Abstract
Background:
Aspergillus, a widespread fungus in the natural environment, poses a significant threat to human health by entering the human body via the airways and causing a disease called aspergillosis. This study comprehensively analyzed data on aspergillosis in published articles from mainland China to investigate the prevalence of Aspergillus, and risk factors, mortality rate, and underlying condition associated with aspergillosis.
Methods:
Published articles were retrieved from Google Scholar, PubMed, and Science Direct online search engines. In the 101 analyzed studies, 3558 Aspergillus isolates were meticulously collected and classified. GraphPad Prism 8 was used to statistically examine the epidemiology and clinical characteristics of aspergillosis.
Results:
Aspergillus fumigatus was prominently reported (n = 2679, 75.14%), followed by A. flavus (n = 437, 12.25%), A. niger (n = 219, 6.14%), and A. terreus (n = 119, 3.33%). Of a total of 9810 patients, 7513 probable cases accounted for the highest number, followed by confirmed cases (n = 1956) and possible cases (n = 341). In patients, cough emerged as the most common complaint (n = 1819, 18.54%), followed by asthma (n = 1029, 10.48%) and fever (1024, 10.44%). Of total studies, invasive pulmonary aspergillosis (IPA) was reported in 47 (45.53%) studies, exhibiting an increased prevalence in Beijing (n = 12, 25.53%), Guangdong (n = 7, 14.89%), and Shanghai (n = 6, 12.76%). Chronic pulmonary aspergillosis (CPA) was reported in 14 (13.86%) studies. Among the total of 14 studies, the occurrence of CPA was 5 (35.71%) in Beijing and 3 (21.42%) in Shanghai. Allergic bronchopulmonary aspergillosis (ABPA), was reported at a lower frequency (n = 8, 7.92%), Guangdong recorded a relatively high number (n = 3, 37.5%), followed by Beijing (n = 2, 25.0%), and Shanghai (n = 1, 12.5%). Percentage of death reported: IPA had the highest rate (n = 447, 68.87%), followed by CPA (n = 181, 27.88%) and ABPA (n = 14, 2.15%). Among the aspergillosis patients, 6220 had underlying conditions, including chronic lung disease (n = 3765, 60.53%), previous tuberculosis (n = 416, 6.68%), and organ transplant or organ failure (n = 648, 10.41%). Aspergillosis was also found in patients using corticosteroid therapy (n = 622, 10.0%).
Conclusion:
This review sheds light on the prevalence patterns of Aspergillus species, risk factors of aspergillosis, and gaps in surveillance that could be helpful for the control and treatment of aspergillosis and guide the researchers in future studies.
Registration:
This systematic review was prospectively registered on PROSPERO: Registration ID CRD42023476870.
Introduction
Aspergillus, a fungus that is widely dispersed in the natural environment, possesses remarkable adaptability and can be transmitted easily. This microbe infiltrates the human body through the airways, giving rise to a range of illnesses called aspergillosis. 1 This fungal disease, caused by several species within the Aspergillus genus, manifests as diverse clinical signs and symptoms, each presenting unique challenges for diagnosis.2,3 Aspergillus fumigatus is acknowledged as the primary causative agent of aspergillosis, accounting for approximately 70–80% of reported cases. 4 Nevertheless, aspergillosis has also been associated with non-fumigatus species, including A. flavus, A. niger, A. nidulans, A. terreus, and A. versicolor.2,3 The dynamic interplay of fungal diversity contributes to the complex nature of the disease landscape. Aspergillus species cause various forms of aspergillosis, including allergic bronchopulmonary aspergillosis (ABPA), chronic pulmonary aspergillosis (CPA), and invasive pulmonary aspergillosis (IPA).2,3 ABPA represents a hypersensitive reaction triggered by the inhalation of Aspergillus spores, typically affecting individuals with underlying respiratory conditions, such as asthma or cystic fibrosis. ABPA is characterized by airway inflammation and has the potential to lead to pulmonary disease. CPA, on the other hand, is a persistent and progressive mycotic infection that primarily targets the respiratory system. It is frequently observed in individuals with a history of preexisting pulmonary conditions or compromised immune responses.2,3 Simple aspergillosis or aspergilloma Occurs when a ball of Aspergillus grows in the lungs or sinuses but usually does not spread to other parts of the body. 3 The condition presents in the form of pulmonary cavities, nodules, or fibrosis, often necessitating the prolonged administration of antifungal treatment.
