Abstract
Background:
There is growing evidence of fungal infections associated with COVID-19. The development of cryptococcosis in these patients has been infrequently reported. However, it can be life-threatening.
Objective:
To identify cases of COVID-19 patients who developed cryptococcosis and to compare baseline characteristics and management between those who survived and those who died.
Methods:
We conducted a scoping review using PubMed, Scopus, Web of Science, and Embase to identify studies that reported patients with COVID-19 and cryptococcosis. No language restriction was applied. Single case reports, case series, and original articles were included. It is important to note that ‘n’ refers to the total number of individuals with the specified variable.
Results:
A total of 58 studies were included. Among these studies, 51 included individual patient data, detailing information on a total of 65 patients, whereas eight studies reported the proportion of cryptococcosis in COVID-19 patients. One study provided both individual and aggregate case information. From individual patient data, the majority were male (73.9%; n = 48) with a median age of 60 years (range: 53–70). Severe COVID-19 and multiple comorbidities, led by arterial hypertension and diabetes mellitus, were frequently reported, but few had classic immunosuppression factors. On the other hand, HIV status, either negative or positive, was reported in just over half of the patients (61.5%; n = 40). Most were admitted to the intensive care unit (ICU) (58.5%; n = 31), received mechanical ventilation (MV) (50.0%; n = 26), and developed disseminated cryptococcosis (55.4%; n = 36). Secondary infection, mainly bacterial, was reported in 19 patients (29.2%). Mortality was 47.7% (n = 31). Of the studies that reported the proportion of cryptococcosis in COVID-19 cases, the majority were descriptive studies published as conference abstracts.
Conclusion:
Cryptococcosis in COVID-19 patients has been reported more frequently. However, it is still not as common as other fungal infections associated with COVID-19. Few patients have some classic immunosuppression factors. The factors associated with mortality were male sex, age, ICU admission, MV, secondary infections, and lymphopenia.
Keywords
Background
Cryptococcosis is a fungal infectious disease caused by the yeast Cryptococcus spp., which is ubiquitous in nature and can invade any organ. 1 Traditionally, the risk factors associated with their infection are related to an impaired immune system including advanced human immunodeficiency virus (HIV)/acquired immunodeficiency syndrome, solid organ transplant (SOT) recipients, hematological malignancy, decompensated liver cirrhosis, prolonged medication for any illness, and other disorders which suppress the immunity of the individual such as rheumatic diseases.1,2
Immune dysregulation caused by the SARS-CoV-2 virus leads to a series of complex changes in both innate and acquired immunity, characterized by a cytokine storm, such as tumor necrosis factor and interleukins (mainly IL-1 and IL-6), that can lead to widespread tissue damage, secondary to the deregulated inflammatory cascade. 3 The function of Natural Killer (NK) cells is reduced in COVID-19, mainly in severe cases, which leads to a poor rapid response to infected immune cells by this innate response. 4 T-cell dysfunction and compromised antiviral immunity contribute to impaired viral clearance, while the virus may also induce immunosuppression, hindering an effective defense mechanism.5,6 Subsequently, compromising host immunity in COVID-19 patients increases the risk of reactivation of latent diseases or the development of new opportunistic infections. Indeed, with the use of multiple immune-modulating drugs for COVID-19 along with COVID-19-related immunosuppression, the risk of fungal infections is worryingly growing.7–9 As a result, mortality has risen in COVID-19 patients due to fungal infections.10,11 The most common COVID-19-associated fungal infections are candidiasis, aspergillosis, and mucormycosis.12,13 Nonetheless, Cryptococcus, like all opportunistic fungi, is becoming more frequent, especially in patients with COVID-19 admitted to the intensive care unit (ICU) and with an immunosuppression factor according to some reports.14–16
This study aimed to conduct a comprehensive scoping review through case reports, case series, and epidemiological studies to identify research gaps in the epidemiology, clinical features, and treatment outcomes of patients with COVID-19 who developed cryptococcosis. A secondary aim was to compare patient characteristics between those who survived and those who died, as well as to determine the feasibility of another form of evidence synthesis such as systematic reviews based on the currently available scientific literature.
