Abstract
Background:
Whole body 18F-fluorodeoxyglucose positron emission tomography/CT (WBP) may be an important tool for the management of infective endocarditis (IE) by identifying areas of occult primary or metastatic infection. However, the optimal use of this study in patients with IE is unknown.
Objectives:
Compare clinical characteristics and outcomes in patients who did and did not have WBP as part of their endocarditis management, and describe the impact that WBP has on the management of IE.
Design:
Retrospective cohort study.
Methods:
We performed a protocolized chart review of hospitalized patients with suspected IE who were discussed by a multidisciplinary endocarditis team at a tertiary care center between June 2018 and January 2022.
Results:
Among 427 patients, there were 114 patients (26.7%) in the WBP group and 313 patients (73.3%) in the non-WBP group. The WBP group was significantly more likely to have end-stage renal disease, intracardiac prostheses, and cardiac devices, while the non-WBP group was more likely to have flail leaflet or paravalvular abscesses. There were no statistically significant differences in mortality, hospital readmission, or length of stay between the two cohorts. The WBP group was more likely to receive longer antibiotic courses and had higher rates of suppressive antibiotics following treatment courses (p < 0.001). The use of WBP directly affected management in 44.6% of those patients, especially when performed to evaluate intravascular prostheses and grafts. Changes in management included further workup, performance of a source control procedure, or a change in the antibiotic regimen.
Conclusion:
WBP plays an important role in identifying metastatic foci of infection and directly impacting the management of patients with confirmed or suspected endocarditis. Infected intravascular prostheses were effectively identified via WBP, and as a result, these patients were prescribed longer courses of antibiotics and suppressive antibiotics.
Keywords
Introduction
Infective endocarditis (IE) is a disease characterized by infection of endocardial tissue or endocardial prosthetic materials and devices. In addition, peripheral foci of infection such as septic emboli, mycotic aneurysm, and metastatic infection to other organs often occur. 1 Despite multidisciplinary advances, IE global incidence and mortality have increased over the last 30 years, with one-year mortality as high as 30%.2–4 This trend is present in many high-income countries, including the United States, where there has been a sharp increase in mortality in the last 3–5 years. 3 Increasing population age, use of prosthetic cardiac devices, and intravenous drug use are thought to be factors contributing to these trends.2,3
The use of 18F-fluorodeoxyglucose (18F-FDG) positron emission tomography (PET) has emerged as an important imaging modality for the diagnosis and management of IE and its complications over the last 20 years. 5 18F-FDG is a glucose analog that is taken up in metabolically active inflammatory cells, which express high levels of the glucose transporters and thus detect areas of active inflammation, which could indicate the presence of active infection in tissues. 6 The use of cardiac PET for the diagnosis of prosthetic valve and cardiac implantable electronic device-related endocarditis is supported by a large body of literature and is part of diagnostic recommendations of IE by the 2023 European Society of Cardiology and the Duke-International Society for Cardiovascular Infectious Diseases.7–9
On the contrary, there is less literature that has investigated the use of whole body positron emission tomography (PET) scan (WBP) to detect extracardiac infectious foci or sources of infection associated with endocarditis.10–12 WBP is promising for this purpose because IE-related metastatic foci are typically detected through multiple imaging modalities such as magnetic resonance imaging (MRI), computed tomography scan (CT), and ultrasound of various anatomic sites, which may require several days to complete or be limited by the presence of hardware in the case of MRI. 13 In contrast, WBP can provide total body imaging with a single test much more rapidly, with the exception of the brain, where high basal rates of glucose uptake lower the accuracy of PET imaging. 14 In addition, WBP has demonstrated an advantage in the detection of asymptomatic occult infection. A prospective cohort study in 2014 showed that WBP was the only initially positive test for peripheral infectious foci in 55.5% of patients, and led to significantly more diagnoses of extracardiac complications than those who did not get WBP. 15 In other studies, WBP detected infectious foci in 24%–28% of cases where there was no prior clinical suspicion, further supporting the value of this study.16,17 A French prospective cohort study demonstrated that WBP played a significant role in the diagnostic criteria of IE in 15% of patients and directly impacted management in 26.4% of patients. 18 Nevertheless, there are few studies that assess comparative outcomes between the patients who did and did not get WBP as part of their IE management.
