Abstract
Invasive fungal infections (IFIs) are associated with high mortality rates and mostly affect patients with compromised immunity. The incidence of IFIs is increasing worldwide with the expanding population of susceptible patients. Candida and other yeast infections represent a major component of IFIs. Rare Candida/yeast infections have also increased in recent years and pose considerable diagnostic and management challenges as they are not easily recognized by routine phenotypic characteristic-based diagnostic methods and/or by the automated yeast identification systems. Rare Candida/yeasts also exhibit reduced susceptibility to antifungal drugs making proper management of invasive infections challenging. Here, we review the diagnosis and management of 60 cases of rare Candida/yeast IFIs described so far in Kuwait, an Arabian Gulf country in the Middle East. Interestingly, majority (34 of 60, 56.7%) of these rare Candida/yeast invasive infections occurred among neonates or premature, very-low-birth-weight neonates, usually following prior bacteremia episodes. The clinical details, treatment given, and outcome were available for 28 of 34 neonates. The crude mortality rate among these neonates was 32.2% as 19 of 28 (67.8%) survived the infection and were discharged in healthy condition, likely due to accurate diagnosis and frequent use of combination therapy. Physicians treating patients with extended stay under intensive care, on mechanical ventilation, receiving broad spectrum antibiotics and with gastrointestinal surgery/complications should proactively investigate IFIs. Timely diagnosis and early antifungal treatment are essential to decrease mortality. Understanding the epidemiology and spectrum of rare Candida/yeast invasive infections in different geographical regions, their susceptibility profiles and management will help to devise novel diagnostic and treatment approaches and formulate guidelines for improved patient outcome.
Introduction
Invasive fungal infections (IFIs) mostly affect individuals with compromised immunity such as patients living with human immunodeficiency virus (HIV), patients with cancer receiving chemotherapy, and organ transplant recipients in tertiary care settings and are associated with considerable morbidity and high mortality rates.1–3 The progress in medical and surgical procedures during the last three decades and increasing use of antifungal prophylaxis have managed to prolong the survival of critically ill patients at the extremes of age (neonates and >65-year-old subjects) in intensive care unit (ICU) settings but have also resulted in expanding the number of patients susceptible to IFIs.4–7 Candida and other yeasts are a significant cause of IFIs worldwide.8–13 Epidemiological studies carried out in many countries including Kuwait have shown that nearly 92% of all Candida/yeast invasive infections are caused by only five species comprising Candida albicans, Candida parapsilosis, Candida tropicalis, Nakaseomyces glabratus (also known as, aka, Candida glabrata), and Pichia kudriavzevii (aka Candida krusei), while rare Candida/yeast species usually cause <1% of all invasive yeast infections.14–17 Although C. albicans is the most common causative agent of invasive candidiasis in susceptible patients, majority of Candida infections are now caused by non-albicans Candida or other yeast species in many geographical settings.11,14–18 Recent years have also witnessed the emergence of many rare Candida/yeasts as human pathogens.11,19–22 Severely ill patients with prior immunosuppression, mechanical ventilation, total parenteral nutrition, hematological malignancies, and chronic lung disease have been shown to be more prone to develop fungemia due to rare yeasts.20–26
Recent emergence of Candida auris, a growing health threat, has been phenomenal and is attributed to its environmental adaptation to increasing temperatures as a result of the global warming, which have allowed it to tolerate mammalian thermal barrier.27–31 Although initially identified only in 2009, 32 C. auris has spread rapidly across various countries on all inhabited continents and has also caused major outbreaks in health care facilities that have been very difficult to control and manage clinically, chiefly due to its often multidrug-resistant nature.33–36 Other multidrug-resistant yeasts have also emerged in recent years and include N. glabratus, P. kudriavzevii, Meyerozyma guilliermondii (aka Candida guilliermondii), Candida haemulonii species complex, Clavispora lusitaniae (aka Candida lusitaniae) and Kluyveromyces marxianus (aka Candida kefyr).37–42
Rare Candida/yeast infections pose considerable diagnostic and management challenges as they are not easily recognized by routine diagnostic laboratory procedures based on phenotypic characteristics including growth on chromogenic media.20–22,43–47 Automated systems such as Vitek 2 yeast identification system (Vitek 2) or matrix-assisted laser desorption ionization time-of-flight mass spectroscopy (MALDI-TOF MS) tests such as Vitek MS also occasionally fail to identify rare yeast species accurately due to lack of reference data warranting identification by molecular methods.21,47–50 Rare Candida/yeast species also exhibit reduced susceptibility to one or more antifungal drugs.20–22,50 Furthermore, pediatric patients and neonates compared to adults are distinct in terms of predisposing factors, disease presentation and epidemiology of Candida/yeast invasive diseases, and in their response to antifungal drugs.14,15,18 Thus, proper treatment of rare yeast invasive infections is extremely challenging and so these infections are usually associated with higher mortality rates compared to infections by other more common Candida/yeast species.20–22,47
Kuwait is an Arabian Gulf country in the Middle East. Kuwaiti nationals comprise only about one-third of the total population of nearly 4 million individuals. The remaining subjects are expatriate residents originating from >50 countries. The vast majority of the expatriates originate from developing countries of South Asia (mainly India, Bangladesh, Pakistan, Nepal, and Sri Lanka), Southeast Asia (mainly the Philippines) and Africa (mainly Egypt, Sudan, and Ethiopia).51,52 The laboratory capacity building during the beginning of the new millennium and the establishment of a dedicated mycology laboratory within the Department of Microbiology, Faculty of Medicine, Kuwait University employing state-of-the-art molecular diagnostic procedures enabled us to accurately identify rare Candida/yeast pathogens isolated from various clinical specimens of patients hospitalized in different government hospitals in Kuwait. In this article, we present an overview of the diagnosis and management of invasive Candida/yeast infections described so far in Kuwait. In order to keep pace with recent developments, 42 we have first described the infections caused by rare Candida species followed by infections by yeast species previously included among Candida genus and concluded the write-up by including infections by other rare yeast species. The cumulative clinical details, diagnostic methods needed for identification, antifungal treatment duration, and outcome for patients with rare Candida/yeast infections are presented in Table 1. Patient-specific details are provided in Supplemental Table S1. The antifungal susceptibility testing (AST) data for rare Candida/yeast isolates described here are shown in Table 2. We also briefly discuss the new antifungal drug pipeline that may help in reducing the mortality from rare Candida /yeast infections in the future.