IPA stands out as the most severe form of fungal invasion, posing a significant risk of mortality and imposing a substantial burden on affected individuals. 5 The spectrum of aspergillosis, comprising ABPA, CPA, and IPA, encompasses a wide range of clinical manifestations associated with the condition. Each of these manifestations requires specific approaches for diagnosis and treatment that cater to its characteristics. The enhancement of clinical outcomes in aspergillosis depends on two crucial factors: the swiftness of diagnosis and the precision of antifungal therapy. Unfortunately, several diagnostic techniques, such as sputum culture, are characterized by the need for considerable effort and inherent limitations in efficacy. 6 Therefore, physicians frequently resort to writing prescriptions for empirical antifungal drugs. Given this context, evaluating both local and global risk factors and antifungal surveillance become crucial for the judicious selection of appropriate pharmacological drugs and treatment procedures.
The World Health Organization (WHO) recently released a fungal priority pathogen list, categorizing A. fumigatus as the third most critical pathogen. A. fumigatus plays a significant role in the prevalence of aspergillosis. However, it is crucial to recognize that non-fumigatus strains have also been identified in various regions throughout mainland China.7 –10 Additionally, several review articles on Aspergillus species have been published in different regions of China.11–13 However, it is worth noting that comprehensive systematic analyses of Aspergillus species from mainland China are lacking. Hence, in this study, we extracted data from published sources to analyze the prevalence of Aspergillus species and associated risk factors of aspergillosis in China. The study findings provide future directions for researchers by identifying crucial facts, such as the specific types of Aspergillus prevalent and the geographical regions with higher incidence rates. Additionally, the study mentions its limitations, including potential biases in the collected data and variations in reporting standards. Furthermore, the findings provide insights into the characteristics of aspergillosis that are relevant for healthcare officials and clinicians, aiding disease control and management.
Methods
This systematic review was prospectively registered on PROSPERO: Registration ID CRD42023476870.
Literature search and selection
To compile published research articles on Aspergillus species in mainland China, a comprehensive search was performed using NCBI PubMed, Google Scholar, and Science Direct engines. The process adhered to the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines for systematic reviews and meta-analyses (Figure 1). 14 The search was performed using the keywords including ‘Aspergillus’ AND ‘Antifungal susceptibility/resistance’ and ‘Aspergillosis’ AND ‘Antifungal susceptibility/resistance’ in the context of mainland China. In addition, the existing scientific literature was exhaustively reviewed by scrutinizing the bibliographies of the initially downloaded articles, comprising all studies on Aspergillus species in mainland China.

Study identification and selection based on PRISMA guidelines.
All relevant articles were directly downloaded from the online databases, and duplicates were subsequently removed. Initially, the titles and abstracts of the articles were reviewed to select only articles on Aspergillus in mainland China. The articles were searched and collected independently by two researchers (SK and HB), and the chosen papers were then processed for inclusion and exclusion based on their full text.
Inclusion criteria
The selected article featured patients with aspergillosis and/or clinical samples containing Aspergillus isolates. These reports also provide data on the total number of isolates and affected patients. All the articles included in this study were published in mainland China and written in English between January 2012 and December 2022.
Exclusion criteria
Articles were omitted if they were unrelated to Aspergillus isolates collected from human patients, contained incomplete data, or were reviews unrelated to mainland China.
Data collection
Comprehensive data from each article were collected initially in an Excel spreadsheet (2017) by three independent researchers (SK, HB, and MNK). The collected data included the study’s title, year of publication, geographical region, sampling period, total sample size, whether the subjects were inpatients or outpatients, the patient’s age and gender distribution, gender, types of infection or co-infection, underlying diseases, mortality rates, identification methods used, and the antifungal susceptibility test (AST) method utilized. The data provided in the articles were carefully tabulated and counted in terms of both numerical counts and percentages. The sample collection was specifically categorized by the year, and pediatric patients were defined as individuals who were under 18 years old. In this analysis, data related to susceptibility to antifungal treatment or wild-type isolates were included, while data on susceptible dose-dependent and intermediate were not considered.