Based on the literature that is now available, WBP is emerging as an important modality for the diagnostic criteria of endocarditis.8,9 However, WBP is a time and labor-intensive study. Because 18F-FDG normally accumulates in myocardial tissue, a pre-study preparation consisting of a high-fat, low-carbohydrate ketogenic diet typically starting 24 h prior to the study, followed by a 12-h fast, is required to allow for the acquisition of the best cardiac images. 6 With increasing use and growing interest in WBP for the management of IE, further research is needed to understand which patients will benefit the most from the use of WBP and its impact on outcomes such as mortality and other aspects of clinical management.
This study seeks to compare patient characteristics, IE disease characteristics, and clinical outcomes between patients who did and did not receive WBP as part of their management of IE. The goal is to identify groups of patients with possible or definite endocarditis who will benefit the most from having this study performed. For the group of patients who underwent WBP, we provide a descriptive summary of indications, findings, and resultant management changes related to WBP to enhance understanding of the role of WBP in IE management.
Methods
Study design
This is a retrospective cohort study comparing patients with suspected infectious endocarditis who had WBP performed as part of their management and those who did not. Study participants were hospitalized patients at the University of Michigan Hospital, a 1100-bed tertiary referral center located in Ann Arbor, Michigan. All patients hospitalized for suspected IE and followed by the inpatient infectious disease consult team are discussed weekly by the multidisciplinary endocarditis team (MET), who represent various subspecialties, including infectious diseases, cardiology, cardiac surgery, neurology, nuclear medicine, and addiction medicine, among others, to optimize patient care. All patients who were discussed by the MET for suspected or definite endocarditis between June 2018 and January 2022 were included in this study. Definite endocarditis was determined by the modified Duke criteria. 19 PET scan results were not part of the Duke criteria at the time this study was performed. Patients who were discussed at multiple MET conferences for a single episode of endocarditis or hospitalization were considered to be a single case. If a patient was evaluated for a new episode of endocarditis over this time period, each hospitalization was considered a separate endocarditis case. Outpatients with suspected endocarditis discussed at the conference were not included in the study. In addition, those patients who were still admitted to the hospital with incomplete outcome information at the end of the time period were excluded. The study was conducted and reported in accordance with the STROBE guidelines. 20 (Supplemental 6).
Variables and data collection
Independent variables collected included demographic information (i.e., age, gender, ethnicity, etc.), comorbidities (i.e., length of stay, acuity of admission, comorbidities, recent emergency department use (LACE) index, presence of HIV, and end-stage renal disease on dialysis), risk factors for endocarditis (i.e., presence of long-term intravenous access at admission, history of intravenous drug use, and presence of endocardial devices or prostheses), and endocarditis characteristics (i.e., causative microorganism, location of infected valve, and complications of endocarditis). LACE index is a marker for predicting the risk of 30-day readmission, and also has been used for predicting mortality after hospital admission as well. 21 It was used as a marker of comorbidity and medical complexity in our study, as it is automatically calculated in our system and takes into account the Charlson comorbidity index as one of the variables. Dependent variables included hospital outcomes, including length of admission, duration of antibiotic therapy, use of suppressive antibiotics, 6-month readmission rates, and in-hospital and 6-month mortality rates. Descriptive data regarding WBP indication, findings, and features of post-imaging management were also collected to better understand the use of WBP. The WBP scanner used was the Siemens BIOGRAPH 128 Vision 600 Edge PET/CT (Malvern, Pennsylvania). Patients underwent metabolic preparation prior to the study, consisting of a high-fat, low-carbohydrate ketogenic diet typically starting 24 h prior to the study, followed by a 12-h fast. Images were interpreted by board-certified radiologists who specialize in nuclear imaging. All data was collected manually via RedCap through a protocolized review of the electronic health record by the study team members after a pilot period.
Bias
In addition to the analysis of the full cohort, we performed additional analysis matching by propensity scores to reduce the amount of bias by confounders between groups. To decrease the interpretation bias of chart review, definitions, and data abstraction processes were protocolized. Any conflicting interpretations during the data collection were discussed to resolution as a group.
Statistical methods
Descriptive statistics were used for comparisons between the WBP and non-WBP cohorts.
