Abstract
Background:
Methicillin-susceptible Staphylococcus aureus (MSSA) is a leading cause of infective endocarditis (IE), associated with high morbidity and mortality. While antistaphylococcal penicillins (ASPs) are considered the standard treatment, cefazolin has emerged as a potential alternative due to its pharmacokinetic advantages and lower toxicity profile.
Objectives:
To assess the efficacy and safety of cefazolin compared with ASPs in adult patients with MSSA infective endocarditis.
Design:
Systematic review and meta-analysis.
Data sources and methods:
We systematically searched MEDLINE, Embase, CENTRAL, Scopus, and Web of Science up to 2024. We included cohort studies and non-randomized trials comparing cefazolin and ASPs in adult patients with MSSA IE. Risk of bias was evaluated using the ROBINS-I tool. Meta-analyses were performed using random-effects models to estimate pooled odds ratios (ORs) and standardized mean differences (SMDs).
Results:
Seven cohort studies involving 1685 patients (305 cefazolin; 1380 ASPs) were included. Thirty-day mortality, reported in two studies (n = 1083), was lower with cefazolin (OR 0.49; 95% CI 0.29–0.83; I² = 0%). Ninety-day mortality (three studies, n = 475) showed no significant difference (OR 1.00; 95% CI 0.63–1.57; I² = 0%). No significant differences were observed in relapse rates (OR 1.01; 95% CI 0.34–3.02; I² = 12.5%), hospital stay duration (SMD –0.06; 95% CI –0.27 to 0.14), or bacteremia duration (SMD –0.96; 95% CI –1.93 to 0.01). Safety data suggested a lower incidence of adverse events with cefazolin, although definitions varied across studies. Risk of bias was moderate in most studies.
Conclusion:
Cefazolin demonstrated comparable efficacy to ASPs for the treatment of MSSA IE, with a potential reduction in short-term mortality and a favorable safety profile. These findings support cefazolin as a viable therapeutic alternative, but randomized controlled trials are needed to confirm its effectiveness and safety.
Trial registration:
PROSPERO registration number: CRD42024593515.
Plain language summary
Staphylococcus aureus is a common bacteria that can cause a serious infection of the heart called infective endocarditis. When the bacteria are not resistant to antibiotics (a condition called methicillin-susceptible Staphylococcus aureus, or MSSA), two types of antibiotics are usually considered for treatment: cefazolin (a type of cephalosporin) and antistaphylococcal penicillins (such as nafcillin or cloxacillin). There has been ongoing debate about which option is more effective and safer.
We conducted a systematic review of published studies to compare cefazolin and antistaphylococcal penicillins in patients with MSSA infective endocarditis. All the studies we found were observational, meaning they were not randomized clinical trials. Our findings suggest that cefazolin may be as effective as, or potentially better tolerated than, antistaphylococcal penicillins. However, because the included studies were not randomized and had some risk of bias, our confidence in the results is moderate to low. More high-quality research is needed to confirm these findings and guide clinical decisions.
Introduction
The annual incidence of infective endocarditis (IE) has shown a significant increase, driven by the rising number of nosocomial infections, broader use of implantable cardiac devices, and intravenous drug use.1,2 Despite advances in medical care, in-hospital mortality remains high, with estimates ranging from 20% to 30% at 6 months. 3 These patients often require prolonged hospitalizations, as the standard course of intravenous antimicrobial therapy extends up to 43 days on average. 4 Treatment complexity further contributes to a readmission rate of approximately 12%, primarily due to recurrent bacteremia. 5
Staphylococcus aureus is one of the most clinically significant pathogens in IE due to both its high prevalence and its association with severe complications.6,7 Once S. aureus is identified as the causative organism, determining its antibiotic susceptibility is essential to guide targeted therapy. Cohort studies have reported that over 75% of S. aureus isolates in IE are methicillin-susceptible.6,7
For patients with methicillin-susceptible S. aureus (MSSA) endocarditis, antistaphylococcal penicillins (ASPs) are considered the first-line treatment.6,7 However, due to their short half-life, it needs administration four to six times daily, increasing catheter handling and the risk of adverse events. 8 Consequently, cefazolin has been proposed as an alternative. Cefazolin exhibits potent in vitro activity against S. aureus, 9 with more favorable pharmacokinetic properties, including a longer half-life that allows for three daily doses. Additional benefits include lower sodium content, feasibility of postdialysis administration, and reduced cost. 10 In contrast, ASPs have been associated with higher rates of adverse events, including interstitial nephritis. 11
Two meta-analyses have demonstrated that cefazolin is noninferior to ASPs in terms of mortality and clinical failure for MSSA bacteremia.12,13 Accordingly, both European and American clinical guidelines include cefazolin as an alternative for MSSA endocarditis, particularly in patients with penicillin allergy.6,7 Nonetheless, concerns persist regarding the potential for higher treatment failure with cefazolin due to the in vitro “inoculum effect”—a phenomenon whose clinical significance in IE remains uncertain.9,14
The objective of this review is to systematically assess the existing literature to evaluate the efficacy and safety of cefazolin compared to ASPs in adult patients with MSSA endocarditis, in terms of all-cause mortality at 30 and 90 days. At the time of conducting this study, no systematic reviews were identified that specifically addressed this clinical question in the scenario of endocarditis, it has been addressed only in bacteremia. This review aims to provide clinicians with evidence-based insights to support clinical decision-making and contribute to the optimization of therapeutic strategies in the management of this serious infection.
