Abstract
Background:
Fungal contamination of hospital water distribution systems has been implicated in outbreaks of healthcare-associated infections.
Objectives:
To evaluate the prevalence of fungi in the water distribution system of a tertiary hospital in Nigeria.
Design:
This was a descriptive cross-sectional study.
Methods:
Swabs and water samples were collected from taps and faucets in the hospital categorized into low (Accidents and Emergency Unit, Children Emergency Unit, Acute Stroke Unit and the 24 in-patient hospital wards) and high-risk (Renal Dialysis Unit, Central Sterile Services Department, Theatres and Intensive Care Units (ICUs)) units based on the vulnerability of patients being managed there. The membrane filtration method for water analysis was used. Where possible, isolates cultured were identified to species level. In total, 105 water and 49 swab samples were collected for analysis.
Results:
All analysed water samples grew fungi. A total of 289 (high-risk; n = 178; low-risk; n = 111) and 76 fungi isolates were recorded from water and swab samples, respectively, with 31 different species identified. Aspergillus was the most predominant genus with five different species: Aspergillus niger (9.9%), terreus (4.4%), flavus (3.3%), fumigatus (8.8%) and versicolor (2.20%) isolated. Twenty-five and 18 species of fungi were identified in the low and high-risk units, respectively. The labour ward (n = 46; 25.8%) and modular theatre (n = 47; 42.3%) were the most contaminated units. Cladosporium spp. and Paecilomyces spp. were the most frequently isolated fungi in the low and high-risk units, respectively. The dialysis centre (n = 9; 8.1%) and renal transplant theatre (n = 7; 6.31%) had the lowest contamination rates in the high-risk units. Aspergillus niger, Cephalosporium curtipes, Penicillium chrysogenum and Penicillium glabrum were each identified in 4/6 units from which swabs were taken. The facility had no documented protocol for its water safety and quality.
Conclusion:
Our data reveal a high rate of contamination of hospital water sources by fungi, some of which are known to cause life-threatening infections. For better water treatment and water tank cleaning and disinfection, a standard protocol is advised. Ensuring that the water distribution systems in hospital settings are free of fungal contaminants is important to prevent the possibility of waterborne mycosis outbreaks.
Introduction
Drinking water should be safe for human use and consumption. 1 It should also be devoid of pathogenic microorganisms and any other substances capable of causing harm to the health of the consumers, particularly the at-risk population.1,2 The presence of fungi in water has been associated with various deleterious effects including adverse taste and odour, allergies and mycotoxicity by toxigenic fungal species.3,4 Reported channels of waterborne fungi transmission include inhalation while showering or in the sauna, ingestion via cavities such as the cornea, skin lacerations, oral cavities and invasive procedures.3,4 The World Health Organization guidelines for drinking water safety and quality have thresholds for determining water potability for other microorganisms except fungi. 1 Most countries, including Nigeria, do not include fungi as a priority pathogen in the indicator organisms for safe drinking water list.1,2 Water microbial standards in most countries, however, are focused on faecal material as risk indicators for the presence of pathogenic bacteria, viruses, intestinal protozoa and helminthic parasites. However, there are other paths for the entrance of most fungi into water distribution systems. 5 In the last two decades, environmental factors such as water and air have thus been explored as possible sources of mycoses in hospital water distribution systems.6,7 Currently, there are no standards in the public domain for regulating fungi in water except the 100 CFU/mL limit in Sweden’s water safety regulation.1,8
The earliest reports of fungal contaminants’ presence in hospitals’ water distribution systems have been traced back to the 1990s from studies conducted in the USA and Europe. 9 Other studies have been conducted that further support these findings.6,10,11 Some studies have through the use of advanced molecular techniques, demonstrated evidence of waterborne transmission of fungal infections by correlating the genotypic similarities of fungal species isolated from patient samples and those isolated from the water distribution system in patients’ rooms.9–11 These and many more studies have pointed to water distribution systems in healthcare settings as a potential reservoir and source of nosocomial infections.9–11 Despite the number of reported studies, fungi are still not regarded as an important microbial contaminant of water, particularly in hospital settings, where the population at risk of infections is managed. 1
Water outlets such as taps and showers are proven sources of outbreaks of nosocomial mycoses due to inhalation of aerosolized mould conidia, spores and small hyphal fragments.12–15 This evidence thus serves as a pointer to the need for the possible adoption of fungi into the group of indicator microorganisms often used to access and assure water safety and quality. This is of top priority in healthcare settings, being a pivotal factor in the core components for efficient infection prevention and control as stated by the WHO provisions for water safety and hygiene in hospitals. 1
Several studies within the community settings in Nigeria have investigated the microbial contamination of water distribution systems but with an emphasis on bacterial and parasitic pathogens. 16 Data on fungal contamination of water sources particularly from hospital settings in Nigeria is sparse in the literature. Notably, there is no previous report of study aimed at evaluating water distribution systems of hospitals in Lagos for fungal contaminants. Contrastingly, sufficient data from other climes show contamination of water systems by fungi is not uncommon and has serious health implications.3,4,7,12–15 This index study therefore aimed to determine the prevalence of fungi and the species distribution in the water distribution system of a tertiary hospital in Nigeria.