Chi-squared test and Student’s T-test were used for comparisons of statistical significance between categorical and numerical variables, respectively. Subgroup analysis on hospital outcomes was performed on independent variables with significant differences between the two cohorts. Descriptive statistics were used to evaluate the indications for, results of, and management changes resulting from imaging within the WBP group. Multivariate logistic regression was used to evaluate those most likely to benefit from WBP. Observations with missing data were not censored. Propensity score matching analysis was performed using PROC PSMATCH in SAS. Matching was performed with average treatment effect on the treated (ATT) weighting after matching without replacement. All statistical analysis was performed on SAS 9.4 (Cary, North Carolina) by the study team.
Results
Patient demographics and endocarditis risk factors
There were 427 patients included in our study; 114 patients (26.7%) had WBP performed as part of their IE evaluation (WBP group), and 313 patients (73.3%) did not have WBP performed (non-WBP group). Among the full cohort, 61.1% met definite Duke criteria, 31.6% met possible Duke criteria, and 7.3% met rejected Duke criteria. There were no significant differences in demographics, such as age, gender, or race, between the two groups. The WBP group was significantly more likely to have end-stage renal disease compared to the non-WBP group (20.2% vs 10.2%, p = 0.01). Otherwise, the two groups had similar rates of comorbidities such as diabetes, the presence of HIV, and active malignancy, as well as LACE scores. The WBP groups were also more likely to have intracardiac prostheses (57.9% vs 35.5%, p < 0.001) and cardiac devices (25.4% vs 15.7%, p = 0.01). The non-WBP group was more likely to have a history of intravenous drug use within 6 months of admission (10.9% vs 0.9%, p < 0.001). These results are further summarized in Table 1.
Patient demographics, risk factors, and endocarditis characteristics.
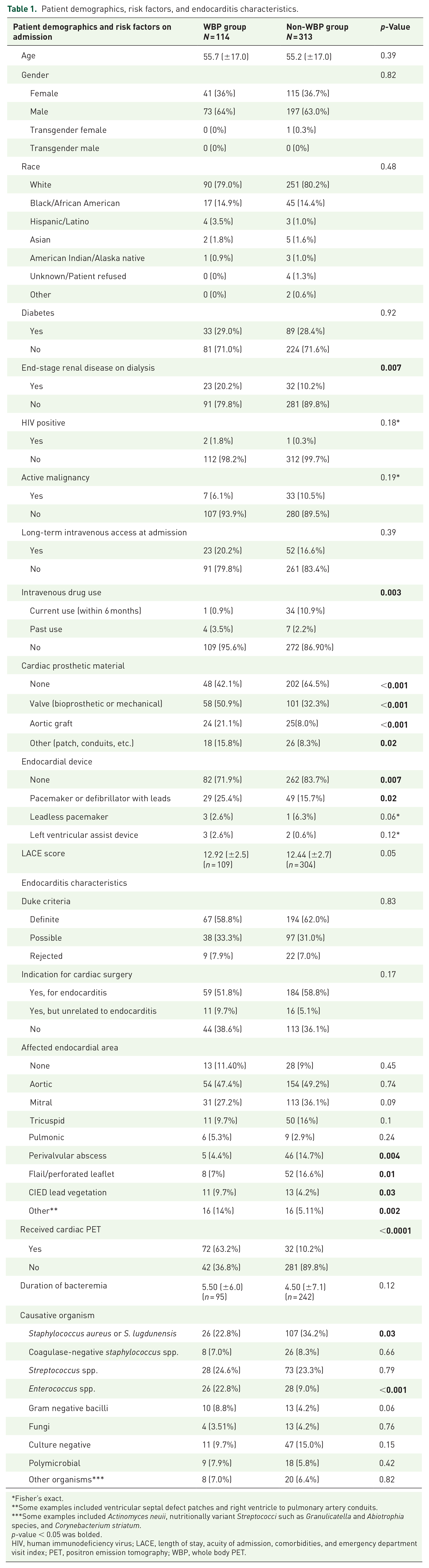
Fisher’s exact.
Some examples included ventricular septal defect patches and right ventricle to pulmonary artery conduits.
Some examples included Actinomyces neuii, nutritionally variant Streptococci such as Granulicatella and Abiotrophia species, and Corynebacterium striatum.
p-value < 0.05 was bolded.
