Abstract
Background:
Lower respiratory tract infections (LRTIs) pose a significant threat to global health, causing more than 2 million deaths worldwide. This menace is intensified by the alarming increase in drug resistance, which limits the availability of effective antibiotics for bacterial respiratory infections. Consequently, there is an urgent demand for alternative therapeutic options. Phage therapy (PT) has re-emerged as a promising therapeutic approach and as an adjunct to antibiotic treatment.
Objective:
This systematic review synthesises the application of PT for LRTIs in humans, providing unified and updated data on the evaluation of the safety and efficacy of PT for LRTIs.
Design:
Systematic review.
Data sources and methods:
Following the PRISMA guidelines, a comprehensive search strategy was carried out (spanning January 2000 – February 2024) in four databases: PubMed, Scopus, ScienceDirect and Web of Science to retrieve published records of PT for LRTIs in humans only. The reference list of each included study was evaluated for possible inclusion of other relevant articles.
Results:
Among the 18 records that fulfilled the inclusion criteria, 70 patients were administered PT. Microbiologically, 71.42% (n = 50/70) of the patients improved; with either the eradication of the pathogen or a decrease in bacterial load, whilst 15.71% (n = 11/70) did not record any improvement. About 5.71% (n = 4/70) recorded a partial/incomplete improvement, whilst 7.14% (n = 5/70) of the patients microbiological outcomes were unspecified. Clinically, up to 74.29% (n = 52/70) of the patients improved, whilst 10.00% (n = 7/70) of the patients showed no improvement. Another 2.86% (n = 2/70) recorded partial/incomplete improvement, whilst 12.86% (n = 9/70) were uncategorized. Phage titres that yielded positive outcomes ranged from 105 to 1012 PFU/mL. Studies that achieved a substantial phage titre at the site of infection frequently observed notable improvements. Regarding the safety of PT, 77.78% (N = 14/18) of the studies did not record any adverse effects after PT was administered, whilst 16.66% (n = 3/18) of the studies reported adverse effects.
Conclusion:
Based on recently published data originating mainly from observational studies, PT has shown considerable efficacy and safety in the treatment of LRTIs. However, there is a lack of uniform methodologies and protocols across different PT cases in the management of LRTIs. Consequently, there is a need for additional clinical studies to establish standardised pharmacokinetic elements and an overall protocol for PT. By doing so, we can fully unlock the potential of PT in effectively managing clinical bacterial infections, including LRTIs.
Keywords
Introduction
Lower respiratory tract infections (LRTIs) are a group of infections that affect the airways and structures below the larynx, including the trachea, alveoli and lungs.1,2 These infections often lead to various respiratory conditions such as bronchitis, pneumonia, tuberculosis, acute exacerbations of Chronic Obstructive Pulmonary Disease (COPD), cystic fibrosis exacerbations and bronchiolitis. The cumulative effects of these conditions make LRTIs one of the leading causes of death and morbidity across all age groups. 3 In 2016, a systematic analysis of the Global Burden of Disease found LRTIs to be responsible for over 2 million deaths worldwide in people of all ages. 3 Furthermore, in 2019, 489 million cases of LRTIs were reported, resulting in 2.5 million deaths. 4 Another systematic analysis revealed that in 2019, there were 1.5 million resistance-related deaths from respiratory infections, making it the most burdensome infectious syndrome. 5 LRTIs are caused by diverse organisms including viruses, fungi and bacteria. Among these aetiological causative agents, bacteria are significant contributors to the disease burden globally, particularly in relation to pneumonia and bronchitis, the most common LRTIs.1,3,6,7 Bacteria that are often responsible for LRTIs and are also known to acquire significant multidrug resistance include Streptococcus pneumoniae, Mycobacterium spp., Pseudomonas aeruginosa, Haemophilus influenzae, Klebsiella pneumoniae, Acinetobacter baumannii and Staphylococcus aureus.8,9
The multitude of bacterial pathogens causing LRTIs is not the sole concern; the escalation of antibiotic resistance among these pathogens has surpassed expectations. Many of these bacteria have become resistant to numerous antibiotics and are developing pan-drug-resistance, exhibiting resistance to multiple drugs, including those considered the last line of defense.10,11 Compounding the issue, these bacterial pathogens can be acquired both in healthcare settings and within the community, indicating our regular exposure to them.12,13 In addition to the antibiotic resistance crises, the slow pace of antibiotic discovery and innovation has intensified the need for alternative antibacterial therapeutics. Consequently, there has been a resurgence of interest in bacteriophage (phage) therapy as a promising approach to combat bacterial infections.
