Abstract
Background:
A predictive scoring system used in patients with acute respiratory distress syndrome (ARDS) known as Age, PaO2/FiO2, Plateau Pressure Score (APPS) has been externally validated to predict mortality in Asian and Caucasian populations. Its performance in Latin American and COVID-19 patients still needs to be done.
Objective:
To assess the association between APPS and mortality, as well as APPS performance in predicting mortality within 28 days of intensive care unit (ICU) admission in patients with ARDS due to COVID-19.
Design:
Analytical observational retrospective cohort study.
Methods:
We conducted a retrospective cohort study in patients diagnosed with ARDS according to the Berlin criteria who were admitted to the ICU. We evaluated the prognostic performance of APPS using the area under the receiver operating characteristic curve (AUC-ROC), and association with mortality was measured using the Cox proportional hazards regression models.
Results:
A total of 271 patients were analyzed. The AUC for 28-day mortality was 0.78 (95% CI: 0.73–0.84). In the Cox proportional hazards model, after adjusting for sex and categorized Charlson Comorbidity Index, it was found that grade 2 APPS (aHR: 3.67, 95% CI: 2.14–6.30, p < 0.001) and grade 3 APPS (aHR: 7.95, 95% CI: 3.72–17.02, p < 0.001) were associated with a higher hazard of 28-day mortality.
Conclusion:
The APPS scoring system has a good prognostic performance for mortality in patients diagnosed with COVID-19-induced ARDS.
Plain language summary
When someone is severely ill with COVID-19 and has trouble breathing, doctors often use a score called APPS to predict how likely they are to survive. The APPS score looks at three things: the patient’s age, their oxygen levels (measured by the PaO2/FiO2 ratio), and the pressure needed by a ventilator to help them breathe (plateau pressure). We studied whether this score works well in predicting the survival of patients with COVID-19 who were admitted to intensive care units. By reviewing the medical records of 271 patients, we found that the APPS score is quite good at predicting who might not survive the next 28 days. Patients with higher APPS scores had a greater risk of dying within this period. This finding helps doctors better understand a patient’s condition and possibly tailor their treatment more effectively.
Introduction
Acute respiratory distress syndrome (ARDS) is characterized by diffuse alveolar damage, which leads to pulmonary edema secondary to increased endothelial permeability to fluids and proteins. 1 Consequently, rapid progressive respiratory failure ensues, accompanied by refractory hypoxemia, and manifested by radiological alveolar infiltrates, resulting in the need for mechanical ventilation and high mortality rates. 2 Since Ashbaugh et al. 3 first described this syndrome diagnostic criteria for ARDS have evolved over time, 4 culminating in the current Berlin criteria established in 2012. 2
Prior to the official declaration of the COVID-19 pandemic by the World Health Organization, 5 the incidence of ARDS ranged between 10 and 86 cases per 100,000 people. 6 It accounted for 10.4% of admissions to Intensive Care Units (ICUs) and occupied 42% of ICU beds for at least 4 weeks. 7 In the context of COVID-19, studies have shown that 12% of all individuals diagnosed with the virus required ICU admission.8,9 Similarly, between 67% and 85% of individuals with SARS-CoV-2 pneumonia admitted to the ICU developed ARDS.10,11 This is concerning, as ARDS serves as a significant barrier to patient recovery, 12 with an in-hospital mortality rate of 20%–40% within the first 28 days of ICU admission in European countries.7,13–16 In Latin America, ARDS is a common reason for ICU admission, 17 with estimated 30-day ICU mortality rates reaching up to 48%. 18 In a multicenter study in Peru, a 90-day mortality rate of 55.6% was reported for patients who developed ARDS in the ICU. 19
Several scoring systems exist for assessing the severity of a patient in the ICU. For instance, the Acute Physiology And Chronic Health Evaluation II (APACHE-II) scoring system is frequently used to predict mortality. 20 Other systems such as SOFA and SAPS-II have also been utilized to evaluate disease severity and predict outcomes.21,22 However, these systems assess the general condition and are not disease-specific. For ARDS, there are more targeted scoring systems that may more accurately predict mortality risk, such as Murray’s Lung Injury Score (LIS), 23 and the PaO2/FiO2 severity grades based on the Berlin criteria. 2 Nonetheless, Murray’s score has been criticized for its lack of specificity and adequate validation, 24 and although the Berlin criteria are validated for ARDS diagnosis, some authors argue that they lack inclusion of other important parameters. 25