HIV, human immunodeficiency virus; LACE, length of stay, acuity of admission, comorbidities, and emergency department visit index; PET, positron emission tomography; WBP, whole body PET.
Characteristics of infective endocarditis
The two cohorts had similar durations of bacteremia, proportions of Duke criteria classification, and indications for surgery. While specific valvular involvement was similar between the two groups, the non-WBP group was more likely to have endocarditis complicated by a perivalvular abscess (14.7% vs 4.4%, p = 0.004) and flail leaflet (16.6% vs 7%, p = 0.01). On the other hand, the WBP group was more likely to have implantable device lead vegetations (9.7% vs 4.2%, p = 0.03) and infections of other endocardial prostheses such as patches, conduits, and grafts (14% vs 5.11%, p = 0.002). In addition, cardiac-specific PET scans were significantly more likely to be performed in the WBP group (63.2% vs 10.2%, p < 0.001).
In cases where a causative organism was identified, Staphylococcus aureus (or lugdunensis) was the predominant species, followed next by various Streptococcus species for both groups (Table 1). Infection with S. aureus (or lugdunensis) was significantly more likely in the non-WBP group (34.2% vs 22.8%, p = 0.03), while infection with Enterococcus species was more likely in the WBP group (22.8% vs 9%, p < 0.001).
Patient outcomes
There were no statistically significant differences in outcomes such as mortality, hospital readmission in 6 months, infection relapse in 6 months, length of stay, or ICU stay between the WBP and the non-WBP groups (Table 2). Cardiac surgery was performed in 137 (43.8%) of the patients in the non-WBP group, while 38 (33.3%) of the patients in the WBP group had surgery; this difference had a trend toward statistical significance (p = 0.05). There were also statistically significant differences between the two groups in medical management, with the WBP group more likely to receive longer antibiotic courses (mean 6.36 ± 2.2 weeks vs mean 5.23 ± 2.2 weeks, p < 0.001) and higher rates of suppressive antibiotics planned following treatment course (30.7% vs 13.4%, p < 0.001).
Outcomes between WBP and non-WBP groups.

p-value < 0.05 was bolded.
ICU, intensive care unit; IE, infective endocarditis; WBP, whole body positron emission tomography.
We then performed a propensity score-matched analysis, where the matching variables included age, end-stage renal disease, intravenous drug use (IVDU), prosthetic material, endocardial device, infection with Enterococcus, and infection with S. aureus (or lugdunensis). The standardized mean differences were ±0.25, and the variance ratio of the weighted matched study was close to 1. There were 114 patients in the WBP group who were matched to 228 patients in the non-WBP group. Propensity score-matched analysis showed a longer duration of antibiotic therapy in the WBP group. No other outcomes were significantly different between the groups (Supplemental 5).
Whole body PET scan findings
The mean time between antibiotic initiation to the date that WBP was performed was 11 days. Among the 114 patients in the WBP group, the most common reason for WBP was to “evaluate for extracardiac sites of infection” (n = 83, 72.8%), with other reasons being “to evaluate for seeding of grafts and prostheses” (n = 31, 27.2%) and “at request of surgical team for operative planning” (n = 4, 3.5%). The most common clinically important findings included pulmonary infections such as pneumonia or septic pulmonary emboli (n = 32, 28.6%), infection in bones and joints (n = 17, 14.9%), and skin and soft tissue infections (n = 15, 13.4%). There were seven patients (6.1%) who were found to have a vascular graft infection. Nonspecific findings with unclear clinical significance (n = 35, 30.7%) or noninfectious incidental findings (n = 50, 43.9%) were common. Patient management was changed as a result of the WBP findings in 50 of the 114 patients (44.6%). These changes included further infectious or noninfectious workup (n = 32, 28.6%), the performance of a source control procedure (n = 12, 10.7%), and a change in the antibiotic regimen, including duration or antibiotic choice (n = 11, 9.8%). These findings are outlined in Table 3.
Whole body PET characteristics (N = 114).

There included polyps or nodules in lungs (n = 4), colon (n = 4), prostate (n = 2), thyroid (n = 1), and soft tissue (n = 1).
