Abstract
Drug-resistant tuberculosis (DR-TB) poses a major threat to control of tuberculosis worldwide. Diagnosis and treatment of DR-TB are considerably more difficult than for drug-susceptible tuberculosis (TB) and require higher level infrastructure and proficiency from laboratory specialists and clinicians. The World Health Organization (WHO) End TB Strategy calls for early diagnosis and initiation of appropriate treatment of all persons of all ages with any form of drug-susceptible TB or DR-TB. This requires ensuring access to WHO-recommended rapid diagnostics and universal drug susceptibility testing (DST) for all persons with signs and symptoms of TB. There are a number of laboratory tools available for diagnosis of DR-TB, including phenotypic culture-based DST as well as molecular methods. Optimal and complementary use of the available diagnostic tools at the different levels of the tiered network of TB laboratories, as well as correct interpretation of the diagnostic results provided by them is critical for accurate and timely diagnosis of DR-TB thus enabling effective treatment and care of patients.
Background
Worldwide in 2015, there were an estimated 10.4 million new incident TB cases. TB remains the leading infectious disease causing mortality with an estimated 1.4 million TB deaths in 2015, and an additional 0.4 million deaths resulting from TB disease among people living with HIV. W02016a) [World Health Organization (WHO), 2016a]. Multidrug-resistant (MDR)-TB and extensively drug-resistant (XDR)-TB are major threats to global TB control. Ending the global TB epidemic will be achievable over the next 20 years only if there is intensive action by all countries that have endorsed the End TB Strategy and its ambitious targets [WHO, 2015a]. It requires a paradigm shift from focused actions that gradually reduce the incidence of TB to enhanced, multisectoral actions that have been shown to drive down the epidemic at a rapid pace. WHO-recommended rapid TB diagnostics and drug susceptibility testing (DST) should be available to all persons with signs and symptoms of TB and no longer only prioritized for persons at greater risk of MDR-TB and-or HIV-associated TB.
MDR-TB is a major global public health problem, and threatens progress made in TB care and prevention in recent decades. Drug resistance in Mycobacterium tuberculosis is caused by selection of naturally occurring genomic mutants. There are two ways that people get drug-resistant-TB (DR-TB). First, acquired DR-TB occurs when TB treatment is suboptimal because of inadequate policies and failures of health systems and care provision, poor quality of TB drugs, poor prescription practices, patient non-adherence or a combination of the above. Second, primary DR-TB results from the direct transmission of DR-TB from one person to another. Globally, an estimated 3.9% of new cases and 21% of previously treated cases had MDR-TB or rifampicin-resistant TB in 2015 [WHO, 2016a]. In 2015, of the 580 000 people new eligible for MDR-TB treatment, only 125,000 (20%) were enrolled. XDR-TB has been reported in 117 countries. About 9.5% of patients with MDR-TB have XDR-TB globally. However, XDR-TB is more common among MDR-TB patients in some countries in eastern Europe [WHO, 2016a].
The End TB Strategy calls for early diagnosis and prompt treatment of all persons of all ages with any form of drug-susceptible TB or DR-TB. This requires ensuring access to WHO-recommended rapid diagnostics and universal access to DST for all patients with signs and symptoms of TB. WHO defines universal access to DST as rapid DST for at least rifampicin, and further DST for at least fluoroquinolones and second-line injectable agents in all TB patients with rifampicin resistance [WHO, 2016b]. The purpose of this article is to review available methods for the detection of TB and DR-TB that can be used at different levels of the laboratory network in high TB burden settings.
In 2016, the diagnostic technology landscape continues to look promising; however, there are no new technologies of suitable accuracy for use at the point of care. The majority of technologies in the pipeline are molecular tests that are being developed for use in reference or intermediate laboratory level only [WHO, 2016a]. The greatest need for new TB diagnostics is for rapid and sensitive tests that can be implemented where sputum smear microscopy is being performed. Until new technologies for diagnosis of TB and DR-TB become available, the use of existing WHO-recommended diagnostic techniques must be accelerated and their use fully optimized. This will require ensuring that laboratories have adequate infrastructure and human-resources capacity. Additionally, there must be clear country-level policies on using these recommended tests in the most effective screening and diagnostic algorithms, depending on each country’s specific TB and MDR-TB epidemiology.
