Abstract
Background:
Tuberculosis (TB) remains a global public health priority, with 10.5 million new cases and 1.5 million deaths reported in 2023. Current diagnostic methods face limitations in sensitivity, biosafety, and accessibility, particularly in low-resource settings.
Objective:
This study evaluates the diagnostic accuracy of a novel, Polymerase Chain Reaction (PCR) platform (Orange G3 TBC), comparing it with the WHO-endorsed GeneXpert Ultra system in Oruro, Bolivia.
Design:
We conducted a randomized, double-blind study.
Methods:
The study included 71 clinical samples (67 sputum samples and 4 cerebrospinal fluid samples) from patients with presumptive TB. All samples were tested with GeneXpert Ultra and the Orange G3 TBC platform, which incorporates a unique biosafe processing system. Statistical analysis included sensitivity, specificity, predictive values (positive predictive value (PPV) and negative predictive value (NPV)), likelihood ratios (LR+ and LR−), and correlation measures.
Results:
Orange G3 TBC demonstrated strong performance metrics compared to GeneXpert Ultra: sensitivity = 90%, specificity = 97%, diagnostic efficiency = 96%, PPV = 82%, and NPV = 98%. Statistical analysis showed a high correlation between the two methods (Pearson’s correlation = 0.834, Kappa = 0.832, LR+ = 27.3, LR− = 0.103).
Conclusion:
The Orange G3 TBC platform offers comparable diagnostic accuracy to GeneXpert Ultra. The system is adaptable to resource-limited settings, making it a viable alternative for TB diagnosis in endemic regions.
Plain language summary
Tuberculosis (TB) is a major global health problem today, causing 10.5 million new cases and 1.5 million deaths by 2023. Rapid and accurate diagnosis is particularly challenging in settings with limited medical resources. In this study, we evaluated a new diagnostic tool called Orange G3 TBC against the widely used GeneXpert Ultra test, recommended by the WHO. We examined 71 patient samples (primarily sputum and cerebrospinal fluid) with suspected TB. Both tests were performed on each sample. Orange G3 TBC showed similar accuracy to GeneXpert Ultra, detecting TB in 90% of positive cases and correctly ruling it out in 97% of negative cases. It provided robust predictive values and a high level of agreement with the reference method. It is noteworthy that Orange G3 TBC includes a more biosecure sample handling system, making it adaptable for use in resource-limited clinics. These findings suggest that the Orange G3 TBC test could be a reliable and more accessible option for TB diagnosis in areas where the disease is common and advanced laboratory infrastructure is lacking.
Introduction
Tuberculosis (TB) is an infectious disease caused by Mycobacterium tuberculosis (M. tuberculosis) and is responsible for causing more than 1 billion deaths in the last 200 years. 1 The global burden of TB is concentrated in developing countries, particularly in Latin America, Africa, and some regions of Asia. 2 Despite the worldwide impact of this disease, case detection rates remain very low in many endemic settings, posing a significant obstacle to TB control efforts. In 2023, the World Health Organization (WHO) reported 10.5 million cases of TB and 1.5 million deaths. 2 The risk of TB transmission is linked to social and economic forces, including low schooling, overcrowding, malnutrition, and occupational exposure. 3
Diagnostic confirmation of TB remains a significant challenge in low-income and low-resource settings. There is likely a large number of undetected cases that foster ongoing chains of TB transmission that ultimately lead to incalculable human suffering. 2 Bolivia reports approximately 8000 cases of TB each year, with 200 TB-related deaths in 2023. 4 Smear microscopy has been the standard tool for diagnosing TB in Bolivia for decades. 5 This test has multiple limitations, including its low sensitivity and risk of transmission to laboratory personnel, and it offers a limited value in diagnosing extrapulmonary TB and identifying cases of pulmonary TB in children. 6 GeneXpert is an advanced molecular technology with the potential for rapid diagnosis of TB. 7 Since 2010, the WHO has endorsed this advanced molecular tool for diagnosing TB due to its high-pooled sensitivity and specificity.7,8 The use of GeneXpert for routine TB diagnosis increases health system testing costs compared to smear microscopy. 7 Multiple factors must be considered when choosing a molecular test. Even with similar comparisons, continuous and uninterrupted access to the supplies and ease of maintenance are paramount.