Bacteriophage is a virus and a natural predator of bacteria that infects and replicates using the bacteria’s machinery to produce phage progeny, resulting in bacterial cell lysis. 14 Phage therapy (PT) is a treatment approach that involves the use of a single phage or a phage cocktail (multiple phages) for managing bacterial infections. Currently, PT is primarily limited to compassionate treatment, where it is personalised and tailored to the specific needs of each patient. Among the benefits that influence the adoption of PT is its high host specificity, allowing phages to target and kill specific bacterial pathogens without damaging the normal microflora of the host. 15 The continuous isolation and identification of diverse phages with additional antibacterial enzymatic activities have also contributed to their therapeutic potential, enabling synergistic combinatorial activity of phages with other antibacterial agents, including antibiotics and peptides that elicit greater antibacterial activity in therapeutic applications.16–20 The unique ability of phages to destroy biofilms and re-sensitise bacteria to antibiotics is another phenomenon that contributes to its success in treating bacterial infections.21,22 These immense benefits have influenced the adoption of PT as a compassionate treatment option in some parts of the world and many clinical trials are underway. 23
Although PT has demonstrated effectiveness in preclinical and some clinical applications, its widespread adoption as a standard therapeutic approach requires critical evaluation of several key components. Currently, the application of PT is an experimental endeavour rather than an established clinical practice. This stems from the uncertainties surrounding its key contributing components and the lack of a suitable legal and regulatory framework in Western medicine.23–26 The pharmacokinetics (PK) and pharmacodynamics (PD) of PT are among the significant contributing components for its successful application in clinical settings, although research is insufficient in this area. Defining the PK and PD of active PT involves determining the appropriate phage particle dosage, considering factors such as multiplicity of infection (MOI), adsorption, biodistribution, metabolism, and elimination.23,27,28 Currently, these key components, safety and efficacy data for PT in LRTIs are fragmented across various clinical studies. This systematic review, therefore, synthesises the application of PT for LRTIs among humans, providing an updated understanding of the modifications required for these key components and the general evaluation of the safety and efficacy of PT in the management of LRTIs.
Methods
Search methods
Following the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines (Supplemental Table 2), a comprehensive search for peer-reviewed literature was conducted using four databases: PubMed, Scopus, ScienceDirect and Web of Science. The search strategy involved combining keywords such as “phage therapy,” “management,” “respiratory infections” and “pulmonary infections” using Boolean operators. Adjustments to the search terms were made based on the requirements of each database. The initial search was performed on 13 February 2024, and subsequently repeated on 16 March 2024. The reference list of each included study was evaluated for the possible inclusion of other relevant papers.
Inclusion and exclusion criteria
The review included full-text articles, clinical case series and case reports published in English from January 2000 to February 2024. The included studies encompassed investigations that involved the use of PT for managing bacterial respiratory infections in humans, where reports of patient improvement or deterioration were provided.
We excluded studies that were published in any language other than English. We also excluded in vitro studies, animal studies and studies that were not peer-reviewed. Articles that were published before the year 2000, bibliographic materials such as systematic and narrative reviews, meta-analyses, conferences, book chapters and poster presentations were also excluded. Our study was conducted and reported in consideration of the PRISMA statement. 29
Data extraction
Python v 3.10.10 and Pandas v.2.2.0 were used to deduplicate results obtained from the four databases. Data retrieved from the selected studies included study method, demographics of the study subjects (age and gender of patients), route of phage administration, dosage and treatment regimen (duration of treatment and frequency of dosage), medical history, causative respiratory bacteria involved, use of antibiotics during PT and the characteristics of the phage used.
Quality assessment
The quality assessment of the included studies in this systematic review involved a thorough evaluation conducted by all authors. Given the substantial variation in study designs, including case reports, cohort studies, and clinical cases, the assessment focused on multiple factors. These included transparency of reporting, ethical considerations, an indication of preclinical assessment of phages and the reported outcomes. Additionally, core items from the STROBE checklist 30 that are quite applicable to these study designs were considered (Supplemental Table 1).
Results
Study characteristics
The search for articles published between January 2000 and February 2024 across the four databases resulted in a total of 1625 articles: 1476 in PubMed, 42 in Scopus, 53 in Web of Science and 54 in Science Direct. After deduplication, the number of articles was reduced to 1577. These articles were then screened based on their titles and abstracts, leading to the exclusion of 1538 records. The remaining 39 articles were then subjected to full-text screening to assess their eligibility, and only 16 articles fulfilled the inclusion criteria. Two additional articles were obtained from a manual search. In all, 18 studies fulfilled the inclusion criteria and as such, were included in this review (Figure 1). With regard to the study approach, 72.22% (n = 13/18) of the studies were case reports,31–43 11.11% (n = 2/18) were case series,44,45 5.56% (n = 1/18) was a cohort study, 46 5.56% (n = 1/18) was a retrospective study 47 and 5.56% (n = 1/18) was a clinical trial 48 (Table 1).