Methods
Nature of the study
This was a descriptive cross-sectional study aimed at evaluating the prevalence of fungi in the water distribution system of a tertiary hospital in Nigeria.
The study was conducted between January and September 2019, at Lagos University Teaching Hospital (LUTH), a 761-bed facility, which admits a minimum of 4250 patients annually. LUTH is one of the two tertiary hospitals in Lagos State, Nigeria, which has an estimated population of 22 million people and has several full compliments of specialist units. The hospital has an infection control unit domiciled in the Department of Medical Microbiology. Ethical approval (Approval Number: ADM/DCST/HREC/APP/2751) was obtained from the Health Research and Ethics Committee of LUTH.
Study sites
For this study, the hospital was stratified into high and low-risk areas based on patients’ susceptibility to infection. Areas where patients with severe immunocompromised status and those under intensive care were being managed were classified as high-risk areas. In contrast, areas where patients were not under intensive care and with a non-compromised immune status were classified as low-risk.
High-risk areas: Renal Dialysis Unit, Central Sterile Services Department (CSSD), Theatres and ICUs.
Low-risk areas: Accidents and Emergency Unit, Children Emergency Unit, Acute Stroke Unit (ASU) and the 24 in-patient hospital wards.
There are three main sources of water used in the hospital. These include:
(i) Water supply from the Lagos State Water Corporation (Major supply).
(ii) Hospital main borehole (augments shortfall in supply from Lagos State Water Corporation).
(iii) Dedicated boreholes (some Wards).
The hospital water distribution systems
Water flows from the state water corporation through large underground pipes that run through a canal into a large ground aeration reservoir. The supply, however, is unreliable and or inconsistent, hence the hospital’s main borehole, which also pumps water into the aeration reservoir is made available. This supplements the shortage in water supply from the state water corporation facility. The water is filtered and transferred into twin iron tank reservoirs from where it is transferred into elevated tanks and later pumped into the central tank(s) positioned at the top of each hospital ward/block via the dedicated boreholes in the wards.
The water supply is not consistent; therefore, reservoir tanks are commonly used for water storage in different sections of the hospital.
There is no established protocol for water treatment and maintenance of the water storage facilities, but some units namely, Renal Dialysis Centre (RDC), Renal Transplant Theatre and Central developed their protocol (chlorination and filtration) for water treatment.
The hospital wards have four floors, and on each floor, faucets, taps and showers are available from which samples were randomly collected.
The hospital water is used for washing, bathing (including burns and trauma patients) and cleaning but not for consumption purposes in the areas investigated.
Sample collection
Water samples (200 mL each) were collected into sterile containers aseptically from all major water outlets in the hospital, following standard procedures as described by the American Public Health Association. 17 Swabs stick samples were collected from taps, faucets or shower surfaces to determine if these are colonized, or if it is just the water that is contaminated. Before water collection was done, the tap, faucets and shower heads were sprayed with ethanol and flamed until complete combustion of the ethanol. Water was allowed to run for 30 s before a sample was collected from each collection point. Collected samples were placed inside an ice chest and transported to the laboratory for analysis within 2 h of collection. The selection of collection points was done randomly at each of the sampling points to ensure that ⩾50% of water outlets were covered.
Swab stick samples of tap and faucet mouths were also collected from the following hospital units; Accidents and Emergency Theatre (AET), AET ward, ASU, Labour Ward Theatre (LWT), Renal Dialysis Theatre (RDT) and RDC.