Methods
We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for scoping reviews (PRISMA-ScR) to secure adequate reporting of the study. 17
Eligibility criteria
Studies that met the following criteria were included: (a) patients (⩾18 years old) who acquired cryptococcosis concurrently or after COVID-19; (b) who had individual patient data available including epidemiologic information, diagnoses and underlying conditions, medications, laboratory test results, and disease outcomes, and (c) any other studies or abstracts that reported cryptococcosis in COVID-19 patients. Articles were excluded if (a) patients acquired COVID-19 after cryptococcosis, (b) did not provide basic individual patient data such as sex and age, or (c) did not provide the total number of COVID-19 and cryptococcosis cases. To have the largest number of studies, there were no language restrictions or full-text availability since the conference proceedings were also included.
Epidemiologic information included sex, age, and reporting country. Diagnosis of cryptococcosis (histopathology, cultures, and serological tests) and related information (site of infection, species) as well as COVID-19 severity was also collected. Underlying conditions included comorbidities such as arterial hypertension (HTN), diabetes mellitus (DM), obesity, among others; and immunosuppressive factors such as HIV, SOT, cirrhosis, autoimmune diseases (lupus, rheumatoid arthritis, autoimmune hemolytic anemia, multiple sclerosis), and hematologic malignancies. Information regarding the patient’s admission to the ICU, use of mechanical ventilation (MV), and infections during hospitalization was also collected. Treatment for COVID-19 included immunosuppressive drugs (corticosteroids, tocilizumab) and antivirals (remdesivir), while for cryptococcosis included antifungals in monotherapy or combination antifungal therapy (CAT). Results of laboratory tests included specifically, total lymphocyte count and CD4 cells. Finally, the outcome of the disease was included, such as those patients who survived and those who died.
Information sources and search strategy
We performed a comprehensive search in four sources (PubMed, Scopus, Web of Science, and Embase). Our search strategy included terms related to COVID-19 and cryptococcosis. The complete and reproducible search strategy for each database is available in Supplemental Material 1. All searches were performed on 6 August 2023.
Study selection
Documents were exported to Endnote X9 (Philadelphia, PA, USA) and duplicates were removed. Two independent researchers (AQL and MP) evaluated whether the retrieved documents met the eligibility criteria for inclusion or not. Any discrepancy was resolved by discussion between reviewers. The latter is valid both for the review stage of only titles and abstracts and for the review stage of the full text.
Data extraction and synthesis
For each study, one researcher independently extracted data. Unclear information was discussed between two reviewers (AQL and MP) before reaching a final decision. For the synthesis, the articles were divided into two groups: (1) studies with individual patient data and (2) studies with the total number of COVID-19 and cryptococcosis cases. For the first group, continuous variables were presented as median and interquartile range (IQR), whereas categorical variables were presented as frequency and percentage. According to the extraction of individual information from the included cases, two cohorts were formed to compare patient characteristics and other outcomes of interest between those who survived and those who died. To compare proportions, the chi-square test (X2) and Fisher’s exact test were used, whenever appropriate. To compare continuous variables, the Mann–Whitney U test was used. A p value of ⩽0.05 was considered statistically significant. All data analyses were conducted on RStudio software version 4.3.0 (Boston, MA, USA). In addition, the severity of COVID-19 was scored for each case based on the patient’s symptoms at the time of COVID-19 diagnosis. Thus, patients were classified into five categories: an asymptomatic status as well as mild, moderate, severe, and critical illness. The criteria for each category are based on the National Institutes of Health (NIH) COVID-19 treatment guidelines. 18 For the second group, a summary of the results of each study was presented separately.
Results
Selection
We evaluated 600 references, of which 58 studies met the inclusion criteria. The PRISMA-ScR flowchart is shown in Figure 1. Of these, 51 studies contain individual patient data, reporting a total of 57 patients. On the other hand, eight studies reported the total number of cases of COVID-19 and the proportion of patients with cryptococcosis. One study provided both individual and aggregate case information.