Methods
Design and registration
This systematic review was conducted in accordance with the Cochrane Collaboration guidelines for systematic reviews of interventions and adhered to the 2020 “Preferred Reporting Items for Systematic Reviews and Meta-Analyses” (PRISMA) checklist, a detailed checklist documenting adherence to each item of the guideline is provided in Supplemental Table 3. 15 The study protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO) under the identifier CRD42024593515.
Eligibility criteria
Study design
We included cohort studies, case–control studies, quasi-experimental studies, nonrandomized controlled trials, and randomized controlled trials.
Study population
Eligible studies included male or female participants aged ⩾16 years with a diagnosis of native or prosthetic valve endocarditis (defined according to Duke criteria or their modifications), with isolation of MSSA from blood cultures or cardiac valve specimens. Patients had to have received definitive antibiotic therapy with either cefazolin or an ASP (oxacillin, nafcillin, dicloxacillin, flucloxacillin, or methicillin), regardless of dose, frequency, or duration. Studies involving patients treated with these antibiotics for indications other than infective endocarditis or those with polymicrobial infections were excluded.
Outcomes
Studies were required to report at least one of the following outcomes:
Primary outcomes: (1) All-cause mortality at 30 and 90 days; (2) Serious adverse events, as defined by the World Health Organization, new embolic events during treatment, and duration of bacteremia.
Secondary outcomes: (1) Relapse, defined as recurrence of infection within 2 weeks after completing treatment with the same microorganism and identical antimicrobial susceptibility profile; (2) all-cause mortality at 1 year; (3) length of hospital stay; (4) nonserious adverse events.
Search strategy
A comprehensive electronic search was performed through November 2024 in the following databases: Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, Embase, and Scopus. No language restrictions were applied. The specific search strategies used for each database are detailed in Supplemental Table 1.
Study selection
Study selection was performed in two phases by two independent reviewers. In the first phase, titles and abstracts were screened, followed by full-text evaluation. Disagreements were resolved by a third reviewer.
Data analysis
Data extraction was performed independently by two reviewers using a standardized Excel® database. Extracted variables included eligibility criteria, intervention, and comparator characteristics (type, dose, frequency), demographic and clinical features (age, sex, site of infection, immunosuppression, heart failure, vegetations), complications (shock, intracardiac abscess, fistulas, valvular perforation, aneurysms, or pseudoaneurysms), and effect estimates for all reported outcomes.
Risk of bias assessment
Risk of bias was assessed independently by two reviewers using the Cochrane ROBINS-I tool for nonrandomized studies. 16 Discrepancies were resolved by consensus with a third reviewer, ensuring consistency and minimizing selection and assessment bias.
Statistical analysis
For dichotomous outcomes (e.g., 30- and 90-day mortality), a meta-analysis using a random-effects model was conducted to estimate the intervention effect compared to the comparator. Binary event data (event counts and sample sizes for each group) were analyzed using the metabin function from the “meta” package in R, applying the Mantel–Haenszel method. 17 A random-effects model was chosen due to expected clinical and methodological heterogeneity across studies.
