Abstract
Background and aim:
With conflicting data from previous observational studies on the relationship between hepatitis B virus (HBV) or hepatitis C virus (HCV) infection and pancreatic cancer (PC), we decided to conduct a systematic review and meta-analysis in order to evaluate any potential association.
Design:
This is a systematic review and meta-analysis.
Methods:
We conducted a search of three databases (PubMed, Embase, and Web of Science) from the time of their creation up to June 2023. The summary results, including hazard ratio (HR) with 95% confidence interval (CI), were pooled using a generic inverse variance method and a random-effects model. Furthermore, subgroup and sensitivity analyses were conducted.
Results:
In this meta-analysis, 22 cohort studies with a total of 10,572,865 participants were analyzed. Meta-analysis from 15 cohort studies revealed that HBV infection was correlated with an increased risk of PC (HR = 1.53, 95% CI: 1.40–1.68, p < 0.00001) with no heterogeneity (I2 = 0%, p = 0.49). Meta-analysis from 14 cohort studies showed that HCV infection was associated with an increased risk of PC (HR = 1.82, 95% CI: 1.51–2.21, p < 0.00001). Most of our subgroup analyses yielded similar results. Meta-analysis from four cohort studies indicated that co-infection with HBV and HCV was linked to an increased risk of PC (HR = 2.32, 95% CI: 1.40–3.85, p = 0.001) with no heterogeneity observed (I2 = 0%, p = 0.60). The results of sensitivity analyses were robust.
Conclusion:
Our meta-analysis showed that HBV/HCV infection or co-infection with HBV and HCV was associated with an increased risk of PC. Future prospective cohort studies need to take into account various ethnicities and any confounding factors, as well as investigate the potential mechanisms of PC development in those with HBV/HCV.
Trial registration:
Open Science Framework registries (No: osf.io/n64ua).
Introduction
Globally, hepatitis B virus (HBV) and hepatitis C virus (HCV) infections are a major public health concern, with an estimated 316 million and 71 million long-term infections, respectively.1–3 It is estimated that approximately one-fourth of males and 8% of females with chronic hepatitis B (CHB) that was contracted in the perinatal period will pass away due to cirrhosis or hepatocellular carcinoma caused by HBV in untreated patients. 1 Similarly, it is thought that 10–20% of individuals who remain chronically infected with HCV will experience complications, such as cirrhosis, liver failure, or hepatocellular carcinoma, over a period of two to three decades. 3 Growing epidemiological evidence indicated that the clinical and economic burden of HBV or HCV infection was not only linked to the incidence and mortality of liver-related diseases, but may also be associated with extrahepatic malignancies, such as non-Hodgkin lymphoma, cholangiocarcinoma, and pancreatic cancer (PC).4,5
PC is a highly fatal condition, and it is the seventh most common cause of cancer deaths among men and women worldwide.6,7 Over the past 25 years, its global burden has increased drastically, doubling in magnitude. 8 In the United States, the prognosis for those diagnosed with pancreatic cancer is poor, with only 10% of patients surviving for 5 years after diagnosis. 6 The majority of patients, 80–85%, suffer from unresectable or metastatic diseases, and even those with locally resectable tumors have a 5-year survival rate of only 20%. 6 In view of the poor treatment effect, low survival rate, and high mortality rate of PC, it is of great public health significance to determine the risk factors of PC and to prevent early PC according to the risk factors.
Recently, the association between HBV or HCV infection and PC has been the subject of increased interest. Previous meta-analyses9–17 had extensive debates regarding the association between HBV/HCV and PC risk; however, these analyses were mainly composed of hospital-based case-control studies and few cohort studies, which decreased the accuracy and dependability of the results. Since the prior meta-analyses were published, a variety of high-quality cohort studies have been published, yet the results remain inconsistent and inconclusive. In addition, to explore the causal connection of this noninterventional association, the most suitable type of observational research is cohort studies. Unfortunately, no meta-analysis that only includes cohort studies has been identified yet. Therefore, we conducted a systematic review and meta-analysis of cohort studies to comprehensively and precisely evaluate the relationship between HBV/HCV infection and PC risk, thereby providing information for better prevention of PC in clinical practice.