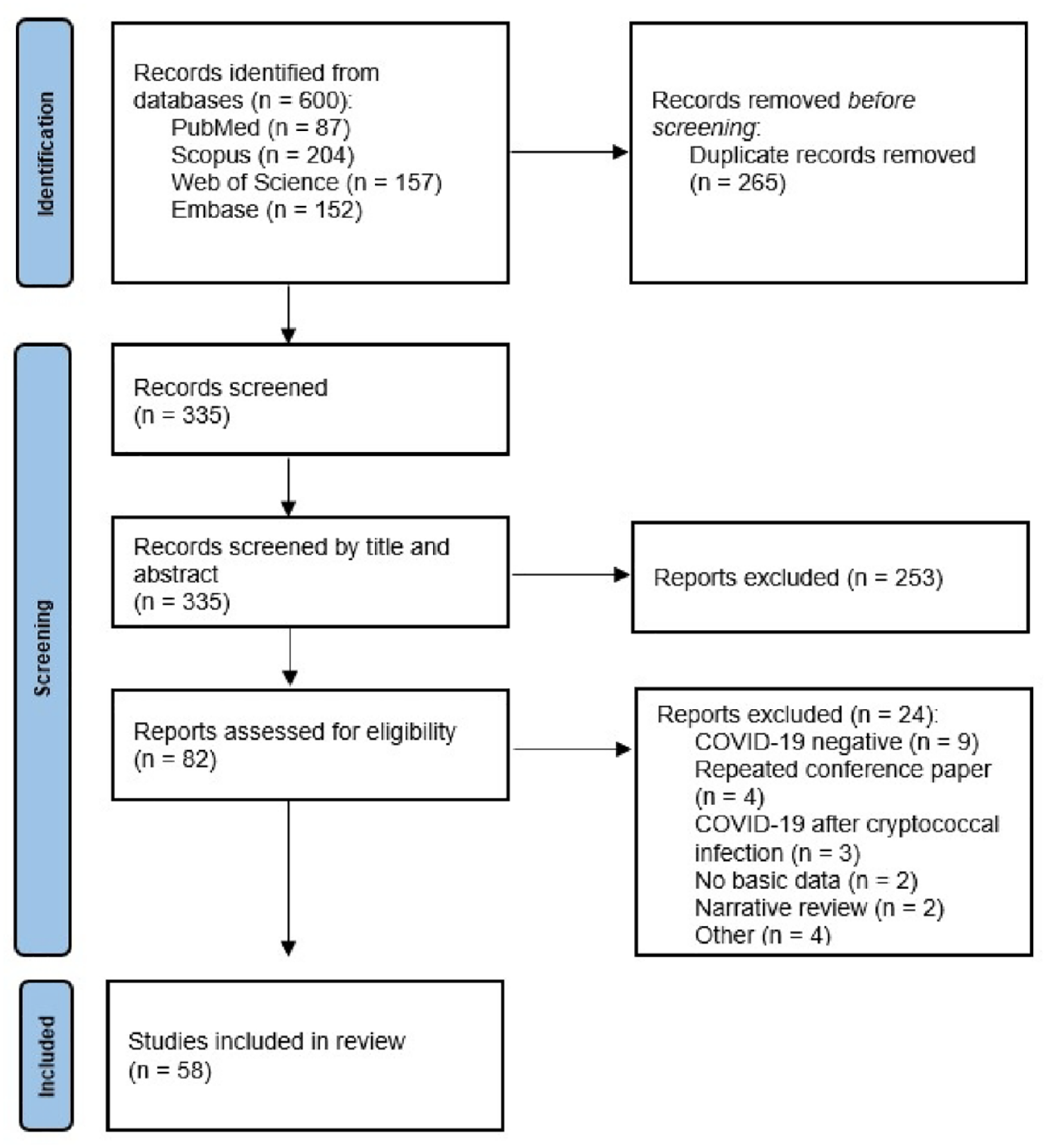
PRISMA-ScR flowchart of study selection.