Some examples of other findings included: abdominal mesh uptake, venous thromboembolism, ureteral calculus, intra-abdominal cysts, esophagitis, and an enterovesical fistula.
PET, positron emission tomography.
In addition, we looked at the changes in management stratified by the reason for WBP being performed. This demonstrated that among patients for whom WBP was performed to evaluate for seeding of grafts or prostheses, there were higher rates of source control procedures, avoidance of unnecessary removal of grafts or prostheses, and change in antibiotic plan. (Supplemental 4). In comparison, patients for whom WBP was performed to evaluate for extracardiac sites of infection, such as portal of entry or metastases, had higher rates of further infectious workup being performed. The overall rate of change in management was higher among patients who had WBP to evaluate for seeding of grafts or prostheses (n = 21, 67.7%).
One of the more common ways that WBP impacted patient management was by informing about additional infectious work-ups. In one such case, a patient had non-focal diffuse pain and WBP revealed unexpected multifocal infection in the aortic arch, multiple spinal disks, the glenohumoral joint, cavitary lung lesions, and the iliacus muscle. This led to further workup of these infectious complications and an iliacus drain placement. In seven patients, WBP was able to confirm or rule out an extracardiac infected graft (such as abdominal aortic endograft, aortoiliac graft, or arteriovenous graft), which informed plans for long-term antibiotic suppression. In addition, WBP was able to identify infection of orthopedic hardware or vascular grafts in patients who were not candidates for surgical removal of hardware, which led to a prolongation of the therapeutic antibiotic course or a change in antibiotic management (such as the addition of rifamycins). Finally, there were multiple cases where an incidental noninfectious finding, such as a colonic mass or ureteral stone, required further evaluation. Key case descriptions are found in Supplemental 1.
Subgroup analysis
Post-hoc analysis included comparison of outcomes between the WBP and non-WBP groups for prespecified sub-groups. (Supplemental 2). These subgroups included prolonged bacteremia (5 or more days), the causative organism being S. aureus (or lugdunensis), end-stage renal disease, presence of intracardiac prostheses, presence of endocardial devices, positive cardiac surgery indication, definite Duke criteria, and WBP without a cardiac PET. For all of these sub-groups, we consistently found that there would be a notable increase in antibiotic duration and rates of suppressive antibiotics for the WBP group. For the definite Duke criteria sub-group, we found the WBP cohort also had a longer length of stay (27.8 vs 22.8 days, p = 0.020). Among those with an endocardial device present, surgery was more likely to be performed among non-WBP group (41.2% vs 9.4%, p = 0.002). For patients with end-stage renal disease, in-hospital mortality was higher among the WBP group (39.1% vs 12.5%, p = 0.02), although 6-month mortality remained similar between groups.
WBP outcome associations with patient characteristics
Multivariate logistic regression was performed to look at the relationship between the likelihood of WBP changing the management and prespecified and post-hoc exploratory variables, including age, gender, comorbidities (i.e., end-stage renal disease and long-term IV access at admission), IE risk factors (i.e., long-term IV access, cardiac prostheses), and IE characteristics (i.e., causative organism, duration of bacteremia, indication for surgery) (Supplemental 3). Presence of long-term IV access was the only variable associated with increased likelihood of WBP changing management on univariate analysis (OR 2.7, p = 0.04), but this association was statistically nonsignificant on multivariate regression (OR 4.2, p = 0.06). We found that patients with long-term IV access were more likely to get a source control procedure (17.4% vs 8.8%) and further infectious workup (30.4% vs 17.6%) compared to the rest of the WBP cohort. Among the four patients who had a change to the source control plan, three of them had end-stage renal disease, and WBP helped identify infected vascular prostheses (arteriovenous graft and dialysis catheter) that required removal.
Discussion
To our knowledge, this is the largest study to compare patient characteristics and outcomes between cohorts of patients who did and did not have WBP performed as part of IE management. We were able to evaluate how WBP impacted patient care, including the reason it was performed and how management was impacted by the results of WBP, which are important factors when considering how best to utilize this comprehensive nuclear medicine diagnostic study. As part of this study, we identified patients with vascular prostheses as those who may benefit most from the use of WBP when managing IE.