Materials and methods
Our study protocol has already been registered with Open Science Framework (https://osf.io/n64ua). This research was conducted in accordance with the Preferred Reporting Items for Systematic reviews and Meta-Analysis (PRISMA) 2020 reporting guidelines. 18
Search strategy
Systematic searches of the PubMed, Embase, and Web of Science databases were conducted from the time of their inception up to 29 June 2023, without language restriction. The main search terms were described as follows: ‘hepatitis B’, ‘hepatitis C’, ‘HBV’, ‘HCV’, ‘pancreatic’, ‘pancreas’, ‘neoplasms’, ‘neoplasm’, ‘cancer’, ‘cancers’, ‘carcinoma’, ‘tumour’, ‘tumor’, ‘tumours’, ‘tumors’, and ‘adenocarcinoma’. We conducted a comprehensive search by combining Medical Subject Heading terms with text word searching. The complete search strategy for PubMed can be found in the Supplemental materials online (see Supplemental Table S1). Additionally, we also checked all applicable references of original research and review articles to ensure the comprehensiveness of the search.
Study selection criteria
Studies that were eligible for inclusion were: (1) cohort studies that investigated the association between HBV or HCV infection and risk of PC; (2) the exposure was HBV or HCV infection; (3) the outcome was the incidence rate of PC; (4) the confirmation methods of HBV/HCV and PC should be described; (5) studies should present hazard ratios (HRs), standardized incidence ratios (SIRs), incidence rate ratios (IRRs), or relative risks (RRs) with corresponding 95% confidence intervals (CIs), or data for their calculation. The exclusion criteria were as follows: (1) case-control or cross-sectional studies; (2) duplicate publications, abstracts, editorials, comments, letters, animal studies, reviews, and meta-analyses; and (3) studies with insufficient data. Two researchers independently screened all eligible studies based on inclusion and exclusion criteria. All disagreements were handled through mutual consent.
Data extraction
The first author’s surname, publication year, study design, geographic region, sample size, population characteristics, diagnostic methods for HBV/HCV and PC, follow-up time, HRs/SIRs/IRRs/RRs with their 95% CIs, and adjusted confounders were all extracted from the data. Two researchers independently extracted the data from the eligible studies and any disagreements were resolved by a third researcher.
Quality assessment
To assess the methodological quality of the studies included, we used the Newcastle-Ottawa Scale (NOS). 19 This scale evaluates a study based on three domains, with a maximum of four stars for selection of participants, two stars for comparability of study groups, and three stars for ascertainment of outcomes of interest, for a total of nine stars. We determined that studies with a score of nine stars had a low risk of bias (high quality), those with a score of seven or eight stars had a medium risk (moderate quality), and those with a score of six or less had a high risk of bias (low quality). 20
Statistical analysis
DerSimonian and Laird proposed the generic inverse variance method to calculate the pooled HRs with 95% CIs. 21 Taking into account the potential disparity between study settings and populations across different studies, the random-effects model was employed. As the outcome of interest was not frequent, the risk estimation of SIRs, HRs, IRRs, and RRs was the same. 22 The adjusted RRs/HRs/SIRs/IRRs reported were the ones that had the most confounders taken into account. We used the Cochran’s Q-test (p ⩽ 0.10) and I² statistic to assess the statistical heterogeneity. I2 values of 0–25% show insignificant heterogeneity, 26–50% demonstrate low heterogeneity, 51–75% signify moderate heterogeneity, and 76–100% represent high heterogeneity. 23 Subgroup analyses were conducted according to study location, study design, confirmation methods of HBV/HCV, study quality, and publication year to assess any potential factors that may affect the overall results and identify potential sources of heterogeneity. Sensitivity analyses were conducted by sequentially excluding each of the studies that were included. If the aggregate meta-analysis contained 10 or more studies, the potential publication bias was assessed by inspecting the funnel plots and performing Begg’s 24 and Egger’s tests. 25 A p value of less than 0.05 was used to determine statistical significance. The Review Manager software 5.3 (The Cochrane Collaboration, Copenhagen, Denmark) and STATA/SE software (Version 12.0, STATA Corporation, TX, USA) were both utilized to conduct all statistical analyses.