A PRISMA flow chart of the study search and study selection.
Studies evaluating the effectiveness and safety of phage therapy in respiratory and pulmonary infections.

Dedrick et al. compassionately administered phages to 20 patients suffering from resistant Mycobacterial disease. It should be noted that 18 patients suffered from respiratory infections and were included in this review, whilst the remaining two patients suffered from skin infections, and thus were excluded.
Samaee et al. carried out a double-blind clinical trial involving 60 patients; 30 of which were administered phages and the other 30 placebos. Results of the patients who were administered the phages were included in this review, whilst results of the patients who were given placebos were excluded.
NS, Not Stated.
Most of the respiratory infections (38.89%) in the studies selected for this review were caused by P. aeruginosa (n = 7/18),35,36,38,39,44,47,48 whilst Achromobacter spp. accounted for 27.78% (n = 5/18)31,32,34,37,43 of the cases. Other pathogens that were identified as causative microorganisms were; Burkholderia multivorans, B. dolosa, A. baumannii, S. aureus (co-infection) and M. abscessus (Table 1).
Phage therapy was administered to 70 patients across the 18 studies. The age range of the patients who received PT in the reviewed studies ranged from 10 to 88 years. However, two studies did not state the exact ages of the patients under treatment.46,48 A proportion of 22.22% (n = 4/18) of the studies were conducted on minors only,31,32,34,37 whilst the remaining studies (77.78%) included patients above the age of 18. Most of the patients (55.56%) had a medical history of cystic fibrosis (n = 10/18).31–34,36,37,43,44,46,47 Other medical conditions reported include COPD, advanced bronchiectasis, pneumonia, interstitial lung disease and primary ciliary dyskinesia (Table 1).
Phage therapy characteristics
Out of the 18 articles, 61.11% (n = 11/18) used phage cocktails to treat patients,31,34,36,37,39–41,44–46,48 whilst 38.89% (n = 7/18) administered either a single lytic phage, or did not state whether a single phage or combination of phages was used32,33,35,38,42,43,47 (Table 2).
Characteristics of phages used in the studies for the treatment of infection.
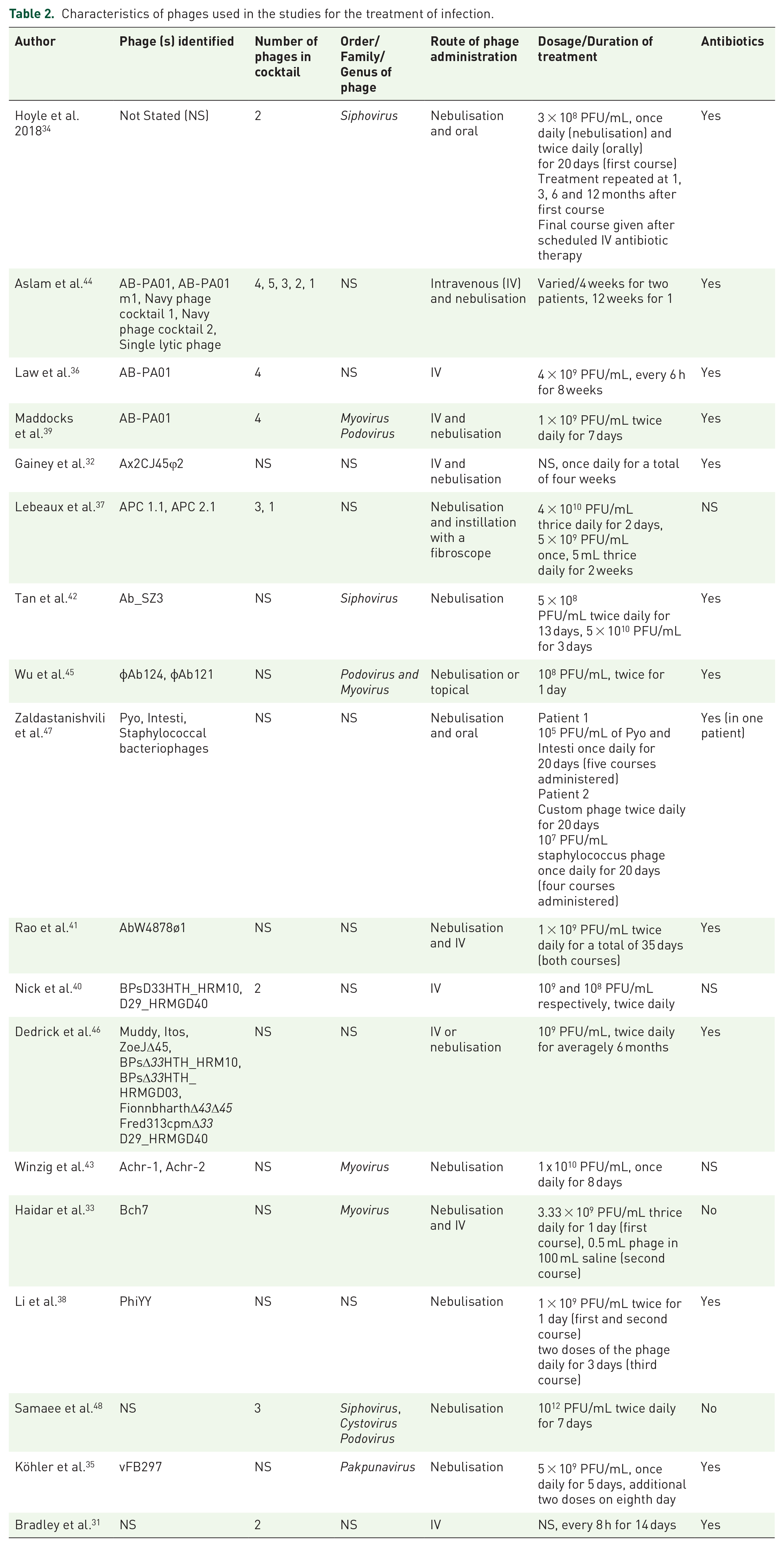
