Abstract
Objective:
The optimal confirmatory tests for diabetes mellitus (DM) in patients with tuberculosis (TB) vary across populations. This study aimed to evaluate the performance of two confirmatory tests for DM against the oral glucose tolerance test (OGTT) as the reference test in adult Ugandans with recently diagnosed TB.
Methods:
A total of 232 adult participants receiving TB treatment underwent initial screening for DM with random blood glucose (RBG) measurement. Participants with a RBG level ⩾6.1 mmol/l received additional screening with fasting blood glucose (FBG), laboratory-measured glycated haemoglobin (HbA1c) and an OGTT. Using the latter as the gold standard and reference test, we evaluated the diagnostic accuracy of laboratory-measured HbA1c and FBG.
Results:
Of the 232 participants initially screened for DM using RBG measurement, 117 participants (50.4%) had RBG level ⩾6.1 mmol/l and were scheduled to return for additional blood glucose testing. Of these, 75 (64.1%) participants returned for FBG and HbA1c measurements. A diagnosis of DM was made in 32 participants, corresponding to a prevalence of 13.8% [95% CI 9.9–18.9].
The areas under the curve (AUC) for FBG and laboratory-measured HbA1c were 0.69 [95% CI 0.47–0.90] and 0.65 [95% CI 0.43–0.87], respectively. The sensitivity and specificity of a FBG level of ⩾7 mmol/l were 57.1% [95% CI 18.4–90.1] and 74.6% [95% CI 62.5–84.5], respectively, whereas the sensitivity and specificity for laboratory-measured HbA1c of ⩾6.5 mmol/l (48 mmol/mol) were 14.3% [95% CI 0.40–57.9] and 95.3% (86.9–99.0%), respectively.
Conclusion:
FBG may be better than laboratory-measured HbA1c in confirming DM in adult Ugandans with recently diagnosed TB. However, because of the small study sample size, larger studies evaluating the diagnostic utility of these diabetes screening tests in adult Ugandans with TB are needed to confirm these findings.
Plain language summary
Diabetes mellitus (DM) is a common condition in patients with tuberculosis and proactively screening for the condition is encouraged in all adult patients with tuberculosis. In this study, a total of 232 adult Ugandans with recently diagnosed tuberculosis were screened for DM using random glucose test, fasting blood glucose test, glycated haemoglobin test and an oral glucose tolerance test (OGTT), as the gold-standard and reference test. Compared with the OGTT, a fasting blood glucose test was noted to be a better screening test for diabetes mellitus than glycated haemoglobin in these patients and may be used as a follow-up test to random blood glucose in the screening and diagnosis of DM in adult Ugandans with tuberculosis.
Keywords
Introduction
Diabetes mellitus (DM) adversely affects treatment outcomes in patients with tuberculosis (TB). It is associated with an increased risk of early mortality, delayed sputum conversion, the onset of multi-drug resistant TB and TB disease recurrence.1–4 Because of this clinical influence, early diagnosis of DM and initiation of optimal glucose-lowering therapy in patients with TB, especially during the intensive phase of TB treatment is integral to improving treatment outcomes. 2
It is also important to note that, due to the pro-inflammatory state induced by TB disease, transient stress hyperglycaemia is very common in patients with TB and often misclassified as overt DM.5,6 The majority of patients with stress hyperglycaemia revert to normal glucose status after treatment of TB.7,8
The International Union Against TB and Lung Disease (The Union) guidelines for the management of TB and DM advocate for the universal screening of patients with TB for DM. 6 However, the optimal screening and confirmatory tests to be used vary across populations and are not known in some clinical settings like in Uganda. The guidelines recommend the use of random blood glucose (RBG) measurement as the initial screening test for DM and either fasting blood glucose (FBG) or glycated haemoglobin (HbA1c) if the RBG is ⩾6.1 mmol/l. 6 A 75-g oral glucose tolerance test (OGTT), considered the gold standard test for diagnosing DM due to its high sensitivity, is expensive and cumbersome to perform in clinical settings. 9 Both FBG and HbA1c tests are simple and relatively cheap to perform.6,9,10
No study has investigated the diagnostic accuracy of the commonly available laboratory tests for DM in adult patients with TB in Uganda. We, therefore, aimed to evaluate the diagnostic performance of two confirmatory tests for DM in an adult Ugandan population with recently diagnosed TB at three tertiary healthcare facilities offering comprehensive TB treatment services.
