for inclusion in the SOC group based on the previously defined matching criteria. Patients were screened in reverse chronological order from the date of admission to evaluate for inclusion in the SOC group.

Patient enrollment in the study.
Baseline characteristics are shown in Table 1. The majority of patients included in the study were adult males, with a mean age of 54 years. Community-onset infections predominated, and the most common sources of MSSA bacteremia were catheter-related (28%) and skin and skin structure infection (24%). Organisms identified at the highest frequency in polymicrobial bacteremia episodes were Enterobacterales spp., Streptococcus spp., coagulase-negative Staphylococcus spp., and Enterococcus faecalis.
Baseline and clinical characteristics.

Other organisms in blood cultures: Bacillus spp. (n = 3), Corynebacterium spp. (n = 3), Haemophilus influenzae (n = 1).
In the APBL group, 33 unique patients received SOC (17 oxacillin only, 13 cefazolin only, and 3 both oxacillin and cefazolin sequentially).
Other antibiotics received: linezolid, clindamycin, gentamicin, rifampin, levofloxacin, ciprofloxacin, daptomycin, ceftaroline, and doxycycline.
AKI, acute kidney injury; APBL, antipseudomonal beta-lactam; IQR, interquartile range; SD, standard deviation; SOC, standard of care (cefazolin/oxacillin).
Complicated S. aureus bacteremia was twice as common in the SOC group as in the APBL group (80% vs 40%, p < 0.0001). In addition, hardware-associated infection and osteoarticular infection were more frequently identified in the SOC group compared to the APBL group (20% vs 4% for each, p = 0.01). On the other hand, cancer was more common in the APBL group (26% vs 10%, p = 0.04), as was polymicrobial bacteremia (34% vs 12%, p = 0.009). Most patients received cefepime and/or vancomycin during their treatment course. The median duration of vancomycin was 5 days (IQR 3–11) in the APBL group compared to 3.5 days (IQR 2–5) in the SOC group (p = 0.0084). The median duration of therapy for MSSA bacteremia was 45 days (IQR 31–50) in the SOC group compared to 30 days (IQR 19–43.5) in the APBL group (p = 0.0020). There were 33 patients in the APBL group who received SOC during the treatment course, with a median time to initiation of 79 h (calculated as the time from index blood culture collection to the first dose of SOC). SOC was administered for a median of 7.5 days (IQR 4–14.5). Infectious diseases (ID) consultation was performed for all 50 patients (100%) in the SOC group compared to 42 patients (84%) in the APBL group (p = 0.0058).
Table 2 includes the study outcomes. No statistically significant difference was found in the composite primary outcome of clinical failure between the APBL and SOC groups (6% vs 2%, p = 0.62). Similar outcomes were found in both groups for 60-day mortality, ICU and hospital length of stay, readmission within 30 days, and time to mortality. With respect to adverse events in the total study population, 53% of patients met criteria for AKI, 20% of patients experienced hepatotoxicity, and 7% of patients developed C. difficile infection during the study period; however, no statistically significant differences were identified between groups for any of these outcomes. The median time to blood culture clearance was prolonged at 95 h (IQR 49.5–144) in the SOC group compared to 65.5 h (IQR 42.3–117.3) in the APBL group; however, this finding did not reach statistical significance (p = 0.17).
Study outcomes.