For continuous outcomes (e.g., duration of bacteremia, hospital stay), random-effects meta-analyses were conducted using standardized mean differences (SMDs). Inputs included means, standard deviations, and sample sizes for each group. SMDs and their variances were calculated using the “metafor” package in R. 18 A restricted maximum likelihood (REML) method was applied to fit the random-effects model and account for between-study variability.
Heterogeneity was assessed using statistical metrics including Tau², I², and Chi² tests. An I² > 40% or p-value < 0.10 was considered indicative of significant heterogeneity. All analyses were performed using RStudio version 4.4.1. 19 For missing data, study authors were contacted directly.
Sensitivity analysis
A subgroup analysis was conducted to explore the robustness of the findings among studies exclusively enrolling patients with a confirmed diagnosis of MSSA IE.
Results
Search process
A total of 5323 records were identified through the database search. In addition to peer-reviewed databases, we searched gray literature sources including medRxiv and ClinicalTrials.gov to identify unpublished or ongoing studies. No additional eligible records were found. After removing 2588 duplicates, 2735 titles and abstracts were screened. Of these, 27 articles were selected for full-text review. Seven were excluded due to lack of full-text availability (i.e., conference abstracts or electronic posters), two were excluded based on study design, six did not include the target population, two lacked the appropriate comparator, and two did not report relevant outcomes. Ultimately, seven observational cohort studies were included in this systematic review. The selection process is illustrated in the PRISMA flow diagram (Figure 1).13,20–25 Study characteristics are summarized in Table 1, and reasons for exclusion are detailed in Supplemental Table 2.
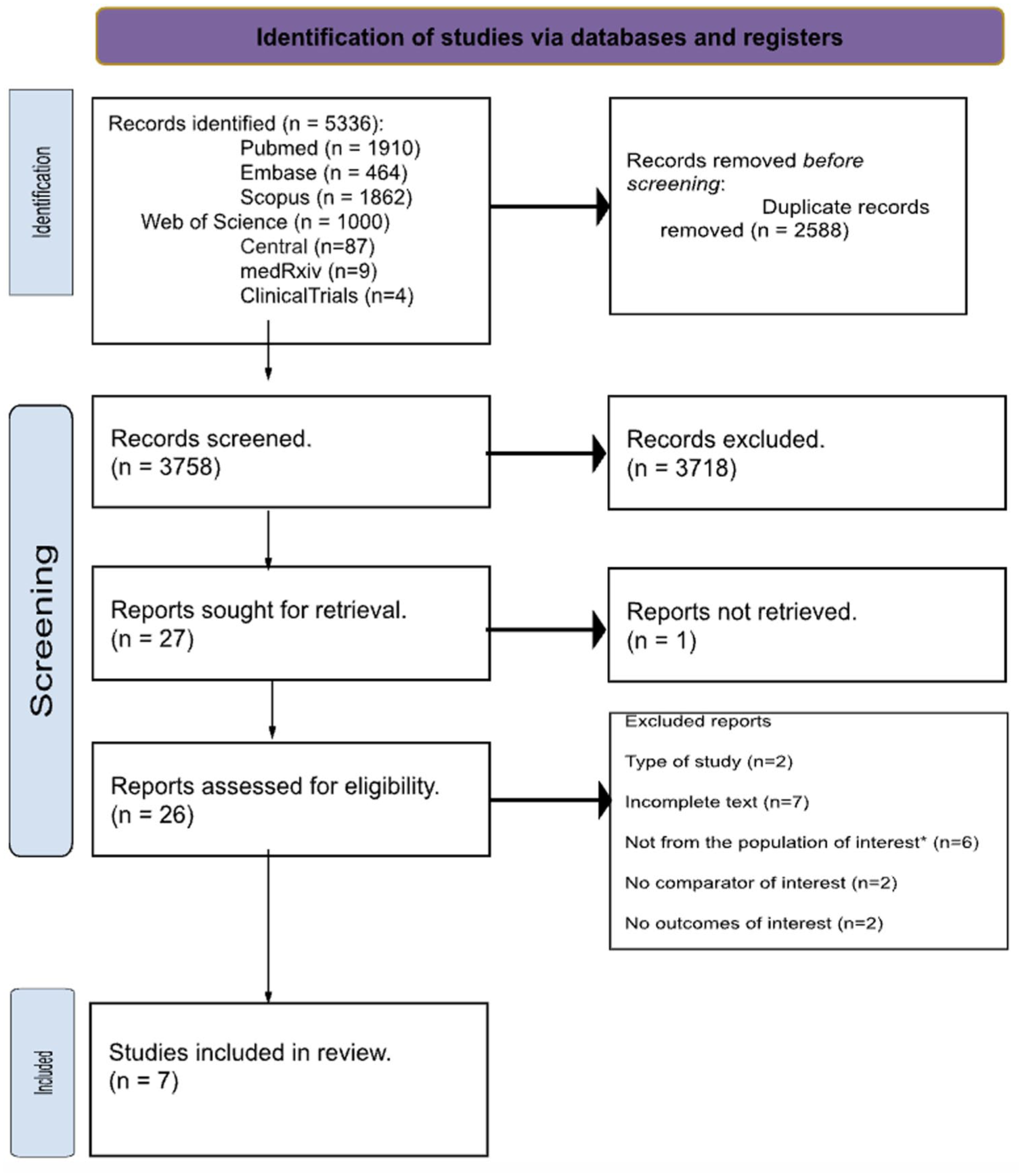
PRISMA flow diagram.