Laboratory analysis of samples
Collected water samples were processed using the membrane filtration (MF) technique as described previously. 11 A volume of 100 mL water sample was filtered through a sterile membrane filter with 0.45 µm pore size and 47 mm diameter. The membrane filter was removed with the aid of sterile forceps and placed on Sabouraud dextrose agar (SDA) plates supplemented with chloramphenicol (50 mg/L) and gentamycin (0.5 mL/L). Plates were incubated at room temperature and 37°C. The plates were examined daily for any visible growth of fungi and kept for a maximum of 2 weeks. Subculture of distinct colonies was performed to obtain pure colonies. Identification of the isolates was done using standard procedures for the identification of fungi as already described in existing literature using mycology atlas. 18 The procedure was carried out within a biosafety cabinet to avoid airborne contamination of plates and plates were properly sealed before incubation. An SDA plate inoculated with sterile water was exposed within the biosafety cabinet and then later incubated to serve as a control. Lactophenol-cotton blue staining, Indian ink stain for encapsulated fungi, urease test for Cryptococcus, Germ tube test (for Candida albicans) and chromogenic agar test (for Candida species) were also done as part of the identification of the fungal isolates.
Data analysis
Descriptive analysis and computation of data were done using Microsoft Excel 2010 and SPSS version 27.19,20
Results
A total of 105 water samples and 49 swabs from various sections of the hospital were randomly collected for fungal analyses. Every single sample grew at least a fungal isolate. A total of 178 and 111 fungal isolates were recovered from water samples in low and high-risk areas, respectively, from which 31 different species of fungi were identified in this study (Tables 1 and 2). Seventeen different species of fungi were identified from 76 fungal isolates cultured from the swab samples (Table 3). Details of the findings of this study are presented in Tables 1–3.
Distribution of fungal isolates in water samples from low-risk areas.
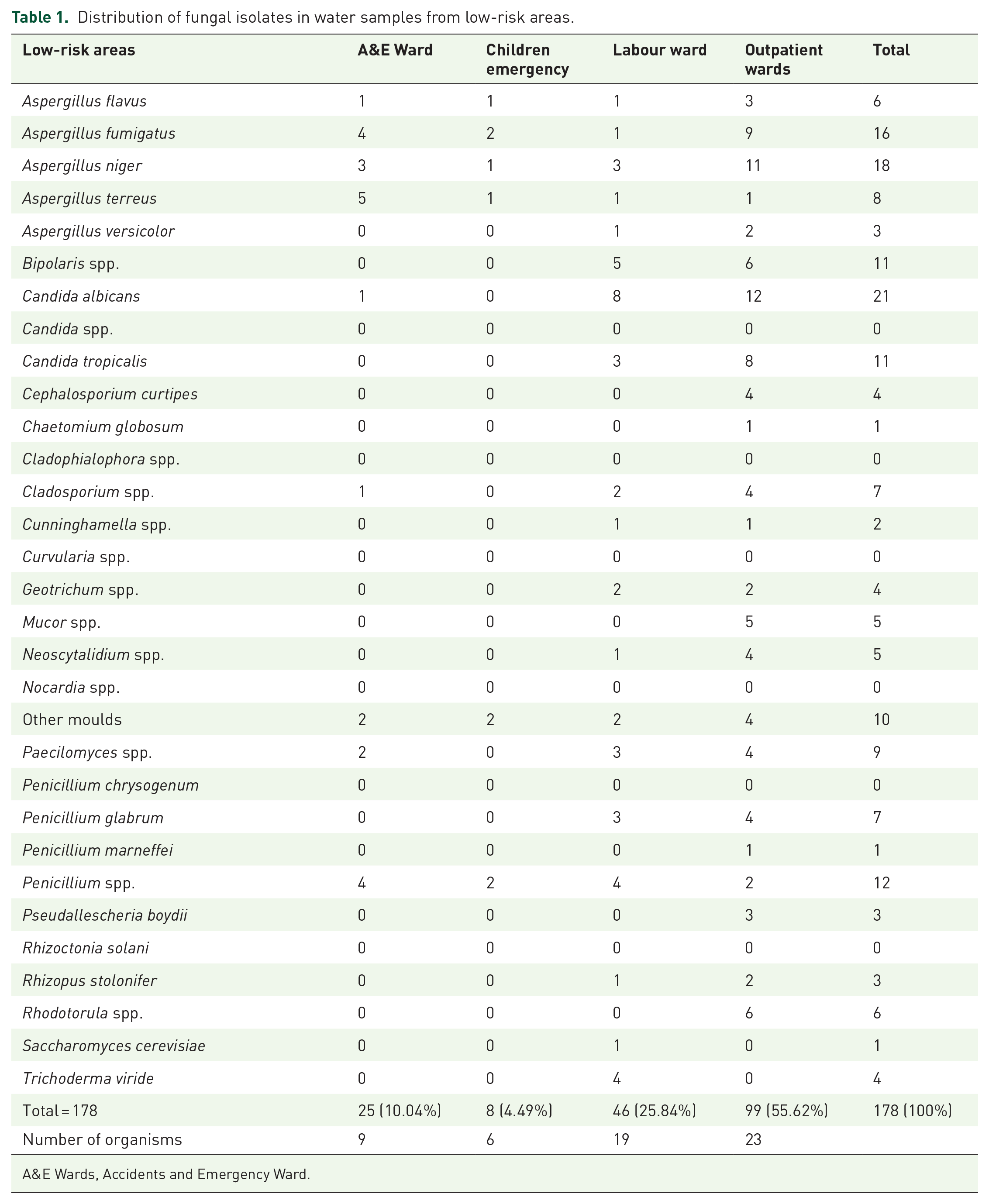
A&E Wards, Accidents and Emergency Ward.