Brief summary of clinical details, treatment duration and outcome for patients with rare Candida/yeast invasive infections.
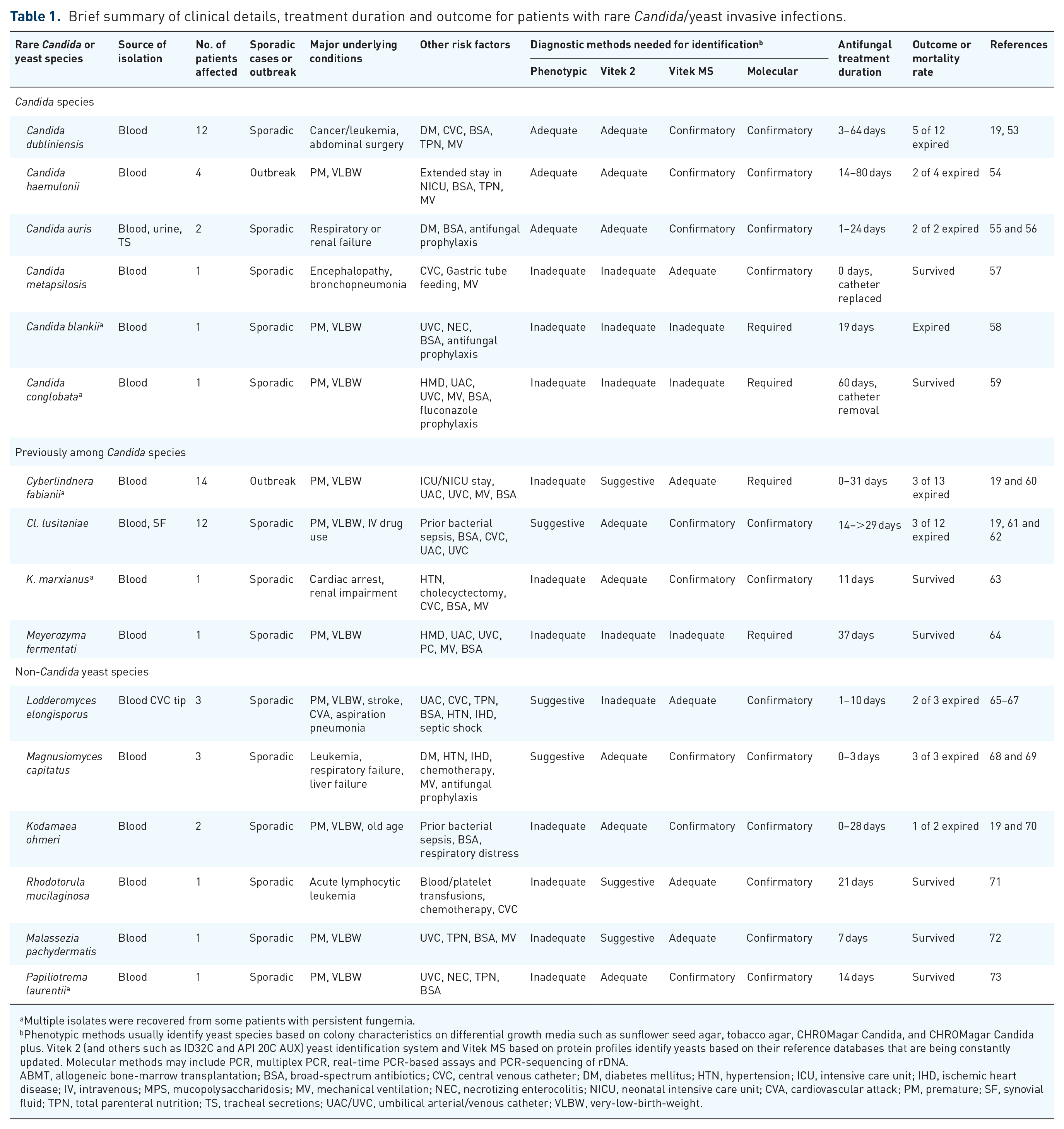
Multiple isolates were recovered from some patients with persistent fungemia.
Phenotypic methods usually identify yeast species based on colony characteristics on differential growth media such as sunflower seed agar, tobacco agar, CHROMagar Candida, and CHROMagar Candida plus. Vitek 2 (and others such as ID32C and API 20C AUX) yeast identification system and Vitek MS based on protein profiles identify yeasts based on their reference databases that are being constantly updated. Molecular methods may include PCR, multiplex PCR, real-time PCR-based assays and PCR-sequencing of rDNA.
ABMT, allogeneic bone-marrow transplantation; BSA, broad-spectrum antibiotics; CVC, central venous catheter; DM, diabetes mellitus; HTN, hypertension; ICU, intensive care unit; IHD, ischemic heart disease; IV, intravenous; MPS, mucopolysaccharidosis; MV, mechanical ventilation; NEC, necrotizing enterocolitis; NICU, neonatal intensive care unit; CVA, cardiovascular attack; PM, premature; SF, synovial fluid; TPN, total parenteral nutrition; TS, tracheal secretions; UAC/UVC, umbilical arterial/venous catheter; VLBW, very-low-birth-weight.
Antifungal susceptibility testing data for rare Candida/yeast isolates.

Values indicative of reduced susceptibility/resistance are shown in bold.
Multiple isolates from the same patient from the same or different sites.
AFG, andulafungin; AMB, amphotericin B; AST, antifungal susceptibility testing; CFG, caspofungin; 5-FC, 5-flucytosine; FLU, fluconazole; ITR, itraconazole; MCN-AM, MICRONAUT-AM EUCAST broth microdilution-based method; MFG, micafungin; N.D., not done; POS, Posaconazole; SF, synovial fluid; TS, tracheal secretion; VOR, voriconazole.
Invasive infections caused by rare Candida species
C. dubliniensis candidemia
C. dubliniensis, a pathogenic yeast species closely related to C. albicans was identified as a distinct species in 1995. 74 Although C. dubliniensis is prevalent throughout the world, it is usually found as oral carriage or causes oropharyngeal infection in people living with HIV but rarely occurs in the oral microflora of normal healthy subjects. 75 While C. albicans is usually the dominant cause of candidemia, C. dubliniensis causes <1% of candidemia cases.14,16,76 Since C. albicans and C. dubliniensis share many phenotypic characteristics including germ-tube formation, differential media such as sunflower seed agar and tobacco agar have been developed for their rapid differentiation in routine diagnostic laboratories.77,78 The clinical details, antifungal treatment given and outcome were described for 11 of 14 candidemia patients diagnosed during 2002–2010 in an earlier study 53 and an additional case was described more recently. 19 All (including six elderly and three pediatric) patients were immunocompromised subjects with one to several risk factors for IFIs (Table 1 and Supplemental Table S1). All isolates were uniformly susceptible to all antifungal agents tested except one isolate that was resistant to 5-flucytosine (Table 2).