Statistical analysis
All data, including counts, numerical values, and percentages, were meticulously analyzed using an Excel spreadsheet. The mortality rates and underlying diseases were scrutinized using GraphPad Prism 8. Furthermore, the number and percentage of patients with underlying diseases were calculated. Various types of infection, including IPA, CPA, APBA, simple aspergillosis, and cerebral aspergillosis (CBA), were identified and categorized. The number of pathogens involved in co-infection was also carefully documented. Statistical analysis and graphical representation were performed using GraphPad Prism v8.0.2. Three researchers independently reviewed the data to mitigate any potential bias.
Results
Study characteristics
The systematic review encompassed a total of 101 articles, collectively containing 3558 Aspergillus isolates. Notably, a high number of articles were published in the years 2017, 2019, and 2021 (15, 14, and 13 articles, respectively). The study period ranged from January 2012 to December 2022, as mentioned in Supplemental Figure S1.
Among the various regions in China, the highest number of articles was from East China (n = 37, 36.6%), followed by North China (n = 24, 23.76%) and South China (n = 19, 18.81%). In terms of sub-regions, Beijing contributed a significant number of articles (n = 21, 20.79%), closely followed by Guangdong (n = 18, 17.82%). The reports mention a total of 9810 patients, with males accounting for 69.0% and females for 29.07%. Regarding the overall number of cases, probable cases accounted for the highest number (approximately 7513 cases), followed by confirmed cases at (n = 1956) and possible cases (n = 341).
In the published articles, the median age of adults, along with the 95% confidence interval (95% CI), was calculated as 73.5 years (56–75), while that of children was 16 years (13.5–18). Among the 35 studies that reported mortality rate, a notably high mortality rate (18.81%) was observed in cases of IPA, with an interquartile range (IQR) of 3.0–20.5%. The mortality rate was calculated for infections caused by both A. fumigatus and non-fumigatus strains, as indicated in the studies (see Table 1). The culture methods used to identify Aspergillus isolates were specifically mentioned in 28 (27.72%) articles. The clinical manifestations, laboratory tests, and diagnosis are summarized in Table 1.
Characteristics associated with aspergillosis, including co-infections, patient types, age groups, gender, mortality rates and identification methods.

The percentages of some variables are more than 100 because some studies mention more than one variable, and we calculated each study individually for every variable.
ABPA, allergic bronchopulmonary aspergillosis; BAL, bronchoalveolar lavage; CLSI, clinical laboratory, and standard institutes; COPD, chronic obstructive pulmonary disease; CBA, cerebral aspergillosis; CPA, chronic pulmonary aspergillosis; CT, computed tomography; EORTC/MSG, European Organization for Research and Treatment of Cancer/Mycoses Study Group; ERS, European Respiratory Society; ESCMID, European Society of Clinical Microbiology and Infectious Diseases; ESICM, European Society of Intensive Care Medicine; EUCAST, European Committee of Antimicrobial Sensitivity Testing; GM, Galactomannan; IDSA, Infectious Diseases Society of America; IPA, invasive pulmonary aspergillosis; MALDI-TOF-MS, Matrix-assisted laser desorption ionization–time of flight mass spectrometry; NM, not mentioned; PCR, Polymerase Chain Reaction.
In patients, cough was the most common complaint (1819, 18.54%), followed by asthma (1029, 10.48%), fever (1024, 10.43%), sputum (939 patients, 9.58%), wheezing (578, 5.90%), dyspnea (545, 5.56%), hemoptysis (245, 5.50%) and chest pain (231, 2.36%). Furthermore, the details of the various techniques used to identify Aspergillus isolates are mentioned in Table 2.
Overview of Aspergillus species, the spectrum of aspergillosis, and underlying diseases.