The Age, PaO2/FiO2, and Plateau Pressure Score (APPS) introduced by Villar et al. in 2016. 26 This score is applied 24 h after ARDS diagnosis in the ICU to determine the severity and predict 28-day mortality risk. The score is out of 9 points and requires only three clinical data points, making it simpler and quicker to use than other scoring systems. According to the score, patients can be stratified into three categories of escalating risk; scores of 3–4 indicate mild risk, 5–7 moderate risk, and 8–9 severe risk, corresponding to estimated mortality probabilities of 7.5%, 40.9%, and 90.1%, respectively. 26
Some researchers like Hwang et al. 24 have evaluated APPS with satisfactory results, although others have concluded that the system has an acceptable performance in predicting mortality. 27 It is important to note that APPS has only been applied in Anglo-Saxon and Asian populations, with etiologies for ARDS that differ from SARS-CoV-2.24,26,27 Therefore, extrapolating these findings to COVID-19-related ARDS and to the Latin American population is not feasible. Additionally, evaluating the prognostic performance of this simple and quick score would facilitate its consideration for clinical practice and, potentially, the timely identification of ARDS patients at high risk of mortality. For this reason, the aim of this study was to assess the prognostic performance of APPS for 28-day mortality in patients with COVID-19-related ARDS admitted to the ICU.
Methods
Study design and context
A retrospective cohort study was conducted, which included patients admitted to the ICU of Clínica San Pablo, located in Lima, Peru, between March 2020 and December 2021. This private clinic’s ICU has duly accredited intensivist physicians and nurses. During the COVID-19 pandemic, it offered 21 fully-equipped beds exclusively for treating patients with ARDS due to SARS-CoV-2. The physician-to-patient ratio was 1:6 (exceptionally 1:7), and the nurse-to-patient ratio was 1:2, although at times it had to resort to a 1:3 ratio. This allowed for rigorous and continuous monitoring of vital functions and ventilation parameters, with records taken every hour in the patient’s medical history from the time they were admitted to the ICU. The clinic is also accredited by the Joint Commission International, and all its patients have electronic medical records, from which the necessary data for the study were obtained.
A protective mechanical ventilation protocol, defined as 4–8 mL/kg predicted body weight was applied to all the patients in the ICU. To assure the ideal range of tidal volume, a volume-controlled ventilation mode was initiated in every patient and maintained in most of them, only switching to pressure-controlled ventilation if there was a risk of ventilator-induced lung injury. Such risk was implied if an elevation of either peak or plateau pressure was detected. Cutoff values to decide a switch to pressure-controlled ventilation were >32 cm H2O for plateau pressure and >35 cm H2O for peak pressure.
Study sample
A sample size calculation was performed for the main AUC-ROC test for reference purposes. The minimum sample size required was 218 patients. A database was constructed from ICU patients who met the selection criteria. The inclusion criteria used in this study were: ARDS diagnosis based on the Berlin criteria performed by attending physicians from the intensive medicine service, a positive result from a molecular test for SARS-CoV-2, and complete data for the variables of interest. A patient with an ICU stay of less than 24 h or missing data in regards to APPS score parameters or ARDS diagnostic criteria resulted in exclusion. Follow-up censoring was applied if any patient either died after the 28-day observation period ended or was discharged from the ICU within this time interval.
Outcome
The outcome was mortality. Any cause of death within 28 days of ARDS diagnosis in the ICU was considered.
Age, PaO2/FiO2 and Plateau Pressure Score
The APPS score was analyzed with a scale ranging from 3 to 9, stratified by severity: 3 to 4 (mild), 5 to 7 (moderate), and 8 to 9 (severe). The score was calculated based on age ranges, PaO2/FiO2, and Plateau pressure, as proposed by Villar et al. (Table 1). 26 The calculation used the PaO2/FiO2 and Plateau pressure closest to 24 h after admission to the ICU.
Age, PaO2/FiO2 and Plateau Pressure Score.

The total score is equal to the sum of the scores of each tertile category, based on the values obtained 24 h after the diagnosis of acute respiratory distress syndrome.