Although all bloodstream isolates were correctly identified by phenotypic methods including germ-tube formation and colony characteristics on sunflower seed agar and the diagnosis was confirmed by Vitek 2 and molecular methods, 5 of 12 patients expired including 4 with central venous catheter (Table 1). The high mortality was likely due to multiple comorbidities and other risk factors, prior episodes of bacteremia and delays in specific diagnosis as two patients died before culture results became available and another two died soon after the commencement of treatment (Supplemental Table S1).19,53 Treating physicians should consider C. dubliniensis as one of the possible pathogens causing catheter-related infections for rapid diagnosis and improved outcome. Late complications and fatal outcome have also been reported in patients whose blood cultures had become negative for C. dubliniensis after antifungal therapy.79,80
Mini outbreak of candidemia due to C. haemulonii
C. haemulonii species complex comprises several rare and recently described species such as C. haemulonii, C. duobushaemulonii, C. pseudohaemulonii, and C. vulturna and the notorious multidrug-resistant pathogen, C. auris.35,41,81 These organisms colonize humans in the community and hospital settings and many clinical isolates show reduced susceptibility to one or more antifungal drugs.41,81 Candida khanbhai, another novel species closely related to C. haemulonii complex, isolated from the nasal swab sample in Kuwait and bloodstream of a candidemia patient in Malaysia, has also been described recently. 82 Interestingly, colonies of C. khanbhai on CHROMagar Candida Plus medium are indistinguishable from C. auris. 82
The first documented candidemia case by C. haemulonii occurred in a nearly full-term, neonate who had been in the neonatal ICU (NICU) for 42 days, had received broad-spectrum antibiotics for multiple episodes of bacteremia and was on mechanical ventilator and total parenteral nutrition through central lines (Table 1 and Supplemental Table S1). While this patient was in the NICU, three other premature, very-low-birth-weight (VLBW) neonates who were in the same NICU for 30 to 76 days with multiple episodes of bacteremia also developed C. haemulonii fungemia resulting in a mini outbreak (Table 1). Repeat isolates were obtained from cases 3 and 4. The isolates were identified as Rhodotorula glutinis by the Vitek 2 and as C. haemulonii by PCR-sequencing of the internal transcribed spacer (ITS) region and D1/D2 domains of rDNA. 54 CHROMagar Candida™ supplemented with Pal’s medium is now available for phenotypic identification of C. haemulonii. 83 All isolates showed reduced in vitro susceptibility to amphotericin B, fluconazole, and itraconazole (Table 2). 54 Two of four neonates survived the infection including one case treated with fluconazole alone even though the isolate showed in vitro resistance to this drug (Table 1 and Supplemental Table S1). 54 This is consistent with other reports showing successful treatment of fungemia cases caused by both, in vitro fluconazole-susceptible and -resistant strains by fluconazole therapy.84,85 Treatment with amphotericin B is not very effective against C. haemulonii 84 as is also evident by fatal outcome of the neonate in Case 3 (Supplemental Table S1). Other C. haemulonii complex members (e.g. C. duobushaemulonii) have also been isolated from blood samples of hospitalized patients in Kuwait but the clinical details and treatment given were not available. 86
C. haemulonii invasive infections are more common in developing countries in tropical regions. This yeast colonizes the skin, predominantly in women, and catheters provide the main portal of entry for fungemia. 81 Physicians treating neonates in tropical areas should consider the diagnosis of C. haemulonii fungemia and treatment should include, if echinocandins are not available, fluconazole or other azoles, even if the isolate exhibits reduced in vitro susceptibility.84,85
Emergence, spread and outbreak of C. auris infections in Kuwait
C. auris is a recently recognized yeast that was first isolated from the ear canal of a female patient in Japan in 2009 but soon spread to many other countries.32,87 This novel yeast has attracted worldwide attention as many isolates exhibit reduced susceptibility to azoles and/or amphotericin B resulting in multidrug-resistant status of many strains and has been recognized as an urgent threat to public health by the Centers for Disease Control and Prevention (CDC) of USA. 30 This notorious organism has been isolated from >50 countries worldwide and has also caused outbreaks in healthcare facilities with devastating results and significant infection control challenges.35,88 Two C. auris fungemia cases are noteworthy and their details together with other important development are briefly included in this review. The first case of C. auris fungemia in Kuwait was diagnosed in May, 2014 in a 27-year-old female (Table 1 and Supplemental Table S1). She was being treated with broad-spectrum antibiotics for septic shock when she developed fungemia. 55 The blood culture yeast isolate was initially identified as C. haemulonii, reflecting the inability of Vitek 2 in 2014 to correctly identify it or other rare/emerging yeast pathogens. Species-specific identification was achieved by PCR-sequencing of the ITS region of rDNA. In vitro AST data showed that the isolate was resistant to fluconazole but was susceptible to voriconazole, caspofungin, and amphotericin B (Table 2). Although the treatment was started with liposomal amphotericin B (150 mg/day), the patient died soon afterward from multiorgan failure. 55
The initial isolation in 2014 was followed by identification of other C. auris isolates. A retrospective study identified 158 of 166 isolates, forming pink-colored colonies on CHROMagar Candida and identified as C. haemulonii by Vitek 2, as C. auris. 86 C. auris spread rapidly thereafter and was isolated from all major government hospitals except Maternity Hospital within 4 years of its first isolation in Kuwait. 89 The C. auris outbreak, which started in a major secondary care hospital, could only be contained but not eradicated despite intensive infection control measures. 33 By 2018, it had emerged as a major bloodstream yeast pathogen, surpassing the fourth ranked N. glabratus. 19 Patients with C. auris candidemia had multiple comorbidities, the duration of hospital stay before onset of fungemia varied from 5 to 93 days and the mortality was >50%, which is consistent with the data reported from other countries.30,33,90 All C. auris isolates in Kuwait were resistant to fluconazole and bloodstream isolates were generally susceptible to amphotericin B and echinocandins.33,89 However, some urine and respiratory isolates exhibited reduced susceptibility to echinocandins and amphotericin B and high-level amphotericin B resistance was shown to involve ERG6 gene alterations and concomitant changes in total cell sterol profiles.3,33,89,91 A whole genome sequence-based recent study also showed that resistance to echinocandins due to FKS mutations developed in patients during treatment. 92 The second important C. auris fungemia case in Kuwait involved multiple isolates from an immunocompromised patient with chronic lung transplant rejection (Table 1 and Supplemental Table S1). 56 This study showed that echinocandin resistance due to multiple genotypes may emerge in the same patient during treatment that is consistent with recent recommendations of multiple colony AST for proper management of invasive Candida infections.56,93