Methods
Study setting and population
This study was part of the TB and DM comorbidity study that enrolled adult Ugandans (aged ⩾18 years) with a recent bacteriological, radiological or clinical diagnosis of TB (<2 months from the time of diagnosis). Participants were either treatment naïve or initiated on TB treatment and had provided written informed consent. The participants were recruited from outpatient TB treatment centres of three tertiary healthcare facilities located in Central Uganda from January 2022 to January 2023. Participants who could not comply with the scheduled study visits were excluded.
Study procedures
After providing written informed consent, a pre-tested case report form was used to collect the relevant sociodemographic and medical history of each participant. Using standardised study methods, anthropometric measurements like weight, height, waist circumference, hip circumference, body mass index (BMI) and waist:hip circumference ratio (WHR) were performed and documented.
Assessment of the glycaemic status
The Union and World Health Organization (WHO) guidelines were used to guide the screening and diagnosis of DM.6,9 At the enrolment visit, participants were initially screened by measuring the RBG level using a One Touch Select Plus Flex® glucometer, LifeScan, Inc. No further blood glucose testing was performed if the RBG was <6.1 mmol/l. Additional blood glucose testing was performed for participants with a RBG level ⩾6.1 mmol/l using the 75-g OGTT, FBG and laboratory-measured HbA1c. To measure the FBG level, participants were requested to fast for at least 8 h before their clinic visits. After measurement of the FBG, the OGTT was performed using 75 g of anhydrous glucose, and blood glucose levels were measured 2 h after ingestion of the glucose solution.
Measurement of all blood glucose levels was done using a One Touch Select Plus Flex® glucometer. The laboratory HbA1c measurement was done using an electro-chemiluminescence immunoassay manufactured by Roche Diagnostics Limited, Germany on a Cobas 6000 C-model SN 14H3-15 machine (Hitachi High Technologies Corporation, Tokyo, Japan).
Study outcomes
A diagnosis of DM was made based on any of the following: (1) pre-existing history of DM (either treatment naïve or on any glucose-lowering therapy); (2) FBG ⩾7 mmol/l; (3) a blood glucose level of ⩾11.1 mmol/l 2 h after a 75-g OGTT or (4) laboratory- measured HbA1c level ⩾6.5% or 48 mmol/mol. 11
Statistical analysis
To describe the characteristics of all study participants, we used proportions for the categorical variables and medians with interquartile range (IQR) for the continuous variables. The categorical and continuous variables were compared using chi-square and Kruskal–Wallis tests, respectively.
The sensitivity and specificity of the FBG and laboratory-measured HbA1c for detecting DM using the 75-g OGTT as the reference test were calculated. Receiver operating characteristic curves were used to evaluate the diagnostic performance of FBG and laboratory-measured HbA1c against the results of the OGTT. The positive predictive values (PPVs) and negative predictive values (NPVs) and accuracy for predicting DM by each test were also calculated. The diagnostic accuracy was assessed by the area under the curve (AUC). All analyses were done using STATA statistical software version 15 College Station, TX, USA: StataCorp LLC.
Ethics considerations
The study received ethical and regulatory approval from the Infectious Diseases Institute, Research and Ethics Committee (IDIREC REF 014/2021), and the Uganda National Council for Science and Technology (HS1707ES). All study sites offered administrative approval before the study initiation. All participants enrolled in the study provided written informed consent. For participants who could not read and write, a thumbprint was used to express informed consent in addition to written informed consent offered by an impartial witness representing the illiterate participant.
All methods were carried out in accordance with the ethical standards of the Declarations of Helsinki.