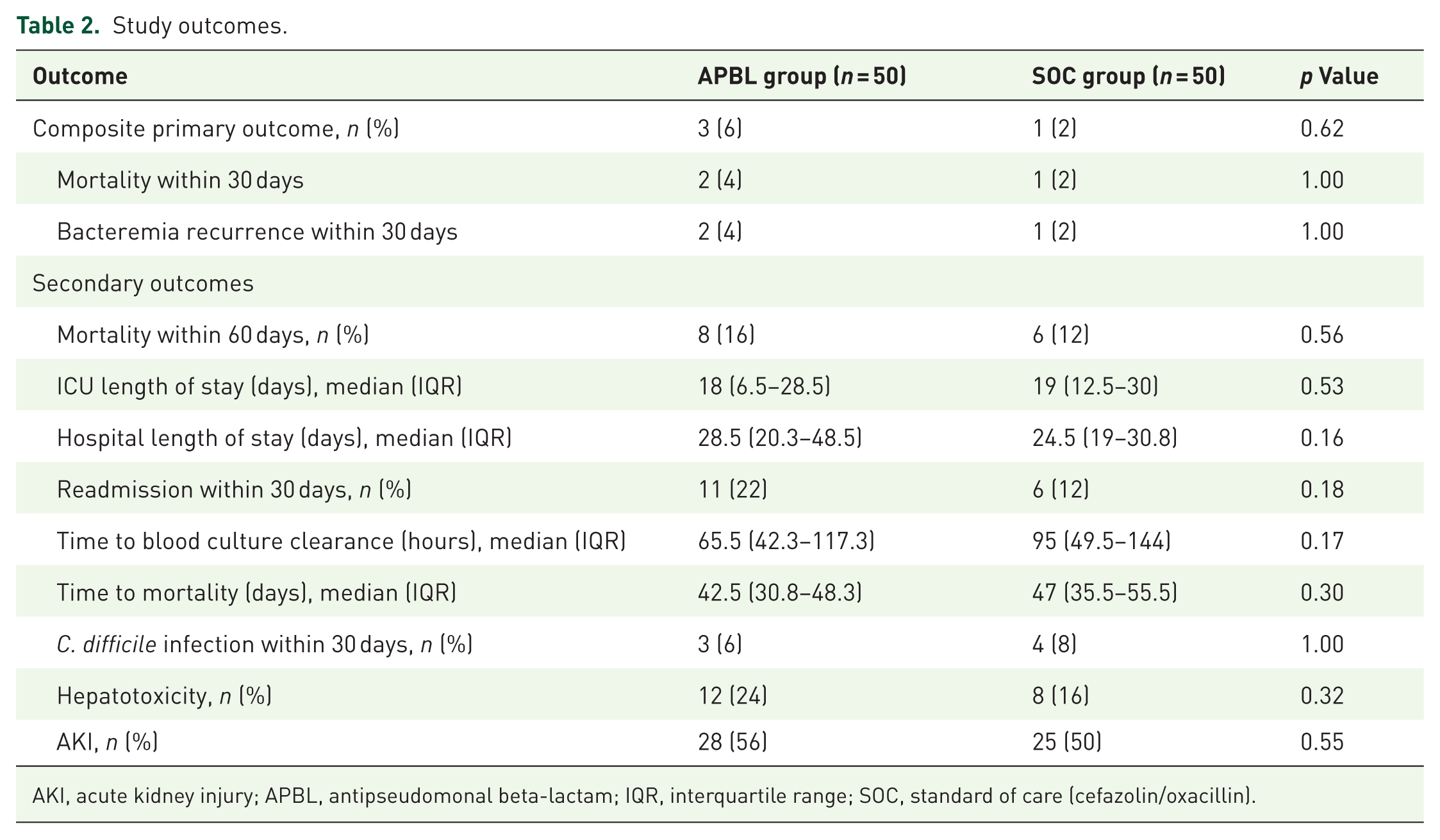
AKI, acute kidney injury; APBL, antipseudomonal beta-lactam; IQR, interquartile range; SOC, standard of care (cefazolin/oxacillin).
There were six pediatric patients included in the study (three patients in each group). Evaluation of these patients revealed most had complicated MSSA bacteremia with catheter-related infection as the source and achieved source control (n = 4/6, 67%). In addition, the median total treatment duration was 22.5 days, and no patients met the primary composite outcome of clinical failure. For secondary outcomes, no pediatric patients died within 60 days, and three patients (50%) were readmitted within 30 days.