Some recent findings regarding C. auris invasive infections are noteworthy and treating physicians and microbiologists should be aware of these developments. Timely diagnosis and accurate AST of C. auris is crucial for effective management of invasive infections. Accurate diagnosis even in resource-limited settings is now greatly facilitated by the development of phenotypic methods such as CHROMagar Candida Plus, expanded databases of Vitek 2 and Vitek MS, and a variety of molecular methods.30,46,83 The in vitro AST data should be interpreted with caution as several, particularly commercial, methods either overestimate or underestimate resistance of C. auris to fluconazole and amphotericin B.94,95
C metapsilosis fungemia
C. metapsilosis and C. orthopsilosis together with C. parapsilosis form the C. parapsilosis complex. Simple species-specific PCR and/or multiplex PCR assays were developed in Kuwait to identify the two cryptic species in C. parapsilosis sensu lato isolates.96,97 These studies identified bloodstream infections by C. orthopsilosis but clinical details, treatment given and outcome were not reported.96,97 Although initial studies did not detect invasive cases due to C. metapsilosis, one fungemia case in a 10-year-old female was subsequently detected. 57 She had progressive encephalopathy, epilepsy, and developed bronchopneumonia in the pediatric ICU (PICU). Other risk factors included CVC, mechanical ventilation, endotracheal tube placement, and gastric feeding (Table 1). The blood culture isolate was initially identified as C. parapsilosis by colony characteristics on CHROMagar Candida and Vitek 2 but was later confirmed as C. metapsilosis by PCR-sequencing of the ITS region of rDNA (Supplemental Table S1). 57 The isolate was susceptible to the antifungal drugs tested (Table 2). The catheter was removed but she did not receive any specific antifungal treatment and survived the infection. However, she subsequently had episodes of bronchopneumonia and candidemia due to C. albicans and eventually expired.
C. metapsilosis colonizes the skin and oral cavity that may provide the source of infection in individuals with catheters and/or mechanical ventilation. Specific identification is achieved by Vitek MS or molecular methods and catheter removal may suffice in most cases, as was noted in this case, since this is the least virulent member of the C. parapsilosis complex. 98
C. blankii fungemia
C. blankii, a non-fermenting yeast mostly used in biotechnological research, is an emerging, often fatal, opportunistic pathogen that recently caused an outbreak among low/VLBW neonates in India.99,100 C. blankii usually has lower susceptibility to azoles and may also exhibit reduced susceptibility to echinocandins or amphotericin B.99–101 A case of persistent fungemia due to C. blankii has also been described from Kuwait in a preterm, VLBW neonate receiving antifungal prophylaxis during multiple episodes of bacteremia (Table 1). 58 He was also empirically treated twice with liposomal amphotericin B (AmBisome) (Supplemental Table S1). He later developed persistent septicemia due to a bacterial and a yeast pathogen, the latter initially forming pink and subsequently dark metallic blue colonies (similar to C. tropicalis) on CHROMagar Candida. It was identified as Stephanoascus ciferii by Vitek 2 and as C. blankii by PCR-sequencing of rDNA. 58 Despite treatment with AmBisome for 16 days and then with AmBisome plus caspofungin for 5 days in addition to broad-spectrum antibiotics, the neonate expired due to polymicrobial septicemia (Table 1). The AST data showed that the isolates exhibited reduced susceptibility to fluconazole (minimum inhibitory concentration, MIC 12–16 μg/mL) but appeared susceptible to other antifungal drugs by Etest (Table 2). 58
Isolation of yeast during breakthrough infection from VLBW neonates with the yeast initially forming pink and subsequently dark metallic blue colonies on CHROMagar Candida and showing clusters of budding yeast with pseudohyphae during slide culture on cornmeal agar should alert microbiologists to use molecular methods to confirm its identification.
C. conglobata fungemia
The first case of persistent C. conglobata bloodstream infection occurred in a preterm, VLBW (930 g) male neonate in Kuwait and the etiologic role of C. conglobata in bloodstream infection was proven unequivocally for the first time (Table 1 and Supplemental Table S1). 59 He had two episodes of bacteremia requiring treatment with broad-spectrum antibiotics and was given fluconazole empirically before blood culture from peripheral vein yielded a yeast that was identified as M. farinosa by Vitek 2 and as C. conglobata by PCR-sequencing of the ITS region and D1/D2 domains of rDNA. 59 The diagnosis was further confirmed by detection of C. conglobata DNA in serum samples. Despite treatment with AmBisome for 7 days, blood culture was still positive and so caspofungin was added to the treatment regimen (Supplemental Table S1). Although the blood sample collected on Day-6 post combination therapy was positive for C. conglobata DNA by PCR and blood cultures were also positive for C. conglobata after 7-days and 17-days post combination therapy, the overall condition of the baby improved. The AST data showed that C. conglobata isolates appeared susceptible to amphotericin B, azoles, echinocandins, and 5-flucytosine (Table 2). The catheters were removed on Day-18 post combination therapy. The blood cultures eventually became negative after continued treatment with AmBisome and caspofungin for 25 days, and the baby was discharged with a weight of 2690 g in a healthy condition (Table 1). 59
Isolation of yeast during breakthrough infection from VLBW neonates with the yeast forming pink colonies on CHROMagar Candida 86 and showing well-branched pseudohyphae with budding yeast during culture on cornmeal agar 59 should prompt microbiologists to use molecular methods to confirm its identification for appropriate treatment.