The Orange G3 TBC is a platform for nucleic acid amplification that provides important advantages in diagnosing TB. It was developed in Argentina in 2009 using a device designed to avoid the risk of airborne transmission to the operator. The platform has been validated in previous studies9–11 and authorized for clinical diagnostic use by the Argentine National Agency for Drugs and Medical Technology (ANMAT). 12 This diagnostic system utilizes tools that provide biosecurity for the operator and offers simple, easy-to-follow instructions for its use. These features allow technology to be implemented in low-income and remote settings.
Herein, we conducted a comparative study of the Orange G3 TBC platform (developed and manufactured in Argentina, Orange Solutions S.R.L.), utilizing GeneXpert Ultra for comparison in the Oruro Department, Bolivia (3700 m above sea level). Orange G3 TBC presents results categorized according to the bacilli load of the sample analyzed by thermocycling and fluorometry, which is classified as detectable when the algorithm fluorescence number is higher than the negative value plus the half-negative value, nondetectable when the algorithm sample number is a negative number, and indeterminate, when the algorithm number is between both.
This study evaluates an innovative diagnostic platform in a TB-endemic setting in South America. By addressing critical barriers such as affordability and accessibility, the Orange G3 TBC platform has the potential to meet the urgent need for advanced diagnostic tools capable of transforming TB control efforts globally.
Materials and methods
Study design and sample collection
The objective of this study was to compare the diagnostic accuracy of the Orange G3 TBC system with the GeneXpert Ultra system in individuals with a presumptive diagnosis of TB. Orange G3 TBC is a diagnostic platform that could be used in resource-limited settings in Bolivia and elsewhere where the GeneXpert Ultra is not widely available. Orange G3 TBC offers results categorized according to the bacilli load of the sample analyzed by thermocycling and fluorometry, which is classified as detectable when the algorithm fluorescence number is higher than the negative value plus the half-negative value, nondetectable when the algorithm sample number is negative, and indeterminate, when the algorithm number is between both. GeneXpert results for the detection of M. tuberculosis are also presented as positive, negative, and indeterminate, 7 which was the object of comparison in this study. The Orange G3 TBC system uses an algorithm validated in previous studies conducted with Orange G3 TBC and the academically agreed-upon gold standard, which is the bacillus culture.9–14 The bacillus load is correlated with the algorithm value of each sample analyzed. All positive samples are analyzed for isoniazid and rifampicin resistance utilizing the same purified DNA. We evaluated clinical specimens from patients having a clinical picture compatible with TB who were seen in March–April 2025 at rural and interurban health centers in Oruro City, Bolivia. The reporting of this study conforms to the STARD statement. 15
We examined 67 sputum specimens and four cerebrospinal fluid samples. The sample size was considered adequate to estimate the diagnostic performance of the test (n = 71). Specifically, it was assumed that a sample including at least 10 diseased cases would allow the sensitivity to be estimated with an acceptable standard error (±10%–15% at 95% CI), assuming an expected sensitivity of 90%. Furthermore, the observed sample yielded a LR+ with a sufficiently narrow confidence interval for exploratory purposes.
All specimens were analyzed using GeneXpert Ultra, Cepheid technology, and by a balanced hemi-nested fluorometric PCR system (Orange G3 TBC) that could preserve worker safety and produce a relatively pure material free of potential inhibitors. The samples were analyzed by smear microscopy and then delivered to the Central TB Laboratory in Oruro City, Bolivia (the only laboratory in Oruro, out of 50, with the capability to perform GeneXpert). The validation parameters between Orange G3 TBC and microscopy are detailed in Garberi et al., 10 which showed a sensitivity of 94.7% for Orange G3 TBC against a sensitivity of 46%–78% for smear detection. Orange G3 TBC DNA purification was performed by liquefying the sputum in a sealed recipient using a three-way stop cock (Figure 1), and then inactivating the sample by submerging the recipient at 88°C for 20 min, which inactivates all bacteria. 11 All samples were analyzed in a randomized, double-blind, and independent manner using the GeneXpert Ultra and Orange G3 TBC systems.

Diagram outlining the steps of the Orange G3 TBC in the diagnosis of TB.