Laboratory methods available in the diagnosis of DR-TB
There are a number of currently available WHO-recommended diagnostic techniques for detection of resistance of M. tuberculosis isolates that are suitable for complementary use at the different levels of the tiered network of TB laboratories. DST determines whether a population of M. tuberculosis bacilli is susceptible to particular anti-TB agents: a result indicating that the strain is susceptible to particular agents means that treatment with those agents will most likely be successful, and a result indicating that a strain is resistant means that there is a high possibility that treatment with those agents will fail and, therefore, other agents should be used. Thus, using standardized and reliable DST for M. tuberculosis provides guidance for treating patients appropriately and effectively.
Phenotypic methods
Phenotypic, culture methods are based on assessment of ability of M. tuberculosis to grow in culture media containing a critical concentration of specific anti-TB agents (which indicates resistance) or, conversely, its inability to grow in the same media (which indicates susceptibility). The critical concentration is generally different from the minimum inhibitory concentration (MIC). The MIC may vary from strain to strain, but the critical concentration is usually higher than the MIC distribution of wild strains of M. tuberculosis. A critical concentration is defined as the lowest concentration of drug that inhibits the growth of wild strains of M. tuberculosis that have never been exposed to TB drugs, while at the same time not inhibiting clinical strains of M. tuberculosis that are considered to be resistant, for example, from patients who are not responding to therapy [Ängeby et al. 2012].
In order to perform phenotypic DST, mycobacteria are initially grown in a variety of solid or liquid culture media. Most frequently used media are Löwenstein–Jensen, Middlebrook 7H10 Agar, Middlebrook 7H11 enriched agar and Middlebrook 7H9 broth. The latter is used as a medium for the mycobacterial growth indicator tube (MGIT) automated M. tuberculosis culture system (Becton Dickinson Diagnostic Systems, Sparks, MD, USA). Bacterial growth on solid medium can be identified visually (i.e. by identifying specific characteristics of the colonies) or in liquid medium by automated detection of fluorescence which indicates a reduction in oxygen tension due to bacterial growth. All positive cultures must be typed to confirm the detection of M. tuberculosis complex (MTBC) and to exclude the presence of any nontuberculous mycobacteria or other bacteria prior to performing a DST.
The indirect proportion method is the most common method using solid media for testing the susceptibility of M. tuberculosis isolates [Canetti et al. 1963]. In this method, a defined inoculum is used to inoculate the drug containing media and two 10-fold serial dilutions of the inoculum are used to inoculate the drug-free control medium. The growth (i.e. the number of colonies) on a control medium without an anti-TB agent is compared with the growth present on a medium containing the critical concentration of the anti-TB agent being tested [Canetti et al. 1963]. Resistance is defined when at least 1% of growth is observed at the critical concentration of drug in the culture medium. Other methods, the absolute concentration and resistance ratio methods require additional controls and the use of a standardized inoculum [Canetti et al. 1963]. As these methods have not been adequately validated for all anti-TB agents, they are currently not recommended for routine use.
Commercial liquid culture systems for DST reduce the time to result to as little as 10 days, compared with the 28–42 days needed for DST using solid media. Because liquid culture systems are more rapid and can reduce the time to detection of resistance, they may contribute to improved patient management [WHO, 2007].