Phages were administered to patients by different means. Nebulisation was the most common method of administration, as it was used in 77.78% (n = 14/18) of the studies.31,33–48 In addition to this method of administration, other methods employed included topical application, oral ingestion, instillation using a fibroscope and intravenous (IV) administration. Patients received the phages through either a single method of administration or a combination of two methods (Table 2).
Phages were dispensed at various frequencies across the 18 studies: once, twice, thrice or four times daily. The duration of PT varied across the studies: ranging from just a day to beyond a year (with several courses administered, not continuously). In some studies, multiple rounds of PT had to be administered to patients due to relapse or lack of improvement after the first round. Phages were also given at different concentrations, ranging from 105 PFU/mL to 1012 PFU/mL (Table 2).
In the majority of the studies (72.22%, n = 13/18) PT was administered alongside antibiotics.31,32,34–36,38,39,41,42,44–47 A proportion of 11.11% (n = 2/18) of the studies stated that they did not administer antibiotics during PT,33,48 whilst 16.67% (n = 3/18) did not specify whether or not antibiotics were given.37,40,43 Antibiotics administered during PT included colistin, piperacillin-tazobactam, tobramycin, trimethoprim-sulfamethoxazole and ciprofloxacin (Table 2).
Efficacy of PT
In the 70 patients treated across the 18 studies, microbiological and clinical improvements were observed in most patients, either after a single or multiple rounds of PT. PT was reported to be successful if the patient’s condition improved microbiologically and/or clinically. Generally, eradication was defined by the authors as the absence of the causative microorganism in all samples taken repeatedly for bacterial culture throughout the follow-up period.32,37–42,48 Partial improvement was generally defined as a reduction in bacterial burden.35,36,43,45,47 Clinical improvement referred to the resolution of signs and symptoms of the patients, which ranged from stabilisation of forced expiratory volume in one second (FEV1)32,34,43 and improved oxygenation 39 to resolution of fever36,48 and decreased inflammation, 44 among others. No improvement meant that there was no reduction in bacterial load or that clinical signs and symptoms were not resolved or did not improve at the end of PT. However, Dedrick et al. 46 stated in their study that fixed definitions of treatment outcomes could not be established, as patients had different underlying conditions with complex infections caused by diverse mycobacterial species.