Orange G3 TBC processing
Orange G3 is based on an end-point fluorometric thermal cycler dedicated to the diagnosis of infectious diseases. The system processes samples through a series of steps, including (1) sample liquefaction (for sputum material); (2) biological inactivation; and (3) DNA purification. Concerning genetic targets and primers, the targets used are described in previous studies.10,11 The target of amplification is a segment of IS6110 insertion MTB fragment.
The process occurs in a closed system of syringes, filters, and absorbents, eliminating environmental contact and enhancing biosafety (Figure 2). The final step yields concentrated genetic material for amplification. DNA was isolated by a simplified Boom method 14 and stored at −20°C until amplification. DNA amplification was performed in a TB thermocycler-fluorometer (Figure 3). The double-stranded DNA was fluorometrically detected using custom-designed software and a DNA intercalating fluorochrome. The entire reaction was conducted in a single tube that remained sealed after sample addition to minimize cross-contamination with amplicons.

Flow diagram of the DNA extraction procedure of the Orange G3 TBC.

Thermocycler Orange G3 TBC for amplification of the Mycobacterium tuberculosis genome.
To test the simplicity of our procedure, a team with only 2 days of training performed sample processing and assays. Samples were processed using our improved extraction methodology (Orange G3 TBC biosafety procedure for TB samples, followed by silica-based DNA purification).
Statistical analysis
We performed statistical analysis using McNemar chi-square, Pearson’s correlation, and Kappa measure to evaluate the differences between Orange G3 TBC and GeneXpert Ultra, and determine positive predictive and negative predictive values (PPV and NPV, respectively), percent of error, diagnosis efficiency, specificity, sensitivity, and likelihood ratios (LR+ and LR−).
Results
From the 71 clinical specimens tested, GeneXpert Ultra identified 10 positive and 61 negative samples. The Orange G3 TBC platform identified 11 positive and 60 negative samples (Table 1). Nine positive samples and 59 negative results were concordant between both platforms. Orange G3 TBC showed a strong correlation with GeneXpert Ultra results.
Comparison of results using Orange G3 TBC and GeneXpert Ultra in the diagnosis of active TB (n = 71).