Epidemiology of COVID-19 patients with cryptococcosis
Table 1 is based on individual patient data. Of the total number of patients, the male sex was affected in 48 cases while the median age was 60 years, with statistically significant differences between the two groups: alive versus dead in both variables (p value = 0.0203 and 0.0332, respectively). Of 60 patients with available information, 31 were from the United States15,16,19–40 (51.6%), 8 from Peru 41 (13.3%), 7 from Brazil14,42–47 (11.6%), and 2 from India48,49 and Italy50,51 (3.3% each), while the rest were from Canada, 52 Colombia, 53 China, 54 the Czech Republic, 55 the UK, 56 Uganda, 57 Qatar, 58 Lebanon, 59 Spain, 60 and Germany 61 (n = 1; 1.7% each).
Characteristics of COVID-19 patients with cryptococcosis.
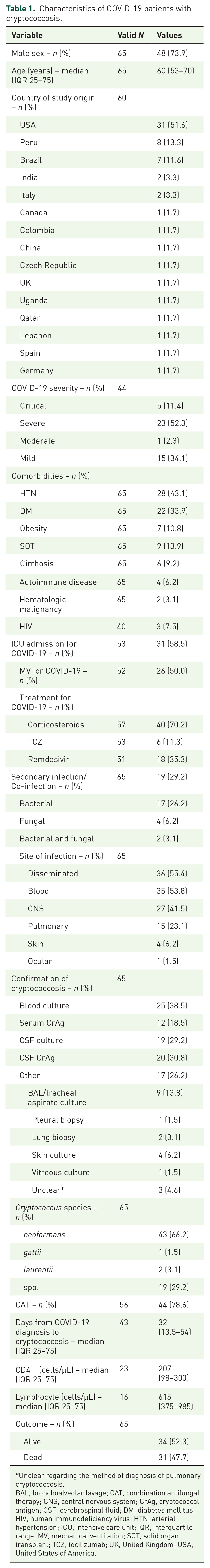
Unclear regarding the method of diagnosis of pulmonary cryptococcosis.
BAL, bronchoalveolar lavage; CAT, combination antifungal therapy; CNS, central nervous system; CrAg, cryptococcal antigen; CSF, cerebrospinal fluid; DM, diabetes mellitus; HIV, human immunodeficiency virus; HTN, arterial hypertension; ICU, intensive care unit; IQR, interquartile range; MV, mechanical ventilation; SOT, solid organ transplant; TCZ, tocilizumab; UK, United Kingdom; USA, United States of America.
COVID-19 severity
According to the scale of severity of signs and symptoms of COVID-19, 18 the majority of the cases were severe (n = 23; 52.3%).
Comorbidities, ICU admission, and MV for COVID-19
A large percentage of patients had some comorbidity (n = 57; 87.7%), the most frequent being HTN and DM. Similar mortality rates were found in patients with any comorbidity in general or specific comorbidities in contrast to those without them. Of 36 patients hospitalized when the COVID-19 diagnosis occurred, the vast majority were admitted to the ICU (n = 31; 86.1%) and underwent MV (n = 26; 72.2%). A higher mortality rate was reported among those admitted to ICU than those who were not (81.5% versus 34.6%). Similar results were obtained for MV (76.9% versus 23.1%).
History of immunosuppression
In 22 patients (38.8%), some history of immunosuppression including SOT, cirrhosis, HIV, autoimmune diseases (lupus, rheumatoid arthritis, autoimmune hemolytic anemia, multiple sclerosis), hematologic malignancies, or immunosuppressive drugs for any reason other than COVID-19 infection was reported. However, no mortality differences were found between those who died and those who survived, nor when the causes of immunosuppression were analyzed separately (Table 2).
Risk factors associated with death in patients with COVID-19 and cryptococcosis.

Including SOT, cirrhosis, HIV, autoimmune diseases (lupus, rheumatoid arthritis, autoimmune hemolytic anemia, multiple sclerosis), hematologic malignancies, or immunosuppressive drugs for any reason other than COVID-19 infection.
CAT, combination antifungal therapy; CNS, central nervous system; DM, diabetes mellitus; HIV, human immunodeficiency virus; HTN, arterial hypertension; ICU, intensive care unit; IQR, interquartile range; MV, mechanical ventilation; SOT, solid organ transplant; TCZ, tocilizumab.