Results
Baseline characteristics of all study participants
Table 1 summarises the sociodemographic, clinical and anthropometric characteristics of all participants and those with and without TB and DM comorbidity. A total of 232 participants were enrolled in the study. Of these, 69% were females.
Sociodemographic, clinical and anthropometric characteristics of all participants, and those with and without diabetes-tuberculosis comorbidity.

Assessment of smoking or alcohol use based on a history of ever smoking or alcohol ingestion.
BMI calculated as weight (kg)/height (m)2.
WHR calculated as waist circumference (cm)/hip circumference (cm).
All continuous variables are expressed using medians and interquartile ranges. All categorical variables expressed as absolute numbers and percentages (n, %). p-Values for the categorical and continuous variables calculated using chi-squared test and Kruskal–Wallis test, respectively.
BMI, body mass index; DM, diabetes mellitus; EPTB, extrapulmonary tuberculosis; TB, tuberculosis; WHR, waist:hip ratio.
The median (IQR) age, BMI and RBG of all study participants was 35 (27–42) years, 19.2 (17.6–21.3) kg/m2 and 6.1 (5.5–7.2) mmol/l, respectively.
Compared with those with TB alone, participants with TB and DM comorbidity were more likely to be older [median (IQR): 42.5 (37.0–53.5) years versus 33.5 (25.0–42.0) years, p < 0.001], to reside in a semi-urban area (28.1% versus 12.9%, p = 0.04), to be former smokers (34.4% versus 16.9%, p = 0.04) and less likely to have HIV co-infection (18.8% versus 41.8%, p = 0.01).