Specifically, diagnostic performance metrics for Orange G3 TBC included a sensitivity of 90%, a specificity of 97%, a diagnostic efficiency of 96%, a NPV of 98%, and a PPV of 82% (Table 2). Statistical analysis revealed strong agreement between the two diagnostic methods. Pearson’s correlation coefficient was calculated at 0.834 (95% CI 0.644–1.000, p < 0.001), indicating substantial agreement between the platforms (Table 2). Additionally, a LR+ = 27.3 (95% CI 6.9–107.9), which is evidence in favor of identifying the disease when the test is positive, and a LR− = 0.103, demonstrate evidence against having the disease when the test is negative.
Clinical validity indexes of Orange G3 TBC in the diagnosis of active TB (n = 71).
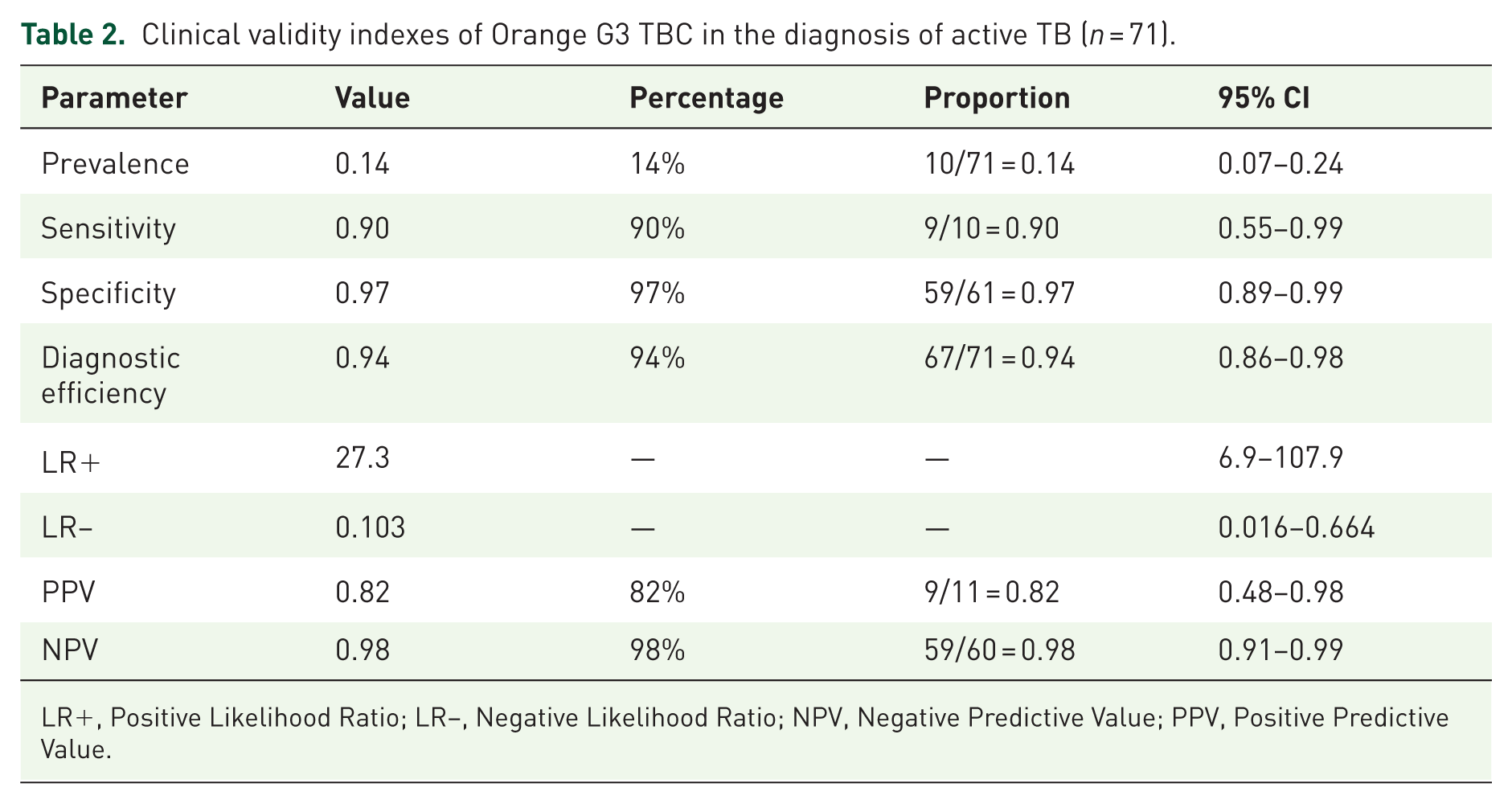
LR+, Positive Likelihood Ratio; LR−, Negative Likelihood Ratio; NPV, Negative Predictive Value; PPV, Positive Predictive Value.
Discussion
TB can be prevented and treated; however, its reemergence in the post-COVID-19 era, along with the concomitant spread of drug-resistant strains, represents a significant global public health concern. To get there, it is crucial to have high-quality, safe, and affordable diagnostic tools to diagnose the many unidentified cases in low-income and resource-constrained settings where TB is highly prevalent.16,17 In the Americas, Peru and Bolivia report the highest burden of TB, with incidence rates of 100 to 299 per 100,000 population per year. 2 Similar to other high-incidence settings, Bolivia faces critical limitations in reducing the burden of disease due to the limited availability of current diagnostic capabilities. Despite the WHO recommendation for upscaling the implementation of molecular diagnosis 15 for TB, only a few laboratories have access to GeneXpert, and most of them require traditional approaches such as smear microscopy, which have low sensitivity and pose risks of operator exposure.
The need for speed is also an important factor when the results of tests positively impact the decision-making related to infection control regarding patient isolation and therapeutic management of patients with active pulmonary TB. In these situations, molecular diagnostic methods can provide the data needed more quickly and offer a significant operational advantage for clinicians. GeneXpert is a rapid, accurate, and low-complexity point-of-care molecular diagnostic testing that the WHO has recommended. 7 Despite the benefits of scaling up the use of GeneXpert in many settings, its widespread use is limited. In Oruro, Bolivia, a province with high rates of TB transmission, only one out of 50 laboratories with the capability to process samples for TB diagnosis has GeneXpert technology. The other 49 laboratories employ smear microscopy.