Other variables
Baseline characteristics were also collected, including sociodemographic features and patient comorbidities, using the Charlson Comorbidity Index (CCI). The CCI was categorized into four severity levels with scores of 0, 1–2, 3–4, and ⩾5.28,29 Length of UCI stay within the follow-up period and the length of hospital stay from hospital admission to hospital discharge or death were also recorded. Likewise, arterial blood gas analysis data, mechanical ventilation indicators, and adjuvant therapies such as proning were collected.
Statistical analysis
Data description was expressed as mean with standard deviation, median with 25–75 percentiles (p25–p75), or frequencies and percentages, as appropriate. The normality of numerical variables was assessed by observing skewness, kurtosis, quantile plots, and histograms, and then applying the Shapiro–Wilk test. Data were grouped by 28-day outcome after ICU ARDS diagnosis into survivors and non-survivors, and according to their APPS severity. Bivariate analysis was performed to identify statistically significant differences for each group. These baseline characteristics were evaluated using Pearson’s Chi-square or Fisher’s exact test for categorical variables, and Student’s T-test, Mann–Whitney U test, ANOVA, or Kruskal–Wallis test for numerical variables, depending on the type of variable and assumption adherence. ROC AUCs were constructed to evaluate APPS performance, considering mortality at 28 days as the outcome. Additionally, survival curves were constructed according to APPS severity using the Kaplan–Meier method, compared using the log-rank test.
To evaluate the association between APPS and mortality, crude Hazard Ratios (HRc) were initially calculated through Cox regression models. For calculating adjusted Hazard Ratios (HRa), an epidemiological approach was followed. Confounding variables, such as sex and categorized CCI, were included in the multivariable model. Age was not included in the regression model as it is a component of APPS. Compliance with the proportional hazard assumption was evaluated using the log-log plot of survival and the Kaplan–Meier and predicted survival plot. The presence of multicollinearity was assessed with the correlation coefficient matrix. All estimates were presented with their respective 95% confidence intervals (CI 95%), and p-values <0.05 were considered significant.
Ethics
The present study received approval from the Ethics Committee for Clinical Research of Clínica San Pablo (No. 015/UID/CHSP-2021), as well as from the Ethics Committee of the Universidad Peruana de Ciencias Aplicadas (FSC-CEI/728-11-20). The ethics committees waived the need for informed consent because data extraction was performed directly from the electronic medical records retrospectively and no patient contact was made. The protocol was also indexed in the Peruvian health research registry-PRISA (EI00001614) and was conducted according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material).
