Abstract
Background:
Mycobacterium avium complex lung disease (MAC-LD) is a chronic, progressive, potentially life-threatening infection. Some cases are refractory to standard guideline-based therapy (GBT), and sputum cultures are persistently positive for acid-fast bacilli. Although an early identification of treatment-refractory MAC-LD is crucial, its risk factors remain unknown.
Objectives:
We aimed to identify the risk factors for refractory MAC-LD in response to initial GBT.
Design:
A retrospective single-center study was conducted involving consecutive patients with MAC-LD who were diagnosed between 2006 and 2024 and received initial GBT.
Methods:
Refractory MAC-LD was defined as sputum culture positivity at least 6 months after the initial GBT. Prognostic factors were identified using Cox proportional hazards analysis, and risk factors for refractory MAC-LD were examined using logistic regression analysis.
Results:
Of the 201 patients with definite MAC-LD, 35 (17.4%) had refractory MAC-LD. Patients with refractory MAC-LD had a significantly lower body mass index (BMI), more cavitary lesions on high-resolution computed tomography (HRCT), and higher mortality (log-rank test, p = 0.006) compared to those with non-refractory MAC-LD. A multivariate analysis adjusted for age and sex showed that refractory MAC-LD (adjusted hazard ratio (HR): 2.76; 95% confidence interval (CI): 1.10–6.95; p = 0.030) and cavitary lesions on HRCT (adjusted HR: 2.77; 95% CI: 1.34–5.70; p = 0.005) were significantly associated with all-cause mortality. In addition, a multivariate analysis revealed that lower BMI (odds ratio (OR): 0.68; 95% CI: 0.55–0.85; p < 0.001) and cavitary lesions on HRCT (OR: 2.52; 95% CI: 1.15–5.50; p = 0.020) were independent risk factors of refractory MAC-LD.
Conclusion:
Low BMI and cavitary lesions on HRCT are risk factors for refractory MAC-LD.
Introduction
Nontuberculous mycobacteria (NTM) are ubiquitous environmental bacteria that exist in soil and water, and more than 200 species of NTM have been identified.1,2 Several species of NTM are infectious to humans and cause chronic respiratory disease. The Mycobacterium avium complex (MAC), which includes M. avium and M. intracellulare, is often the causative agent, and the chronic lung disease caused by it is called MAC lung disease (MAC-LD).2–6 Although MAC-LD is generally considered a chronic respiratory disease associated with a reduced quality of life, its clinical course is diverse, ranging from long-term stable cases without treatment to fatal outcomes.7,8 Furthermore, the prevalence of MAC-LD has been increasing worldwide, posing a serious health problem.9–12
According to the American Thoracic Society (ATS)/European Respiratory Society (ERS)/European Society of Clinical Microbiology and Infectious Diseases (ESCMID)/Infectious Diseases Society of America (IDSA) clinical practice guidelines for the treatment of NTM, updated in 2020, at least 12 months of treatment with multidrug regimens is recommended. 4 However, the percentage of initial treatment success is reported to be 33%–60%,13,14 with many cases being refractory or recurrent.15,16 Therefore, developing a more effective treatment or management strategy for MAC-LDs is necessary. According to the latest guidelines, refractory MAC-LD is defined as sputum culture positivity after 6 months of guideline-based therapy (GBT). 4 Recently, amikacin liposomal inhalation suspension (ALIS), a liposomal formulation of amikacin, was reported to significantly improve the culture conversion proportion when added to the GBT for refractory MAC-LD. 17 Although identifying patients at high risk for developing refractory MAC-LD is crucial, the risk factors and prognostic impact of refractory MAC-LD remain unknown.
Therefore, in this study, we aimed to investigate the prevalence, clinical characteristics, and prognosis of refractory MAC-LD and clarify the risk factors of refractory MAC-LD.