Results
Study selection
After an initial systematic search, a total of 5090 records were identified (1025 from PubMed, 3443 from Embase, and 615 from Web of Science, and 7 from other sources). After eliminating duplicates, the initial screening process yielded 3830 records. Subsequently, the full texts of 86 studies were acquired and further evaluated. As a result, 22 of them fulfilled the criteria and were included in the meta-analysis.26–47 The PRISMA flowchart of study selection process is summarized in Figure 1.

PRISMA flowchart of study selection process.
Study characteristics and quality assessment
Table 1 provides an overview of the main characteristics of the included studies. This meta-analysis included 22 Cohort studies with a total of 10,572,865 participants, exploring the association between HBV infection and PC risk (15 studies),26,27,30–34,36–38,41–43,46,47 HCV infection and PC risk (14 studies),26,28,29,31,32,35–37,39,40,42–45 and simultaneous infection of HBV and HCV and PC (4 studies).26,42,43,45 All included studies were published from 2006 to 2022. Nine studies27,30,31,37,38,41,42,46,47 were from Asia (Korea, China, and Japan), five studies29,32,33,36,39 from Europe (Denmark and Sweden), seven studies28,34,35,40,43–45 from North America (US and Canada), and one study 26 from Oceania (Australia). Of the studies included, 16 were retrospective cohorts26,28,29,31–34,36,37,39,40,42–46 and 6 were prospective cohorts.27,30,35,38,41,47 The sample sizes of the studies ranged from 4349 to 2,421,302 individuals. HBV/HCV infection was confirmed by laboratory test (detecting serum markers) and International Classification of Diseases (ICD) codes. Regarding PC verification, the majority of studies were based on ICD codes, with the exception of one that utilizes histopathology and imaging. Studies that provided follow-up time had an average follow-up time of 2.3–16 years, while some of the included studies did not provide follow-up time. The online Supplemental Table S2 summarizes the other characteristics (source of study subjects, study continents, mean years of participants, adjusted confounders, and corresponding data). Supplemental Table S3 provides a detailed assessment of the NOS. Nine studies30–32,35,36,41–42,44,47 have been rated as high quality with a score of 9, 13 studies26–29,33,34,37–40,43,45,46 have been rated as medium quality with a score of 7 or 8, and no study was rated as low quality.
Main characteristics of included studies.

Anti-HBc, anti-hepatitis B core antigen; Anti-HBs, anti-hepatitis B surface antigen; Anti-HCV, anti-hepatitis C virus; HBsAg, hepatitis B surface antigen; HBV, hepatitis B virus; HCV, Hepatitis C virus; ICD, international classification of diseases; NOS, Newcastle-Ottawa Scale; PC, pancreatic cancer; RNA, ribonucleic acid.
Association between HBV infection and risk of PC
Fifteen cohort studies26,27,30–34,36–38,41–43,46,47 explored the association between HBV infection and risk of PC. We found that HBV infection was correlated with an increased risk of PC (pooled HR = 1.53, 95% CI: 1.42–1.68, p < 0.00001), with no heterogeneity (I2 = 0%, p = 0.49) (Figure 2).

Forest plot of overall meta-analysis of association between HBV infection and risk of pancreatic cancer.
In order to explore potential factors affecting the overall results, we performed several subgroup analyses in different ways. When conducting subgroup analyses based on study design, confirmation methods of HBV infection, study quality, and publication years, the results were consistent with the overall pooled results. In a subgroup analysis stratified by study location, HBV infection was seen to be associated with an increased PC in Asia (n = 9, HR = 1.53, 95% CI: 1.37–1.71, p < 0.00001) and North America (n = 2, HR = 1.47, 95% CI: 1.02–2.13, p = 0.04), whereas no significant association was observed in Europe (n = 3, HR = 1.51, 95% CI: 0.91–2.52, p = 0.11) and Oceania (n = 1, HR = 0.70, 95% CI: 0.30–1.63, p = 0.41). The results of the subgroup analyses were displayed in Table 2 and Supplemental Figures S1–S5.
Subgroup analyses of association between HBV and risk of PC.

CI, confidence interval; HBV, Hepatitis B virus; HR, hazard ratio; ICD, international classification of diseases; PC, pancreatic.
Additionally, to further validate the stability of our results, we conducted a sensitivity analysis to assess the impact of each study on the overall estimate by omitting one study at a time, which revealed that none of the individual studies could significantly alter the summary of risk estimates in this analysis. The results of sensitivity analyses were presented in Supplemental Table S4. With regard to publication bias, there was no substantial publication bias by Begg’s funnel plot, Begg’s and Egger’s tests (Supplemental Figure S6, pBegg = 0.843, pEgger = 0.174).
