Abstract
Tuberculosis is of particular concern in lung transplant recipients. We present the case of a patient who received a double lung transplant from a deceased donor from Mexico and developed disseminated tuberculosis 60 days post-transplant manifested as tenosynovitis, liver abscesses, and subcutaneous nodules with no definitive lung allograft involvement. The recipient did not have evidence of tuberculosis on explanted lungs, had a negative interferon gamma release assay pre-transplant, and did not have risk factors for this infection.
Keywords
Introduction
Tuberculosis (TB) is a leading cause of morbidity worldwide. In the United States, the incidence of active TB remains low with most cases occurring in people born outside the country. Most cases in the United States result from untreated latent tuberculosis infection (LTBI), which is not a reportable infection. 1
Solid organ transplant (SOT) recipients are at risk for active TB with an incidence of 20–74 times higher than the general population.2,3 SOT recipients have higher risk of death, 4 risk of graft dysfunction, and frequently have therapy limitations due to hepatotoxicity and drug–drug interactions.2,5 Disseminated infection is associated with higher mortality 4 and occurs in 22–39% of transplant recipients with active TB. 6
Early detection of TB on SOT recipients can be challenging due to protean clinical presentations. We present the case of a lung transplant recipient with disseminated TB that presented as tenosynovitis, subcutaneous nodules, and hepatic abscesses without evident lung graft involvement. We describe previous cases of tuberculous tenosynovitis in SOT. We highlight limitations in screening of TB in solid organ recipients and donors and the importance of recognizing TB in the differential diagnosis of disseminated infections on immunocompromised hosts.
Case presentation
A 60-year-old White, non-Hispanic woman with history of bilateral lung transplant due to severe emphysema presented with painful occipital subcutaneous nodules and a painful right index finger 60 days post-transplant. Induction immunosuppression consisted of basiliximab and maintenance immunosuppression was prednisone, tacrolimus, and mycophenolate. Histologic examination of native lungs showed changes related to smoking (severe emphysema and bronchiolitis), bronchiectasis, and non-necrotizing granulomatous inflammation suggestive of hypersensitivity pneumonitis. Acid-fast bacilli (AFB) and Grocott Methenamine Silver (GMS) stains were negative for organisms. Two days after transplant, the right lung graft was removed due to lobar torsion and infarction. Histologic examination of explanted graft confirmed infarction and AFB stains were also negative. Patient was discharged home on post-operative day 13. Thirty days post-transplant she had declining pulmonary function tests and was treated for presumed rejection with high-dose methylprednisolone for 3 days and rituximab due to presence of donor-specific antibodies. She developed parainfluenza infection on post-transplant day 54 and received methylprednisolone and intravenous immunoglobulin. Sixty days post-transplant she developed painful nodes in the occipital area and painful swelling of the right index finger. Two weeks later, she presented to clinic and was started on linezolid and ceftriaxone for cellulitis. Shortly after completing a 2-week course of antibiotics, pain worsened and she developed nausea, vomiting, chills, and diaphoresis prompting hospital admission. Social history was notable for residing in Kansas, and never having lived or traveled outside the United States. She had dogs and cats but denied bites or scratches. She denied exposure to livestock or other animals, recent water exposures, remote traumatic injuries, gardening, or other outdoor work. She denied consumption of unpasteurized dairy products. She had no history of incarceration or homelessness.
On examination, she was afebrile and well-appearing. She had two exquisitely tender, non-mobile nodules in her occiput without overlying skin discoloration or drainage. There was no cervical, axillary, or inguinal lymphadenopathy. Her right index finger was edematous and erythematous, with limited range of motion. Right-sided breath sounds were absent and left lung was clear to auscultation. There was no abdominal tenderness, hepatosplenomegaly, or rash. Laboratory studies were notable for leukopenia with 3 × 109 cells/liter with lymphopenia of 0.6 × 109 cells/liter.
Non-contrasted chest computed tomography showed new, numerous hypoattenuating liver lesions. Abdominal magnetic resonance imaging (MRI) showed multiple rim-enhancing hepatic lesions suggestive of multifocal abscesses. Right-hand MRI showed a peripherally enhancing 1.3-cm fluid collection along the palmar aspect of the index finger associated with the flexor tendon sheath. Transthoracic echocardiogram and brain MRI were unremarkable. Extensive infectious work-up remained negative.