Methods
Study design
This was a single-center, retrospective cohort study conducted at Seirei Hamamatsu General Hospital. We initially identified 501 adult patients with a diagnosis of MAC-LD from the hospital database, through data extraction from electronic medical records, between January 1, 2006, and March 31, 2024. A comprehensive chart review was performed to re-evaluate these diagnoses according to the latest official ATS/ERS/ESCMID/IDSA clinical practice guidelines. 4 Patients were included as definite MAC-LD cases if they met all three diagnostic criteria: clinical (pulmonary symptoms consistent with NTM infection), radiological (nodular or cavitary opacities on chest radiograph, or multifocal bronchiectasis with multiple small nodules on high-resolution computed tomography (HRCT) scan), and microbiological (two positive sputum cultures or one positive bronchoscopy culture for MAC). 4 In addition, these patients must have received initial GBT for at least 6 months to be included in the study. Based on these guidelines, refractory MAC-LD was defined as positive sputum cultures despite initial GBT for >6 months.4,17 Patients with no history of treatment, those with initial treatment for less than 6 months, previously treated patients, and those who did not meet the diagnostic criteria were excluded. GBT is a treatment method outlined in the latest clinical practice guidelines, and is a combination of the following drugs: clarithromycin/azithromycin, rifampin/rifabutin, and ethambutol with/without amikacin or streptomycin.
We identified 201 patients with definite MAC-LD who underwent their initial course of GBT for at least 6 months. The prevalence of refractory MAC-LD in these patients was investigated, and we analyzed the risk factors of refractory MAC-LD and prognostic differences between refractory and non-refractory MAC-LD. The prognostic factors for all-cause and respiratory-related deaths were also examined. The committee granted a waiver of informed consent for this study due to its retrospective nature, as the research posed minimal risk to participants and was based solely on medical records review. This study was conducted and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 18
Data collection
We conducted a comprehensive chart review to collect the following data: age, sex, body mass index (BMI), smoking status, clinical data at diagnosis, including serological findings, presence of cavitary lesions at diagnosis on HRCT, acid-fast bacillus smear positivity, sputum culture positivity at diagnosis and 6–12 months after the initial treatment, bacterial species, treatment, and outcomes. In this study, we utilized Mycobacteria Growth Indicator Tube (MGIT)® (Becton-Dickinson, Sparks, MD, USA) for NTM cultivation, matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS) for species identification, and Broth MIC NTM® (Kyokuto Pharmaceutical Industrial Co., Ltd., Tokyo, Japan) for susceptibility testing of MAC isolates. All patients underwent drug susceptibility testing at the time of species identification. Two pulmonologists and a radiologist who were blinded to the clinical data independently reviewed the HRCT images and reached a consensus on the presence of cavitary lesions at diagnosis through discussion. The observation period was calculated from the date of diagnosis of MAC-LD, which is defined as the date of the second positive sputum culture or the date of a positive bronchoscopy culture, to the last visit (date of censoring or death). Patients who survived until August 31, 2024 were censored.
Statistical analysis
Continuous and categorical variables are expressed as mean ± standard deviation or median (interquartile range) and number (percentage), respectively. The differences between refractory and non-refractory MAC-LD were assessed using the Mann–Whitney U test, Fisher’s exact test, or chi-squared test. Logistic regression analysis was used to detect the risk factors for refractory MAC-LD. The cumulative proportions of all-cause mortality and respiratory-related mortality were estimated using the Kaplan–Meier method. For this study, respiratory-related deaths were defined as those resulting from chronic respiratory failure, pneumonia, lung cancer, and hemoptysis. To assess the intergroup differences in cumulative survival and cumulative incidence of respiratory-related death, the log-rank test and Gray’s test were used, respectively. Cox proportional hazards regression analysis was used to analyze the prognostic factors for all-cause mortality and respiratory-related death. Refractory MAC-LD was considered a time-dependent covariate. Hazard ratios (HR), 95% confidence interval (CI), and p values were calculated. To assess the independent association of refractory MAC-LD, we included clinically important variables (e.g., previously reported prognostic factors) in the univariate and multivariate models. All statistical analyses were performed using EZR version 1.36 (Saitama Medical Center, Jichi Medical University, Saitama, Japan); statistical significance was set at p < 0.05.