Association between HCV infection and risk of PC
Fourteen cohort studies26,28,29,31,32,35–37,39,40,42–45 reported the relationship between HCV infection and risk of PC. The meta-analysis showed that HCV infection was linked to an increased risk of PC (pooled HR = 1.82, 95% CI: 1.51–2.21, p < 0.00001). Moderate heterogeneity was observed in this analysis (I2 = 65%, p = 0.0004) (Figure 3).
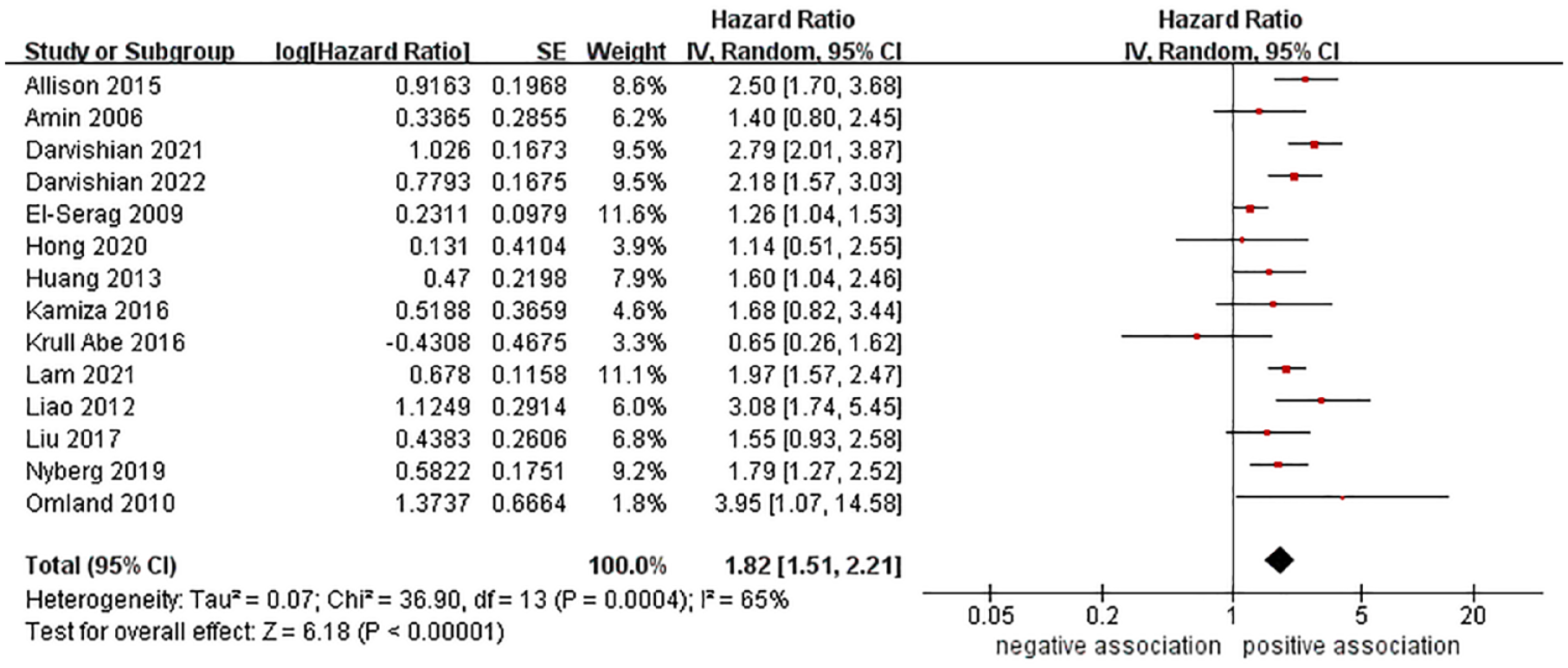
Forest plot of overall meta-analysis of association between HCV infection and risk of pancreatic cancer.