Of the 232 participants initially screened for DM using RBG measurement, 117 participants (50.4%) had RBG level ⩾6.1 mmol/l and were scheduled to return for additional blood glucose testing. Of these, 75 (64.1%) participants returned for re-assessment. The participant characteristics of those who did not return for re-testing and those did, did not vary greatly.
A diagnosis of DM was made in 32 participants, corresponding to a prevalence of 13.8% [95% CI 9.9–18.9]. Of the 32 participants diagnosed with DM, 7 (9.5%) had a blood glucose level ⩾11.1 mmol/l after the OGTT, 8 (10.7%) had a laboratory-measured HbA1c ⩾6.5% and 22 (29.3%) had a FBG of ⩾7.1 mmol/l.
Diagnostic performance of the confirmatory tests
Supplemental Tables 1, 2 and Figures 1, 2, summarise the number of participants diagnosed with DM by each test and its diagnostic performance when compared with the 75-g OGTT as the reference test.
The AUC for FBG and laboratory-measured HbA1c was 0.69 [95% CI 0.47–0.90] and 0.65 [95% CI 0.43–0.87], respectively (Figures 1 and 2).
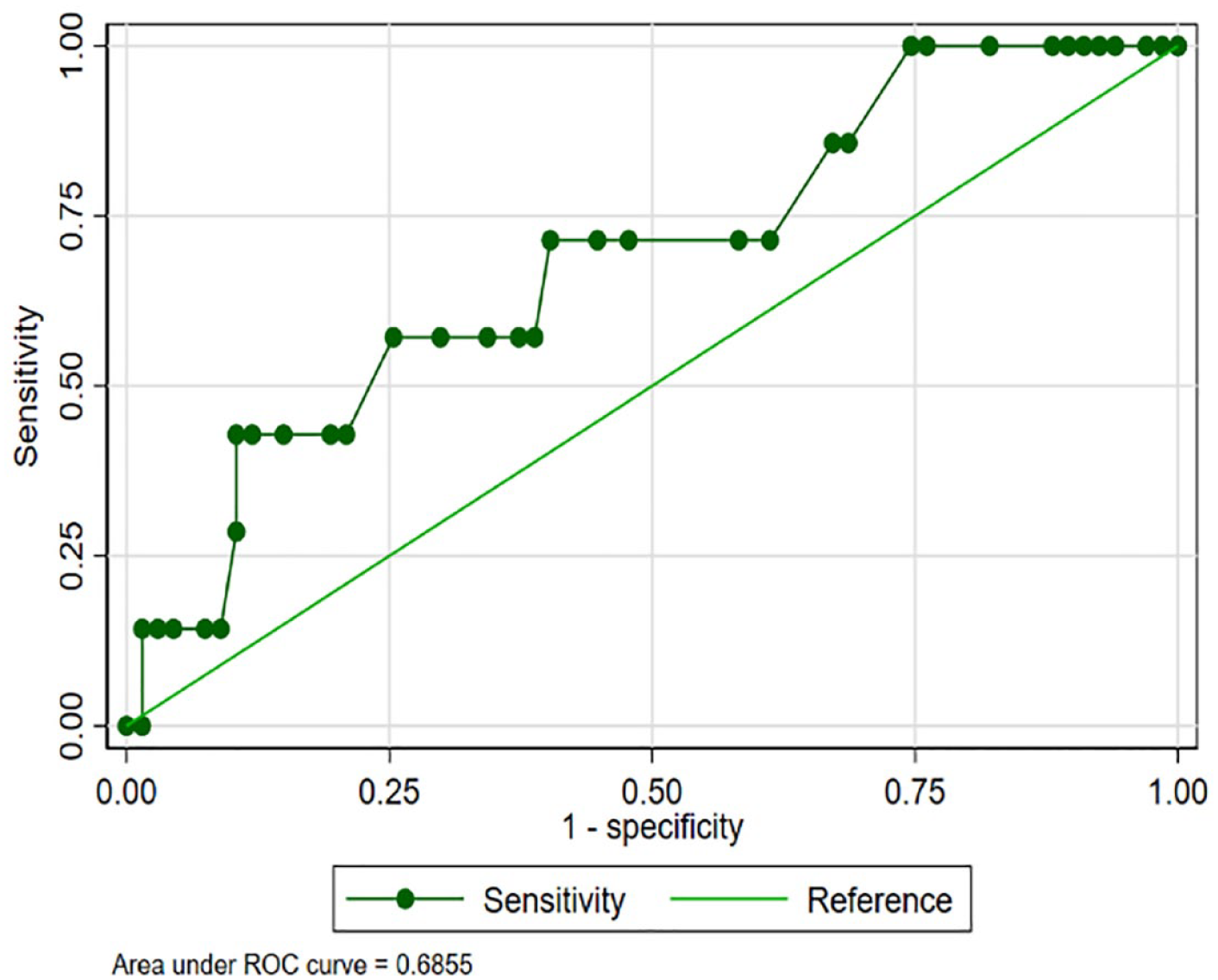
Diagnostic accuracy of fasting blood glucose test when compared with the oral glucose tolerance test as the reference test.
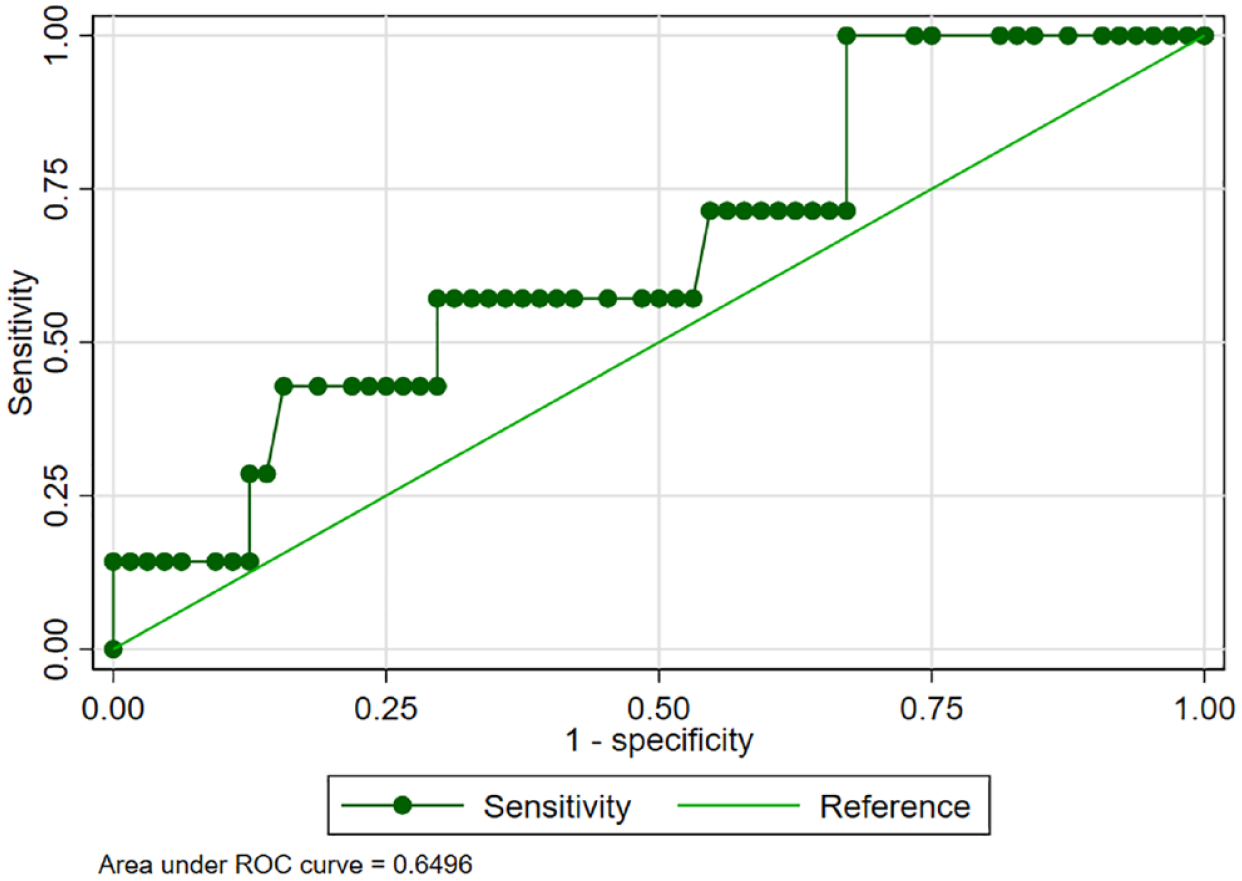