Phenotypic DST for first-line agents (isoniazid and rifampicin) and selected second-line anti-TB drugs (kanamycin, amikacin, ofloxacin, levofloxacin) are generally reliable and reproducible across various settings. New drugs for the treatment of MDR-TB such as bedaquiline and delamanid are recommended for use by WHO under specific conditions and may be added to a core MDR-TB regimen [WHO, 2016e]. Other drugs are being re-purposed (notably linezolid and clofazimine) in the shorter standardized MDR-TB regimen [WHO, 2016e]. It will therefore be necessary to have DST methods developed and validated by the WHO TB Supranational Reference Laboratory Network to allow for the reliable detection of resistance to these drugs. Other anti-TB agents such as the later generation fluoroquinolones (moxifloxacin and gatifloxacin), capreomycin, thioamides, cycloserine and pyrazinamide are becoming increasingly important in the treatment of DR-TB, and there is a need for their critical concentrations to be re-evaluated [Zignol et al. 2016].
WHO has initiated a systematic process to re-assess critical concentrations of anti-TB drugs. It is envisaged that current critical concentrations of selected drugs may be revised, and critical concentrations for the new and re-purposed drugs will be defined in 2017. Evidence is emerging that the detection of resistance-conferring mutations may have better accuracy, at least for some anti-TB agents, such as rifampicin and pyrazinamide. WHO is therefore in the process of reviewing the accuracy of sequencing of different genes associated with drug resistance to inform recommendations for the use of genotypic DST methods.
Molecular (genotypic) methods
Molecular (genotypic) methods detect specific DNA mutations in the genome of the M. tuberculosis, which are associated with resistance to specific anti-TB drugs. Molecular methods have considerable advantages for programmatic management of DR-TB, in particular with regard to their speed, the standardization of testing, their potentially high throughput and the reduced requirements for laboratory biosafety.
Molecular tests for detecting drug resistance to rifampicin alone or in combination with isoniazid have been recommended for use by WHO since 2008. These tests currently include the Xpert MTB/RIF assay (Cepheid, Sunnyvale, CA, USA) and two commercial line probe assays (LPAs), the MTBDRplus assay (Hain Lifescience, Nehren, Germany) and the Nipro NTM + MDRTB detection kit 2 (Nipro Corporation, Tokyo, Japan). LPAs offer the advantage of being able to detect mutations associated with resistance to both isoniazid and rifampicin but are accurate only on sputum smear–positive specimens or cultured isolates of the MTBC. Phenotypic resistance to rifampicin and isoniazid highly correlates with resistance-conferring mutations detected by LPA. Both the Xpert MTB/RIF assay and the LPAs target mutations in the rifampicin resistance determining region of the rpoB gene, which are almost exclusively associated with rifampicin resistance with very high sensitivity [WHO, 2016c, 2013]. The detection of rifampicin resistance–conferring mutations using either Xpert MTB/RIF or LPA compares well with phenotypic DST methods using solid media and appears to be more reliable compared with liquid culture phenotypic DST [Van Deun et al. 2013].
Resistance conferring mutations in inhA and KatG genes account for approximately 90% of isoniazid resistance detected by phenotypic DST methods [WHO, 2016c]. It is important to note that different mutations are associated with different levels (MICs) of resistance to isoniazid. Mutations in the promotor region of the inhA gene are normally associated with low-level resistance to isoniazid and with cross-resistance to the class of thioamides (ethionamide and prothionamide) [Vilcheze et al. 2006; Morlock et al. 2003]. The presence of a katG 315 mutation alone is associated with elevated MICs [Machado et al. 2013; Kambli et al. 2015]. Although resistance associated with katG concerns almost always the same mutation (i.e. Ser315Thr), MIC levels vary considerably, with a mean around 5 µg/ml for the peak serum concentration after a normal dose of the drug. With high dose isoniazid this peak increases proportionally. Only a minority of strains with this mutation then exceeds therapeutically achievable levels [Böttger, 2011]. Mutations outside the hotspot regions are uncommon, but the presence of double mutations in the coding region and promotor regions of the inhA gene has been reported to be associated with high-level isoniazid resistance [Rieder and Van Deun, 2016].