Characteristics of included studies.

Defined as death from any cause during hospitalization or within 1 month after discharge.
Defined as positive blood cultures for the same microorganism within 6 months of the initial episode.
Defined as an increase in serum creatinine ⩾1.5× baseline or ⩾0.3 mg/dL absolute increase.
AST or ALT ⩾3× upper limit of normal (ULN) if baseline normal, or ⩾1.5× ULN if baseline abnormal.
Persistent MSSA-positive blood cultures >7 days after starting beta-lactam therapy.
Increase in serum creatinine ⩾0.3 mg/dL within 48 h or ⩾50% at any time from baseline.
ALT, Alanine aminotransferase; AST, Aspartate aminotransferase; IE, infective endocarditis; MSSA, Methicillin-susceptible Staphylococcus aureus; ULN, upper limit of normal.
Included studies
Seven cohort studies were included, encompassing a total of 1685 participants—305 in the intervention group (cefazolin) and 1380 in the comparator group (ASPs). Of the seven studies, four included only patients with a confirmed diagnosis of infective endocarditis,20–22,25 while the remaining three focused on patients with MSSA bacteremia, within which a subgroup had a diagnosis of endocarditis.23,24,26
Patient allocation to treatment groups (cefazolin vs ASPs) was generally based on the timing or predominance of antimicrobial therapy. For example, Herrera-Hidalgo et al. assigned patients based on the antibiotic received for ⩾75% of treatment duration, 21 while Destrem et al. included those who received at least ten consecutive days of either agent. 22 Flynt et al. classified patients who received ⩾72 h of cefazolin or nafcillin therapy. 24 Davis et al. 24 and Lee et al. 26 included only those who received the respective antibiotic as definitive treatment, postsusceptibility confirmation. Lefèvre et al. 20 excluded patients who received both agents to avoid treatment overlap, while Lecomte et al. 25 included patients who received at least one dose of the assigned antibiotic and excluded those treated with both.
For further details, see Table 1.
Risk of bias
None of the included studies were rated as having an overall low risk of bias. All were assessed as having a moderate risk of bias. At the domain level, the most common concerns were related to confounding and missing data. Detailed results of the risk of bias assessment are presented in Supplemental Table 3 and illustrated in Figures 2 and 3.