Diagnostic accuracy of the laboratory-measured HbA1c when compared with the oral glucose tolerance test as the reference test.
Using a FBG cut-off value of ⩾7 mmol/l, the sensitivity and specificity were 57.1% [95% CI 18.4–90.1] and 74.6% [95% CI 62.5–84.5], respectively. A laboratory-measured HbA1c cut-off value of ⩾6.5% (48 mmol/mol) had a sensitivity and specificity of 14.3% [95% CI 0.40–57.9] and 95.3% (86.9–99.0), respectively. Despite having a lower sensitivity than FBG, the laboratory-measured HbA1c test had a better PPV [25.0%, (95% CI 0.6–80.6) versus 19.0%, (95% CI 5.4–41.9)]. Both tests had comparable NPV [91.0% (95% CI 81.5–96.6) for HbA1c and 94.3% (95% CI 84.3–98.8) for FBG] (Table 2).
Diagnostic accuracy of the screening tests for diabetes mellitus when compared with a 75-g oral glucose tolerance test as the gold standard test.

AUC, area under the curve; CI, confidence interval; FBG, fasting blood glucose; HbA1c, glycated haemoglobin; NPV, negative predictive value; PPV, positive predictive value.
Discussion
Our study is the first to evaluate the performance of laboratory-measured HbA1c and FBG tests in confirming DM in adult Ugandans with recently diagnosed TB using a 75-g OGTT as the gold standard and reference test. It showed that FBG performed better than the laboratory-measured HbA1c as a confirmatory test for DM in this study population. However, the laboratory-measured HbA1c test had a higher specificity than FBG.