Results
Patient characteristics
The clinical characteristics of patients with refractory and non-refractory MAC-LDs are shown in Table 1. The median age of the patients was 65.0 years, and 56 (27.9%) were males. Of the 201 patients, 35 (17.4%) were classified as having refractory MAC-LD. None of the patients had macrolide-resistant MAC. Compared with patients with non-refractory MAC-LD, those with refractory MAC-LD had significantly lower BMI (19.3 kg/m2 vs 18.5 kg/m2, p = 0.001), more frequent cavitary lesions on HRCT (15.1% vs 28.6%, p = 0.006), and higher proportion of patients receiving amikacin or streptomycin with initial treatment (1.8% vs 17.1%, p = 0.001). Other factors, including bacterial species, sputum smear positivity at diagnosis, percentage of patients receiving macrolide/ethambutol/rifamycin regimen, and percentage of patients receiving surgical resection, did not differ between the two groups. No patients received ALIS treatment in this study.
Baseline characteristics, treatment, and outcomes of patients with refractory and non-refractory MAC-LD.
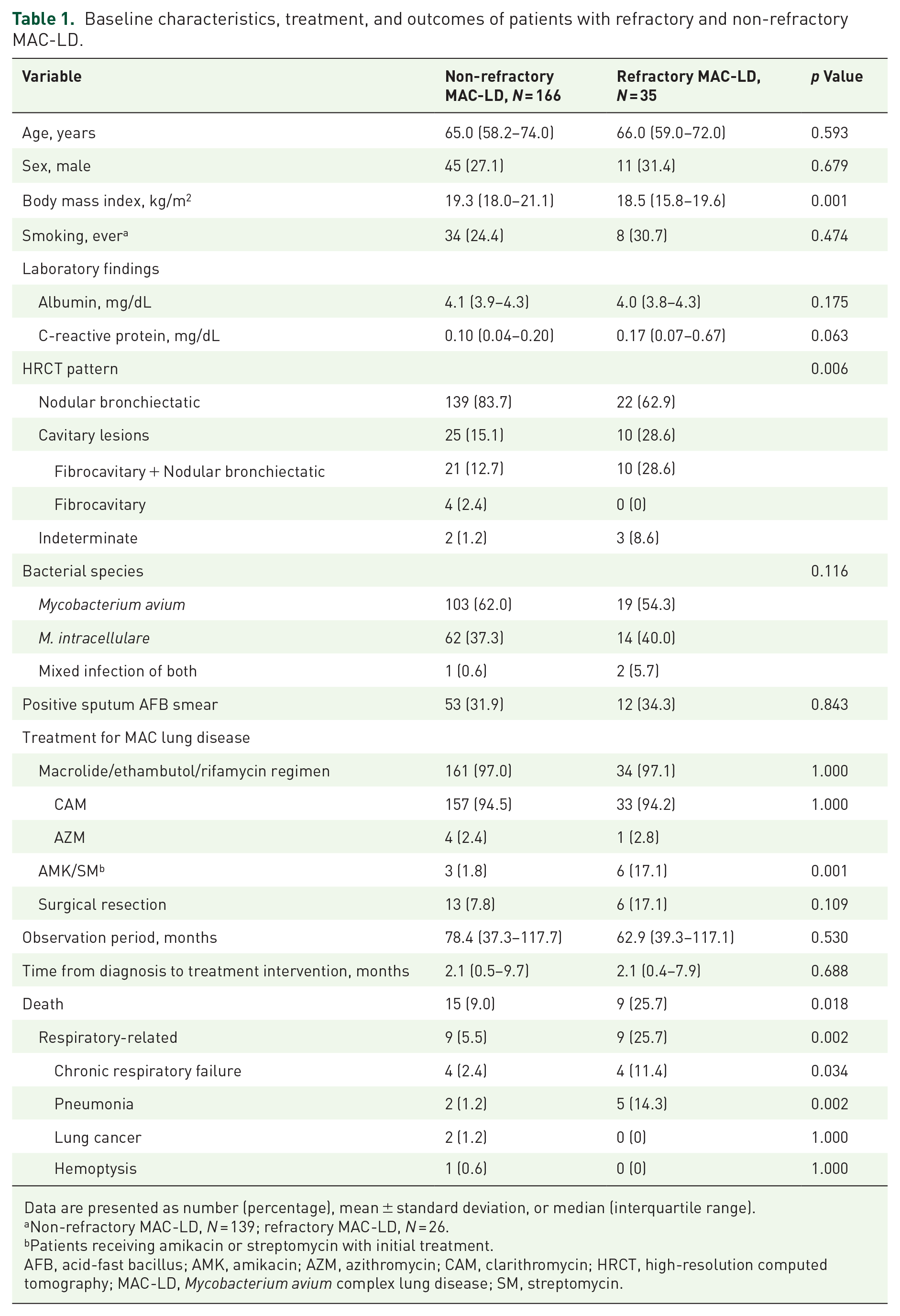
Data are presented as number (percentage), mean ± standard deviation, or median (interquartile range).