Distribution of fungal isolates in water samples from high-risk areas.

AET, Accidents and Emergency Theatre; CSSD, Central Sterile Services Department; DU, Dialysis Unit; LWT, Labour Ward Theatre; MT, Modular Theatre; NNU, Neonatal ICU; RTT, Renal Transplant Theatre.
Distribution of fungal isolates in swab samples from different units.

A&E Wards, Accidents and Emergency Ward.
Distribution of fungal isolates in water samples from low-risk units
A total of 178 isolates were recovered from water samples in the low-risk areas. Within the low-risk areas, the outpatient wards and children’s emergency unit had the highest (n = 99; 55.6%) and least (n = 8; 4.5%) number of isolates, respectively. Twenty-three different species of fungi were identified from the outpatient wards while only six different species of fungi were identified from the Children Emergency Unit (CEU) (Table 1). Twenty-four different species of fungi were identified from water samples from the low-risk units (Table 1). Aspergillus niger (n = 18; 10.1%), Aspergillus terreus (8; 4.5%), Aspergillus fumigatus (16; 9.0%), Aspergillus flavus (6; 3.4%) and Penicillium spp. (12; 6.7%) were the most frequently isolated fungi, each occurring in all the low-risk areas studied, while the least occurring ones were Cephalosporium curtipes (4; 2.3%), Chaetomium globosum (1; 0.6%), Penicillium marneffei (1; 0.6%), Pseudallescheria boydii (2; 1.7%), Saccharomyces cerevisiae (1; 0.6%) and Trichoderma viride (4; 2.3%), each occurring only in outpatient wards, except T. viride and S. cerevisiae which only occurred in the labour wards. Aspergillus was the most predominant genus with five species identified altogether including, A. flavus (n = 6; 3.4%), A. fumigatus (n = 16; 9.0%), A. niger (18; 10.1%), A. terreus (8; 4.5%) and Aspergillus versicolor (n = 3: 1.7%) (Table 1). The most abundant fungi species of all the isolates was C. albicans representing (21; 11.8%) of all isolates recorded in the low-risk areas (Table 1).
Distribution of fungal isolates in water samples from high-risk units
A total of 111 isolates were recorded from water samples in the high-risk units. The modular theatre had the highest number of isolates (n = 47; 43.9%), while the least (n = 4; 3.60%) number of isolates was recorded in CSSD. In the high-risk units, 19 different species of fungi were identified from the water samples analysed (Table 2). Paecilomyces spp., was the most frequently isolated fungi, occurring in 6 of the 7 (85.7%) high-risk units studied, followed closely by A. flavus, and A. niger each occurring in 5 (71.4%) high-risk units, while Nocardia spp., Bipolaris spp., A. versicolor, Penicillium glabrum and S. cerevisiae were the least frequent, each occurring in only 1 (14.3%) of the high-risk units (Table 2). Aspergillus was the most predominant genus with three species reliably identified from the high-risk areas, including A. flavus (n = 10; 9.0%), A. fumigatus (n = 15; 13.51%) and A. niger (n = 11; 9.91%) (Table 2). The modular theatre had the highest number of fungi species isolated from a single unit (13 of 19 species) followed closely by AET (10 of 19 species), while the CSSD had the least number (3 species) of fungi species isolated from a single unit (Table 2).