Discussion
S. aureus bacteremia affects a wide variety of patient populations, and the selection of appropriate therapeutic regimens is of utmost importance given the significant morbidity and mortality associated with these infections. 1 IDSA guidelines indicate that drugs of choice in the management of MSSA infections are beta-lactams, such as cefazolin or antistaphylococcal penicillins (e.g., oxacillin or nafcillin). 9 In the setting of polymicrobial infections or coinfections, patients may receive broad-spectrum agents such as APBLs for management of MSSA bacteremia. In this study, no significant differences were found in 30-day all-cause mortality or 30-day bacteremia recurrence among patients who received APBLs versus SOC for MSSA bacteremia. Similarly, there were no significant differences in 60-day mortality, length of hospital and ICU stay, readmission rates within 30 days, time to mortality, and rates of adverse events between the groups.
Several previous studies have evaluated outcomes associated with different therapeutic agents in MSSA bacteremia. McDanel et al. evaluated empiric and definitive therapy with vancomycin versus beta-lactams (including APBLs) in over 5600 patients admitted to Veterans Affairs hospitals with MSSA bacteremia. The authors reported similar rates of 30-day all-cause mortality with empiric regimens between groups; however, beta-lactams were associated with 35% lower mortality than vancomycin in definitive therapy. Within the beta-lactam group, piperacillin–tazobactam was one of the most common agents evaluated, comprising 32% and 15% of beta-lactams administered in empiric and definitive regimens, respectively. 11 Findings in our study are similar to those from a prior analysis by Aleissa et al. that reported no difference in rates of 60-day mortality and bacteremia recurrence among patients with febrile neutropenia and MSSA or penicillin-susceptible S. aureus (PSSA) bacteremia who received broad-spectrum beta-lactams (i.e., piperacillin-tazobactam, cefepime, or meropenem) vs narrow-spectrum beta-lactams (i.e., cefazolin, oxacillin, or nafcillin). The study also evaluated combination beta-lactam therapy (defined as concomitant broad-spectrum and narrow-spectrum beta-lactam therapy for at least 72 h) and found increased 60-day mortality with combination therapy when compared to broad-spectrum therapy in a multivariable logistic regression. 4 Some studies have found increased mortality with the use of APBLs when compared to cefazolin or antistaphylococcal penicillins for treatment of MSSA bacteremia. A retrospective cohort study by Paul and colleagues compared outcomes between cloxacillin or cefazolin versus other beta-lactams (APBLs included from most to least frequent: ceftazidime, piperacillin-tazobactam, carbapenems, and cefepime) for treatment of adults with MSSA bacteremia, and found a reduction in 30-day mortality with empiric use of cloxacillin/cefazolin. Specific to APBLs, an evaluation of empiric antibiotic groups in this study also reported increased mortality when beta-lactam-beta-lactamase inhibitors, including piperacillin-tazobactam, were compared directly to cloxacillin/cefazolin. 2 Another retrospective cohort study by Beganovic et al. found decreased 30-day mortality among adult patients who received exclusive nafcillin, oxacillin, or cefazolin compared to piperacillin–tazobactam for treatment of MSSA bacteremia (2.1% vs 20.8%, respectively, in the propensity-score matched cohort). Although our study found a similar 30-day mortality rate in the SOC group (2%), lower mortality was observed in the APBL group (4%). However, direct comparison of this outcome is limited by differences in APBLs used (i.e., piperacillin–tazobactam in the Beganovic study vs varied regimens that could include a combination of piperacillin–tazobactam, meropenem, and/or cefepime in our study). 3 Lastly, Najia et al. conducted a retrospective analysis to evaluate outcomes of APBLs for treatment of MSSA bacteremia or pneumonia in patients with polymicrobial or concomitant infections. The study included 45 patients and found no differences in treatment success rates ranging from 50% to 70% among APBLs (i.e., cefepime, piperacillin–tazobactam, and meropenem). While APBLs were not directly compared to other beta-lactams, the authors acknowledged lower rates of treatment success compared to cefazolin and nafcillin/oxacillin in the literature, which was felt to be related to inclusion of polymicrobial and concomitant infections in the study. 12