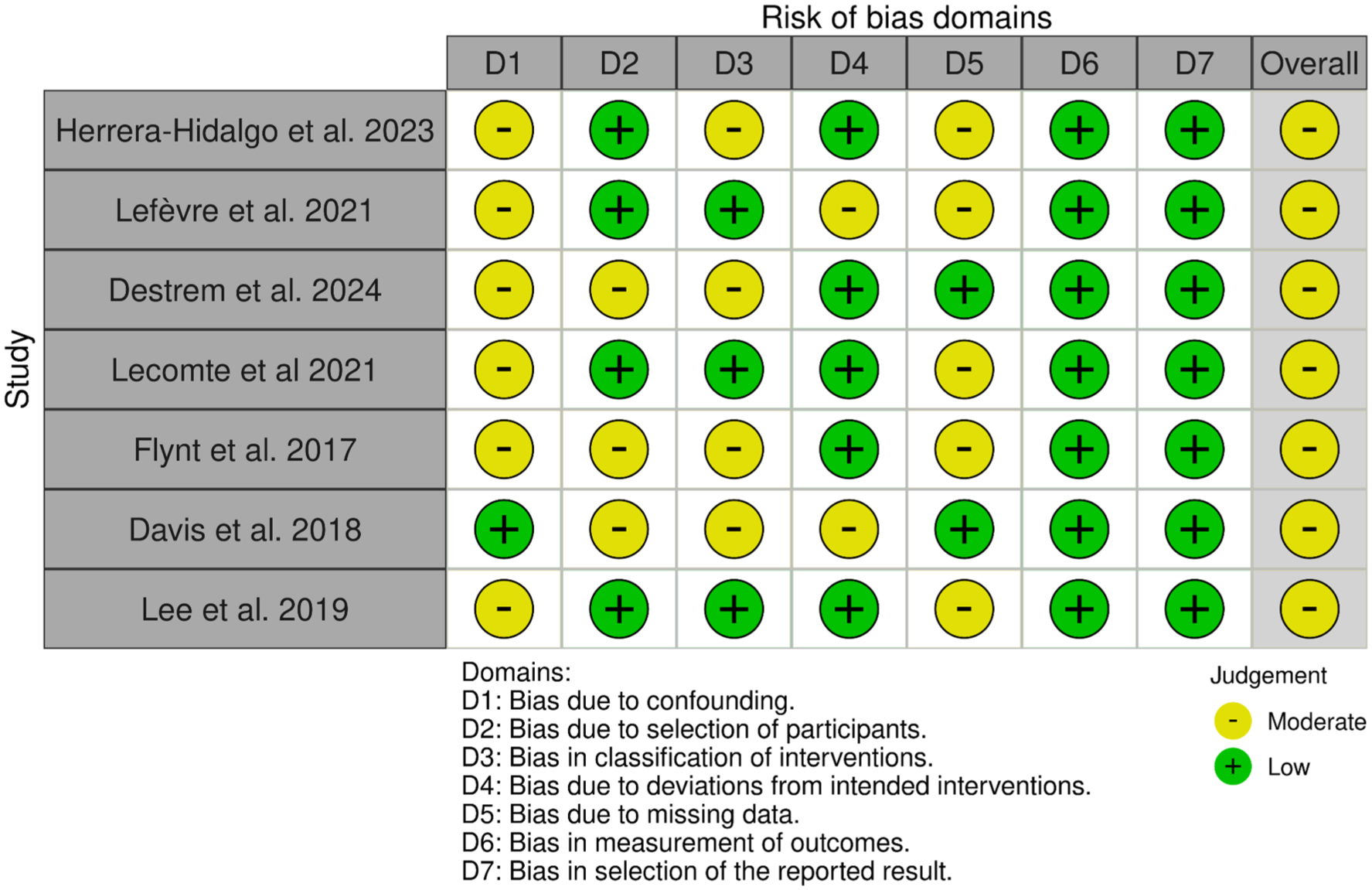
Summary of the risk of bias according to the domains of ROBINS-I.

Overall assessment of the risk of bias of the included studies.
Primary outcomes
30-Day mortality
This outcome was evaluated in two of the included studies.19,21 A meta-analysis using a random-effects Mantel–Haenszel model demonstrated a significant reduction in 30-day mortality among patients treated with cefazolin compared to those receiving ASPs (n = 1083; OR 0.49; 95% CI 0.29–0.83; I² = 0.0%; p = 0.85; Figure 4).

(a) Pooled 30-day mortality and (b) 90-day mortality.
90-Day mortality
This outcome was assessed in three of the included studies.20,22,25 A meta-analysis using the Mantel–Haenszel random-effects model revealed no statistically significant difference between groups (475 patients; OR 1.00; 95% CI 0.63–1.57; I² = 0.0%; p = 0.39) (Figure 4). To explore the robustness of these findings, we performed a sensitivity analysis restricted to studies exclusively enrolling patients with a confirmed diagnosis of MSSA IE. Pooled estimates were recalculated using the same random-effects model (192 patients; OR, 0.76; 95% CI, 0.42–1.38; I² = 0.0%; p = 0.76), again showing no statistically significant difference between treatment groups (Supplemental Figure 1).
Serious adverse events
No serious adverse events were reported in any of the studies included in this review.