To investigate the potential sources of heterogeneity among the studies and assess the reliability of the overall results, we conducted several subgroup analyses. In a subgroup analysis stratified by study design, the meta-analysis findings revealed that HCV infection was associated with a heightened risk of PC in retrospective cohort studies (n = 12, HR = 1.84, 95% CI: 1.51–2.23, p < 0.00001), yet in prospective cohort studies, the association was positive but not statistically significant (n = 2, HR = 1.36, 95% CI: 0.37–5.07, p = 0.64). In a subgroup analysis stratified by study location, HCV infection was seen to be associated with an increased PC in North America (n = 6, HR = 1.98, 95% CI: 1.53–2.57, p < 0.00001) and Europe (n = 3, HR = 1.67, 95% CI: 0.78–2.30, p = 0.002), whereas no significant association was observed in Asia (n = 4, HR = 1.49, 95% CI: 0.78–2.86, p = 0.23) and Oceania (n = 1, HR = 1.40, 95% CI: 0.80–2.45, p = 0.24). When conducting subgroup analyses based on HCV confirmation methods, study quality, and publication year, the results were consistent with the overall pooled results. The results of the subgroup analyses were presented in Table 3 and Supplemental Figures S7–S11.
Subgroup analyses of association between HCV and risk of PC.

CI, confidence interval; HCV, Hepatitis C virus; HR, hazard ratio; ICD, international classification of diseases; PC, pancreatic cancer.
In addition, we carried out sensitivity analysis by deleting any single study and combining the remaining studies. The results revealed that no considerable alteration was observed when any of the studies were excluded, indicating that our results are robust. Supplemental Table S5 provides the results of the sensitivity analyses. Regarding publication bias, no substantial publication bias was observed by Begg’s funnel plot and Begg’s and Egger’s tests (Supplemental Figure S12, pBegg = 0.324, pEgger = 0.665).
Association between HBV and HCV co-infection and risk of PC
Four cohort studies26,42,43,45 provided data concerning the association between HBV and HCV co-infection and risk of PC. The meta-analysis indicated that co-infection with HBV and HCV was linked to an increased risk of PC (pooled HR = 2.32, 95% CI: 1.40–3.85, p = 0.001), with no heterogeneity observed (I2 = 0%, p = 0.60) (Figure 4). To further validate the stability of the results, sensitivity analysis was conducted by removing individual studies one by one and the results showed no significant changes, demonstrating that the results were robust.

Forest plot of association between HBV and HCV co-infection and risk of pancreatic cancer.
Discussion
Summary of principal findings
This meta-analysis collates the current evidence from 22 cohort studies (involving 10,572,865 participants) to investigate the association between HBV/HCV infection and risk of PC. Meta-analysis from 15 cohort studies revealed that HBV infection was correlated with an increased risk of PC (HR = 1.53, 95% CI: 1.40–1.68, p < 0.00001). Similarly, meta-analysis from 14 cohort studies showed HCV infection was also associated with an increased risk of PC (HR = 1.82, 95% CI: 1.51–2.21, p < 0.00001). Further subgroup analyses showed that the pooled HRs were similar, regardless of study location, study design, confirmation methods of HBV/HCV, study quality, and publication year, although some of the results did not attain statistical significance due to limited number of studies. Meta-analysis from four cohort studies indicated that co-infection with HBV and HCV was linked to an increased risk of PC (HR = 2.32, 95% CI: 1.40–3.85, p = 0.001). The above-mentioned results were further validated by sensitivity analyses.