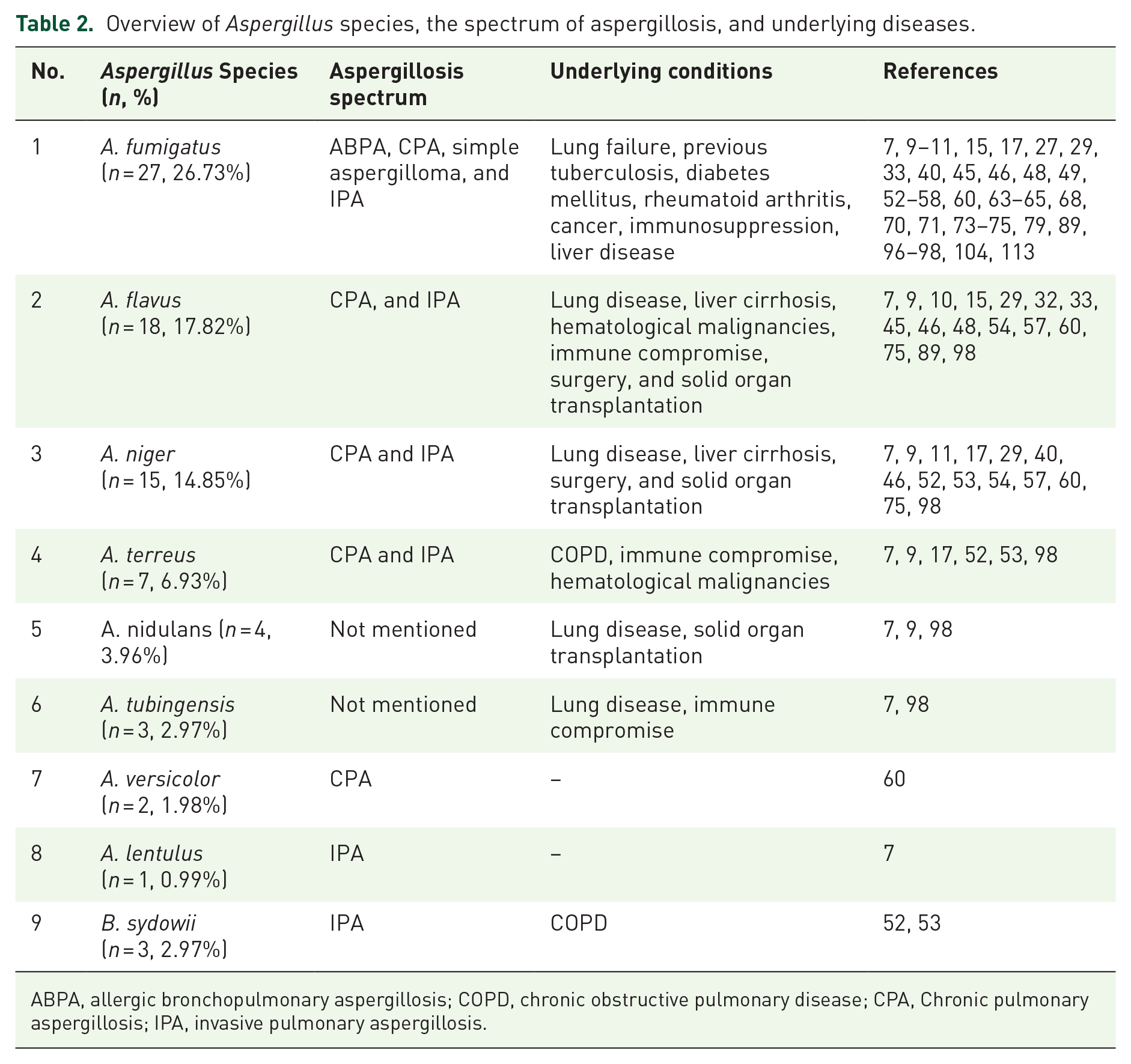
ABPA, allergic bronchopulmonary aspergillosis; COPD, chronic obstructive pulmonary disease; CPA, Chronic pulmonary aspergillosis; IPA, invasive pulmonary aspergillosis.
Prevalence of Aspergillus species
The prevalence of various Aspergillus isolates is shown in Figure 2(a). A. fumigatus was prominently reported (n = 2679, 75.14%), followed by A. flavus (n = 437, 12.25%), A. niger (n = 219, 6.14%), A. terreus (n = 119, 3.33%), A. nidulans (n = 35, 0.98%), A. tubingensis (n = 21, 0.58%), and A. sydowii (n = 18, 0.50%), encompassing all Aspergillus isolates [Figure 2(a)]. Mainland China was divided into seven regions, and the prevalence of Aspergillus in each region was studied. North China exhibited the highest proportion of Aspergillus isolates (56.14%), followed by East China (26.23 %), South China (10.0%), Northwest China (7.24%), Central China (3.85%), and Northeast China (3.54%); see Figure 2(b).

Aspergillus species, prevalence, and percentage of reported death rates (a) percentage of Aspergillus species. The number of isolates is indicated over the bar, (b) distribution of Aspergillus species in different regions of mainland China. The colors of the bars indicate specific regions. (c) Percentage of reported death rates, with various types of aspergillosis.