Compared to previous research, this study has several shared and distinct features in patient populations and study design. Aside from the study by Aleissa et al., which focused solely on patients with febrile neutropenia and malignancy, cancer rates across the remaining studies were comparable to those seen here. In addition, the most common sources of MSSA bacteremia (excluding primary or unknown source) were catheter-related and skin and skin structure infections, consistent with this evaluation.2–4 In contrast to previous studies limited to adults, our evaluation also included pediatric patients with MSSA bacteremia, which is a group that may experience different clinical features and outcomes. In children, S. aureus bacteremia is associated with a higher frequency of osteoarticular infections and lower mortality rates compared to adults. 13 These differences suggest that outcomes may not be directly comparable between groups, underscoring the need for more data to guide treatment decisions in this underrepresented population. In addition, unlike the study by Paul et al., differences between empiric and definitive treatment regimens were not evaluated in our study. Instead, comparator groups were determined based on receipt of at least 14 days of consecutive therapy, as patients often switch between different antibiotics during their treatment course. This study also differed from Beganovic et al., as outcomes were not evaluated based on exclusive treatment with one therapeutic agent. The decision to combine cefazolin and oxacillin into one group in this study was based on previous analyses demonstrating similar outcomes between the two agents for treatment of MSSA bacteremia.14,15
One notable finding in this study included a longer median time to blood culture clearance in the SOC group compared to the APBL group by about 30 h (95 h vs 65.5 h, respectively); however, this result did not reach statistical significance. This is related to the finding of twice as many patients with complicated MSSA bacteremia in the SOC group, as one component of the criteria for complicated S. aureus bacteremia is prolonged time to culture clearance beyond 2–4 days following the initial set of blood cultures. 9 In addition, patients in the SOC group experienced more deep-seated infections (including hardware-associated and osteoarticular infections), which may explain the longer total treatment duration utilized in this group, as these infections typically require prolonged durations of therapy. The median total treatment duration was longer than anticipated at 30 days in the APBL group, despite only 40% of patients with complicated MSSA bacteremia. The authors were unable to identify the definitive cause of this finding through data collected; however, it is possible that these patients may have been treated for other infections that required longer durations of therapy, which could, in turn, explain the reason for the selection of APBLs. In addition, the inclusion criteria requiring at least 14 days of inpatient therapy with study antibiotics may have selected for more complicated patients for reasons outside of MSSA bacteremia. Patients in the SOC group transitioned to targeted therapy with cefazolin or oxacillin at a median of 54.5 h (IQR 42–85) after the index date. This is reflective of the average turnaround time for susceptibility information to be available in the electronic health record from the index date. There were more patients in the APBL group with polymicrobial bacteremia, which was expected, as it was anticipated that APBLs would be employed to cover additional pathogens beyond MSSA. Patients in the APBL group also received, on average, 1.5 additional days of vancomycin compared to patients in the SOC group. The longer duration of vancomycin may, in part, reflect provider hesitancy to discontinue vancomycin and rely on APBL monotherapy for treatment of MSSA bacteremia due to a paucity of data to support this approach.
One strength of this study included the matching of patients based on three variables expected to contribute to the most confounding. 6 This was performed to prevent large variability in the types of patients included between groups that would make comparison of clinical and microbiological outcomes difficult. In addition, many patients received antibiotic agents from both groups at different times throughout their MSSA bacteremia treatment course. This is reflective of real-world practice, as patients may initially receive broad-spectrum therapy prior to de-escalation once MSSA bacteremia is identified, or may switch between antibiotics based on their clinical course. Inclusion of patients with polymicrobial bacteremia is also considered a strength, as this is reflective of real-world practice and represents a group of patients who may be more likely to receive broad-spectrum antimicrobial agents beyond antistaphylococcal penicillins or cefazolin for treatment of MSSA bacteremia. This study adds to the limited body of literature examining APBLs in the management of MSSA bacteremia.