Secondary outcomes
Relapse of endocarditis
This outcome was assessed in five of the included studies.20–22,24,25 Due to variability in the operational definitions of relapse, a meta-analysis was not performed. Herrera-Hidalgo et al. defined relapse as repeat positive blood cultures with the same microorganism as the initial episode, reporting relapse rates of 3.6% in the cloxacillin group and 2.6% in the cefazolin group, with no statistically significant difference (p = 0.782). 21 Lefevre et al., 20 without providing a specific definition, reported a 5.3% relapse rate in the cefazolin group and none in the ASP group (p = 0.494). Destrem et al. defined relapse as a new diagnosis of endocarditis within 90 days of completing antimicrobial therapy caused by the same microorganism, with rates of 4.3% in the cloxacillin group and 1% in the cefazolin group, also without statistical significance (p = 0.396). 22 Lecomte et al. defined relapse as a new episode of infective endocarditis by the same microorganism within 1 year of follow-up, reporting a relapse rate of 2.7% in the ASP group and none in the cefazolin group (p = 0.574). 25 Lastly, Flynt et al. assessed relapse as part of a composite outcome including all-cause mortality at 30 days, MSSA recurrence within 60 days postdischarge, or microbiological failure, reporting relapse rates of 36.4% in the cefazolin group and 18.2% in the ASP group. 24 Overall, no consistent differences in endocarditis relapse rates were observed between cefazolin and ASP-treated groups. However, the variability in definitions and evaluation criteria limits cross-study comparability. Definitions are summarized in Supplemental Table 4.
Length of hospital stay
This outcome was reported in two studies.19,23 Herrera-Hidalgo et al. reported similar lengths of stay between treatment groups: 36.5 days in the cefazolin group and 37.7 days in the cloxacillin group. 21 Lecomte et al. 25 observed a mean hospital stay of 34 days in the cefazolin group and 36 days in the oxacillin or cloxacillin group. A random-effects meta-analysis revealed a pooled standardized mean difference (SMD) of –0.06 days (95% CI: –0.27 to 0.14), indicating no statistically significant difference in hospital stay duration between cefazolin and ASP groups (Figure 5).

(a) Pooled length of hospital stay and (b) pooled duration of bacteremia.
Duration of bacteremia
This outcome was reported in two studies.22,25 A random-effects meta-analysis was performed, yielding a pooled standardized mean difference (SMD) of –0.96 days (95% CI: –1.93 to 0.01) when comparing patients treated with cefazolin versus those treated with ASPs. This difference was not statistically significant (Figure 5).
Nonserious adverse events
Five studies evaluated this outcome, four of which reported a lower incidence of adverse events among patients treated with cefazolin. In the study by Herrera-Hidalgo et al., nonserious adverse events occurred in 1.75% (1/57) of patients receiving cefazolin compared to 3.72% (20/537) in the ASPs group. Reported events included severe hypokalemia, hematologic toxicity, phlebitis, vasculitis, and other complications. 21 In the study by Destrem et al., adverse events were observed in 24.5% (24/98) of patients in the cefazolin group versus 34% (32/94) in the ASP group, primarily related to acute kidney injury and elevated liver transaminases, with no statistically significant differences between groups. 22 Lecomte et al. 25 reported no adverse events in the cefazolin group, while the ASP group experienced an 8.3% (13/157) incidence, including acute kidney injury, rash, cytopenias, and gastrointestinal disturbances. In Flynt et al., 24 acute kidney injury occurred in 27.3% (3/11) of cefazolin-treated patients versus 50% (11/22) in the ASP group. In contrast, Lefèvre et al. reported a higher rate of adverse events in the cefazolin group—68.4% (26/38) versus 51.4% (18/35) in the ASP group—mostly associated with acute kidney injury, hepatocellular injury, and elevated bilirubin levels. 20 Definitions of adverse events are summarized in Supplemental Table 4.
Discussion
The objective of this systematic review was to evaluate the efficacy and safety of cefazolin compared to ASPs in adult patients with MSSA infective endocarditis. Seven cohort studies were included in the analysis. The findings suggest a significant reduction in 30-day mortality with cefazolin; however, this difference did not persist at 90 days. No statistically significant differences were observed in relapse rates, hospital length of stay, or duration of bacteremia. In terms of safety, cefazolin showed a slightly more favorable profile, with a trend toward fewer adverse events, although the differences were not statistically significant.