Regarding the types of aspergillosis in total studies, IPA was reported in the highest number of studies (n = 47, 46.53%). Among the regions, Beijing accounted for a relatively high number (n = 12, 25.53%), followed by Guangdong (n = 7, 14.89%), Shanghai (n = 6, 12.76%), Sichuan (n = 5, 10.63%), Zhejiang (n = 5, 10.63%), Jiangsu (n = 4, 8.51%), and Hunan (n = 3, 6.38%). CPA was reported in 14 (13.86%) studies. A high number of studies came from Beijing (n = 5, 35.71%), followed by Shanghai (n = 3, 21.42%), Guangdong (n = 2, 14.28%), Jiangsu (n = 2, 14.28%), Shandong (n = 1, 7.14%), and Zhejiang (n = 1, 7.14%). Furthermore, ABPA was reported at a lower frequency (n = 8, 7.92%), Guangdong recorded a relatively high number (n = 3, 37.5%), followed by Beijing (n = 2, 25.0%), Shanghai (n = 2, 25.0%), and Zhejiang (n = 1, 12.5%).
IPA associated with Aspergillus, A. fumigatus was reported in the highest number of studies (n = 17, 16.83%), followed by A. flavus (n = 9, 8.91%), A. niger (n = 2, 1.98%), and A. lentulus (n = 1, 0.99%). CPA caused by Aspergillus, A. fumigatus reported an increase in number of studies (n = 7, 6.93%), followed by A. flavus (n = 4, 3.96%), A. niger (n = 4, 3.96%), A. versicolor (n = 1, 0.99%), and A. terreus (n = 1, 0.99%). ABPA was associated with A. fumigatus (n = 10, 9.90%). Regarding percentage of death reported, IPA had the highest rate (n = 447, 68.87%), followed by CPA (n = 181, 27.88%), ABPA (n = 14, 2.15%), CBA (n = 7, 1.07%), while simple aspergillosis had no death report [Figure 2(c)].
Regarding co-infections with Aspergillus, a high number of articles were reported by fungi (n = 9, 14.28%) and viruses (n = 9, 14.28%) followed by bacteria (n = 8, 12.69%). The total number of pathogens is mentioned in Figure 3.
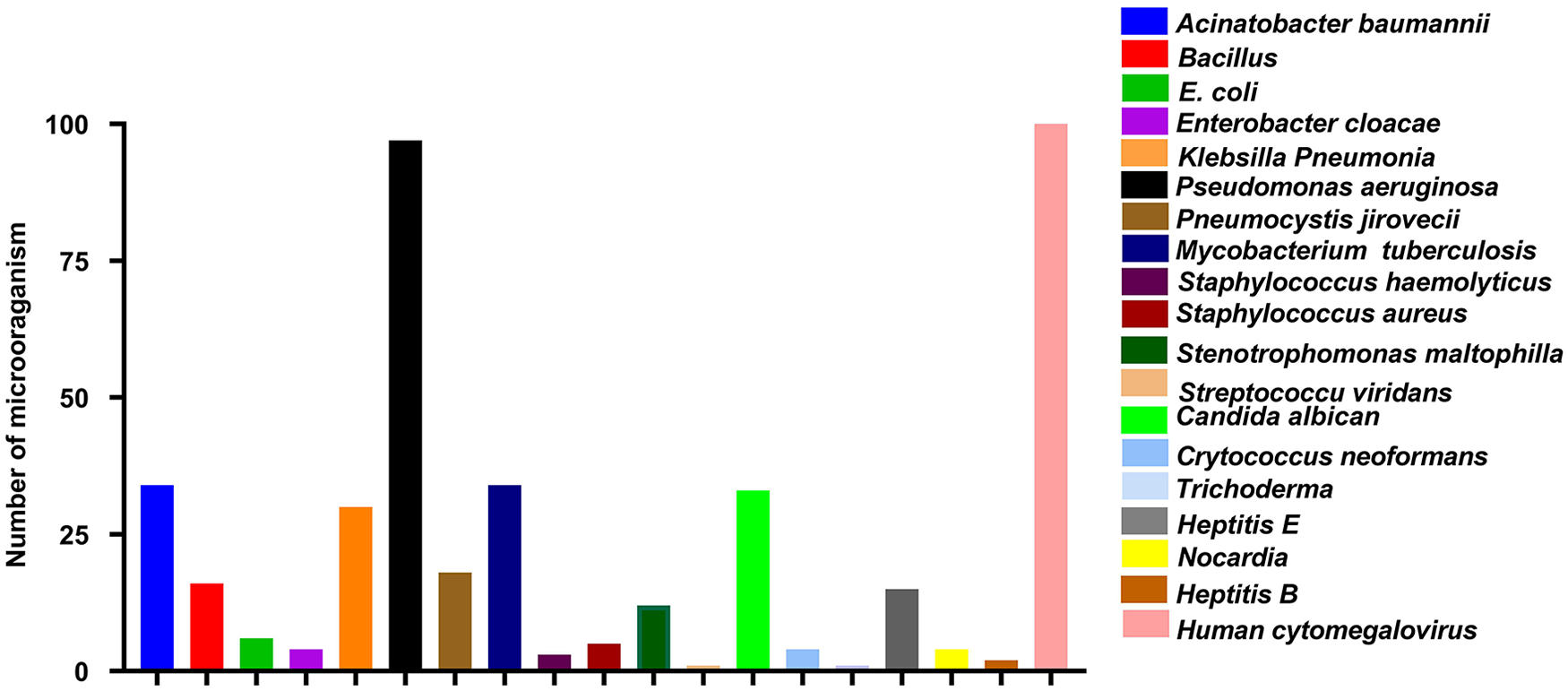