Limitations of this study include a small sample size and the retrospective design. Although few pediatric patients were included, and conclusions remain limited in this group, the findings may offer preliminary insights, given the overall lack of pediatric data in the literature. In addition, since the majority of patients received cefepime in the APBL group, findings related to APBLs may be more applicable to cefepime than piperacillin–tazobactam or meropenem. While 98% of patients in the APBL group received cefepime at some point during their treatment course, 38% (n = 19) received cefepime alone. No sample size calculation was performed a priori due to the limited number of patients expected for inclusion in a single-center study. Potential confounding variables among groups are also considered a limitation. Patients in both groups received multiple agents with activity against MSSA throughout their treatment course, making interpretation of the results difficult. For instance, patients in the APBL group received a median of 7.5 days of SOC, representing 25% of this group’s median total treatment duration of 30 days. Consequently, outcomes in each group cannot be exclusively linked to either APBL or SOC therapy. However, the requirement for patients to receive at least 14 consecutive days of the study drug(s) was intended to help eliminate some of these concerns. Fourteen days was selected as this is recommended as the minimum duration of antibiotic treatment for S. aureus bacteremia. 9 This did, however, limit the number of patients eligible for inclusion. Concurrent administration of vancomycin and other antibiotics (including linezolid and clindamycin) with APBLs was possible; however, the median duration of these agents was short relative to the total duration of therapy in the APBL group (i.e., 5 days for vancomycin and 6 days for other antibiotics compared to the median total treatment duration of 30 days). In addition, the criteria used to determine recurrent bacteremia (i.e., growth of MSSA in any repeat blood cultures within 30 days after initial blood culture clearance) did not consider the skip phenomenon. This phenomenon has been reported in S. aureus bacteremia and refers to intermittently positive blood cultures occurring at least 1 day after prior negative cultures. 16 As a result, patients exhibiting this pattern with ongoing active infection may have been incorrectly classified as having recurrent bacteremia. However, given the limited number of patients identified with recurrent bacteremia, accounting for the skip phenomenon is unlikely to have had a meaningful impact on the study findings. Investigators also did not establish causality of adverse effects (i.e., AKI, hepatotoxicity, and C. difficile infection) to the study antibiotics. Significant differences in patient characteristics were identified between the APBL and SOC groups. There were more patients with polymicrobial bacteremia in the APBL group compared to the SOC group. While this is an expected finding, it is important to note that polymicrobial bacteremia is often associated with higher mortality rates than monomicrobial bacteremia. 17 Patients in the SOC group also experienced a higher frequency of complicated bacteremia and more deep-seated infections, indicating this group consisted of patients with more severe infections. Lastly, there were more patients with cancer in the APBL group, which may have biased this group toward worse clinical outcomes. Although outcomes were similar between groups in this study, it is possible that these confounders could contribute to differences in outcomes in a larger population.
Conclusion
This study found no significant differences in mortality and rates of bacteremia recurrence between patients who received APBLs versus SOC for treatment of MSSA bacteremia. Although SOC is preferred for targeted treatment of MSSA bacteremia, there were no concerning safety or efficacy signals in a small cohort of patients who received at least 14 consecutive days of APBLs. In addition, the results of this study suggest that substituting an APBL for SOC temporarily in patients who require broadened therapy during treatment of MSSA bacteremia may not worsen outcomes. The generalizability of these findings to pediatric patients may be limited by the small sample size and heterogeneity in age within this group. It is important to recognize the impact of APBLs in driving antimicrobial resistance, and without a compelling indication to utilize an APBL in MSSA bacteremia (such as polymicrobial infections), SOC is considered ideal. 18 Future studies with larger patient populations should be conducted to further clarify the outcomes associated with the use of APBLs in patients with MSSA bacteremia.
Supplemental Material
sj-docx-1-tai-10.1177_20499361251395527 – Supplemental material for Evaluation of outcomes associated with antipseudomonal beta-lactams for treatment of methicillin-susceptible Staphylococcus aureus bacteremia: a retrospective cohort analysis
Supplemental material, sj-docx-1-tai-10.1177_20499361251395527 for Evaluation of outcomes associated with antipseudomonal beta-lactams for treatment of methicillin-susceptible Staphylococcus aureus bacteremia: a retrospective cohort analysis by Madison Fielding, Kathryn DeSear, Vidhu Kariyawasam, Kalen Manasco, Lisa Vuong, Veena Venugopalan and Barbara A. Santevecchi in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