Non-refractory MAC-LD, N = 139; refractory MAC-LD, N = 26.
Patients receiving amikacin or streptomycin with initial treatment.
AFB, acid-fast bacillus; AMK, amikacin; AZM, azithromycin; CAM, clarithromycin; HRCT, high-resolution computed tomography; MAC-LD, Mycobacterium avium complex lung disease; SM, streptomycin.
Association of refractory MAC-LD with all-cause mortality
The cumulative survival was significantly lower in patients with refractory MAC-LD than in those without refractory MAC-LD (p = 0.006; Figure 1). Similarly, we found that the 5-year cumulative incidence of respiratory-related deaths was higher in patients with refractory MAC-LD than in those with non-refractory MAC-LD (2.3% vs 12.9%, p < 0.001; Figure S1). Univariate Cox proportional hazards regression analysis of all-cause mortality showed that older age (HR: 1.08; 95% CI: 1.03–1.13; p < 0.001), males (HR: 6.55; 95% CI: 2.79–15.40; p < 0.001), lower serum albumin (HR: 0.32; 95% CI: 0.14–0.69; p = 0.004), higher serum C-reactive protein (CRP; HR: 1.15; 95% CI: 1.03–1.29; p = 0.011), refractory MAC-LD (HR: 2.99; 95% CI: 1.30–6.86; p = 0.009), and cavitary leison on HRCT (HR: 2.58; 95% CI: 1.28–5.18; p = 0.007) were significantly related to increased mortality (Table 2). In a multivariate analysis adjusted for age and sex, refractory MAC-LD (adjusted HR: 2.76; 95% CI: 1.10–6.95; p = 0.030) and cavitary lesions on HRCT (adjusted HR: 2.77; 95% CI: 1.34–5.70; p = 0.005) were independent prognostic factors for all-cause mortality. In addition, the multivariate Cox proportional hazards regression analysis of respiratory-related death, adjusted for age and sex, revealed that serum CRP (adjusted HR: 1.21; 95% CI: 1.02–1.43; p = 0.021) and refractory MAC-LD (adjusted HR: 4.82; 95% CI: 1.65–14.05; p = 0.003) were independent prognostic factors of respiratory-related death (Table S1).

Kaplan–Meier survival curves. The 5-year cumulative survival of patients with refractory MAC-LD (n = 35) and those with non-refractory MAC-LD (n = 166) were 87.1% (95% CI: 68.5–95.1) and 97.0% (95% CI: 92.2–98.9), respectively (p = 0.006 by log-rank test).
Cox proportional hazards regression analysis of all-cause mortality.

Time-dependent covariate.
AFB, acid-fast bacillus; CI, confidence interval; HR, hazard ratio; HRCT, high-resolution computed tomography; MAC-LD, M. avium complex lung disease.