There were major differences between the demographics of the groups who had WBP and those who did not. One possible explanation for the WBP group having a higher likelihood of cardiac prostheses and devices involves the collinearity with cardiac PET; we found that the order form for cardiac PET scan in our electronic health system included a check box option to perform a WBP as well, and it’s possible the ease of this primed users to tack on additional testing. It would make sense for those with more risk factors for IE or the presence of cardiac prostheses and devices to require cardiac PET for a more detailed diagnosis and characterization of the disease. In addition, it is possible that patients with more urgent surgical indications, such as paravalvular abscess and flail leaflet, would forgo time-intensive diagnostics such as WBP and would go directly to cardiac surgery. This is further suggested by the higher rates of cardiac surgery observed in the non-WBP group, although the difference was not statistically significant. Higher rates of IVDU in the non-WBP group may be explained by the fact that they had a known source of bacteremia and would be more likely to have right-sided endocarditis, which may reduce the risk of metastatic sites outside of the lung.
We found that the WBP group trended toward receiving less surgery and longer duration of antibiotic regimens, including long-term oral antibiotic suppression. The observation that 45% of the patients who underwent WBP had their management impacted by the imaging results may be related to the fact that WBP helped clinicians optimize the nonsurgical management of IE, such as antibiotic plans and extracardiac source control. This then brings to question what patient factors may predict those most likely to benefit from a WBP. Our multivariate logistic regression showed an association between the presence of long-term IV access and increased likelihood of management being impacted by the result of WBP, driven by an increase in source control procedures via removal of infected vascular catheters and grafts, and further infectious workup. Taken together with our finding of increased change in management when WBP was performed to evaluate for seeding of grafts and prostheses, these illustrate the value that WBP provides for the identification of infected vascular prostheses in the setting of infective endocarditis.
There is an emerging literature on the use of FDG-PET/CT in diagnosing vascular graft infections. Systematic review and meta-analysis by Mahmoodi et al. found FDG-PET/CT have sensitivity and specificity of 92% and 76%, respectively, in diagnosing vascular graft infections. 22 The European Association of Nuclear Medicine has adopted FDG-PET/CT as a useful imaging modality in the diagnosis of vascular graft infections in their guidelines, although there is some heterogeneity in the interpretation of these studies when evaluating FDG uptake and pattern. 23 While the current study was not designed with the strict focus of evaluating patients with vascular grafts and IE, we believe our results point to the benefit that WBP can have for these patients. Infected grafts may be the result of metastatic seeding or may be a portal of entry. Identification of these sites of infection is important because if not managed appropriately with removal of the device or suppressive oral antibiotics, relapse of infection is common.
In our review of the literature, the study by Kestler et al. 15 in 2014 is the only paper comparing the cohorts of patients who did and did not get WBP in the management of IE. There are methodological differences between our studies, as their study compared a prospective cohort of 47 patients, matched against a historical control of 94 patients by affected valve and causative pathogen. While we found a lower proportion of the WBP cohort receiving cardiac surgery, the opposite was found by Kestler. Methodological differences may account for this discrepancy as Kestler et al. excluded patients who underwent early surgery, had early mortality, or had ICU admission. Other findings were similar between the two studies. We also found that the WBP cohort received longer durations of antibiotics without differences in readmission rates or relapses. Our study adds further value by delineating differences in patient characteristics such as end-stage renal disease, history of injection drug use, mortality, and suppressive antibiotic use as outcomes of interest.
We believe that our cohort was overall representative of patients with IE nationally. According to Wurcel et al., 24 the proportion of patients with IE who use IVDU in the United States was approximately 12% in 2013, having risen from stable rates of 7%–8% in the preceding decade. Percentage of our cohort who had active IVDU was 8.2%, with another 2.6% reporting prior use (greater than 6 months prior to admission). Our cohort also included high rates of IE risk factors, including intracardiac prostheses, endocardial devices, and the presence of long-term IV access at admission. In addition, our cohort included patients with severe illness and high complexity with 48% of patients requiring ICU stay during hospitalization and a mean LACE index of 12.6. 25
Limitations
Observational retrospective cohort design is a limitation of this study, as it prohibits any inference about causality. Retrospective design is also susceptible to confounding by indication; for example, an unmeasured confounder like extracardiac prostheses may have predisposed to both undergoing WBP and requiring suppressive antibiotics. This also limits our ability to make definitive conclusions on the impact on patients with vascular grafts and prostheses, as the study was not designed with this specific characteristic in mind, and the presence of this variable was not systematically collected. However, we believe this investigation sheds light on how and on whom WBP is currently being used, and provides valuable information on how it is impacting patient care. Another limitation is that the use of cardiac PET was strongly associated with WBP use as noted above, and may be another confounding variable. Attempts to analyze those who underwent WBP but not cardiac PET were limited by reduced sample size.