Microbiologically, 71.42% (n = 50/70) of the patients who underwent PT improved (there was either a complete eradication of the pathogen or a decrease in bacterial load), whilst 15.71% (n = 11/70) did not record any improvement. Only 5.71% (n = 4/70) of the patients recorded a partial/incomplete improvement, whilst it was not stated whether 7.14% (n = 5/70) of the patients improved microbiologically or not. Clinically, 62.86–74.29% (n = 44–52/70) of the patients improved, whilst 10.00% (n = 7/70) of the patients did not. A proportion of 2.86% (n = 2/70) recorded partial/incomplete improvement, whereas 12.86% (n = 9/70) were unaccounted for. It is worth highlighting that some studies reported patient outcomes within a range (eg. 20-28 patients improved), as shown in Table 1. 48
In the majority of the studies (66.67%, n = 12/18), PT was administered in just a single course, with duration ranging from a day to several months, depending on the study.31,34,35,39,40,42,44–48 In the remaining studies (33.33%, n = 6/18), multiple courses of PT were prescribed33,37,38,41,43; either no improvement was observed in the patient, or the patient relapsed after initial/temporal improvement. The initial time of recovery ranged from 3 days to 2 months during or post-PT in 83.33% (n = 15/18) of the studies.31,32,35,36,38–48
Phage resistance was reported in less than half of the studies (44.44%, n = 8/18).35,36,37,38,42,44,45,47 Approximately 38.89% (n=7/18)30–33,38,40,42 did not specify whether phage resistance was encountered, whilst 16.67% (n = 3/18)40,46,48 of the studies reported no resistance. It is plausible that the development of phage resistance could potentially result to fitness trade-offs in the studies where bacteria resensitivity to antibiotics occurred.
Safety of PT
Across all the studies conducted, the safety profile of PT varied from mild side effects to the absence of any adverse effects on the patients. Of the studies reviewed, 5.56% (n = 1/18) of the studies did not report whether adverse effects occurred, 34 while 77.78% (N = 14/18) of the studies did not observe any adverse effects after PT was administered.31–33,36,37,39–44,46–48 However, 16.66% (n = 3/18) of the studies reported physiological changes that might be ascribed to side effects after PT administration.35,38,45 In the first study, one out of the four patients who received PT experienced an atypical cytokine storm 4 h after receiving treatment, which manifested as a transient fever with a dramatic increase in interleukins 6 and 8 (IL-6 up to 596.32 pg/mL and IL-8 up to 112.05 pg/mL). The situation was resolved a day later, but it was not specified whether any intervention was carried out to assuage the problem. 45 In another study, the patient experienced a transient fever post-PT, with a temperature of 38.7°C. After treatment with indomethacin, the temperature returned to normal (36.5°C) within a few hours. 38 In the third study, during the first 2 days of PT, the patient’s oxygen saturation levels dropped, accompanied by an initial rise in C-reactive protein (CRP) and a febrile event. 35
Studies in which patients were followed up after treatment accounted for 61.11% (n = 11/18) of the articles included in this manuscript, with follow-up periods ranging from 1 month to 2 years.31,32,36–40,42–45 In 33.33% (n = 6/18) of the studies, it was not stated whether patients were followed up after PT.34,35,41,46–48 One study involved a single patient who died before treatment ended. 33 As a result, this patient is neither classified as followed-up nor unfollowed up. The death of the patient was attributed to other underlying conditions, such as candidemia and pulmonary aspergillosis.
Phage pharmacokinetics, pharmacodynamics and host immune response
The quantity of phages reaching the target site is not exactly the same as the initial phage titre administered, which is often dependent on the route of administration. 49 In this review, four studies33,35,38,44 recorded a drop in phage titre after PT administration but Tan et al. 42 rather observed an increased in phage titre over time. In one of the patients treated by Aslam et al., 44 the authors were unable to detect any viable AB-PA01 or AB-PA01-m1 phages in serum samples taken approximately 30 min after PT was administered. However, 4 × 107 PFU/mL of phage was obtained from bronchoalveolar lavage (BAL) samples 3 days after AB-PA01 was administered intravenously, and on day 29 after the patient had been administered inhaled AB-PA01. Köhler et al. 35 also presented findings where there was an increase in phage titre after 3 days of PT, and a subsequent reduction in viable phages. Conversely, phage particles were found in BAL fluid samples an hour after treatment with a titre of 5 × 103 PFU/mL, increasing to approximately 107 PFU/mL in 15 days, alluding to in situ phage replication. 42
In relation to the host immune response to PT, Dedrick et al. 46 noticed neutralising antibodies in the serum of 8 out of 15 patients tested but Haidar et al. 33 found no evidence of phage-neutralising antibodies up to a week after phage administration. Nick et al. 40 reported that anti-phage neutralising antibody titres to one phage increased with time, but this did not adversely affect clinical improvement during treatment. Inflammatory markers such as white blood cells, procalcitonin, high-sensitivity CRP, temperature, interleukins and cytokines storms were monitored in some of the patients, with variable results observed.35,38,45 One study reported a transient fever (38.7°C) in a patient, which returned to normal (36.5°C) after treatment with indomethacin. 38 While Tan et al., 42 presented no major changes in these markers, there was a dramatic increase in interleukins 6 and 8 in one of the patients treated in another study. 45
Discussion
This systematic review presents a comprehensive synthesis of the application of PT for LRTIs in humans. In addition to a thorough evaluation of the safety and efficacy of PT for LRTIs, this systematic review further offers an updated understanding of the necessary modifications to the key components of PT. To the best of our knowledge, this review is the first of its kind in this specific area of research. However, it is important to note that the available level of evidence is relatively low, as the review mainly presents results from case series and reports, which are subject to significant publication bias. As such, this should be taken into consideration when interpreting the results.