Risk factors of refractory MAC-LD
Univariate logistic regression analysis showed that lower BMI (odds ratio (OR): 0.68; 95% CI: 0.55–0.84; p < 0.001) and cavitary lesions on HRCT (OR: 2.27; 95% CI: 1.43–5.36; p = 0.002) were significantly associated factors for refractory MAC-LD (Table 3). In addition, the multivariate analysis revealed that lower BMI (OR: 0.68; 95% CI: 0.55–0.85; p < 0.001) and cavitary lesions (OR: 2.52; 95% CI: 1.15–5.50; p = 0.020) showed significant associations, even after adjusting for age and sex (Table 3).
Risk factors of refractory MAC-LD by logistic regression analysis.

AFB, acid-fast bacillus; CI, confidence interval; HRCT, high-resolution computed tomography; OR, odds ratio.
Stratified analysis on patients with nodular bronchiectatic pattern on HRCT
To the best of our knowledge, studies focusing exclusively on the nodular bronchiectatic (NB) pattern in refractory MAC-LD are limited. Therefore, we conducted a stratified analysis specifically for patients with NB patterns on HRCT, yielding several noteworthy results. The cumulative survival tended to be lower in the refractory group than in the non-refractory group; however, the difference was not statistically significant (p = 0.086; Figure S2). Univariate Cox proportional hazards analysis identified older age (HR: 1.11; 95% CI: 1.05–1.17; p < 0.001), male sex (HR: 7.79; 95% CI: 2.74–22.1; p < 0.001), and lower serum albumin (HR: 0.25; 95% CI: 0.11–0.60; p = 0.001) as significant factors of increased all-cause mortality. In the multivariate analysis, age (adjusted HR: 1.08; 95% CI: 1.01–1.17; p = 0.023) and male sex (adjusted HR: 6.50; 95% CI: 2.14–19.7; p < 0.001) remained an independent associated factor. Refractory MAC-LD was not significantly associated with increased mortality (Table S2). We further investigated the risk factors of refractory MAC-LD. In the multivariate logistic regression analysis, BMI was identified as a significant associated factor with refractory MAC-LD (OR: 0.68; 95% CI: 0.52–0.90; p = 0.006; Table S3).
Discussion
In this retrospective study, we found that 17% of cases of definite MAC-LD were classified as refractory MAC-LD. Previous studies have used varying definitions of treatment success, making it difficult to estimate the frequency of refractory MAC-LDs. 14 Culture conversion, defined as at least three consecutive negative cultures collected 4 weeks apart, has been used as an indicator of treatment success in previous reports.19,20 However, performing monthly sputum tests in clinical practice is often challenging, making it difficult to collect accurate data for culture conversion. Recently, refractory MAC-LD has been defined as remaining sputum culture positivity after 6 months of GBT, 4 and we adopted this definition in our study. Given the retrospective nature of this study, sputum culture tests may not have been performed at appropriate intervals due to factors such as patients being unable to produce sputum or the clinical judgment of the physician. As a result, some patients who should have been categorized as refractory might have been misclassified as non-refractory. This misclassification could have reduced the study’s ability to detect differences between the two groups, potentially influencing the findings. Nevertheless, similar to previous reports,19,20 our study showed that refractory MAC-LD had a significantly higher mortality than non-refractory MAC-LD. These results suggest the clinical importance of identifying refractory MAC-LD, regardless of the differences in definition.
Previous studies report varying proportions of sputum smear positivity (33.3%–70%)21–23 and cavitary lesions (31.3%–60%)21,22 in refractory MAC-LD. In our cohort, the sputum smear positivity was 34.3%, and cavitary lesions were 28.6% in the refractory group. While these results may be influenced by study design, they are not significantly inconsistent with previous reports.
Current guidelines recommend the use of aminoglycosides in combination therapy for patients with cavitary lesions 4 ; however, not all such patients in our study received them. This may have overestimated the impact of cavitary lesions. Nevertheless, a review of patient charts indicated that some patients achieved favorable treatment outcomes even without aminoglycosides. To more accurately determine the clinical significance of cavitary lesions, prospective studies with standardized treatment protocols are warranted.