WHO recommends that the Xpert MTB/RIF may be used as the initial diagnostic test for all adults and children with signs and symptoms of TB, rather than microscopy and culture [WHO, 2013]. The Xpert MTB/RIF assay can be performed directly on sputum, processed sputum sediment and selected extrapulmonary specimens from adults and children [WHO, 2013]. Although Xpert MTB/RIF is suitable for use at all levels of the health system, implementation in a diagnostic facility requires stable and uninterrupted electrical supply and a comprehensive implementation plan that addresses the challenges associated with instrument maintenance, training, quality assurance and adequate funding [Kambli et al. 2015]. To overcome the challenge of testing at lower levels of the laboratory network, Cepheid continues to develop a new platform called the GeneXpert Omni. The Omni device is expected to be smaller, lighter and less expensive and suitable for use for point-of-care nucleic acid detection. The Omni instrument is expected to come with a built-in 4-h battery; an auxiliary battery that provides an additional 12 h of run time is also available [WHO, 2016a].
WHO recommends the use of commercial LPAs for the detection of rifampicin and isoniazid resistance in sputum smear–positive specimens (direct testing) and in cultured isolates of MTBC [WHO, 2016c]. The use of LPA in routine care should improve the time to diagnosis of DR-TB especially when used for the direct testing of a smear-positive sputum specimen [WHO, 2016c]. Early detection of drug resistance using LPAs can allow for the earlier initiation of appropriate patient therapy with the potential to improve patient health outcomes. The accuracy of these assays in the testing of sputum smear–positive specimens is very high, with interpretable results achieved in almost 95% of the cases [Böttger, 2011]. In sputum smear–negative specimens, however, accuracy is compromised. As a consequence, direct testing of sputum smear–negative specimens is not recommended [WHO, 2016c].
Recently, WHO has recommended the use of second-line LPAs (SL-LPAs) as the initial test for the detection of resistance to fluoroquinolones and second-line injectable drugs (SLID) for patients with confirmed rifampicin-resistant TB or MDR-TB, instead of phenotypic culture-based DST [WHO, 2016d]. The Genotype MTBDRsl assay (Hain Lifescience) is a commercially available SL-LPA that incorporates probes to detect mutations within genes which are associated with resistance to either fluoroquinolones (gyrA and gyrB genes) or SLIDs (rrs and eis promoter genes). The presence of mutations in these regions does not necessarily imply resistance to all the drugs within a particular class (e.g. the fluoroquinolones), and the extent of cross-resistance between drugs is not completely understood [WHO, 2016d].
The accuracy of SL-LPA is such that a positive result for fluoroquinolone resistance (as a class of drugs) or the group of SLIDs can be treated with confidence in excluding the use of these drugs in MDR-TB treatment regimens. However, when the test shows a negative result, phenotypic culture-based DST may still be needed, especially in settings with a high pre-test probability for resistance to either fluoroquinolones and/or SLIDs [WHO, 2016d].
Xpert MTB/RIF remains the only WHO-recommended diagnostic test that can simultaneously detect TB and rifampicin resistance that is suitable for use at lower levels of the laboratory network. LPA is a high-throughput technology that allows up to 48 samples to be processed simultaneously. The complexity of testing limits the use of this technology to central reference laboratory level or regional level laboratories where the appropriate infrastructure can be ensured. Laboratory facilities for LPA require at least three separate rooms – one each for DNA extraction, pre-amplification procedures and amplification and post-amplification procedures. Restricted access to molecular facilities, unidirectional work flow and stringent cleaning protocols must be established to avoid contamination.
Using diagnostic techniques in a tiered laboratory network
TB laboratory services are typically managed through a national TB reference laboratory (NRL) that may or may not be under the national TB control programme (NTP). When a NRL is managed separately from the NTP, coordination between both entities is essential to ensure that programme priorities and strategies are reflected in the NRL activities and vice versa [Global Laboratory Initiative (GLI), 2015].
The number and distribution of TB laboratories within a diagnostic network at country level may vary depending on government and health system structure, geography, population density, disease burden and economy. The network is usually composed of laboratories with various testing capabilities, dependent on location, infrastructure, diagnostic algorithm used and the particular roles and responsibilities assigned to each specific level of laboratory within the network. The greatest need for early access to TB diagnostic testing is often at peripheral level, while more sophisticated testing such as DST is based at regional or central level facilities. The primary role of the TB laboratory network is to ensure quality and accurate diagnostic services for the entire population, which relies heavily on the implementation of efficient and timely specimen referral mechanisms [WHO, 2015b].