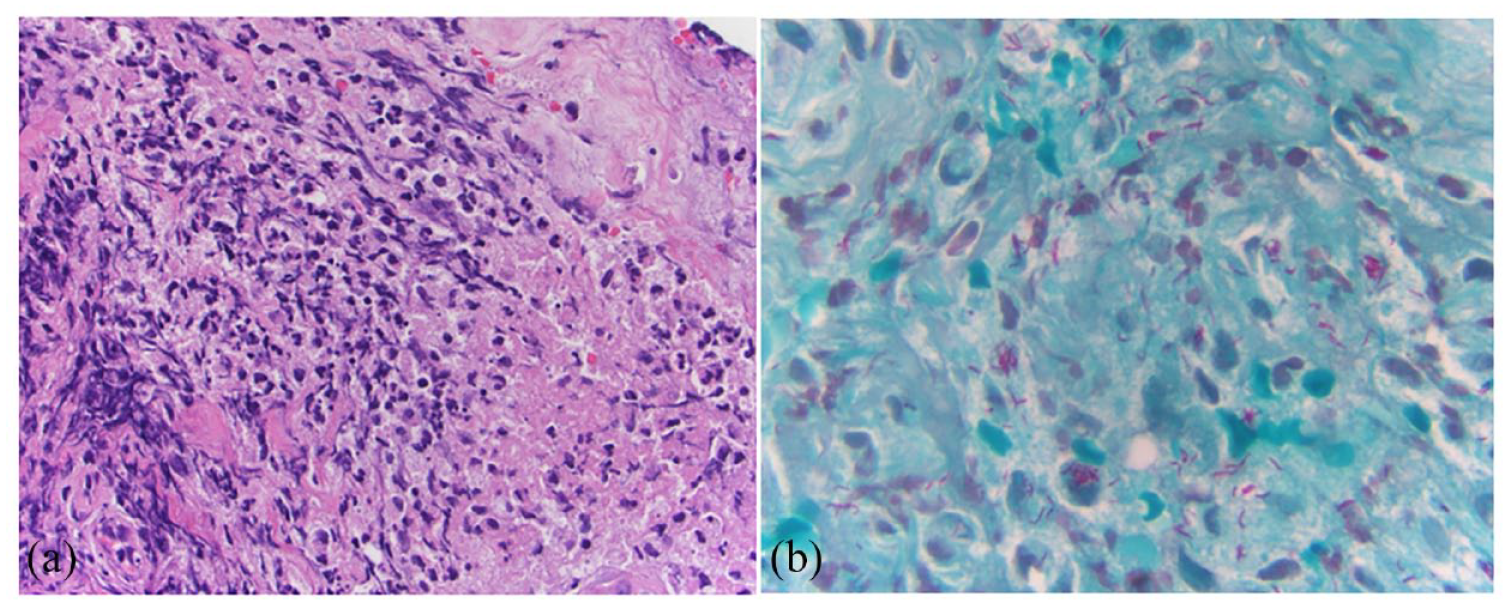
The patient underwent core needle biopsy of liver lesions and finger incision and drainage. Direct staining of finger tissue was positive with 1–9 acid fast bacilli (AFB)/10 high-power field and empiric treatment for disseminated non-tuberculous mycobacteria (NTM) with linezolid, cefoxitin, azithromycin, and moxifloxacin was promptly initiated. Both liver and tendon histopathology noted granulomatous inflammation with AFB (Figures 1 and 2). Finger and liver tissue cultures grew AFB after 4 days identified by matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF) mass spectrometry as
Photomicrographs from the right hand flexor tendon debridement. (a) Acute inflammation and necrosis are present [Hematoxylin & Eosin (H&E), 400×] without granulomatous inflammation. (b) Mycobacteria are present in area of acute inflammation (1000×).

Photomicrographs from a representative rim-enhancing liver lesion. (a) and (b) Liver parenchyma with granulomatous inflammation (H&E, 200×), and AFB stain did not show organisms in these granulomas (not shown). (c) Liver with acute inflammation and no recognizable granuloma (H&E, 400×). (d) Mycobacteria present in area of acute inflammation (AFB, 1000×).