Co-morbidities of other pathogens with Aspergillus. The colors of the bars box indicate specific pathogens.
Underlying diseases
The percentage of studies that mentioned underlying diseases associated with aspergillosis was calculated. The association of IPA with underlying diseases was noteworthy. Studies consistently reported that the most prevalent underlying disease was lung disease (n = 31, 65.95%), followed by liver disease (n = 16, 34.04%), cancer or hematological malignancies (n = 13, 27.65%), and immune compromise (n = 4, 8.51%). Regarding underlying conditions associated with CPA, lung disease was prominent (n = 11, 78.57%), followed by immune compromise (n = 3, 21.42%), solid organ transplantation (n = 3, 21.42%), surgery (n = 2, 14.28%), pneumonia (n = 2, 14.28%), and cancer or hematological malignancies (n = 1, 7.14%). ABPA was associated with underlying lung disease (n = 6, 75.0%), cancer or hematological malignancies (n = 2, 25.0%), and liver diseases (n = 1, 12.5%).
Among the aspergillosis patients, 6220 had underlying conditions. The underlying conditions included chronic lung disease (n = 3765, 60.53%), previous tuberculosis (n = 416, 6.68%), and organ transplant or organ failure (n = 648, 10.41%). Aspergillosis was also found in patients using corticosteroid therapy (n = 622, 10.0%). Other diseases or organ failure in aspergillosis patients included immune compromise (n = 264, 4.24%), pneumonia (n = 311, 5.0%), neutropenia (n = 223, 3.58%), systemic lupus erythematosus (n = 41, 0.65%), cancer (n = 116, 1.86%), leukemia (n = 36, 0.57%), solid tumors (n = 55, 0.88%), acute-on-chronic liver failure (n = 132, 2.12%), and kidney failure (n = 164, 2.63%); see Figure 4.
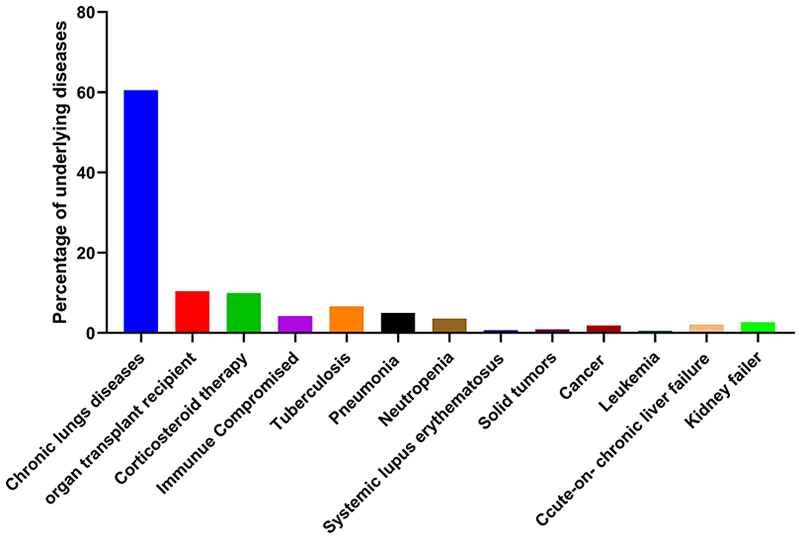
Aspergillosis is associated with different types of underlying conditions in patients.