Most countries have three levels of laboratory services within their networks (Figure 1), and different tests are performed at different levels of the network. This stratification is the result of the specialized nature of the technical procedures, the structure of laboratory management and administration and the need for quality assurance mechanisms [WHO, 2015b].
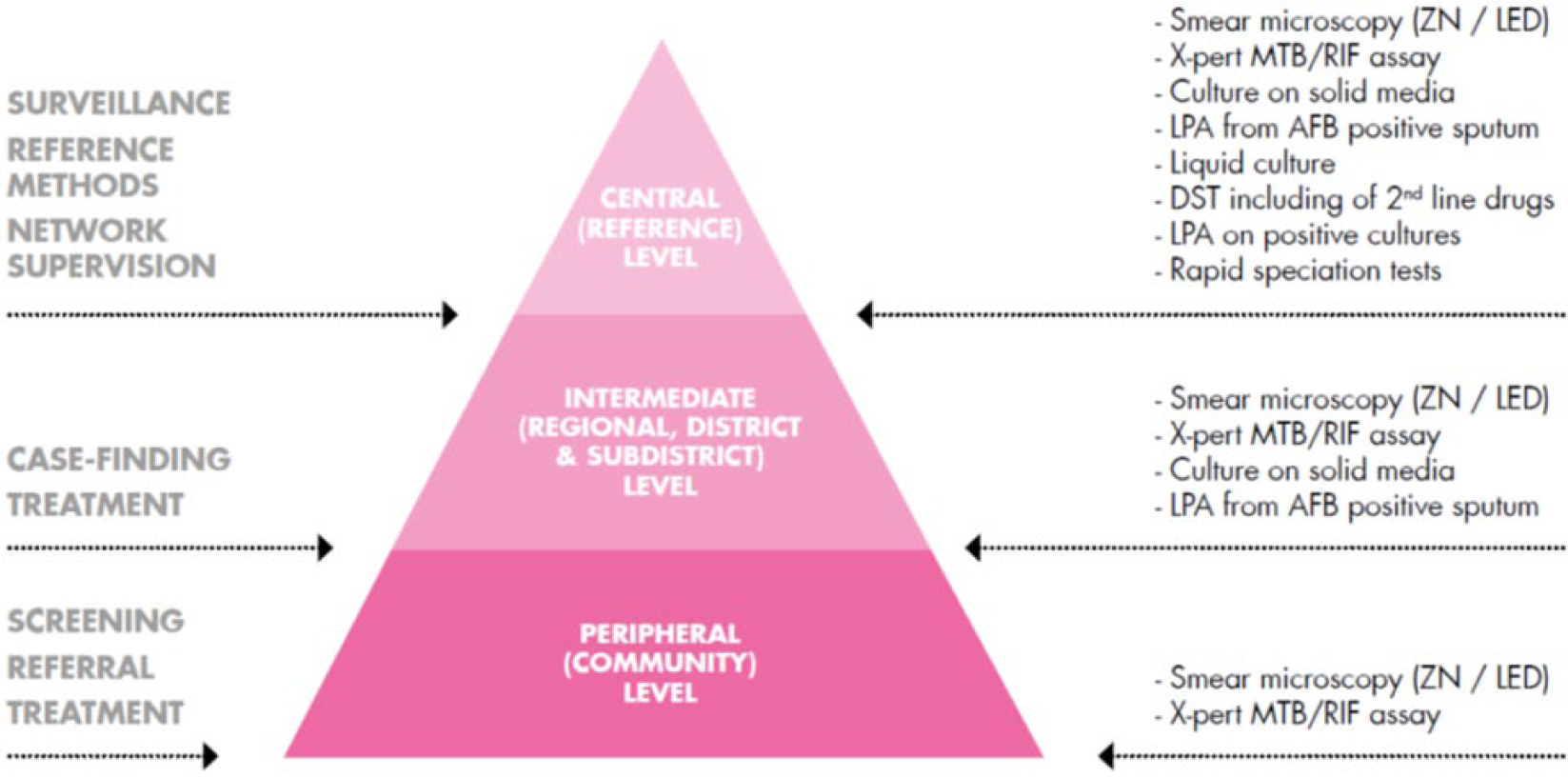
The three tiers of the network of TB laboratories and the responsibilities and the tests offered at each level [WHO, 2015a].