Invasive infections caused by yeasts recently removed from the genus Candida
C. fabianii fungemia cases in Kuwait
Cyberlindnera fabianii (aka Candida fabianii) is an environmental ascomycetous yeast that is found in soil and is used in wastewater treatment and fermentation of alcoholic beverages. Cy. fabianii colonizes the skin and vagina and is an opportunistic pathogen capable of causing bloodstream infections in patients with severe immunosuppression, major surgery, and treatment with broad-spectrum antibiotics.23,102–104 Neonates with low birth weight appear to be particularly susceptible to developing invasive infections.105–108 Cy. fabianii is an emerging yeast pathogen in the Middle East, forms antifungal drug-resistant biofilms and acquires resistance to fluconazole and other drugs rapidly, which allow this yeast to breakthrough antifungal prophylaxis and empiric treatment.102,104,107–110
Cy. fabianii has caused several invasive infections, mostly among neonates including a fungemia outbreak among premature, VLBW neonates in the Maternity Hospital in Kuwait.19,60 The clinical details were available for 14 cases and outcome for 13 cases including all 10 neonates of the outbreak in 2014 (Table 1 and Supplemental Table S1). All neonates involved in the outbreak were on mechanical ventilation in the NICU, had umbilical catheters in place, and were receiving broad-spectrum antibiotics for suspected or confirmed bacteremia when the blood cultures yielded yeast, which was identified as C. utilis by Vitek 2 and as Cy. fabianii by Vitek MS and by PCR-sequencing of the ITS region and D1/D2 domains of rDNA (Table 1). All isolates appeared susceptible to amphotericin B, voriconazole, caspofungin, anidulafungin, and micafungin but showed reduced susceptibility to fluconazole, itraconazole, and posaconazole (Table 2). 60 Fungemia persisted in 6 of 10 neonates as multiple blood culture isolates were recovered, the central venous catheter tips from five neonates also yielded Cy. fabianii and blood samples from three neonates were also positive for Cy. fabianii DNA by PCR (Supplemental Table S1). 60 One neonate expired before blood cultures became positive while nine neonates received treatment with amphotericin B alone (n = 4) or combination therapy (n = 5) and eight of nine neonates survived the infection (Supplemental Table S1). Genotypic relatedness among outbreak isolates was indicated based on data obtained by microsatellite-based and minisatellite-based primers 111 and DNA sequence comparisons of the non-transcribed spacer (NTS-1) between 28S rRNA and 5S rRNA genes. 60
Surveillance cultures from the NICU staff and environment were negative. Although the source of infection was not detected, the outbreak subsided with rigorous hand washing practice and strict compliance with standard infection control measures. 60 However, sporadic cases continued to occur and four other fungemia cases were described subsequently in ICU patients including three patients (a neonate, an infant, and a child) from Maternity Hospital in 2018 (Supplemental Table S1). 19 The isolates showed reduced susceptibility to fluconazole but appeared susceptible to voriconazole, caspofungin, and amphotericin B (Table 2). One patient expired before blood culture results became available, while two neonates treated with amphotericin B alone or combination therapy survived the infection. The outcome was not known for another neonate treated with amphotericin B alone (Supplemental Table S1). 19
Phenotypic methods and Vitek 2 are inadequate for specific identification. Cy. fabianii appeared as ovoid to elongated cells, singly or in pairs, formed white to pink colored colonies on CHROMagar Candida and slide culture on cornmeal agar showed budding yeast cells with pseudohyphae. Rapid identification by Vitek MS or by molecular methods and treatment with amphotericin B alone or in combination with other drugs may help in reducing mortality from invasive Cy. fabianii infections.19,104,60
Invasive Cl. lusitaniae infections
Clavispora lusitaniae (aka Candida lusitaniae) is an emerging opportunistic yeast pathogen that usually affects immunocompromised/immunosuppressed individuals with comorbidities that include immature or compromised immune system, prolonged antibiotic therapy and underlying malignancies, chemotherapy, or bone marrow transplants. 112 Cl. lusitaniae causes nearly 19% of fungemia cases in cancer patients. Although it has lower pathogenic potential and the mortality rate of invasive Cl. lusitaniae infections is usually <5%, some isolates exhibit resistance to amphotericin B. 112 However, none of 28 invasive isolates in Kuwait came from cancer patients and all isolates appeared susceptible to amphotericin B.61,62
The first documented case of invasive Cl. lusitaniae infection in Kuwait was described in a 29-year-old intravenous drug user and the isolate from synovial fluid was identified as Cl. lusitaniae by Vitek 2.
61
Presence of Cl. lusitaniae DNA and high levels of (1–3)-β-
In a retrospective study of 990 available bloodstream isolates, 25 (2.5%) were identified as Cl. lusitaniae by Vitek 2, Vitek MS, and PCR amplification of rDNA. 62 Interestingly, 11 of 25 isolates were recovered from neonates with 9 isolates originating from a single hospital. The ITS region sequences of nine Cl. lusitaniae isolates showed six different haplotypes ruling out an outbreak by a dominant genotype. 62 The clinical details were available for eight of nine neonates. Additionally, 3 of 232 (1.3%) yeast fungemia cases due to Cl. lusitaniae were described 2018. 19 All Cl. lusitaniae isolates were susceptible to fluconazole, voriconazole, caspofungin, micafungin, and amphotericin B (Table 2). Treatment details and outcome were available for 11 neonates and 3 neonates including 2 twins expired giving an overall crude mortality rate of 27.3% (Supplemental Table S1). 62 All three neonates had one or more episodes of bacteremia and two neonates had bacterial septicemia prior to developing candidemia. Cl. lusitaniae has also been involved in bloodstream infections in neonates in Saudi Arabia, another Middle Eastern country in the Arabian Gulf region. 113 On the contrary, Cl. lusitaniae was not isolated from any of the 25 neonates, while eight cases of fungemia were detected among other pediatric patients in an international prospective study of invasive candidiasis. 114
Phenotypic methods showing ovoid to sub-globose budding yeast cells with abundant pseudohyphae and pink-colored colonies on CHROMagar Candida are suggestive, while Vitek 2 is adequate for specific identification of Cl. lusitaniae. All three neonates with unfavorable outcome received amphotericin B either alone or as primary therapy, while replacement of amphotericin B with fluconazole and/or caspofungin improved outcome. 62
K. marxianus fungemia