Similar studies investigating the diagnostic accuracy of FBG and HbA1c for DM in the general Ugandan population and HIV-infected adult Ugandans have also reported that FBG performs better than HbA1c as a confirmatory test for DM.12–14 In the study by Kasujja et al 12 conducted in the outpatient department of a general hospital in Eastern Uganda on 1659 adult Ugandans, the AUC of FBG and HbA1c was 0.89 and 0.86, respectively. The sensitivity of FBG and HbA1c was 69.8% [95% CI 46.3–86.1] and 62.6% [95% CI 41.5–79.8], respectively. Contrary to what we observed in our study, both FBG and HbA1c had comparable and high specificities in this study [99.4% (95% CI 98.9–99.7) for FBG and 98.6 (95% CI 95.4–99.6) for HbA1c]. 12 A finding of a high specificity with HbA1c, as observed in our study, was also reported in another study that investigated the validity of HbA1c to diagnose DM in HIV-infected and uninfected adult patients in rural Uganda. In this study, the specificity of HbA1c was 98% [95% CI 96–99]. 14
Generally, HbA1c has been reported to underperform as a screening and diagnostic test for DM in people of African ancestry with or without clinical factors that affect its performance like anaemia, glucose-6-phosphate dehydrogenase deficiency and haemoglobinopathies.15–17 Its performance is further reduced in non-obese individuals.18,19 It is important to note that our study participants’ median (IQR) BMI was 19.2 (17.6–21.3) kg/m2.
In one systematic review and meta-analysis of 11 studies of 12,925 native adult Africans investigating the diagnostic accuracy of HbA1c when compared with OGTT as a screening test for DM in Africa, HbA1c cut-off of ⩾6.5% (48 mmol/l) had a pooled sensitivity of 57.7% [95% CI 43.4–70.9] and a specificity of 92.3% [95% CI 83.9–96.5], with high heterogeneity across the studies (I2 = 91.8%, p < 0.01). At a cut-off of ⩾6% (42 mmol/mol), the sensitivity increased to 74.9% and the specificity reduced to 71.8%. 17
A global meta-analysis of 37 studies conducted in all regions, with only three studies from Africa, also reported similar findings of a low pooled sensitivity of 50% and a high pooled specificity of 97.3% with HbA1c at a cut-off of 6.5% (48 mmol/mol) when compared with OGTT as the reference test. 20 In another scooping review that assessed studies investigating the HbA1c performance in adults of African descent but living in the United States with normal glucose status, prediabetes or DM, four studies conducted in Africa-born adults showed that HbA1c diagnostic cut-off of ⩾6.5% was suboptimal and associated with under-diagnosis of DM in this particular population. 15
In some studies conducted in Asian, South American and European patients with recently diagnosed TB, HbA1c has been shown to perform better than FBG, contrary to what we observed in our study.21–23 This demonstrates differences in the screening approaches across populations and underscores the need to have screening guidelines for DM in patients with TB individualised to each population or clinical setting.
In one study that screened for DM in 983 adult Indian patients with recently diagnosed TB, using the OGTT as a reference test, the AUC for HbA1c and FBG was 0.75 [95% CI 0.68–0.83] and 0.66 [95% CI 0.58–0.74], respectively. The HbA1c cut-off point of ⩾48 mmol/mol (6.5%) gave a sensitivity of 59.1% and specificity of 91.7%, and the respective values for FBG were 34.8% and 97.5%. However, the PPV of FBG was better than that of HbA1c (56.1% for FBG and 39.8% for HbA1c, p < 0.0001). Both tests had comparable high NPV (96% for HbA1c and 94.2% for FBG). 21
In another multi-country prospective study that recruited 2185 adult patients with TB in Indonesia, Peru, Romania and South Africa, laboratory-measured HbA1c, RBG, Point-of-care glycated haemoglobin (POC HbA1c), FBG and urine dipstick tests were used to screen for DM. Using the laboratory-measured HbA1c as the reference test, the AUC of the POC HbA1c, FBG and RBG were 0.81 [95% CI 0.75–0.86], 0.78 [95% CI 0.70–0.85] and 0.77 [95% CI 0.70–0.83], respectively. The sensitivity and specificity for the POC HbA1c of ⩾6.5% were 59.3% [95% CI 48.5–69.5] and 88.7% [95% CI 87.2–90.2], respectively. The respective values were 35.6% [95% CI 23.6–49.1] and 98.4% [95% CI 97.3–99.1] for FBG of ⩾7 mmol/l and 36.1% [95% CI 26.6–46.5] and 99.3% [95% CI 98.8–99.7] for RBG of ⩾11.1 mmol/l. 22 This shows that the POC HbA1c had the best diagnostic accuracy with both FBG and RBG having high specificities.