Treatment for COVID-19 and secondary infection/co-infection
According to the treatment indicated for COVID-19, corticosteroids were used most frequently (n = 40; 70.2%), followed by remdesivir (n = 18; 35.3%) and tocilizumab (n = 6; 11.3%). About 60% (n = 24) of these patients completed treatment for COVID-19 before the diagnosis of cryptococcosis. However, the time between both mentioned events and the doses and duration of treatment were poorly characterized in the original reports. Individuals who received any medication for COVID-19 showed a greater mortality rate compared to those who did not receive such drugs (77.4% versus 47.1%). However, this difference disappeared when analyzing each treatment individually (see Table 2). Secondary infection/co-infections with bacteria were the most frequent (n = 17; 26.2%), manifesting mainly as findings in cultures, bacteremia, or ventilator-associated pneumonia. Although there were also a few cases of fungi. Those who developed secondary infection/co-infections demonstrated a higher mortality rate in comparison to individuals without them (48.4% versus 11.8%). In all, 16 patients (84.2%) with secondary infections/co-infections received corticosteroids. Notably, the subgroup of secondary infections/co-infections with bacteria also displayed a higher mortality rate in contrast to those without such infections (45.2% versus 8.8%).
Details of each case are shown in Supplemental Material 2.
Cryptococcus site of infection and species
Most cases were disseminated infection (n = 36; 55.4%) defined as two or more non-adjacent organs being simultaneously affected with cryptococcosis. 62 In some cases of disseminated cryptococcosis (n = 17), neurological involvement was ruled out with a lumbar puncture before administering antifungals (n = 8). Lumbar puncture was not performed in 19 patients with disseminated disease. In 10 cases, it was made explicit that it was not performed due to postmortem cryptococcal diagnosis (n = 6) as well as poor prognosis, multiple failed attempts, deferred due to lack of neurological manifestations, or no patient consent (n = 1, each). In the rest (n = 9), all of which were reported in conference abstracts, after obtaining blood cultures or serological tests, it was not reported why lumbar puncture was not performed. There was also involvement of the bloodstream, the central nervous system (CNS), pulmonary, cutaneous, and ocular. No statistical differences were found in any of these cases. The diagnostic methods of cryptococcosis are summarized in Table 1 and greater detail in Supplemental Material 2. The most frequently causing species of cryptococcal infection was Cryptococcus neoformans (n = 43; 66.2%). However, there were also a few cases where C. gattii and C. laurentii were identified.
Cryptococcus therapy, the time between COVID-19 and cryptococcosis diagnoses, and laboratory exams
Individually, of 56 patients, antifungal therapy consisted of polyenes (amphotericin B) in 47 cases (83.9%), azoles (fluconazole, isavuconazole, and intravitreal voriconazole) in 40 cases (71.4%), and flucytosine in 24 cases (42.9%). CAT was observed in 44 cases (78.6%) while the rest received antifungal monotherapy (n = 12; 21.4%). Of the latter, 3 (25%) received polyenes while 9 (75%) azoles. Mortality rates were similar among those who received CAT compared to those who did not (72.0% versus 83.9%). The median time from diagnosis of COVID-19 to cryptococcosis was 32 days. The median and IQR for CD4+ and lymphocytes were 207 (98–300) cells/μL and 615 (375–985) cells/μL, respectively. The lymphocyte count was lower among those who died than those who were alive (400 versus 990 cells/μL).
Mortality
Based on the individual data available for 65 patients, 47.7% (n = 31) of the patients died while 52.3% survived (n = 34). Further details of all studies with individual patient data are shown in Supplemental Material 2.