Results
General characteristics
Figure 1 outlines the selection process of the 271 patients (140 survivors and 131 deceased) who were ultimately included in this study. The 74.2% (n = 201) were male, and the mean age was 58.8 ± 13.3 years (Table 2). Moreover, the mortality rate was assessed based on the APPS score for each specific group of patients. No deaths occurred in the group with a score of 3, whereas 23.5% (n = 16) of those with a score of 4 passed away. The mortality rate increased progressively: 46.3% (n = 38) for a score of 5, 69.1% (n = 38) for a score of 6, 84.4% (n = 27) for a score of 7, 80% (n = 8) for a score of 8 and reached 100% (n = 4) for a score of 9 (Figure 2).

Study flowchart.
General characteristics of survivors and non-survivors with acute respiratory distress syndrome (n = 271). a
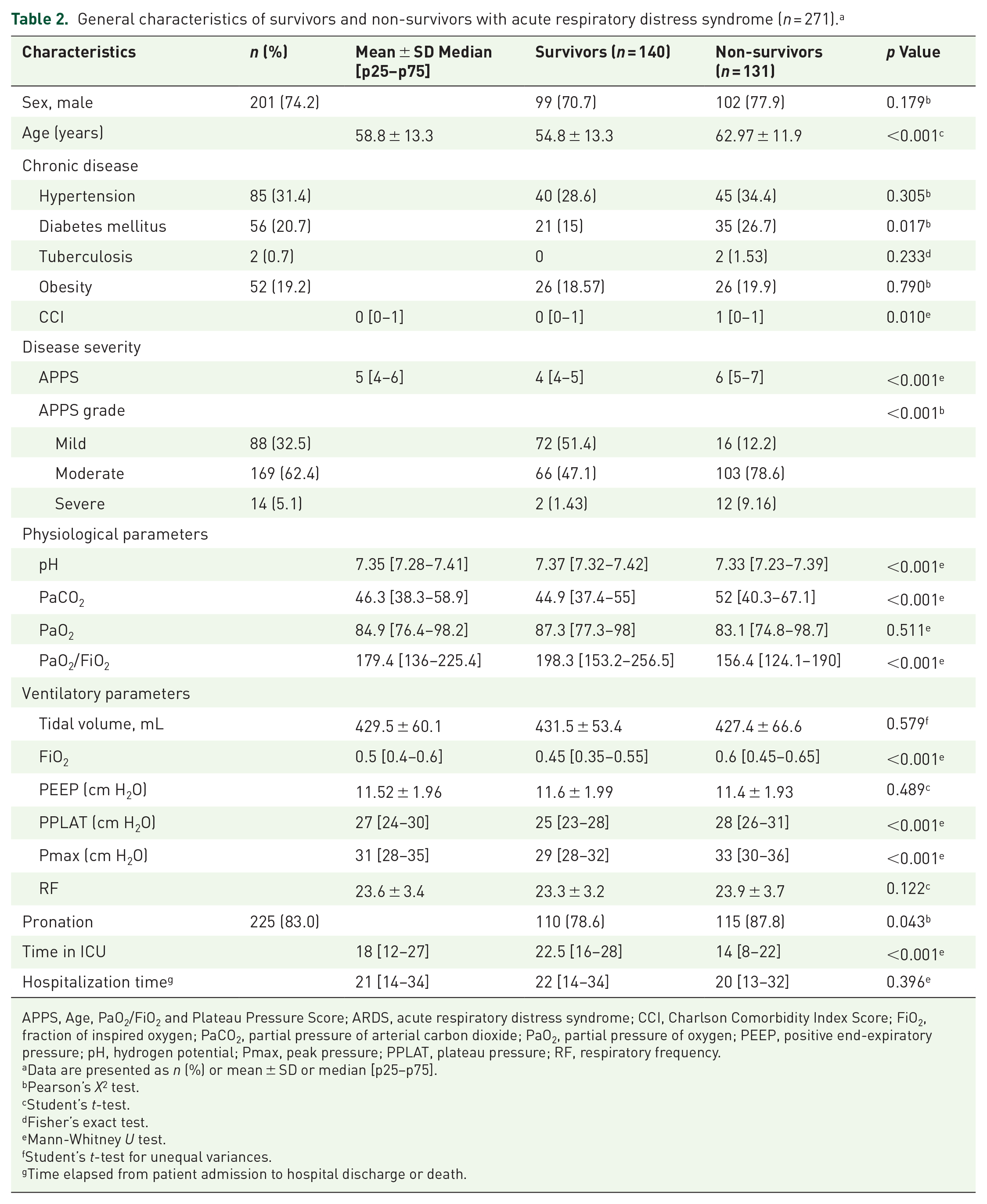
APPS, Age, PaO2/FiO2 and Plateau Pressure Score; ARDS, acute respiratory distress syndrome; CCI, Charlson Comorbidity Index Score; FiO2, fraction of inspired oxygen; PaCO2, partial pressure of arterial carbon dioxide; PaO2, partial pressure of oxygen; PEEP, positive end-expiratory pressure; pH, hydrogen potential; Pmax, peak pressure; PPLAT, plateau pressure; RF, respiratory frequency.
Data are presented as n (%) or mean ± SD or median [p25–p75].
Pearson’s X2 test.
Student’s t-test.
Fisher’s exact test.
Mann-Whitney U test.
Student’s t-test for unequal variances.
Time elapsed from patient admission to hospital discharge or death.

Mosaic plot of mortality and survival according to APPS score.
Characteristics of participants according to 28-day mortality
Age was significantly higher in those who died (62.97 ± 11.9, p < 0.001). Likewise, patients with diabetes mellitus (p < 0.001) and those with severe APPS (p < 0.001) had significantly higher mortality. In addition, the overall APPS score, the CCI score, PaCO2, FiO2, mechanical ventilation parameters, plateau pressure, and peak pressure (Pmax), were significantly higher in individuals with a mortality outcome (p < 0.001). Conversely, pH levels, the oxygenation index (PaO2/FiO2), and ICU length of stay were significantly lower in those who died (p < 0.001) (Table 2).
Characteristics of participants according to APPS severity
Patients with diabetes mellitus (26.2%, p = 0.007) and arterial hypertension (39.6%, p < 0.001) more frequently had a grade 2 APPS. pH levels (p = 0.002), PaO2 (p = 0.003), PaO2/FiO2 (p < 0.001), and tidal volume (p = 0.021) were significantly lower in grade 3. On the other hand, age (p < 0.001), plateau pressure (p < 0.001), and peak pressure (p < 0.001) were higher in grade 3. Lastly, time in ICU (p = 0.048) was significantly longer in grade 1 (Table 3).