Distribution of fungal isolates in swab samples from different units
All 49 (100%) swab samples that were collected from 6 units including AET, Accidents and Emergency Ward, ASU, LWT, RDT and RDC in this study had fungal contaminants. A total of 76 isolates were recorded from which 16 species of fungi were identified (Table 3). The AET and ASU had the highest (n = 20; 26.3%) and lowest (n = 6; 7.9%) number of isolates, respectively (Table 3). The most frequently occurring organisms were Aspergillus niger, C. curtipes, Penicillium chrysogenum and P. glabrum, each identified in 4 (66.7%) of the 6 units from where swabs were obtained (Table 3). Ten (10) fungi species were isolated from AET which is the highest number of species recorded in a single unit, while the least number (n = 2) was isolated from the ASU (Table 3). Penicillium species (n = 22; 28.9%) was the most predominant of all genera identified (Table 3).
Discussion
The presence of fungi in water has been implicated as a plausible source of waterborne nosocomial infections and or outbreaks in hospitals, through the use of advanced molecular techniques to check genetic relatedness between environmental (water and air) isolates and clinical isolates.9,21 An initial attempt at this was made by Anaissie et al. 14 who conducted a 3-year prospective study in a hospital water distribution system and recovered pathogenic Aspergillus species which after molecular typing, was found to be identical to Aspergillus species isolated from a patient. The results of these studies have aroused research interest seeking to explore water distribution systems in hospital settings for the possibility of waterborne outbreaks of fungal infections.22,23
This index study was therefore aimed at investigating the presence of fungi in the water distribution system of a tertiary hospital setting in Nigeria. To the best of our knowledge, this might be the first such study in our setting since most of the studies on microbiological quality testing of water in Nigeria are based solely on bacteriological and parasitological evaluations.
The results of contamination of hospital water distribution systems by fungi have varied from one study to another. Panagopoulou et al. 24 in a study of the water distribution system of a hospital in Greece, despite vigorous sampling and culture of tap water, reported no single (0%) fungal isolate from the water distribution system. Similarly, a 1-year prospective French study involving 750 water samples obtained from 5 different hospitals yielded (<1%) fungal contaminants. 25 However, some authors have reported a higher contamination rate of 56.9% in the potable water supply of a hospital in France. 23
We recorded an absolute (100%) fungal water contamination rate in this index study. Our finding is consistent with some other reports 11 from mycological evaluations of hospital water distribution systems in which very high and near absolute fungal contamination rates were recorded, but contrary to some similar studies in which very low or no contamination rates were reported from the water distribution system of hospitals.26,27 Sautour et al. 23 in a similar finding to ours also reported 100% and 94% contamination rates in 2 units of a French hospital that was investigated for fungal water contamination. These discrepancies between studies could be partly explained by the water supply’s origin because surface water might be more easily contaminated than groundwater.6,28 These differences can also be related to the means of water collection and treatment. 28 For our setting, the absolute contamination rate can be attributed to the lack of standard protocol for treatment and care of the hospital water distribution systems, lack of protocol for the maintenance of water storage facilities, surface water contamination and proximity of the hospital water distribution network to drainage canals. Similar observations and explanations were also deduced from other studies conducted in Belgium and Brazil).6,28,29 The importance of fungal contamination of water is still a subject of debate to date; however, it has been demonstrated to cause significant health impacts in immunocompromised patients.13,15,30 Even, immunocompetent individuals have been reported to be at risk of developing severe allergic and hypersensitive responses when exposed to fungal-contaminated water.3,4,31,32
We identified 31 different species of fungi in this study. Other studies have identified lower numbers and some even higher. For instance, Máira et al. 11 reported a broad diversity of about 70 different species of fungi in the water distribution system of a hospital setting in São Paulo, Brazil from only 84 water samples, while another study in Italy identified 15 species from 26 genera, even though with a significantly higher number (976) of analysed samples. 33 The number of samples and methods seems to be a major factor in fungi recovery from water samples.