Other studies
Half (n = 4; 50%) of these studies were conducted in the United States,63–66 while the rest were conducted in Mexico, 67 Brazil, 14 China, 68 and the UK. 69 Interestingly, only three of these studies (37.5%) were published in their final version in a journal14,63,68 since the rest were conference abstracts64–67,69; two case series,14,69 four descriptive studies,64,65,67,68 and two retrospective cohort studies63,66 were identified. Overall, they had an inclusion interval from February 2020 to April 2022. Although the majority had a population with hospitalized COVID-19, there were two studies with a specific population, only ICU admissions 67 and only people living with HIV (PLWHIV). 69
A descriptive study by Bojorges-Aguilar et al. only included critically ill COVID-19 patients admitted to the ICU, finding that out of 743 only 67 (9%) had an invasive fungal infection, mainly aspergillosis and candidiasis. There were three cases of cryptococcosis, and although a mortality of 48% was reported, it was unknown if it included any of the cases of cryptococcosis. 67 Jewsbury et al. reported a case series of 16 PLWHIV who were mostly controlled (viral load < 200). However, a quarter of these patients died, including the only case of cryptococcal meningitis in this series. 69 Kaleekal et al. reported a descriptive study that included 7508 patients with COVID-19, of which 82 (1.1%) acquired fungal infections, only two due to Cryptococcus. These infections were more frequent in the ICU than in non-ICU and were associated with the use of MV and corticosteroids. 64 Along the same lines, another descriptive study by Swaney et al. whose objective was to report fungal infections in COVID-19 patients, obtained 45 cases (1.7%) of a total of 2639 patients, with only one case of cryptococcosis. Aspergillosis and candidiasis were the most frequent diagnoses, with a mortality rate reaching 60%. The need for MV and ICU admission COVID-19 therapeutics (corticosteroids, remdesivir) was not significantly different among those who survived and expired. 65 A single-center retrospective cohort study by Zahra et al. reported 25 cases of fungemia out of a total of 1398 COVID-19 patients in a period of 3 months, with only one case of cryptococcosis, the rest being Candida species. The fungemia cohort is more likely to require ICU and MV compared to those without fungemia. In the former, mortality was double that of the latter. 66 Martins et al. reported a case series of eight invasive fungal infections from a total of 716 patients with COVID-19. All eight patients died, including the only case of cryptococcosis whose data were included in our bivariate analysis since it also had individual information available. The rest of the cases were candidiasis and aspergillosis. 14 A descriptive study conducted by Zhu et al. that looked for co-infections with respiratory pathogens among COVID-19 cases reported that there was a high percentage of co-infection (94.2%; 242) out of a total of 257 cases. Although the main co-infections were bacterial, there was also a single case of co-infection with Cryptococcus. 68 Unlike the rest of the studies that were more general and included all patients with fungal infections, the cohort study by Chastain et al. was the only one that evaluated the development of cryptococcosis among hospitalized patients with COVID-19. Among 212,479 hospitalized patients with COVID-19, 65 developed cryptococcosis, reporting an incidence of 0.022%. The patient population was divided into two cohorts based on the presence or absence of a diagnosis of cryptococcosis after 3 months of COVID-19 diagnosis. Patients with cryptococcosis were more likely to have received tocilizumab (p < 0.0001) but not dexamethasone (p = 0.0840). MV and mortality were significantly higher among patients with cryptococcosis. 63
Discussion
In the present study, we sought to describe the epidemiology as well as clinical and treatment outcomes of patients with COVID-19 associated with cryptococcosis. Overall, cryptococcosis in COVID-19 patients is not as frequent compared to other fungal infections such as mucormycosis 70 and aspergillosis, 71 widely reported in the literature, in COVID-19 patients, although it can still be life-threatening in light of the high mortality, ranging between 50% and 65%.23,50,72,73 Our analysis showed that the majority of patients were men and around 60 years of age, and in addition, it was determined that both male sex and age are risk factors associated with mortality. The male sex is related to the incidence, severity, and mortality of COVID-19. 74 However, this is also in line with a large cohort study of cryptococcosis among hospitalized patients with COVID-19 where men were mostly affected. 63 In fact, these differences may be related to the protective role of circulating estrogen-mediated hormone levels in adaptive immunomodulation in female patients. 75
Previously, ICU admission and MV were reported as important risk factors in literature reviews on the subject.50,72 Our findings were similar for both variables; in addition to these, we also found that secondary infections/co-infections (the majority being bacterial infections) turned out to be a risk factor associated with mortality. It should be noted that it was difficult to distinguish between both according to the information provided by the papers, thus for the patients in the present review it would be an additional infection to cryptococcosis and COVID-19 and could be either a co-infection or secondary infection. In a recently published systematic review, they consider that there was no clear definition between co-infection and secondary infection; however, a percentage of up to 26% and 19% was found, respectively. 76 What there is greater consensus on is that they increase the mortality rate in COVID-19 patients. 77 Another recent study identified risk factors for bloodstream infections in COVID-19 patients, highlighting in its results the consumption of interleukin inhibitors (i.e. tocilizumab or anakinra) and dexamethasone, among others. 78 Of interest, a high percentage (16/19; 84.2%) of patients with secondary infections/co-infections in our review received corticosteroids.