The organ donor was originally from Mexico and did not have known medical history. The cause of death was cerebral hemorrhage. There was no information available regarding prior TB testing or treatment. No other recipients have thus far developed TB.
Discussion
TB represents a difficult infection to diagnose and treat in SOT recipients with grave implications in morbidity including graft failure and mortality. Up to 95% of active TB cases occur within the first-year post-transplant, with 50–76% of these cases involving the lungs.3,7–9 Lung transplant recipients are at greater risk of graft TB infection as the lung is the primary site of infection.3,10–12 Disseminated infection is associated with higher mortality4,13 and occurs in 22–39% of transplant recipients with active TB,6–8 a stark contrast with the relatively low incidence of disseminated infection in the general population.
Diagnosing active TB in patients with SOT is challenging due to the often unusual and non-specific symptoms, leading to a substantial delay in diagnosis compared to the general population.4,7 The most common symptom of active TB in SOT recipients is fever, but cough and weight loss are also seen. Other manifestations of involvement can be esophagitis, sacroiliitis, dactylitis, skin nodules, and laryngitis. 14
The sites of involvement in this case are uncommon for TB, yet tuberculous tenosynovitis has been described as a site for extrapulmonary infection (Table 1). Infectious tenosynovitis of the digits is more frequently associated with NTM or other bacteria typically after traumatic inoculation; hence, in SOT, recipients should prompt biopsy for histopathologic evaluation due to risk of infection with atypical pathogens.
Tenosynovitis from

CPFX, ciprofloxacin; EMB, ethambutol; INH, isoniazid; LTBI, latent tuberculosis infection; LVX, levofloxacin; PZA, pyrazinamide; RIF, rifampin; SOT, solid organ transplant.
The most common mechanism of developing active TB after SOT is reactivation of recipient LTBI. 5 Exposure after transplant is more common in TB endemic areas and transmission from donors is uncommon with <5% of cases. 10 Thirty-six cases of donor-derived TB, of which 16 were lung transplants, have been reported. 6 Some reports note that donor-derived infections (DDIs) tend to occur in the early post-transplant period (within 3 months) and graft involvement is almost universal.5,19 The risk of developing TB post-transplant from a living donor with LTBI is not well established and some studies suggest it may not be significant.20,21
The mechanism of acquisition of infection in this patient is not clear. Recipient pre-transplant screening was notable for a negative interferon gamma release assay (QuantiFERON-TB Gold Plus), absence of risk factors for TB, and residency in a low prevalence area. Native lung histopathology did not have features of infection. Bronchoscopy was not performed post-transplant due to pneumonectomy; however, expectorated sputum cultures were negative for AFB and there was no radiographic evidence of TB in the graft. Right pneumonectomy histopathology (donor lung) did not have features of infection either. These findings raise suspicion for possible donor transmission from latent infection; however, recipient reactivation despite a negative interferon-γ release assay (IGRA) cannot be completely excluded.
Although donor-derived transmission cannot be confirmed in this case, the possibility illustrates the importance of identifying donors at higher risk for TB to reduce DDI, 6 especially in lung transplant recipients. Evaluation for TB in transplant candidates and living donors is well established. Recommendations for deceased donors include relying on review of medical history, exposures, prior IGRA or tuberculin skin test (TST), and radiographic findings. 2 It is unclear whether this clinical information is readily available at the time of organ procurement. Furthermore, TST is not a feasible test for deceased donors, while IGRA lacks performance data in this setting, with relatively high rates of indeterminate results which may be uninterpretable. 22 More studies are needed to understand the risk of transmission of TB from unrecognized LTBI in deceased donors. 10 It is also important to develop screening assays with adequate performance and technical feasibility for deceased donors.
The most common risk factor for DDI is having a donor from an endemic region. 6 Currently, the US TB screening pathway does not specify information about country of origin only citizenship status which may be unhelpful to identify those at higher risk. 23 With increasing mobility of populations globally, the increased diversity of the organ donor pool will result in a changing epidemiology of DDI.
This case highlights the importance of having a high index of suspicion for TB in the early post-transplant period, particularly in lung recipients, even in low-prevalence areas. Clinical manifestations can be largely atypical and unspecific, with dissemination and absence of pulmonary involvement.