The report of the index study by Schiavano et al. 33 on the water distribution systems of haemodialysis units of eight hospitals in Italy revealed filamentous fungi as the most prevalent fungi, just like in our study. However, this contrasted with the report of Pires-Goncalves et al. 34 in which yeasts (Candida) were the most prevalent fungi in the water distribution systems of a Brazilian hospital. Arvanitidou et al. 26 also reported Aspergillus species and Candida species as the predominant moulds and yeasts, respectively, present in water and dialysate from haemodialysis units in Greece. Aspergillus species, mainly A. niger, A. flavus and A. fumigatus were also the most frequently isolated moulds in our study. At the same time, Candida species were also the predominant yeasts in our study. The Aspergillus and Candida genera have often topped the charts in similar studies6,11,35,36 however, other studies have reported Fusarium as the most predominant fungi species. Moreover, Fusarium was not recovered in our study.13,14 Notable genera of fungi isolated in this study namely Aspergillus, Penicillium and Cladosporium have also been very well reported in other studies on hospital water systems. 37 Other studies have also reported the Aspergillus genus as the most predominant fungal species in high-risk units of hospitals.21,37–40 The solid organ transplantation unit was also the most contaminated in the study of the water distribution network of a hospital’s critical care unit in Greece as reported by Panagopoulou et al. 24 The renal transplant unit in our study also demonstrated a high contamination rate. The implications of contamination in critical care units often come with greater consequences, especially with the greater risk of opportunistic infections caused by most of the fungi recovered in this study. However, these implications are lesser among the immunocompetent population, even though possible.
Even though some high-risk units namely, the Dialysis unit, Renal Transplant Theatre and the CSSD have their water routinely treated by chlorination and use of a water filtration system, these units still had high contamination rates. This indicates that the existing protocol for water treatment and management of water storage facilities in these units is inadequate. The common organisms isolated from these units are also capable of secreting melanin or melanin-like substances in their cell walls, that can resist water treatment. 41 A study has demonstrated the resistance of Cladosporium spp., Aspergillus spp. and Penicillium spp. to chlorine in different water sources so that they remain in the distribution system for long periods. 41 This implies that even with chlorine treatments fungi are still capable of surviving in water. 41 Another possible explanation could be due to the presence of biofilm in pipes and water storage tanks. Fungal contamination of water is further compounded by the formation of biofilms, which increase their ability to survive and thus hamper disinfection procedures.28,41 ‘These structures are generally colonized by bacteria, but they can also trap conidia and other fungal elements’.6,41 A similar study by Mesquita-Rocha et al. 30 also demonstrated the presence of fungi in the high-risk unit of a hospital water distribution even though treatment protocol similarly was in place as observed in this study. A similarity between the water systems in these studies from high-risk units of hospitals6,28,30,41 and ours is that treatment using chlorine and water filtration was routinely carried out, however, this was not enough to stop the growth of fungi. A major reason for this could be attributed to biofilm formation in the pipes which could lead to the reintroduction of fungi even after treatment. Similarly, routine cleaning and maintenance of the plumbing systems and water storage facilities need to be considered due to the likelihood of recontamination.
Attempts have been made to link nosocomial infections with hospital water by comparing the genetic relatedness of species of fungi isolated from patients’ samples with those isolated from the hospital water distribution systems.7,26,35,36,38 A follow-up study could be done to assay for the same in our setting.
Currently, there is no official regulation requiring fungi surveillance of hospital water systems in Nigeria. However, considering the documented studies attesting to the existing risk of fungal waterborne contamination and/or infection,35,36 special attention ought to be paid to water distributions in healthcare settings. Although the water distribution system in the study area has not yet been linked with an outbreak of nosocomial infections, a follow-up study can be done to check for such links, should they exist, using advanced molecular techniques.
Some limitations of our study included:
(1) The lack of a generally acceptable standard for the assessment of fungal contamination in water.
(2) There is no standard method for the evaluation of fungal presence in water. Unlike other organisms such as bacteria, viruses and parasites where there are laid down guidelines for the measurement of the quality of water, there is none in place for fungi so we could not state the level of safety.
(3) Colony count could not be done since this is challenging to do for fungi, especially moulds since they sporulate, so we could not categorize contamination/growth as mild, moderate or heavy.
(4) We could not do a more detailed molecular characterization of the fungal isolates due to a lack of funding. However, this would be done on stored isolates when funding is sourced.
Conclusion
Our results highlight and justify the importance of water treatment and maintenance of water storage facilities and distribution channels in hospitals, especially in high-risk units. The impacts of water contamination and outbreaks are often of grave consequences in resource-limited settings like ours and thus must be given important attention. The results of this study will help provide guidance for an infection control and prevention intervention and possible development of the relevant standard operative procedures and/or policies to be utilized in the maintenance of hospital water distribution networks.
Footnotes
Acknowledgements
We would like to acknowledge the contributions and technical support of the laboratory staff of the Medical Microbiology and Parasitology Laboratory, Lagos University Teaching Hospital and Technologists of the Department of Pharmaceutics and Pharmaceutical Technology, College of Medicine, University of Lagos.