The clinical course of MAC-LD is heterogeneous, with a 5-year mortality of approximately 20%–30%,24,25 and clinical or radiological disease progression is observed in approximately 40% of patients despite treatment with GBT. 26 Previous studies reported that age, sex, hypoalbuminemia, and sputum smear positivity are risk factors for death and disease progression in patients with NTM.26,27 We demonstrated that refractory MAC-LD was independently associated with increased all-cause and respiratory-related mortality, even after adjusting for age, sex, serum albumin, serum CRP, and cavitary lesions. While the majority of previous research on prognostic factors has examined the association with all-cause mortality,19,26–29 our study provides a valuable insight by specifically demonstrating an association with respiratory-related death.
Although we found no significant association between BMI and mortality, consistent with some studies,25,30 this remains controversial.28,31,32 The limited sample size in our study could have reduced the statistical power, making it difficult to detect a significant association. Our inclusion criteria, which required patients to have received initial treatment for at least 6 months, may have introduced selection bias. Furthermore, unaccounted confounders (e.g., comorbidities and socioeconomic status) could explain the discrepancies with prior research, necessitating larger, comprehensive studies to clarify the BMI-prognosis relationship in MAC-LD.
We identified cavitary lesions and low BMI as risk factors for refractory MAC-LD using multivariate logistic regression analysis. A recent meta-analysis reported that cavitary lesions are risk factors for clinical and radiological disease progression in NTM-LD. 26 The presence of cavitary lesions correlates with sputum smear positivity, 33 and is significantly more frequent among patients who fail to achieve culture conversion than among those who successfully achieve culture conversion. 34 Moreover, these lesions are considered irreversible despite treatment.35,36 Continuous bacterial proliferation and poor drug delivery within cavitary lesions contribute to persistent culture positivity, leading to refractory diseases. 37
Furthermore, in patients with NTM and a low BMI, it has been highlighted that the expression of adipokines, such as leptin and adiponectin secreted by adipocytes, differs from that in healthy individuals. 38 Leptin is considered to be lower in patients with NTM and a low BMI.38,39 As it promotes the production of inflammatory chemokines and macrophage function, 38 its deficiency causes macrophage dysfunction, leading to bacterial proliferation and biofilm formation. 2 Furthermore, adiponectin, known for its anti-inflammatory effects, is reported to be higher in patients with NTM than in healthy individuals, and is inversely correlated with BMI.38,39 Thus, patients with a low BMI are thought to be more susceptible to mycobacterial infections,40,41 which is one of the reasons why a low BMI leads to refractory MAC-LD.
Therefore, specific treatment strategies for patients with refractory disease must be established. ALIS, a liposomal form of amikacin, was the first treatment specifically developed for patients with MAC-LDs, with limited treatment options.15,42 The CONVERT trial demonstrated the efficacy of ALIS in refractory MAC-LDs. 17 Currently, ALIS is introduced only after the disease becomes refractory; however, it is often difficult to improve refractory MAC-LD. Given that low BMI and cavitary lesions are risk factors for refractory disease, it may be necessary to consider introducing ALIS earlier in cases with low BMI or cavitary lesions may be necessary. In addition, rehabilitation and nutritional therapy reportedly improve the quality of life and prognosis of patients with chronic respiratory diseases, such as bronchiectasis, interstitial pneumonia, and chronic obstructive pulmonary disease (COPD).38,43–45 Although data on the efficacy of multidisciplinary treatment for patients with MAC-LD with low BMI are lacking, this approach may be beneficial. Future studies should evaluate its potential benefits.
Our study had several limitations. First, the single-center retrospective design and lack of a priori sample size calculation may have introduced selection bias and limited statistical power. Second, we evaluated patients who were able to continue treatment for more than 6 months, excluding those who discontinued treatment due to adverse events or died within the first 6 months. As these patients likely represent a subgroup with more severe disease or poorer prognostic factors, our findings might underestimate the true burden of mortality and treatment refractoriness in the overall MAC-LD population. Furthermore, by focusing only on patients who were able to continue treatment, the study population may disproportionately reflect individuals with milder disease or better treatment tolerance. Third, our study did not examine the complications reported as prognostic factors, such as diabetes, chronic heart disease, chronic kidney disease, COPD, and interstitial lung disease, which may affect the prognosis. Future well-designed, multicenter, and prospective studies with standardized protocols and comprehensive patient representation are needed to address these limitations and ensure adequate statistical power.