Efficacy of PT
Based on the findings obtained from 70 patients, PT in the management of bacterial-associated LRTIs produced favourable results, although interpreting the combined results is challenging due to the heterogeneity of the patient’s conditions and varying PT protocols. The 71.42% microbiological and up to 74.29% clinical improvements in our study reiterate the considerable effectiveness of PT. Similarly, results from a recent phase Ib/IIa randomised, double-blind, placebo-controlled, multicentre study investigating the safety and tolerability of nebulised phage (BX004-A) and its effect on P. aeruginosa burden in CF patients, presented at the 47th European Cystic Fibrosis Conference, showed that PT was considered favourable. The mean change in P. aeruginosa burden in sputum was reported to be −1.42 log CFU/g by day 15 after PT, as compared to −0.28 log CFU/g for the placebo group. 50 In this present review, however, several patients (ranging from 2.86% to 15.71%) partially improved or had no improvement across varying courses of administration and dosage. These key findings suggest more in-depth clinical research to fully understand the dynamics of PT. At least 72.22% of the studies used PT as an adjuvant treatment (alongside antibiotics) making it quite difficult to assess the impact solely made by PT. In cases where neither microbiological nor clinical improvement was observed, the lack of success was suspected to result from factors such as the inability to test for phage viability before administration, the severity of patient’s illness (co-infection and co-morbidity), delayed administration of phage, the patients’s underlying health status, 33 the duration of treatment 38 and low phage titre. 44 Similar to these factors, a review by Aranaga et al. 51 on the use of PT in different drug-resistant infections (such as skin, urinary tract infections, bone and gastrointestinal infections) also attributed the failure of PT to low phage concentrations, co-infections with other species of bacterial strains and deficiencies in the modes of phage administration.
Phage delivery methods
The methods of phage delivery have a significant impact on the success of PT, 52 as their ability to lyse bacterial cells depends on their stability and direct contact with the bacteria within the respiratory environment. The systemic administration of phages via IV can result in phage accumulation at infection sites, including the lungs. 44 On the contrary, several other studies found low phage titre at infected sites following IV and other systemic phage administration, due to low phage bioavailability. This, therefore, suggests that deciding on the phage administration route to use should be based on preclinical tests of that specific phage to identify the most potent route for patients. Nebulisation was the method of delivery in 77.78% (n = 14/18) of the studies in this review. Nebulisation (which involves the conversion of liquid phage suspensions into aerosols) is currently the most used method of conveying phages to the respiratory system. It has been suggested to be highly efficient due to its ability to distribute phages and to penetrate deeply into the respiratory environment.53,54 However, the efficacy of nebulised phages depends on several factors and presents several challenges, such as loss of phage titre and viability.55–57 Reduction in phage titre during nebulisation may occur due to changes in pressure, exposure to the components of the nebuliser and shear force, 58 which can result in decreased efficacy of PT.56,57 Dry powder inhalation has been suggested to be more effective than liquid suspensions 59 due to ease of transport, storage and administration. In an in vivo study conducted by Chang et al. 60 to demonstrate the efficacy of a dry powder phage formulation against MDR P. aeruginosa in a mouse lung infection model, it was observed that the bacterial load in the lungs decreased significantly by 5.3 log10. The authors concluded that it is possible to use dry-powder formulations for the treatment of lung infections caused by P. aeruginosa, however, more clinical trials must be carried out in this regard. Despite significant advancements, there is a lack of comprehensive data on how effectively phages are deposited and distributed on the bronchial tree after nebulisation. This knowledge is crucial for ensuring that phage titre upon reaching these deep, infected areas is sufficient to exert adequate therapeutic effects. 61 Extensive research is needed in this area to accurately predict PT pharmacodynamics (treatment outcomes) and determine optimal phage dosage to achieve maximum results in the treatment of respiratory and pulmonary infections.