The role of immunosuppressive therapy as a source of increased susceptibility of COVID-19 patients to opportunistic infections is still controversial. On the one hand, corticosteroids and TCZ demonstrate important improvements in mortality and the need for MV in patients with COVID-19 for specific conditions in regulated doses.7,9 However, corticosteroids showed worse clinical outcomes in fungal diseases according to a recent study carried out by Li et al. 8 Likewise, a systematic review concluded that tocilizumab therapy significantly increased the risk of fungal co-infections in COVID-19 patients, according to data from eight observational studies (OR = 2.02, 95% CI = 1.05–3.90, p = 0.036). 79 Although our analysis showed that COVID-19 immunosuppressive drugs together were associated with mortality, this difference disappeared when pharmacological therapies were analyzed separately, probably due to the heterogeneity of type of administration (different doses, duration of therapy, time of diseases at start medication, among others). The ideal would be to standardize the dose and duration of these drugs in those who require them, to improve the balance between survival and side effects.
The low proportion of HIV-positive patients in our study (3/40; 7.5%) is consistent with its global reduction in HIV-associated cryptococcal infection likely to be due to antiretroviral therapy expansion. 80 However, the low proportion of patients with immunodeficiencies (22/65; 33.84%) in our review still draws attention, as it differs from modern cryptococcosis cohorts without HIV, in which patients with some immunocompromising conditions are the majority, reaching up to 82.8% in the United States and 60.8% in Australia and New Zealand according to large multicenter studies.81,82 Among previous cases of cryptococcosis in patients with COVID-19 summarized in a literature review, it was reported that 56% of the patients did not have traditional risk factors associated with cryptococcosis. 23 So far, data from a multicenter research network found that cryptococcosis occurred most often in hospitalized patients with COVID-19 who had traditional risk factors, observing a mortality of 36%, which was significantly higher than those with COVID-19 but without cryptococcosis. 63 A recent study that included 69 patients with cryptococcosis following COVID-19 compared the groups of immunocompetent (n = 36) versus immunocompromised (n = 33) observing that the former had a very high mortality at 72%, significantly higher than the 48% mortality observed in the latter (p value = 0.045). 83 In fact, cryptococcosis in COVID-19 patients appears to be a distinct entity, which resembles that of non-HIV patients, in which mortality is much higher than those cases of HIV-associated cryptococcosis.84,85 Interestingly, it would seem that COVID-19 would be a condition that would allow cryptococcal infection as opportunistic, although it is unknown if it is due to the disease itself or due to other factors such as comorbidities or medications. To test the hypothesis that there could be a difference between patients with cryptococcosis with and without COVID-19, a multicenter research network was carried out and found that significantly more patients with COVID-19 had a history of SOT or malignancy compared to non-COVID-19 controls, but not for HIV. 86 Other comorbidities (autoimmune and inflammatory diseases and DM), some of which are risk factors for cryptococcosis as well as corticosteroid use, were also more common among patients with COVID-19 compared to non-COVID-19 controls. Despite this, no differences were noted in terms of ICU admissions and mortality between both groups (with and without COVID-19). 86 Further studies are warranted.