Comparison with previous studies
To the best of our knowledge, this is the first meta-analysis, which focuses solely on cohort studies to examine the relationship between HBV/HCV infection and risk of PC. Previously, in 2013 and 2014, six relevant meta-analyses9–13 with inconsistent results were published, including three meta-analyses9,12,13 on the relationship between HBV/HCV infection and PC risk, and three meta-analyses10,11,14 on the relationship between HBV infection and PC risk. Majumder et al. 14 conducted a smaller meta-analysis of three studies (one cohort and two case-control studies) and showed that individuals who tested positive for hepatitis B surface antigen (HBsAg) had a significantly greater likelihood of developing PC when compared to controls (OR = 1.50, 95% CI: 1.21–1.87). However, the risk of PC was not significantly higher for those with positive anti-hepatitis B core (HBc) status (n = 2, OR = 1.23, 95 % CI: 0.95–1.59). Fiorino et al. 9 conducted a meta-analysis of six studies (four case-control and two cohort studies) and showed that HBsAg positivity was linked to a higher risk of PC (n = 5, RR = 1.18, 95% CI: 1.04–1.33), whereas no significant association was observed between anti-HCV positivity and PC risk (n = 3, RR = 1.16, 95% CI: 0.99–1.3). Xu et al. 13 performed another meta-analysis of eight studies (five case-control and three cohort studies) and showed that chronic hepatitis B and HBsAg positive had a significantly increased risk of PC (n = 6, OR = 1.20, 95% CI: 1.01–1.39) and past exposure to HCV was also linked to an increased risk of PC (n = 5, OR = 1.26, 95% CI: 1.03–1.50). Similar results were observed in another larger meta-analysis 12 of ten studies (eight case-control and two cohort studies) published in 2013. The other two meta-analyses conducted by Li et al. 10 and Wang et al. 11 included eight studies (five case-control and three cohort studies) and ten studies (eight case-control and two cohort studies), respectively. The results showed that individuals infected with HBV were associated with an increased risk of PC compared to individuals not infected with HBV. Among the six meta-analyses mentioned above, only a few cohort studies were included, all of which were included in this study. Some of the previous meta-analyses were due to the limited number of studies included, resulting in insufficient statistical power to show statistically significant findings. In 2019 and 2020, two meta-analyses15,16 were conducted to examine the association between HCV infection and PC. Masarone and Persico 15 conducted a meta-analysis of five studies (four case-control studies and one cohort study), and the result showed that HCV infection was associated with an increased risk of pancreatic adenocarcinoma (OR = 1.60, 95% CI: 1.25–2.04). However, the findings of this meta-analysis were mainly derived from data that had not been adjusted for any confounding factors. Whereafter, Arafa et al. 16 performed a larger meta-analysis of eight cohort and eight case-control studies, which indicated that individuals with HCV infection had a higher chance of developing PC than those without it (OR = 1.51, 95% CI: 1.31–1.74). Notably, this meta-analysis mistakenly included a cohort study on the cancer risk of registered opioid substitution therapy patients. 48 Recently, in 2021, Liu et al. 17 conducted a larger meta-analysis of seven case-control and 10 cohort studies, and the result indicated that individuals with HBsAg or HBV DNA seropositivity were found to have a significantly increased risk of PC, with an RR (95% CI) of 1.39 (1.19–1.63), the similar conclusion was drawn from the subgroup analysis (subgroup by study design, population, and sex ratio) except when stratified by study region: in Europe and Oceania, the RR and 95% CI were 1.44 (0.88–2.34) and 1.47 (0.38–5.71), respectively.
Compared to all previous meta-analyses,9–17 our meta-analysis based on cohort studies further confirms and expands their findings. Previous meta-analyses were mainly composed of hospital-based case-control studies, with most of them only including a small number of cohort studies. We excluded case-control studies that were more susceptible to bias and included newly published, high-quality cohort studies that had not been included in previous studies, providing the latest and most comprehensive high-quality evidence on the link between HBV/HCV infection and risk of PC. In addition, our study further explored the relationship between HBV and HCV co-infection and the risk of PC and found that HBV and HCV co-infection had a higher risk of PC, which suggested that we may strengthen the screening of PC for patients with such co-infection in clinical practice.
Potential explanations and implications
The precise mechanism of how HBV/HCV increases the risk of PC is still a mystery, though there are some potential explanations. First, the liver and pancreas both originate from a single source of endoderm cells and have similar early development stages. The pancreas can serve as a storage place for HBV/HCV, while also providing the perfect conditions for their replication.49–51 Second, the pancreas is situated in the vicinity of the liver, with the common origin of their blood vessels and bile ducts allowing for microorganisms to travel between the two organs. 51 In fact, several studies have demonstrated that during HBV/HCV infection, HBsAg can be identified in pancreatic juice, and the replication of both HBV DNA and HCV RNA can be seen in pancreatic tissue.5,52,53 Third, Inflammatory reactions may play an important role in this process. HBV/HCV infection in pancreatic tissue may induce sustained inflammatory changes, directly promoting carcinogenesis, and/or indirectly inducing carcinogenic effects by promoting systemic inflammatory status and/or being associated with insulin resistance mechanisms. The presence of an inflammatory microenvironment encourages an increase in growth factors and cytokines, which then leads to alterations in driver genes and the stimulation of cancer cell growth and proliferation.32,51 Fourth, HBV and HCV are both carcinogenic viruses that can insert their DNA or RNA into the genome of infected cells, resulting in the disruption of proto oncogenes and tumor suppressor genes, thus causing PC.13,54,55 Fifth, research has shown that Hepatitis B virus X protein (HBx) has a role in increasing pancreatic cancer risk by altering the PI3K-Akt signaling pathway. This alteration leads to an upregulation of ErbB4 and TGF-a, which is linked to the expression of HBx protein. 56 In addition, Fiorino et al. 57 proposed a tensegrity model hypothesis, indicating that the tension integrity model that occurs in the liver and pancreas during HBV- and HCV-induced carcinogenesis is associated with intracellular and extracellular modifications.