Phage pharmacokinetics, pharmacodynamics and host immune response
Active PT, just like many other therapeutic interventions, is determined by key variable components, specifically pharmacokinetics and pharmacodynamics. However, standardisation of these factors is challenging, as their exact mechanisms are not well understood or are sometimes unpredictable. 62 In this review, four studies32,35,37,43 recorded a drop in phage titre after the administration of phages to patients via nebulisation and intravenous therapy. These findings are consistent with that of Petrovic Fabijan et al., 63 who also recorded a gradual decrease of phage titre within days after intravenous phage administration in individuals with severe S. aureus bloodstream infections. In a study where 108 PFU/mL in 10 mL 0.9% NaCl was initially administered through nebulisation, the authors reported a ~10,000-fold reduction in phage load in sputum within a day of PT, and phages could not be detected after 48 h. This suggests a rapid, metabolism, and elimination of phages in the patient. 38 Furthermore, Li et al. reported recurrent episodes of bacteria that remained susceptible to the phage (phiYY), indicating incomplete efficacy in delivering the therapeutic dose to the target site. Additionally, not all the phages present at the target site may adsorb onto the target bacteria, potentially leading to a significant reduction in the MOI and burst size. The rapid clearance of phages also contributed to the reemergence of bacteria, despite their continued susceptibility to the phage treatment. 38 In a much more successful session of PT, phages reached a titre of 5 × 103 PFU/mL in blood and bronchoalveolar lavage fluid an hour after nebulised treatment and gradually increased to a relatively high titre of 107 PFU/mL in 15 days. 42 These heterogenous reports suggest the disproportionate biodistribution of phages and their activity largely depends on the route of administration, and factors such as MOI and adsorption. It is critical to fully understand how phages are adsorbed, distributed and metabolised in human systems. These PK and PD data can be used to improve dosing regimens to ensure that phages reach their intended target organ, thus increasing the effectiveness of PT. 64 The immune response of the host towards PT usually relies on the type of infection and the phage administered. In the context of PT for the management of LRTIs, the phage-induced immune response may include the production of phage antibodies and the triggering of inflammation markers such as white blood cells, increased levels of procalcitonin, high-sensitivity CRP, interleukins (6 and 8), cytokine storms, and elevated temperature resulting in transient fever. Some of these immune responses impact pathogen clearance in infected sites. Not only does PT induces these markers, but many studies have also identified that the administration of phages elicits both innate and adaptive immune responses. Innate responses involve the activation of immune cells such as macrophages, neutrophils, and the complement system, which enhance antimicrobial activity. Adaptive responses, on the other hand, include the production of phage-neutralising antibodies and T cell activations.23,62,65,66 The production of these antibodies can vary depending on factors such as the route of phage administration, duration of therapy, site of infection, and the functionality of the host immune system. However, there are other cases where no antibody production occurs in response to phages. 67 Although some studies have shown that PT can trigger a phage-specific humoral immune response, the mechanisms regulating this response and the impact it may have on the success of PT are not well understood.68,69 Conflicting arguments have been made concerning whether phage-neutralising antibodies indeed influence patient outcomes,70,71 which is consistent with results obtained in this review. In this present study, there was no relationship between neutralisation and patient outcomes in eight patients who underwent treatment, whilst in three patients, it was determined that the presence of these neutralising antibodies possibly led to reduced efficacy of PT. 46 Nevertheless, the host immune response is not exclusively induced by phages; bacterial and toxin contamination during phage formulation, as well as lysis-induced endotoxin release, all contribute to immune response.72–74
The time of recovery after PT varies from patient to patient, depending on several factors, such as the type and severity of the underlying infection, follow-up care and monitoring, complications that may arise from the intervention, and the individual’s immune system function. More than half of the studies (83.33%) reported initial recoveries ranging from a few days to 2 months in some patients during or post-PT, although some patients relapsed afterward and had to undergo treatment again. Documenting specific times of recovery is pertinent for treatment evaluation, monitoring the patient’s progress and making well-informed clinical decision, and as such should be encouraged.