A comprehensive overview of the distribution of Aspergillus species, the spectrum of aspergillosis, and the underlying diseases identified in various studies is presented in Table 1. The percentage and diversity of Aspergillus species are noted along with an in-depth analysis of underlying diseases. This has been done to provide researchers and healthcare workers with valuable insights into various Aspergillus infections and their clinical manifestations.
Discussion
In this study, we systematically examined the prevalence, and characteristics of Aspergillus isolates from mainland China to provide guidance for clinicians and researchers in upcoming studies. Among the Aspergillus species, there was a high rate (75.14%) of A. fumigatus in mainland China. Epidemiological studies of Aspergillus species have been conducted previously in China and other countries.46,99,100 In mainland China, A. fumigatus has been consistently identified as the most prevalent among the Aspergillus species.8,13,76,77,92 Of the non-fumigatus species, A. flavus (12.25%) had the second highest prevalence, followed by A. niger (6.14%), A. terreus (0.82%), and A. nidulans (3.96%). The second most common non-fumigatus pathogen A. flavus has been reported in many countries.101,102 In contrast, in some countries, A. niger and A. terreus have been considered the second most common Aspergillus species. 57
Aspergillosis is mainly attributed to A. fumigatus. Nonetheless, several reported cases in China showed that aspergillosis can also be caused by non-fumigatus species.3,8–10,15–18,33,76,93 This indicates that A. fumigatus is not the sole causative agent of aspergillosis. Further, Aspergillus co-infection with fungal, bacterial, and viral pathogens has been reported in the range of 11.0–15.0%. Co-infections have been identified as one of the contributing factors to aspergillosis complications. The risk of aspergillosis infections is further heightened by the use of broad-spectrum antibiotics, which increase opportunistic infections or contribute to the development of resistant strains. Immunosuppressive drugs and hospital burden also contribute to an increased risk of aspergillosis. 103
In 2011, the ARTEMIS DISK global antifungal surveillance project first reported the identification of the A. fumigatus TR34/L98H mutation in China. Subsequently, this mutation became widespread not only in China but also in other Asian countries and Europe.34,65,104 Furthermore, various mutations in A. fumigatus have been discovered in China, including D343N, I242V, K314T, D262Y, TR34/L98H, TR34/L98H/S297T/F495I, TR46/Y121F/T289A, G432A, M220I, and N248K.16,34,104
These mutations are a serious global health concern, and investigation and proper planning for their control are urgently required. Most samples were collected from hospitalized long-term intensive care unit (ICU) patients, indicating that nosocomial disease is a significant risk factor for aspergillosis. Moreover, a high number of aspergillosis patients in our study were adults, and IPA is the most common type of aspergillosis caused by A. fumigatus. The risk of IPA is also increasing in patients with preexisting pulmonary disorders, such as chronic obstructive pulmonary disease (COPD), a history of tuberculosis, asthma, and lung cancer. 35 Other contributing factors include high doses of inhaled glucocorticoids,105–107 viral infection including influenza and cytomegalovirus,108,109 sarcoidosis, 110 and bronchitis and pneumonia.12,17,19,91 These are recognized as common risk factors for aspergillosis. Risk factors for all Aspergillus-related diseases are mentioned in Tables 1 and 2. To control the horizontal transmission of aspergillosis, health workers must exercise special care in maintaining hygienic conditions within hospital environments.
Several gaps have been identified in this surveillance study that warrant attention. Notably, there is a lack of investigation in certain regions, such as Chongqing, Guizhou, Yunnan, Tibet, and Inner Mongolia. Additionally, crucial data including, age, antifungal susceptibility test (AST), gender, inpatient/outpatient status, standard protocol, and identification method were not reported in 17.82%, 93.80%, 4.76%, 11.88%, 87.12%, and 8.91% of the studies respectively. Furthermore, details regarding the identification of Aspergillus species were not mentioned in 43.56% of the studies. Despite the significance of co-infection as a risk factor, it was not noted in 67.32% of the studies. Moreover, the mortality rate and aspergillosis infection type were not reported in 67.32%. These gaps in the published articles could lead to errors in statistical analysis and pose challenges for practical applications. It is recommended that researchers address these gaps in future studies to boost the reliability and usefulness of the results.