Baseline characteristics according to the degree of severity of APPS (n = 271). a
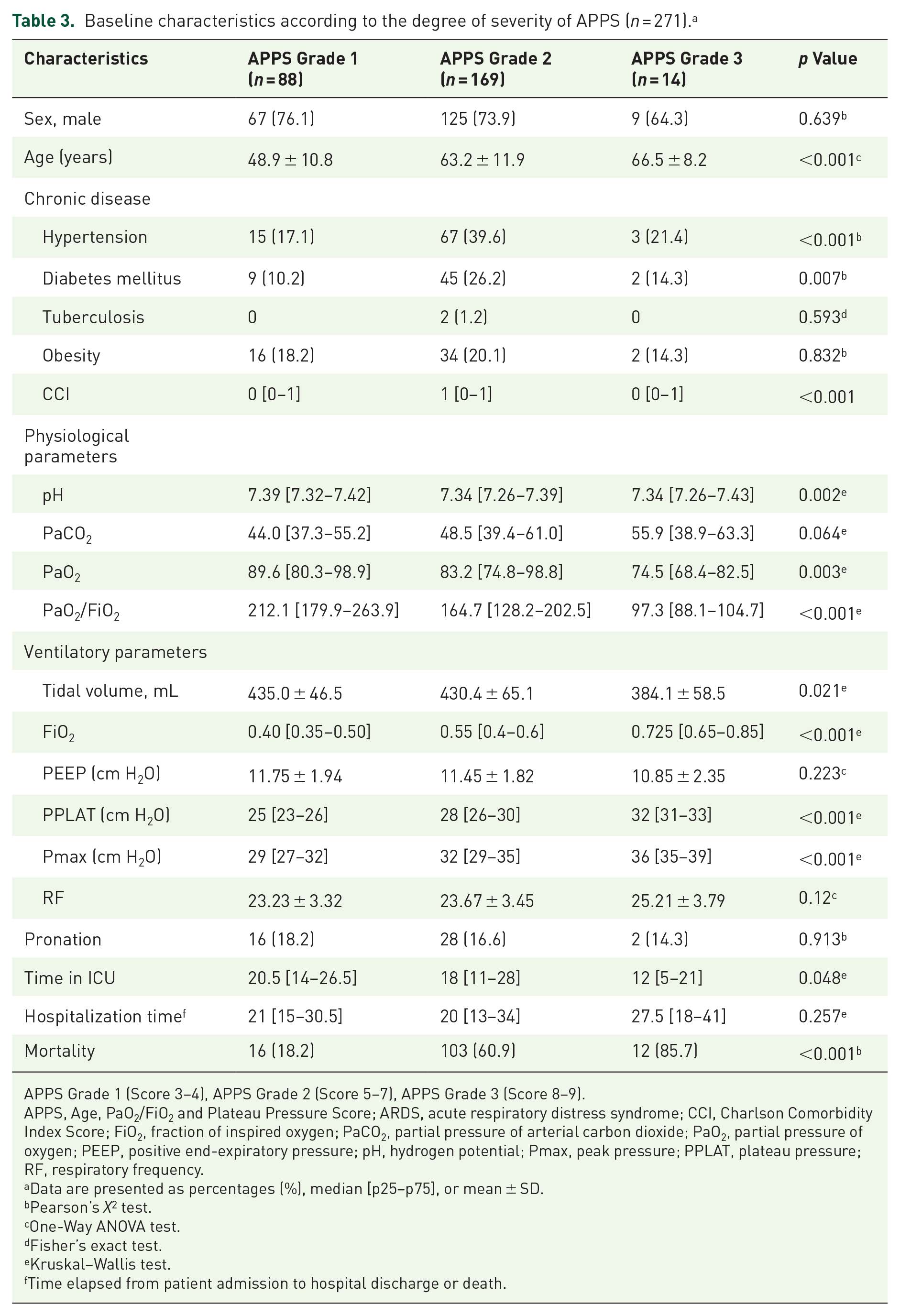
APPS Grade 1 (Score 3–4), APPS Grade 2 (Score 5–7), APPS Grade 3 (Score 8–9).