Phage-antibiotic combinations
The administration of phages in combination with antibiotics has been reported in many studies.19,75,76 These phage-antibiotic interactions can result in antagonistic, additive, or synergistic effects depending on the type of phage and antibiotic used. 19 The most effective combinations can be determined by synography, a process that involves using synograms to test various concentrations of a phage and an antibiotic against a bacterial strain to assess their interactive effects. 77 Among the phage-antibiotic combination effects, synergy has been noted to be highly effective in many studies, resulting in better patient outcomes, compared to using phages or antibiotics as independent treatments. Synergy leads to the repression of resistant cells and re-sensitisation of previously resistant bacteria; however, it is dependent on factors such as the physiologic environment of the host. 77 In this review, at least 72.22% of the studies administered antibiotics alongside PT, which may be a reason for the observed success. It is worth highlighting that in one of the two studies where antibiotics were not used, there was a microbiological improvement after a second course of PT, but the patient succumbed to the infection, which was attributed to other underlying conditions. 33 These observations reiterate the significance of the combined use of phages and antibiotics that have synergistic effects. The noteworthy effects of phage-antibiotic combinations against various bacterial infections have been reported in other studies. Chan et al. reported the treatment of a P. aeruginosa-infected aortic Dacron graft associated with an aorto-cutaneous fistula using ceftazidime and a single course of phage OMKO1. Interestingly, the authors discovered that phage OMKO1 was not only able to treat the infection without any recurrence but also resensitised the bacteria to antibiotics they were previously resistant to. 78
Phage resistance
Bacteria have developed numerous mechanisms to resist phages, which is a major challenge as it reduces the efficacy of PT.79,80 However, resistance may be mitigated by using phage cocktails rather than just a single phage during PT. Eleven out of the eighteen studies used cocktails during PT, and phage resistance was observed during PT in less than half of those studies (45.45%).36,37,44,45,47 Nonetheless, PT has also been observed to be successful despite the presence of some resistant strains in another study. 81 This success is purportedly due to the patients’ immune systems playing a much greater role in the overall effectiveness of PT. 51 In this present review, the efficacy of PT, despite the occurrence of phage resistance in some of the studies, may be explained by incomplete sampling. During the analysis of bacterial populations, it may not be possible to detect every strain present due to sampling limitations, especially in environments that are known to have high bacterial numbers, such as the lung.
Phage concentrations
The efficacy of PT depends on the concentration of phage used. The quantity of phages reaching the target site is not necessarily the same as the initial phage titre administered; lower phage concentrations may lead to undesired outcomes and the development of phage resistance. In this present review, the concentration of phages that yielded positive outcomes among patients with LRTIs ranged from 105 PFU/mL to 1012 PFU/mL. This range of phage concentration aligns with Osman et al., 19 who reported an active concentration of phages ranging from 106 PFU/mL to 1011 PFU/mL for topical and intravenous use of phages at other sites of infections. In a study published in 2019, Jault et al. used a low concentration of phages (102 PFU/mL) to treat burns patients who were infected with P. aeruginosa during a Phagoburn trial. It was observed that the patients recovered very slowly, as compared with another group that received only standard care, thus PT was deemed quite inefficacious at lower phage concentrations. 81 The likelihood that the administration of low titres of phages would result in unsuccessful treatments has also been confirmed by other similar studies.82,83
Limitations
This comprehensive systematic review has certain limitations, primarily stemming from high heterogeneity across the included studies. The articles encompassed a variety of respiratory infections by bacteria and involved patients with diverse medical backgrounds and demographic profiles, which introduces challenges in drawing definitive conclusions regarding the efficacy of PT in managing respiratory infections. Furthermore, the heterogeneity of the data posed difficulties in conducting statistical analyses, resulting in primarily descriptive interpretations of the findings. Another limitation of this study is the inclusion of only articles published in English. This may overlook significant studies published in other languages, especially from regions in Eastern Europe and Russia. As such, critical findings that are relevant to this present review may be missed. Additionally, many of the studies did not state the specific timelines for when the patients fully recovered, which made it challenging to provide the stipulated time for successful treatments. It is also worth noting that, because many of the studies were case reports and the application of PT was strictly among critically ill patients, this may introduce bias in the overall outcome of the evaluation of PT. Another limitation was the assesment of the quality of the included articles. Even though the STROBE checklist is more applicable to cohort, cross-sectional and case–control studies, we applied it to other study designs to enable us to evaluate the quality of the included papers.
Conclusion
It is evident from this review that PT demonstrates successful outcomes with considerable efficacy and safety in managing LRTIs. However, current evidence is somewhat limited as available data is primarily observational and heterogenous. Additionally, there is a lack of uniform methodologies and protocols in various PT cases in managing LRTIs. Therefore, there is a need for further clinical studies to establish standardised pharmacokinetic parameters and an overall protocol for PT. These studies should preferably be conducted on individuals without underlying medical conditions, allowing us to fully exploit the potential of PT in effectively managing clinical bacterial infections, including LRTIs.
Supplemental Material
sj-docx-1-tai-10.1177_20499361241307841 – Supplemental material for Phage therapy in the management of respiratory and pulmonary infections: a systematic review
Supplemental material, sj-docx-1-tai-10.1177_20499361241307841 for Phage therapy in the management of respiratory and pulmonary infections: a systematic review by Patience Sarkodie-Addo, Abdul-Halim Osman, Bill Clinton Aglomasa and Eric S. Donkor in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