Conclusion
This study revealed that patients with refractory MAC-LD had significantly higher mortality than those with non-refractory MAC-LD, mainly due to respiratory-related causes. In addition, low BMI and cavitary lesions on HRCT at diagnosis were identified as risk factors significantly associated with refractory MAC-LD for initial GBT. Comprehensive treatment, including the early induction of ALIS, pulmonary rehabilitation, and nutritional therapy, may be necessary for patients with refractory MAC-LD.
Supplemental Material
sj-docx-3-tai-10.1177_20499361251331676 – Supplemental material for Predictive risk factors of treatment-refractory Mycobacterium avium complex lung disease: a single-center retrospective cohort study
Supplemental material, sj-docx-3-tai-10.1177_20499361251331676 for Predictive risk factors of treatment-refractory Mycobacterium avium complex lung disease: a single-center retrospective cohort study by Takahiko Saito, Yuya Aono, Masato Kono, Masaki Ishige, Takuma Sugiura, Misato Higasa, Fumiya Nihashi, Mineo Katsumata, Hideki Miwa, Yoshihiro Miki and Dai Hashimoto in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-4-tai-10.1177_20499361251331676 – Supplemental material for Predictive risk factors of treatment-refractory Mycobacterium avium complex lung disease: a single-center retrospective cohort study
Supplemental material, sj-docx-4-tai-10.1177_20499361251331676 for Predictive risk factors of treatment-refractory Mycobacterium avium complex lung disease: a single-center retrospective cohort study by Takahiko Saito, Yuya Aono, Masato Kono, Masaki Ishige, Takuma Sugiura, Misato Higasa, Fumiya Nihashi, Mineo Katsumata, Hideki Miwa, Yoshihiro Miki and Dai Hashimoto in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-5-tai-10.1177_20499361251331676 – Supplemental material for Predictive risk factors of treatment-refractory Mycobacterium avium complex lung disease: a single-center retrospective cohort study
Supplemental material, sj-docx-5-tai-10.1177_20499361251331676 for Predictive risk factors of treatment-refractory Mycobacterium avium complex lung disease: a single-center retrospective cohort study by Takahiko Saito, Yuya Aono, Masato Kono, Masaki Ishige, Takuma Sugiura, Misato Higasa, Fumiya Nihashi, Mineo Katsumata, Hideki Miwa, Yoshihiro Miki and Dai Hashimoto in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-jpg-1-tai-10.1177_20499361251331676 – Supplemental material for Predictive risk factors of treatment-refractory Mycobacterium avium complex lung disease: a single-center retrospective cohort study
Supplemental material, sj-jpg-1-tai-10.1177_20499361251331676 for Predictive risk factors of treatment-refractory Mycobacterium avium complex lung disease: a single-center retrospective cohort study by Takahiko Saito, Yuya Aono, Masato Kono, Masaki Ishige, Takuma Sugiura, Misato Higasa, Fumiya Nihashi, Mineo Katsumata, Hideki Miwa, Yoshihiro Miki and Dai Hashimoto in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-jpg-2-tai-10.1177_20499361251331676 – Supplemental material for Predictive risk factors of treatment-refractory Mycobacterium avium complex lung disease: a single-center retrospective cohort study
Supplemental material, sj-jpg-2-tai-10.1177_20499361251331676 for Predictive risk factors of treatment-refractory Mycobacterium avium complex lung disease: a single-center retrospective cohort study by Takahiko Saito, Yuya Aono, Masato Kono, Masaki Ishige, Takuma Sugiura, Misato Higasa, Fumiya Nihashi, Mineo Katsumata, Hideki Miwa, Yoshihiro Miki and Dai Hashimoto in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