APPS, Age, PaO2/FiO2 and Plateau Pressure Score; ARDS, acute respiratory distress syndrome; CCI, Charlson Comorbidity Index Score; FiO2, fraction of inspired oxygen; PaCO2, partial pressure of arterial carbon dioxide; PaO2, partial pressure of oxygen; PEEP, positive end-expiratory pressure; pH, hydrogen potential; Pmax, peak pressure; PPLAT, plateau pressure; RF, respiratory frequency.
Data are presented as percentages (%), median [p25–p75], or mean ± SD.
Pearson’s X2 test.
One-Way ANOVA test.
Fisher’s exact test.
Kruskal–Wallis test.
Time elapsed from patient admission to hospital discharge or death.
APPS and 28-day mortality
Figure 3 showed that APPS had a good discriminative capacity, with an AUC of 0.78 (95% CI: 0.73–0.84). Subsequently, Kaplan–Meier survival curves were constructed, revealing significant differences in cumulative survival rates (p < 0.001) between different APPS grades (Figure 4). Finally, in the Cox proportional hazards model, after adjusting for sex and categorized CCI, it was found that grade 2 APPS (HRa: 3.67, 95% CI: 2.14–6.30, p < 0.001) and grade 3 APPS (HRa: 7.95, 95% CI: 3.72–17.02, p < 0.001) were associated with a higher hazard of 28-day mortality (Table 4).

AUC-ROC for APPS performance for 28-day mortality.

Kaplan–Meier survival curves according to APPS grades.
Cox proportional hazards model for the association between APPS and 28-day mortality.

APPS Grade 1 (Score 3–4), APPS Grade 2 (Score 5–7), APPS Grade 3 (Score 8–9).
95% CI, 95% confidence interval; aHR, adjusted Hazard Ratio, APPS, Age, PaO2/FiO2 and Plateau Pressure Score; cHR, crude Hazard Ratio.
Adjusted for sex and categorized Charlson Comorbidity Index.
Discussion
Main findings
Our study evidenced that the APPS has a good prognostic performance for 28-day mortality in ICU-admitted patients with COVID-19-induced ARDS. In addition, it was observed that as the APPS score increased, the accumulated survival rates decreased, and the 28-day mortality hazard increased.
Comparison with other studies
Since the introduction of this novel scoring system, 26 the predictive performance of the APPS for mortality has been evaluated in Korean, 24 Dutch, 27 and American populations, 30 however, not yet in a Latin American setting. Hwang et al. reported a performance that, while lower, was still deemed acceptable (AUC: 0.70, 95% CI: 0.60–0.81). Bos et al. documented a suboptimal performance (AUC: 0.62, 95% CI: 0.56–0.67). In the original study by Villar et al., results were consistent across both the derivation (AUC: 0.76, 95% CI: 0.70–0.81) and validation cohorts (AUC: 0.80, 95% CI: 0.75–0.85). Huang et al. reported lower but still acceptable predictive values for three outcomes: in-hospital mortality (AUC: 0.69, 95% CI: 0.63–0.75), 30-day mortality (AUC: 0.69, 95% CI: 0.63–0.75), and 1-year mortality (AUC: 0.71, 95% CI: 0.65–0.77).
Interpretation of results
Several factors may account for the variability in prognostic accuracy across different external validation cohorts. First, ethnic variations among these groups could influence outcomes. ARDS involves multiple proinflammatory pathways, incorporating genetic, environmental, and immunomodulatory factors.31,32 Furthermore, polymorphisms related to the immune system and inflammatory processes have been shown to impact the prognosis of patients who develop this condition.33,34 Such factors may account for racial disparities in mortality risks. Second, in the Dutch validation, the APPS was ineffective at predicting mortality in patients with low scores. 27 In contrast, our study was able to discriminate mortality risk beginning from grade 1 of the APPS. Additionally, our findings did not reveal inconsistencies in patients with an APPS score of 3, unlike what was observed in the Korean validation. 24 Third, unlike the Dutch validation, which used maximum airway pressure instead of plateau pressure for APPS calculations, we adhered to the original methodology proposed by Villar et al. 26 Lastly, our study focused solely on patients with COVID-19-induced ARDS, while other studies included various etiological causes of ARDS.24,27
In our study, significant differences were observed in the cumulative survival rates across different APPS grades, paralleling the findings in Hwang et al.’s validation study. 24 Conversely, Bos et al. 27 found no such differences, attributing this to flawed calibration at scores of 4–5, which led to overlapping Kaplan–Meier survival curves for APPS categories 3–4 and 5–7. 27 Both the Korean validation 24 and our study support the reported association between survival time and APPS grades. In addition, although in our study age differences were statistically significant across APPS groups, the overlapping standard deviations indicate a diverse distribution of mortality risk within each age range. This reflects the nature of the APPS score, which combines age with other key clinical parameters to enhance its prognostic ability in assessing mortality risk in COVID-19-associated ARDS.