On the contrary, only the study by Calderon et al. 24 conducted in 136 TB patients and 138 household contacts in Peru reported findings similar to our study. This prospective cohort study noted that, compared to HbA1c, FBG was a better screening test for DM in Peruvian patients with TB. 24 The sensitivity and specificity of FBG in this study was 0.53 [95% CI 0.36–0.70] and 0.54 [95% CI 0.47–0.60], respectively, whereas the corresponding values for HbA1c were 0.47 [95% CI 0.30–0.64] and 0.47 [95% CI 0.40–0.53], respectively. 24 The low performance of HbA1c in this study population with TB could be due to the high co-existing prevalence of anaemia, which directly affects the diagnostic accuracy of the HbA1c test.
Our study had some strengths. It is the first study to assess the diagnostic utility of two blood tests commonly used to screen and confirm DM in an adult population with recently diagnosed TB in Uganda. We used the 75-g OGTT, the gold standard for diagnosing DM, as the reference test when evaluating the diagnostic utility for FBG and laboratory-measured HbA1c.
Our study had limitations too. We did not assess the haemoglobin levels of the participants. The presence of anaemia is one of the factors that can influence the performance of the HbA1c test. We had a relatively small number of study participants with the majority not returning for re-testing despite having an efficient follow-up study mechanism. This may also have introduced selection bias into the study and could have affected our study findings. Loss to follow-up, as reported in our study, has been highlighted as a key limitation in some studies evaluating the diagnostic performance of diabetes tests in TB patients. 24
Conclusion
Our study highlights that the FBG test may be a better test than the laboratory-measured HbA1c to confirm DM in adult Ugandans with recently diagnosed TB. Hence, this readily available and inexpensive test can be used as a follow-up test for confirming DM in adult Ugandan patients with recently diagnosed TB after the initial testing using RBG.
However, due to the small sample size of our study, we recommend that larger studies to evaluate the diagnostic utility of these diabetes screening tests in adult Ugandans with TB should be undertaken to confirm these findings. This will also guide the formulation of context-specific guidelines for screening DM in adult Ugandans with TB.
Supplemental Material
sj-docx-1-tai-10.1177_20499361231216799 – Supplemental material for Diagnostic accuracy of two confirmatory tests for diabetes mellitus in adult Ugandans with recently diagnosed tuberculosis
Supplemental material, sj-docx-1-tai-10.1177_20499361231216799 for Diagnostic accuracy of two confirmatory tests for diabetes mellitus in adult Ugandans with recently diagnosed tuberculosis by Davis Kibirige, Stella Zawedde-Muyanja, Irene Andia-Biraro, Ronald Olum, Susan Adakun, Christine Sekaggya-Wiltshire and Ivan Kimuli in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-xlsx-1-tai-10.1177_20499361231216799 – Supplemental material for Diagnostic accuracy of two confirmatory tests for diabetes mellitus in adult Ugandans with recently diagnosed tuberculosis
Supplemental material, sj-xlsx-1-tai-10.1177_20499361231216799 for Diagnostic accuracy of two confirmatory tests for diabetes mellitus in adult Ugandans with recently diagnosed tuberculosis by Davis Kibirige, Stella Zawedde-Muyanja, Irene Andia-Biraro, Ronald Olum, Susan Adakun, Christine Sekaggya-Wiltshire and Ivan Kimuli in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