Perspectives for new tools for the diagnosis of DR-TB
The WHO End TB Strategy calls for early diagnosis and universal access to DST. However, while rapid DST tests (e.g. LPAs and Xpert MTB/RIF) are available and recommended by WHO as the initial diagnostic test, they are still not yet scaled-up in many high TB burden countries. Currently, the majority of national TB programmes do not offer universal DST for all persons with signs and symptoms of TB, which results in detection of less than one in four cases of DR-TB [WHO, 2016a]. The reasons for slow scale-up of technologies to detect DR-TB may include inadequate funding, delays in changing and implementing TB diagnostic policies, shortage and high turn-over of qualified human resources, weak laboratory and health system infrastructure, inadequate systems for equipment maintenance and biosafety. To overcome these challenges, stronger political commitment coupled with coordinated health system strengthening efforts, tailored to individual country needs, is essential.
The development landscape for TB diagnostics is robust with many manufacturers developing new diagnostic products [WHO, 2014b; Frick et al. 2016]. New tools using molecular technologies such as nucleic acid amplification tests are the most advanced. Technologies under development include tests to detect TB, drug resistance or TB and drug resistance combined. These include microarray-based multiplexing diagnostic platforms, and next-generation sequencing for the simultaneous detection of a large number of resistance-conferring mutations. Microarrays and next-generation sequencing will increasingly become cheaper and easier to perform, and potentially will become a critical component for the detection of drug resistance as newer drugs and drug regimens emerge from clinical trials [WHO, 2014b]. Unfortunately, most tests currently under development are intended for use at the reference or intermediate laboratory level only, requiring dedicated infrastructure and experienced staff.
There are a few commercially available technologies that have similar biosafety requirements as for sputum smear microscopy and are intended for use at the microscopy level [Frick et al. 2016]. However, available performance data for these tests are limited and highly variable, with no data available from independently conducted multi-centre evaluation and/or demonstration studies in different epidemiological settings [Frick et al. 2016]. These are essential to generate the performance data required by WHO to assess and develop recommendations for the use of new TB diagnostic tests.
Diagnostic manufacturers are increasingly expressing the need to be informed about the type of TB diagnostics they should invest in, as well as the potential market size for these products. A set of high priority target product profiles (TPPs) has been developed as an important step to align the needs of end-users with the specifications and targets that product developers should meet [WHO, 2014a]. The discovery of non-sputum-based biomarker tests for TB diagnosis and treatment monitoring as well as next-generation tests for performing DST at the microscopy level remain urgent priorities.
Conclusion
Greater investment in research and development is urgently needed for the development of new tools that will allow for early diagnosis of TB and universal access to DST. In 2014, less than 20% of the estimated annual need of USD340 million was made available to expedite new tools from a diagnostic pipeline that has largely stagnated [Frick et al. 2016]. Until new tools can be developed and assessed for use at the peripheral level, the implementation of existing WHO-recommended diagnostic tools should be fully optimized if the targets in the End TB Strategy are to be met. Xpert MTB/RIF remains the only WHO-recommended diagnostic test that can simultaneously detect TB and rifampicin resistance that is suitable for use at lower levels of the health system. It is unlikely that new technologies suitable for use at the point-of-care will become available in the next few years; hence, National TB Programmes are encouraged to fully optimize the use of currently available tests for the detection of drug resistance and urgently strengthen the laboratory networks at country level, including effective specimen and patient referral mechanisms.
Footnotes
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