We also found that the 28-day mortality hazard increases as the APPS grade increases. This finding aligns with studies that identify ARDS prognostic factors included in the APPS, using Cox proportional hazards regression. For example, the study by Melamed et al., 35 associated age >65 years, PaO2/FiO2 <150, and higher plateau pressure (⩾30) during the first 2 weeks of mechanical ventilation with 30-day mortality in COVID-19-induced ARDS patients.
Clinical relevance
The ease of application and the ability to categorize severity into subgroups are the most significant benefits of APPS. Compared to traditional ICU scores like APACHE-II, 20 or SAPS-II, 22 which contain 13 and 15 clinical and laboratory variables respectively, the three parameters in the APPS are easier and potentially more cost-efficient to use. Categorization into mild, moderate, and severe groups allows for risk stratification, and treatment prioritization, and importantly, reduces the likelihood of clinical trial failures in ARDS. According to Villar et al., the “clinical heterogeneity” of ARDS patients has been a historical reason for the failure of clinical trials. 26 Treating ARDS as a “clinically homogeneous” entity due to a wide mortality rate overlooks the existence of multiple clinical subgroups within the syndrome. Subgrouping allows for precise patient selection and refined objective setting for clinical trials, thereby increasing their success rate.
Limitations
This study has certain limitations that should be highlighted. First, the retrospective single-center cohort design may introduce selection bias, as the sample was not randomized. An observation that should be noted was the male and female disparity among our population. It should be noted that even though our sample was not randomized, we used a census-type method of sampling, which means that we included all the patients who fulfilled our selection criteria and were admitted into our center’s ICU in the established period of time, which was intended to reduce the incidence of systematic errors from a non-probabilistic non-census sampling. Second, not all medical records consistently reported plateau pressure at 24 h; in such cases, the nearest recorded value was used. Unlike previous validation studies that used the Pmax for APPS calculation,24,27 we followed the original study’s methodology by Villar et al. 26 Third, this study only evaluated performance for one ARDS etiology—COVID-19. Despite these limitations, this study shows that APPS has a good prognostic performance for 28-day mortality due to COVID-19-induced ARDS in a Peruvian patient sample. We propose conducting multicenter studies in Latin America to produce more externally valid results and validating this tool for other ARDS etiologies. An updated Berlin definition presented by Estenssoro et al. was proposed to make the criteria more applicable to lower resource settings. These modifications extend the 2012 definition to include non-intubated patients, and severity stratification through SpO2/FiO2 measurement as an alternative to PaO2/FiO2. Despite this update, we did not use this new definition since our setting allowed us to keep using the original 2012 criteria. The authors that proposed the modifications state that these should only apply in lower resource settings and that the original criteria should still be used whenever possible. 36
Conclusion
This study demonstrates that the APPS score has good prognostic performance for predicting 28-day mortality in COVID-19 patients with ARDS, a population primarily characterized by single-organ dysfunction. However, its prognostic accuracy in more diverse ARDS populations, including those with multi-organ failure, requires further validation in non-COVID cohorts.
Supplemental Material
sj-doc-1-tai-10.1177_20499361241306212 – Supplemental material for Prognostic performance of the Age, PaO2/FiO2 Ratio, and Plateau Pressure Score (APPS) for mortality in patients with COVID-19-associated acute respiratory distress syndrome admitted to an intensive care unit
Supplemental material, sj-doc-1-tai-10.1177_20499361241306212 for Prognostic performance of the Age, PaO2/FiO2 Ratio, and Plateau Pressure Score (APPS) for mortality in patients with COVID-19-associated acute respiratory distress syndrome admitted to an intensive care unit by Alfredo A. M. Gutierrez-Zamudio, Rodrigo Alejandro-Salinas, Jose I. Vereau-Robles and Carlos J. Toro-Huamanchumo in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