Given the global burden of HBV/HCV and PC, the findings of this meta-analysis are of great importance in terms of clinical practice. Our meta-analysis has revealed that individuals with HBV/HCV infection are more likely to develop PC, particularly those who are infected with both viruses. Consequently, it is essential to provide these patients with more thorough screening and early management (e.g. vaccination to prevent HBV infection and active antiviral treatment for patients with viral hepatitis) to potentially reduce the risk of PC.
Strength and limitations
The current research has several strengths. First, our meta-analysis of cohort studies is the most current, expansive, and comprehensive to date, investigating the association between HBV/HCV and risk of PC. Second, the majority of the cohort studies we included were of a medium to high quality, thus guaranteeing the dependability of the results. Third, detailed subgroup analysis and sensitivity analysis were performed to assess the reliability of the combined risk estimation, and publication bias tests were carried out to demonstrate no significant publication bias, thus ensuring the stability and reliability of the research results.
Despite this, it is important to take into account the limitations of this study. First, all the studies included adjusted for confounding factors; however, the adjusted confounding factors were not consistent across studies. For instance, some studies did not adjust or only partially adjusted for the common risk factors of PC, such as smoking, drinking, diabetes, chronic pancreatitis, and family history of PC. Additionally, residual and unmeasured confounding factors cannot be completely excluded. These factors could affect the estimation of the effect value of the results. Second, regarding the relationship between HCV infection and PC, moderate statistical heterogeneity was observed (I2 = 65%), which may limit the interpretation of the results. We further performed numerous subgroup analyses to explore the heterogeneity, but did not find any evidence of heterogeneity. We speculate that the heterogeneity may stem from the diversity of population characteristics, different follow-up times, and inconsistent adjustment for confounding factors. Third, the studies included in our analysis used laboratory tests and ICD code to confirm HBV/HCV. We conducted subgroup analysis based on these two methods and found similar results. However, the laboratory testing methods used in the various studies varied. We could not do further analysis due to limited data, but we cannot completely rule out the potential effect of these differences on the results. Fourth, the effects of HBV and HCV infection on the clinical pathological characteristics and long-term survival of resected and unresectable PC remain unknown, and more research is required to improve clinical management in the future. Finally, regarding the relationship between HBV infection and PC, there is a lack of population data from Oceania, Europe, and North America. Similarly, the data from Oceania, Europe, and Asia in regard to the relationship between HCV infection and PC are also scarce. Consequently, further research is required to further validate these results in these regions.
Conclusion
In conclusion, our meta-analysis showed that HBV/HCV infection or co-infection with HBV and HCV was associated with an increased risk of PC. Future prospective cohort studies need to take into account various ethnicities and any confounding factors, as well as investigate the potential mechanisms of PC development in those with HBV/HCV. Physicians should be aware of the potential for PC in these patients in clinical practice.
Supplemental Material
sj-docx-1-tai-10.1177_20499361231212161 – Supplemental material for Association between hepatitis B or hepatitis C virus infection and risk of pancreatic cancer: a systematic review and meta-analysis of cohort studies
Supplemental material, sj-docx-1-tai-10.1177_20499361231212161 for Association between hepatitis B or hepatitis C virus infection and risk of pancreatic cancer: a systematic review and meta-analysis of cohort studies by Jian-Feng Zhao, Qiu-Ping Teng, Yang Lv, Xiao-Yi Li and Yi Ding in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