This study focused on the prevalence of Aspergillus species, mortality rates, and infections associated with aspergillosis in mainland China. However, the impacts of this phenomenon surpass regional boundaries; thus, the study’s insights and guidelines retain significance for researchers and physicians worldwide. The transmission of resistant species through various channels, including humans, water, agricultural products, and animals from the region with drug-resistant reservoir strains.111,113,114 Moreover, the identification of surveillance gaps in this study, will be helpful for guiding future research efforts. The underlying disease and risk factors reported in the study, will also help to control Aspergillus infections.
Limitations
In the ongoing systematic analysis, the prevalence of aspergillosis, whether proven or probable, emerged as an important hurdle within healthcare settings. A large number of publications report probable cases without confirmed identification of Aspergillus species, indicating an identification type bias. The publications were mostly reported in developed regions like Beijing and Shanghai, whereas underdeveloped regions had fewer reports, indicating a bias in publication type. Notably, Aspergillus species exhibit morphological similarities, and molecular identification needs specialized expertise and techniques, which might not be universally available. Moreover, it is important to emphasize that numerous studies have reported a limited number of Aspergillus isolates, typically below 10. This underscores the need to establish a more extensive and varied collection of isolates and conduct AST. Such efforts are essential to facilitate the gathering of dependable and comprehensive data regarding AST.
Furthermore, it is crucial to acknowledge that numerous studies exhibit a significant need for proper treatment regarding the source of Aspergillus isolation, frequently marked by a paucity of outpatient data. Additionally, numerous investigations on aspergillosis are characterized by modest sample sizes, potentially limiting the generalizability of results to broader populations. Achieving a significant sample size poses fundamental challenges that can potentially hinder the statistical impact of the study. These obstacles have the potential to impede the identification of significant characteristics or associations.
Conclusion
In this study, we compiled a comprehensive overview of the prevalence of Aspergillus species, revealing a notable distribution of A. fumigatus and A. flavus in different regions of mainland China. However, it is crucial to emphasize gaps in the present literature, including the absence of research on specific geographical areas such as the northeast and Inner Mongolia. Moreover, our review suggests an absence of comprehensive research on AST for Aspergillus species in a majority of the research articles. The absence of sufficient data limits the thorough analysis of susceptibility patterns, highlighting the need for more extensive investigation of this element in future research. Furthermore, in spite of a rise in mortality and the existence of associated risks in cases of aspergillosis, a significant proportion of the reports failed to provide pertinent data related to these topics. This emphasizes the importance of implementing more extensive reporting methods in future research initiatives to acquire a greater awareness of the mortality related to aspergillosis and its risk factors. Hence, surveillance studies conducted by researchers play a pivotal role in indicating gaps in control measures for aspergillosis. Furthermore, it is imperative for healthcare workers to implement preventive measures as directed by policymakers to effectively reduce the transmission of infections within hospital settings and minimize the emergence of resistance to antifungal drugs. The implementation of a collaborative approach is crucial to improve the understanding of aspergillosis and optimize techniques for its efficient management within clinical settings.
Supplemental Material
sj-docx-2-tai-10.1177_20499361241252537 – Supplemental material for Distribution of Aspergillus species and risk factors for aspergillosis in mainland China: a systematic review
Supplemental material, sj-docx-2-tai-10.1177_20499361241252537 for Distribution of Aspergillus species and risk factors for aspergillosis in mainland China: a systematic review by Sabir Khan, Hazrat Bilal, Muhammad Shafiq, Dongxing Zhang, Muhammad Awais, Canhua Chen, Muhammad Nadeem Khan, Qian Wang, Lin Cai, Rehmat Islam and Yuebin Zeng in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-jpg-1-tai-10.1177_20499361241252537 – Supplemental material for Distribution of Aspergillus species and risk factors for aspergillosis in mainland China: a systematic review
Supplemental material, sj-jpg-1-tai-10.1177_20499361241252537 for Distribution of Aspergillus species and risk factors for aspergillosis in mainland China: a systematic review by Sabir Khan, Hazrat Bilal, Muhammad Shafiq, Dongxing Zhang, Muhammad Awais, Canhua Chen, Muhammad Nadeem Khan, Qian Wang, Lin Cai, Rehmat Islam and Yuebin Zeng in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