Several key considerations emerge when evaluating new diagnostic tools for TB, including performance, diagnostic yield, and technical requirements. New diagnostic methods should be at least as effective as existing tools while being suitable for low-resource countries with the highest TB burden. In terms of diagnostic accuracy, while many nucleic acid amplification (NAA) methods show high sensitivity and specificity in smear-positive samples, their performance is typically lower in smear-negative or extrapulmonary specimens, precisely where improved diagnostics would be most valuable. 7 Current commercial molecular diagnostic kits remain expensive for most TB diagnostic laboratories in low-resource countries. Finally, regarding technical requirements, many molecular procedures require sophisticated equipment and highly skilled personnel, typically only available in developed countries or central laboratory facilities in TB-endemic regions. Until these constraints are adequately addressed, expensive commercial NAA techniques will remain restricted to developed nations or academic and research laboratories with appropriate funding, far removed from TB control programs in high-burden settings. Any new method, whether sophisticated or simple, commercial or in-house, should undergo rigorous evaluation through well-designed clinical trials in low-resource settings where implementation would most benefit TB control efforts.
This study demonstrates the potential of a novel molecular diagnostic device to address these challenges and improve TB control efforts. A fundamental methodological difference between GeneXpert and Orange G3 TBC lies in their sample processing approaches. While GeneXpert analyzes a limited aliquot (0.5–2.0 mL), the Orange G3 TBC platform processes the available sample volume, followed by DNA concentration and purification. This approach theoretically enhances detection sensitivity, potentially explaining discordant results where Orange G3 TBC detected TB in two samples that tested negative with GeneXpert Ultra. As shown in Table 2, Orange G3 TBC results correlate well with GeneXpert Ultra results. Therefore, Orange G3 TBC could be a valuable tool to increase the detection efficiency of TB, particularly in samples with low bacillary loads. Furthermore, this diagnostic platform is user-friendly, increasing the feasibility of its implementation in resource-limited regions, as mentioned in the study by Ssengooba et al. 18
Due to its integrated biosafety design, the Orange G3 TBC system allows complete sample processing without requiring a laminar flow hood. This represents a significant advantage for operator safety, minimizing exposure risk during handling potentially infectious materials. Implementing such devices in low-resource settings could facilitate access to sensitive molecular diagnostics without compromising laboratory personnel’s safety. The order of addition of the reagents plus the exposure of the recipient with the sample to a temperature above 70°C for 15 min guarantees that in the subsequent steps of the DNA extraction procedure, the bacilli are completely inactivated and unable to infect the operator. 13
The diagnostic results obtained in this study are highly similar between GeneXpert Ultra and Orange G3 TBC. A key advantage of the Orange G3 TBC platform is that health operators can learn to process and analyze samples after minimal training. The system requires simple installation conditions, and its biosecurity container represents a significant differentiation from other available methods. TB healthcare workers commonly acknowledge GeneXpert as the only molecular TB diagnostic platform available. Further comparative studies in Latin America and worldwide could raise awareness about alternative options for addressing the global challenge of TB diagnosis. This project was conducted with minimal resources, highlighting the need for increased governmental and institutional support for regional health technology developments.
Conclusion
The objective of this study was to compare the diagnostic accuracy of the Orange G3 TBC system with the GeneXpert Ultra system in individuals with a presumptive diagnosis of TB. It was demonstrated that Orange G3 TBC gets statistically similar results to the ones obtained by GeneXpert Ultra, which was the objective of the diagnostic clinical research.
It is emphasized that the Orange G3 TBC platform demonstrates performance characteristics comparable to the WHO-endorsed GeneXpert Ultra system for TB diagnosis, with biosafety and accessibility in South America, and potentially enhanced sensitivity through whole-sample processing. With minimal training requirements and adaptation to low-resource settings, the use of this system implies the understanding of the steps to be performed, which is acquired during the operator’s training and therefore implies a better performance of the diagnostic process. This Latin American-developed technology offers a viable alternative for expanding molecular TB diagnostics to underserved regions with the highest disease burden.
There are significant limitations to our study, including the testing performed in only one laboratory in one country. The Orange G3 TBC platform for diagnosing TB does not offer detection of rifampicin resistance in one step compared to GeneXpert Ultra.