Our results indicate that cefazolin may reduce 30-day mortality compared to ASPs, this was further supported by a sensitivity analysis restricted to studies exclusively enrolling patients with confirmed IE, in which 90-day mortality remained similar between treatment groups, consistent with findings from Herrera-Hidalgo et al. 21 and Davis et al. 23 The discrepancy between short-term and longer-term mortality may reflect differences in early drug tolerability and confounding by indication could have influenced treatment decisions—whereby more severely ill patients may have been more likely to receive ASPs—thus affecting early outcomes. The attenuation of mortality benefit at 90 days may be due to the influence of underlying comorbidities or complications of endocarditis that become more prominent over time. This temporal trend has also been observed in systematic reviews of MSSA bacteremia, that reported a statistically significant reduction in overall mortality with cefazolin (OR 0.69; 95% CI 0.58–0.82), 25 and a reduction in 30-day mortality (RR 0.7; 95% CI 0.54–0.91), with no significant differences in 90-day mortality (RR 0.7; 95% CI 0.5–1.02). 8 These findings support the potential efficacy of cefazolin in reducing early mortality in MSSA endocarditis, though further research is needed to assess its long-term impact.
The use of cefazolin in high-inoculum infections like endocarditis has historically been limited due to its increased susceptibility to hydrolysis by type A beta-lactamases, which may elevate the minimum inhibitory concentration—known as the “inoculum effect”.9,14 A French case series involving 216 patients reported the inoculum effect in 19% of cefazolin-treated and 38% of oxacillin-treated strains. 27 The presence of the inoculum effect was an independent predictor of 30-day mortality (HR 2.84; 95% CI 1.28–6.30; p = 0.01), with significantly higher mortality observed in cefazolin-treated patients with the inoculum effect (p = 0.01), though not in those treated with oxacillin (p = 0.06). However, no overall mortality difference was found between treatments. Another French study reported a 17.6% prevalence of the inoculum effect without associations with persistent bacteremia, treatment failure, or 90-day mortality. 14 Furthermore, a systematic review of observational studies found no significant impact of the inoculum effect on cefazolin-related mortality or treatment failure in severe MSSA infections. 9
Clinical features associated with high bacterial burden in infective endocarditis include perivalvular abscesses, persistent bacteremia, septic emboli, and rapid decompensation due to heart failure. 28 In this context, the study by Herrera et al.—the one with the greatest weight for 30-day mortality in our review—included patients with such characteristics, yet found no significant difference in mortality between cefazolin and ASP-treated groups. 21 This may indirectly suggest that clinical factors associated with the inoculum effect do not significantly impact outcomes. Therefore, despite its presence in a substantial proportion of cases, current clinical evidence does not support a relevant negative impact of the inoculum effect on cefazolin’s efficacy or associated mortality in MSSA endocarditis.
Guidelines from the European Society of Cardiology (ESC) and the American Heart Association (AHA) recommend cefazolin as an alternative for patients with allergies to ASPs, such as cloxacillin, in the treatment of MSSA endocarditis.7,14 The results of the studies included in this review suggest that cefazolin has comparable efficacy to ASPs, with a potential mortality benefit and a lower incidence of adverse events. However, variability in adverse event definitions and reporting precluded a robust quantitative safety analysis, limiting the strength of conclusions. Further evidence and studies with standardized adverse event definitions are needed to confirm cefazolin’s safety profile in this setting.
The recurrence rate of MSSA endocarditis appears to be around 3%, based on cohort data from Spain. 29 In this review, no significant differences in relapse rates were observed between patients treated with cefazolin and those receiving ASPs, though inconsistencies in operational definitions of relapse limit interpretability. Hospital stays in patients with infective endocarditis are often prolonged due to the need for intravenous antibiotic therapy. A Danish study reported a median duration of 43 days for complete IV therapy and 24 days for partial oral therapy, while a French cohort found an average of 43 days, extending to 47 days in surgical patients and 38 in nonsurgical patients.4,30 In our analysis, hospital stay and bacteremia duration were similar across treatment groups, suggesting that cefazolin as a first-line option does not negatively affect these outcomes.