Kluyveromyces marxianus (aka Candida kefyr), an ascomycetous yeast commonly found in kefir grains, plants, industrial sewage, and fermented dairy products, is an emerging pathogen in immunocompromised patients particularly those with oncohematological diseases.21,42 Consumption of poorly refrigerated dairy products with overgrowth of K. marxianus could be the source of infection and the invasion is usually through damaged mucosa of gastrointestinal tract (mucositis) in susceptible patients.21,115 The first documented case of K. marxianus fungemia in Kuwait was described in a nonhematological 60-year-old patient with severe brainstem dysfunction. The blood culture isolate was correctly identified by Vitek 2 and the diagnosis was confirmed by detection of K. marxianus DNA by PCR and by elevated levels of 1,3-β-
A retrospective study that mainly included immunocompromised/cancer patients from two tertiary care hospitals in Kuwait subsequently identified four other K. marxianus bloodstream isolates during 2011–2018. 117 Sketchy clinical details and outcome were available for two of four fungemia patients and both patients died during their hospitalization. 117 Interestingly, several urine and respiratory isolates exhibited reduced susceptibility to amphotericin B. 117 These isolates showed altered cell sterol contents due to mutations in ERG2 or ERG3 involved in ergosterol synthesis. 118 These studies established ERG2 and ERG3 as gene targets for amphotericin B resistance that has been occasionally reported among invasive K. marxianus isolates from immunocompromised patients including patients with hematological malignancies.115,118
Rapid diagnosis of K. marxianus fungemia is achieved by Vitek 2 and fluconazole/other azoles or echinocandins should be preferred over amphotericin B due to the possibility of reduced susceptibility to amphotericin B.115,117
M. fermentati fungemia in a premature neonate
Meyerozyma fermentati (aka Candida fermentati) closely related to Meyerozyma guilliermondii (aka Candida guilliermondii) and other yeast species such as Wickerhamomyces anomalus (aka Candida pelliculosa) previously included among the genus Candida have also been isolated from fungemia cases in Kuwait.19,42,64 Members of M. guilliermondii complex usually show reduced susceptibility to fluconazole and echinocandins, invasive infections mostly occur in patients with malignancy, immunosuppressive therapy, and/or neutropenia and usually have a favorable outcome due to low virulence of these yeasts.119,120 The first documented case of M. fermentati fungemia, identified as Candida famata by Vitek 2, occurred in a preterm, VLBW neonate (Table 1 and Supplemental Table S1). 64 PCR-sequencing of rDNA was required for correct identification. The culture from the long-line tip also yielded the same yeast. 64 The isolates were susceptible to amphotericin B, voriconazole, caspofungin, and micafungin but exhibited reduced susceptibility to fluconazole and itraconazole (Table 2). He was initially treated with amphotericin B but complete resolution of fungemia required extended duration of combination therapy with liposomal amphotericin B and caspofungin. 64 The clinical details, treatment given, and outcome were not available for the fungemia case due to M. guilliermondii. 19 The W. anomalus fungemia occurred in a 5-month-old infant, the isolate showed reduced susceptibility to fluconazole, and the patient was treated with liposomal amphotericin B for 14 days with favorable outcome. 19
Although Nakaseomyces glabratus (aka Candida glabrata) is a major yeast pathogen in Kuwait 17 and increasing trends of reduced susceptibility to fluconazole, echinocandins, and amphotericin B have been reported, 121 invasive infections by other members of the complex, that is, Nakaseomyces nivariensis (aka Candida nivariensis) and Nakaseomyces bracarensis (aka Candida bracarensis) were not detected. 122 However, N. nivariensis was recently cultured from tracheal aspirate and was identified by molecular methods. 123
Invasive infections caused by other rare yeast species
Invasive infections by other rare yeast species have also increased in recent years due to increasing number of seriously ill and immunocompromised patients and few of these rare yeasts are now regarded as emerging pathogens in some geographical locations including Kuwait.26,124 A brief summary of the rare yeast invasive infections described so far in Kuwait are summarized below.
L. elongisporus as an emerging bloodstream pathogen in Kuwait:
L. elongisporus shares many phenotypic characteristics with C. parapsilosis complex members and so is often misidentified as C. parapsilosis by Vitek 2 or other yeast identification methods. 124 This yeast has been isolated from diverse sources such as citrus concentrate, soil, fermented food products, stored apples, pigeon excreta, marine fish, hospital environments, and human clinical samples including blood.124,125 The first suspected case of fungemia in Kuwait occurred in 2008 (Table 1 and Supplemental Table S1). 65 The isolate cultured from the catheter tip was identified as C. parapsilosis by Vitek 2 and as L. elongisporus by PCR-sequencing of rDNA and was susceptible to antifungal drugs tested (Table 2). The patient was successfully treated by removal of catheter and intravenous fluconazole (400 mg/day). 65 The inability to culture L. elongisporus from the blood samples was attributed to the transient nature of fungemia due to infrequent seeding from the catheter and the low culture positivity that is commonly seen even among patients with proven cases of invasive infections. 116 Unlike C. parapsilosis isolates, L. elongisporus forms turquoise blue colonies on CHROMagar Candida and long ellipsoidal shaped ascospores after 7–10 days of incubation on cornmeal agar, providing a presumptive identification for laboratories lacking molecular diagnostic procedures.65,66 A multiplex PCR assay was developed for rapid and accurate identification and differentiation of L. elongisporus from C. parapsilosis complex members among yeast isolates. 97
Two other cases of L. elongisporus fungemia have been detected in recent years. The first case occurred in a 71-year-old female and despite therapy with caspofungin, the patient died on day 3 of her hospital stay (Table 1 and Supplemental Table S1). 66 The second case occurred in a premature, extremely low-birth-weight neonate, representing the first such case of its kind (Table 1 and Supplemental Table S1). 67 Again, only one dose of liposomal amphotericin B could be administered as the neonate expired due to sepsis on the same day when culture results became available. 67 In both cases, the isolates were identified as C. parapsilosis by Vitek 2 and as L. elongisporus by multiplex PCR and/or PCR-sequencing of rDNA (Table 1) and were susceptible to all antifungal drugs tested (Table 2). These reports66,67 show that L. elongisporus is an emerging pathogen in Kuwait, particularly for patients in ICU settings with compromised/immature immune system, phenotypic methods alone may not be sufficient for accurate identification and rapid diagnosis may be crucial for improved outcome.