Conclusion
In this retrospective cohort study, there were important differences in patient characteristics between the WBP and non-WBP groups, including the presence of intracardiac prostheses, endocardial devices, and types of IE complications. WBP played an important role in identifying metastatic foci of infection and directly impacting patient management in about 45% of the patients who underwent the study. Although there were no major differences in clinical outcomes, such as decreased mortality or length of hospitalization, the WBP group was more likely to have prolonged antibiotic duration and use of suppressive antibiotics related to the detection of infection of intravascular prostheses. Therefore, when considering which patients with IE to select for the performance of WBP, those with intravascular devices appear to be those who may benefit the most.
Supplemental Material
sj-docx-1-tai-10.1177_20499361251336849 – Supplemental material for Use of whole body PET scan in patients with infective endocarditis may impact care of those with intravascular devices: results from a comparative retrospective cohort study
Supplemental material, sj-docx-1-tai-10.1177_20499361251336849 for Use of whole body PET scan in patients with infective endocarditis may impact care of those with intravascular devices: results from a comparative retrospective cohort study by Jason Park, Kaitlyn Simpson, Megan Neils and James Riddell in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-2-tai-10.1177_20499361251336849 – Supplemental material for Use of whole body PET scan in patients with infective endocarditis may impact care of those with intravascular devices: results from a comparative retrospective cohort study
Supplemental material, sj-docx-2-tai-10.1177_20499361251336849 for Use of whole body PET scan in patients with infective endocarditis may impact care of those with intravascular devices: results from a comparative retrospective cohort study by Jason Park, Kaitlyn Simpson, Megan Neils and James Riddell in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-3-tai-10.1177_20499361251336849 – Supplemental material for Use of whole body PET scan in patients with infective endocarditis may impact care of those with intravascular devices: results from a comparative retrospective cohort study
Supplemental material, sj-docx-3-tai-10.1177_20499361251336849 for Use of whole body PET scan in patients with infective endocarditis may impact care of those with intravascular devices: results from a comparative retrospective cohort study by Jason Park, Kaitlyn Simpson, Megan Neils and James Riddell in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-4-tai-10.1177_20499361251336849 – Supplemental material for Use of whole body PET scan in patients with infective endocarditis may impact care of those with intravascular devices: results from a comparative retrospective cohort study
Supplemental material, sj-docx-4-tai-10.1177_20499361251336849 for Use of whole body PET scan in patients with infective endocarditis may impact care of those with intravascular devices: results from a comparative retrospective cohort study by Jason Park, Kaitlyn Simpson, Megan Neils and James Riddell in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-5-tai-10.1177_20499361251336849 – Supplemental material for Use of whole body PET scan in patients with infective endocarditis may impact care of those with intravascular devices: results from a comparative retrospective cohort study
Supplemental material, sj-docx-5-tai-10.1177_20499361251336849 for Use of whole body PET scan in patients with infective endocarditis may impact care of those with intravascular devices: results from a comparative retrospective cohort study by Jason Park, Kaitlyn Simpson, Megan Neils and James Riddell in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-6-tai-10.1177_20499361251336849 – Supplemental material for Use of whole body PET scan in patients with infective endocarditis may impact care of those with intravascular devices: results from a comparative retrospective cohort study
Supplemental material, sj-docx-6-tai-10.1177_20499361251336849 for Use of whole body PET scan in patients with infective endocarditis may impact care of those with intravascular devices: results from a comparative retrospective cohort study by Jason Park, Kaitlyn Simpson, Megan Neils and James Riddell in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
We would like to thank Nuclear medicine experts James Corbett and Benjamin Viglianti for their assistance with the manuscript and Kathleen Shutt for her contribution to statistical analysis.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