Although a meta-analysis could not be performed due to heterogeneity in reporting, the absolute incidence of nonserious adverse events suggests that cefazolin may offer a slightly better safety profile than ASPs. These findings are consistent with prior reviews. One systematic review and meta-analysis found significantly lower risks of nephrotoxicity, acute interstitial nephritis, hepatotoxicity, and treatment discontinuation due to adverse events with cefazolin compared to ASPs. 11 Likewise, Liu et al. found lower risks of nephrotoxicity, hepatotoxicity, and fewer treatment interruptions in patients with MSSA bacteremia treated with cefazolin. 31 However, in the studies by Herrera et al. and Lecomte et al., patients with more severe illness (e.g., ICU admission, septic shock) were more commonly assigned to the ASP group, potentially increasing the likelihood of organ dysfunction.21,25 Conversely, in the study by Lefevre et al., the cefazolin group included the more severely ill patients. 20 Thus, current evidence is insufficient to draw definitive conclusions about the comparative safety of these agents, and randomized prospective trials with standardized definitions are needed to confirm these findings.
This review possesses several methodological strengths, including a systematic and transparent study selection process, a rigorous risk of bias assessment, and the comprehensive inclusion of clinically relevant variables and outcomes. However, important limitations must be considered when interpreting the findings and their potential implications for clinical practice.
Although statistical heterogeneity was assessed, clinical heterogeneity related to differences in patient characteristics, severity of disease, and antibiotic dosing strategies could not be explored in depth due to insufficient reporting in the included studies. For example, the study by Destrem et al. did not specify whether patients were admitted to intensive care units, nor did it report antibiotic dosing or treatment protocols. These limitations may affect the interpretation and generalizability of our findings.
Second, all included studies were judged to have a moderate risk of bias, primarily due to residual confounding and incomplete outcome reporting. As no study met criteria for either low or high risk of bias, we were unable to perform a sensitivity analysis based on study quality. This limitation constrains our ability to explore the influence of methodological rigor on the robustness of the pooled estimates and reduces confidence in the observed associations. Furthermore, the review relied exclusively on observational studies, which inherently carry a risk of confounding, particularly treatment indication bias—where patient characteristics influencing therapeutic decisions may also independently affect outcomes—and this limits causal inference and generalizability.
Third, there was notable heterogeneity in the definition and reporting of key clinical outcomes, such as relapse rates and adverse events. These inconsistencies restricted the feasibility of meta-analysis for several safety endpoints and may lead to under- or overestimation of treatment-related risks. Moreover, some of the evaluated outcomes—including 30-day mortality, hospital length of stay, and duration of bacteremia—were based on small sample sizes and low event rates, thereby limiting the statistical power to detect meaningful differences and increasing the risk of imprecise estimates.
An additional limitation of our review is that we did not systematically assess the funding sources of the included studies, which may have introduced an unmeasured risk of bias. Finally, due to the limited number of included studies (n = 7), a formal assessment of publication bias (e.g., funnel plot asymmetry) was not performed, as current methodological standards discourage such analyses when fewer than 10 studies are available.
As a result, the overall confidence in the findings should be considered moderate to low. Collectively these limitations underscore the need for well-designed randomized controlled trials to more definitively inform clinical decision-making. Until such evidence becomes available, the findings of this review should be interpreted with caution, particularly when applied to patient populations or clinical settings that differ from those represented in the included studies.
Conclusion
The findings of this systematic review suggest that cefazolin demonstrates comparable efficacy to ASPs in the treatment of MSSA infective endocarditis, with similar rates of mortality, relapse, duration of bacteremia, and length of hospital stay. Cefazolin also appears to be associated with fewer adverse events; however, heterogeneity in adverse event definitions and reporting limits the accuracy of this comparison. The inclusion of observational studies with moderate-to-high risk of bias—particularly in domains of confounding and missing data—highlights the need for well-designed randomized controlled trials to confirm these findings and guide clinical decision-making.
Supplemental Material
sj-docx-1-tai-10.1177_20499361251384146 – Supplemental material for Comparative effectiveness of cefazolin versus antistaphylococcal penicillins in methicillin-susceptible Staphylococcus aureus infective endocarditis: a systematic review of observational studies
Supplemental material, sj-docx-1-tai-10.1177_20499361251384146 for Comparative effectiveness of cefazolin versus antistaphylococcal penicillins in methicillin-susceptible Staphylococcus aureus infective endocarditis: a systematic review of observational studies by Laura Juliana Jaime-Ardila, Jorge Leonardo Támara-Rivera, Laura Cristina Nocua-Báez, Candida Díaz-Brochero and Sugeich Del Mar Melendez-Rhenals in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