Fungemia cases due to M. capitatus:
Magnusiomyces capitatus (aka Saprochaete capitatus) and Magnusiomyces clavatus (aka Saprochaete clavatus) are emerging pathogens causing severe infections in patients with profound neutropenia with mortality rates ranging from 40% to 80%.20,26 Magnusiomyces spp. are intrinsically resistant to fluconazole and echinocandins and so patients receiving fluconazole or echinocandin as prophylaxis or empiric treatment are prone to develop breakthrough infections.23,126 A case of breakthrough invasive infection by M. capitatus also occurred in a young girl in Kuwait with relapsing acute myeloid leukemia during chemotherapy following bone-marrow transplantation. 68 The severely neutropenic patient was receiving broad-spectrum antibiotics for several episodes of bacteremia and caspofungin prophylaxis for 24 days. The blood samples as well as urine and tracheal secretions grew yeasts that were identified as M. capitatus by Vitek 2 and PCR-sequencing of rDNA (Table 1 and Supplemental Table S1). The cultured isolates showed reduced susceptibility to caspofungin (Table 2). Despite initiation of treatment with liposomal amphotericin B and removal of Port-A-Catheter, the patient expired on the third day. 68
Two other cases of M. capitatus fungemia occurred in 2018 including breakthrough infection in one patient and both patients died before definitive diagnosis could be made (Table 1 and Supplemental Table S1). 69 The cultured isolates from both patients showed reduced susceptibility to fluconazole and resistance to echinocandins (Table 2). Thus, all three M. capitaus fungemia cases in Kuwait expired, which is consistent with very high mortality rates for invasive infections by this yeast.20,26 Interestingly, Gram-stained smear of the cultured isolates showed numerous arthroconidia fragmenting into rectangular forms. This characteristic could be used for the presumptive identification of the yeast as M. capitatus pending confirmation by Vitek 2 and/or by PCR-sequencing of rDNA for rapidly initiating antifungal treatment with amphotericin B. 69
Fungemia cases due to K. ohmeri:
Kodamaea ohmeri (aka Pichia ohmeri) is frequently mistaken for Candida as they both belong to Saccharomycetales. 22 This yeast has been isolated from various environmental sources, such as sand, seawater, swimming pools, and fruits and as a colonizer from the oral cavity of cancer patients.127,128 K. ohmeri is an emerging human pathogen that causes life-threatening infections in critically ill immunocompromised patients requiring invasive monitoring and intervention with high mortality rates in fungemia cases.22,23,127 The first case of persistent K. ohmeri fungemia occurred in a premature, low-birth-weight neonate (Table 1 and Supplemental Table S1). 70 The AST data showed higher MICs for fluconazole but the isolates appeared susceptible to other antifungal drugs tested (Table 2). She was treated with amphotericin B for a total of 28 days with favorable outcome (Supplemental Table S1). 70
The second K. ohmeri invasive infection was detected in 2018 in a 79-year-old patient (Table 1 and Supplemental Table S1). The isolate was resistant in vitro to fluconazole but appeared susceptible to voriconazole, caspofungin, and amphotericin B (Table 2). Although the underlying condition, comorbidities and other clinical details were not available, no specific antifungal treatment could be given as the patient expired within 2 days of investigations and before the culture results became available. 19
Fungemia cases due to Rhodotorula spp.:
Rhodotorula spp. are environmental yeasts in Asia, contaminate surgical wound site, and are the causative agents in nearly 5% of non-Candida yeast invasive infections in China. 129 Rhodotorula spp., particularly R. mucilaginosa isolates, usually have higher MICs for azoles and echinocandins limiting therapeutic options to amphotericin B and 5-flucytosine. 47 A case of persistent catheter-related R. mucilaginosa fungemia was detected in a 4-year-old female with acute lymphocytic leukemia (Table 1 and Supplemental Table S1). 71 The patient had an episode of bacteremia prior to fungemia and the yeast was identified as R. glutinis by Vitek 2 and as R. mucilaginosa by PCR-sequencing of rDNA. In the absence of AST data, treatment was initiated with voriconazole instead of amphotericin B due to concerns for nephrotoxicity. After 3 weeks of therapy, the patient improved and was discharged from the hospital. However, the patient relapsed and was readmitted in the hospital and blood culture again yielded R. mucilaginosa. R. mucilaginosa DNA was detected in blood and the yeast was also isolated from the tip of Hickman catheter. The AST data showed higher MICs against fluconazole, voriconazole, posaconazole, and caspofungin (Table 2). Despite a higher MIC against voriconazole, treatment was again initiated by voriconazole. With the removal of catheter and continued treatment with voriconazole, the patient improved and subsequent blood cultures became negative. 71 A fungemia case due to another species (R. minuta) was also detected in 2018 by PCR-sequencing of rDNA. However, the clinical details, treatment given and outcome were not available. 19
M. pachydermatis fungemia
Malassezia spp. are lipid-dependent yeasts and colonize the skin and mucous membranes of humans and animals. Systemic infections, reported at increasing frequency in recent years, are mainly caused by three species viz. M. furfur, M. pachydermatis, and M. symphodialis. 130 The risk factors for invasive infections include immature/severely compromised immunity, total parenteral nutrition, break in skin integrity, and invasive surgical procedures.20,130 The first case of M. pachydermatis fungemia was detected in Kuwait in a preterm VLBW neonate. 72 The neonate received broad-spectrum antibiotics for multiple episodes of bacteremia before developing fungemia (Table 1 and Supplemental Table S1). The blood culture isolate was identified as M. pachydermatis by Vitek MS and PCR-sequencing of rDNA and was resistant, in vitro, to fluconazole, caspofungin, and 5-flucytosine (Table 2). 72 The patient was successfully treated with liposomal amphotericin B (Supplemental Table S1). 72
P. laurentii fungemia
Papiliotrema laurentii (aka Cryptococcus laurentii) is an environmental yeast-like organism that was considered as a nonpathogenic species for humans with a normal immune system until recently. However, several invasive cases including fungemia and meningitis attributed to P. laurentii have been described in recent years.131–133 An invasive infection caused by P. laurentii also occurred in a premature, VLBW female neonate in Kuwait (Table 1 and Supplemental Table S1). 73 Repeated blood cultures were identified as P. laurentii by Vitek 2, Vitek MS, and PCR-sequencing of rDNA and showed susceptibility to fluconazole, voriconazole, and amphotericin B but resistance to caspofungin (Table 2). Treatment was started with AmBisome. As the patient also developed bacteremia due to Klebsiella pneumonia during the course of antifungal treatment, combination therapy with broad-spectrum antibiotics, and AmBisome resolved the infection. 73
Invasive infections by Trichosporon spp. have also been detected in Kuwait. Trichosporon spp. are yeast-like organisms that typically cause superficial infections (white piedra), allergic pneumonitis but can also cause invasive infections including fungemia.23,134,135 Trichosporonosis is an emerging infection of severely immunocompromised patients and is mainly caused by T. asahii. Invasive infections including bloodstream infections by T. asahii and T. asteroides have previously been detected in Kuwait; however, clinical details, treatment given, and outcome were not available. 136
The increasing incidence of invasive infections by Candida/yeast species due to increasing number of at-risk (seriously ill, immunocompromised and premature, low-birth-weight neonates) patients presents considerable diagnostic and management challenges for treating physicians due to lack of specific signs and symptoms.22–24,137 Early diagnosis, accurate AST, and early initiation of treatment with appropriate antifungal drugs form the cornerstone of effective management.47–50,93,137 Diagnosing invasive infections by rare Candida/yeast species is even more challenging due to the inability of conventional diagnostic methods to identify these organisms accurately.22,47 Although molecular methods are now regarded as the gold standard for definitive diagnosis, recent advances in phenotypic approaches have also been made that include expanding databases of yeast identification methods such as Vitek 2 and Vitek MS to identify many rare Candida/yeast species.43–45,47,50 Chromogenic media and unique microscopic features could provide presumptive diagnosis of some rare yeast infections for resource-limited settings.46,56,64,65,69 Imaging and biomarker-driven approaches may also help in early diagnosis.47,116,137 Other novel approaches such as detection of fungal volatiles in breath-based biomarker tests are also being developed for the diagnosis of invasive Candida/yeast infections. 138 Treating physicians should consider the possibility of invasive Candida/yeast infections in patients with underlying medical conditions and other major risk factors who are not responding to adequate treatment with broad-spectrum antibiotics.