Lymphopenia associated with COVID-19 occurs as a consequence of a redistribution of peripheral T lymphocytes to the lungs, the main target of the SARS-CoV-2 virus. 87 In addition, in the event of failure to control the virus at the site of infection, functionally exhausted T lymphocytes undergo cell death. Although this effect is temporary, the process of restoration to normal levels in the convalescent period takes several months. 6 In this interval, hosts with depleted T lymphocytes are more vulnerable to cryptococcosis since the fungicidal effect of macrophages promoted by these lymphocytes is lost. 88 Our analysis shows that lymphopenia is associated with mortality in patients who developed cryptococcosis after COVID-19. The review by Pipitone et al. 50 reported that inadequate cryptococcal treatment (non-CAT) and mortality were associated, unlike a meta-analysis in which the duration and type of antifungal therapy (CAT versus monotherapy) were not associated with all-cause mortality in patients with COVID-19 and fungal secondary infections. 89 The latter is similar to what we found in our analysis. Importantly, cryptococcal infection occurred relatively late after COVID-19 diagnosis (32 days, median), which was longer than other studies (10–13 days, median).63,72 This difference may be attributable to delays in the diagnosis or initiation of antifungal therapy for cryptococcosis. The theory in this condition is that Cryptococcus is behaving as an opportunistic infection and has likely been reactivated following lymphopenia or immune compromise at that level due to COVID-19. In this context, symptoms would probably present late.
The fact that only slightly more than half (n = 40; 61.5%) of the included case reports have described the HIV infection status of the patients before suspicion or even after confirming the diagnosis of cryptococcosis is in line with the overall mean completeness of reporting score of 54.4% described by Scaffidi et al. 90 according to the CARE checklist items for COVID-19 case reports. This is even more alarming if one considers that cryptococcal infection is one of the main causes of morbidity and mortality in HIV-positive patients. 1
A scoping review aims to identify and map the available evidence regarding a topic. While it uses a methodology involving a systematic search that is explicit and transparent, it should not be confused with a systematic review which is a study design that also synthesizes the evidence but answers a specific question, whereas the scoping review can be more flexible and open. 91 Definitely, carrying out a systematic review is not feasible for now since few studies do not provide information on a single patient and are published in full text in a journal.
The limitations of the present study include the omission of important data such as the temporality in days of the onset of symptoms, hospital admission, or diagnosis until an outcome occurred, whether it was the patient’s discharge, transfer, or death in the reported clinical cases. One significant limitation of this research is the inherent challenge of obtaining real-time and comprehensive data on the impact of emerging variants of COVID-19 due to the dynamic nature of the pandemic and the evolving landscape of viral mutations. This limitation hinders the ability to make conclusions regarding vaccination status and the saturation of health services. Moreover, many studies were only available as conference abstracts, which limited them to details of relevant clinical data. However, this type of scientific communication becomes important in a relatively recent topic such as the COVID-19 pandemic.
Conclusion
Cryptococcosis in COVID-19 patients has been reported more frequently. However, it is still not as common as other fungal infections associated with COVID-19. There were few patients with any classic immunosuppression factor. Despite this, it was reported that the majority received corticosteroids, although there was poor characterization of the doses and duration of treatment. The high mortality rate (47.7%) was similar to that of cryptococcosis in patients without HIV. The factors that have demonstrated the strongest association with mortality were ICU admission, MV, and secondary infections/co-infections. Studies should adapt to existing reporting guidelines to avoid omissions or improve the quality of the information presented that may be useful for future reviews.
Supplemental Material
sj-docx-1-tai-10.1177_20499361241232851 – Supplemental material for COVID-19 associated with cryptococcosis: a scoping review
Supplemental material, sj-docx-1-tai-10.1177_20499361241232851 for COVID-19 associated with cryptococcosis: a scoping review by Alvaro Quincho-Lopez, Nuvith Poma and Juan José Montenegro-Idrogo in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-2-tai-10.1177_20499361241232851 – Supplemental material for COVID-19 associated with cryptococcosis: a scoping review
Supplemental material, sj-docx-2-tai-10.1177_20499361241232851 for COVID-19 associated with cryptococcosis: a scoping review by Alvaro Quincho-Lopez, Nuvith Poma and Juan José Montenegro-Idrogo in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-3-tai-10.1177_20499361241232851 – Supplemental material for COVID-19 associated with cryptococcosis: a scoping review
Supplemental material, sj-docx-3-tai-10.1177_20499361241232851 for COVID-19 associated with cryptococcosis: a scoping review by Alvaro Quincho-Lopez, Nuvith Poma and Juan José Montenegro-Idrogo in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