Although only four classes of antifungal drugs are currently available, combination therapy could be used to effectively manage the problem of intrinsic or developing drug resistance to reduce the high mortality rates associated with rare Candida/yeast invasive infections. 139 This approach has already been used successfully in some cases in Kuwait.59,62,64,103 Another recent development that may help to improve the outcome of rare yeast infections is the growing pipeline of new antifungal drugs to augment the current antifungal armamentarium.
New antifungal drugs in the pipeline
Echinocandins, regarded as the first-line therapy for invasive Candida/yeast infections in adult populations, may not be so effective in neonatal population and the guidelines and recommendations for the total duration of therapy are also not well defined.140,141 Some antifungal drugs affecting novel targets or improved pharmacological parameters have been developed and have shown promising results in phase II and phase III clinical trials involving patients with invasive Candida/yeast infections. A brief account of the new antifungal drugs in various stages of development is provided below.
In March 2023, the US Food and Drug Administration approved rezafungin, a novel echinocandin with increased half-life (133 h), which allows weekly dosing compared to daily administration for existing echinocandins, for the treatment of invasive Candida/yeast infections in ⩾18-year-old patients with limited or no alternative options.142–145 Once-weekly dosing also makes rezafungin an attractive alternative for prophylactic or empiric treatment of hematological patients. A phase III randomized, double-blind trial (ReSTORE) has shown that once-weekly rezafungin is as effective as daily therapy with caspofungin followed by oral fluconazole. Nearly comparable occurrence of adverse events were seen in both groups of patients with invasive Candida infections. 146 Other studies to evaluate its role in antifungal prophylaxis as well as its safety, tolerability, and pharmacokinetics are also planned or are already being carried out. 147
Ibrexafungerp is an oral and intravenous triterpenoid drug with broad activity against Candida spp., including azole-resistant organisms such as C. auris and echinocandin-resistant isolates.144,148–150 The mechanism of action of ibrexafungerp is similar to echinocandins as it also inhibits 1,3-β-
Other novel antifungal drugs are also in the pipeline. The ATI-2307 is a mitochondrial inhibitor that has shown activity against most Candida spp. isolates and also has the potential for the treatment of infections by drug-resistant organisms.150,157,158 Several other drugs such as GR-2397, oteseconazole, opelconazole, and MAT2203 are also in various stages of clinical development.142,148 These novel antifungals have the potential for expanding the choice of drugs available to improve the management of patients with invasive Candida/yeast infections.
Limitations of review
A limitation of our study is that only previously documented and published cases of invasive rare Candida/yeast infections in Kuwait are reviewed here. Thus, it is possible that some rare Candida/yeast invasive infections in Kuwait may have been missed, which is also a universal problem as diagnosis and treatment of these infections is extremely challenging. Also, the clinical details, treatment given, and outcome were not available for some of the patients described in the studies reviewed here, which may also have affected the epidemiology and outcome of rare Candida/yeast infections in Kuwait.
Conclusion
Considerable improvements were made in the diagnostic services offered to various hospitals of the Ministry of Health in Kuwait by the mycology laboratory in the Department of Microbiology, Faculty of Medicine, Kuwait University. Both traditional and molecular approaches were used for rapid diagnosis of invasive infections by common and rare fungal pathogens and their susceptibility or resistance to antifungal drugs. These advances resulted in the identification of 60 cases of fungemia or other invasive infections due to rare Candida/yeasts from Kuwait that were published as case reports or case series. Majority of these infections occurred among neonates (n = 34) followed by adult patients (n = 19), while the remaining seven cases occurred in pediatric patients. The clinical details, treatment given, and outcome were available for 28 of 34 neonates. All 28 were preterm, low/VLBW neonates receiving broad-spectrum antibiotics for one or more suspected or confirmed episodes of bacteremia in the NICU with many other risk factors for IFIs. The crude mortality rate among these neonates was 32.2% as 19 of 28 (67.8%) survived the infection and were discharged in healthy condition. The neonates were treated with amphotericin B or liposomal amphotericin B alone in nine cases, with liposomal amphotericin B plus azole or caspofungin in nine cases, and with liposomal amphotericin B followed by other antifungals either alone or in combination in six cases. One neonate was treated with fluconazole alone. One neonate died before culture results became available and specific treatment could be initiated while another one expired after only one dose of liposomal amphotericin B was administered. The mortality rate was higher among adult patients. Excluding 8 patients who either did not receive any treatment or only one dose of antifungal drug was administered as they expired before culture results became available, 11 patients received treatment, and 5 died with a crud mortality rate of 45.5%. It is expected that studies such as those reviewed here on the epidemiology of rare Candida/yeast invasive infections in different geographical regions, their susceptibility profiles, and management will help to formulate guidelines for improved patient outcome.
Supplemental Material
sj-docx-1-tai-10.1177_20499361241263733 – Supplemental material for Spectrum and management of rare Candida/yeast infections in Kuwait in the Middle East
Supplemental material, sj-docx-1-tai-10.1177_20499361241263733 for Spectrum and management of rare Candida/yeast infections in Kuwait in the Middle East by Suhail Ahmad, Mohammad Asadzadeh, Noura Al-Sweih and Ziauddin Khan in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
