Abstract
Background:
Ceftolozane/Tazobactam is a β-lactam/β-lactamase inhibitor combination with a high range of efficacy and broad-spectrum action against multidrug-resistant bacterial strains.
Objectives:
The present study aimed to analyze the
Design:
Systematic review and meta-analysis.
Methods:
A systematic literature search was conducted on the Web of Science, Embase, PubMed, Scopus, and Google Scholar electronic databases from the beginning of databases to December 2022 to cover all published articles relevant to our scope.
Results:
At last, 31 publications that met our inclusion criteria were selected for data extraction and analysis by Comprehensive Meta-Analysis Software. The pooled prevalence of Ceftolozane/Tazobactam susceptibility for ESBLs-EC and ESBLs-KP was estimated at 91.3% [95% confidence interval (CI): 90.1–92.5%] and 65.6% (95% CI: 60.8–70.2%), respectively. There was significant heterogeneity among the 31 studies for ESBLs-EC (χ2 = 91.621;
Conclusion:
Based on the meta-analysis results, Ceftolozane/Tazobactam has a more promising
Introduction
Extended-spectrum β-lactamases (ESBLs)-producing Enterobacterales with two predominant pathogens, ESBLs-producing
Ceftolozane/Tazobactam is a β-lactam/β-lactamase inhibitor combination with a broad range of efficacy. It is active against
This study aimed to evaluate the in vitro efficacy of Ceftolozane/Tazobactam as a β-lactam combination agent against ESBLs-producing
Materials and methods
Search strategies
A systematic review was performed using the Preferred Reporting Items for Systematic Reviews and Meta-Analysis recommendations. This systematic review included searching various electronic bibliographic databases such as Web of Science, PubMed, Embase, Scopus, and Google Scholar to identify all related studies published from the beginning of databases to December 2022. The keywords were included in the articles’ title, abstract, or full text. We used a combination of predefined keywords such as ‘Ceftolozane-Tazobactam’ AND ‘
Selection criteria and quality assessment
Two reviewers independently checked the database results with the related keywords. They surveyed the titles, abstracts, and full texts to apply eligibility for inclusion based on the inclusion criterion, and any inconsistencies between reviewers were resolved by debate. There were no restrictions imposed on the language in our search, but the abstract must be available in English at the very least. The research was restricted to cross-sectional publications indexed on the Web of Science, PubMed, or Scopus. Related studies with the following criteria were included in our study:
(1) Antibacterial activity was determined using the standard method, such as broth micro-dilution10–13; (2) MIC 50, MIC 90 (minimum inhibitory concentration at which 50% and 90% of ESBLs-EC and ESBLs-KP isolates were inhibited); and their MIC ranges were reported; and (3) original articles that were performed on clinically derived isolates.
Meanwhile, exclusion criteria were: (1) studies that did not use the antibacterial susceptibility testing method; (2) studies with a sample size of fewer than 10 isolates; (3) studies that were performed on samples with animal or environmental origin; and (4) studies that were performed on
Quality assessment and data extraction
Two researchers assessed eligible studies’ quality separately using a critical appraisal checklist developed by the nine-point Joanna Briggs Institute, and any disagreement was resolved by consensus. 14 Items related to the title and abstract, introduction, methods, results, discussion, and other information were determined, and a score was assigned to each item.
Then, for all eligible studies, the following data were extracted: the first author’s name, date of publication, date of study performing, the geographical distribution of clinical samples, sample size, MIC results, and antibiotic susceptibility rate.
Statistical analysis
Meta-analysis was performed using the random effects model to estimate the pooled prevalence and corresponding 95% confidence interval (CI). Heterogeneity between studies was evaluated using Cochran’s
Results
We selected 31 eligible studies for inclusion in our meta-analysis. A flowchart depicting the literature searches and study selection process is provided in Figure 1. The main detailed characteristics of each included article are presented in Tables 1 and 2.
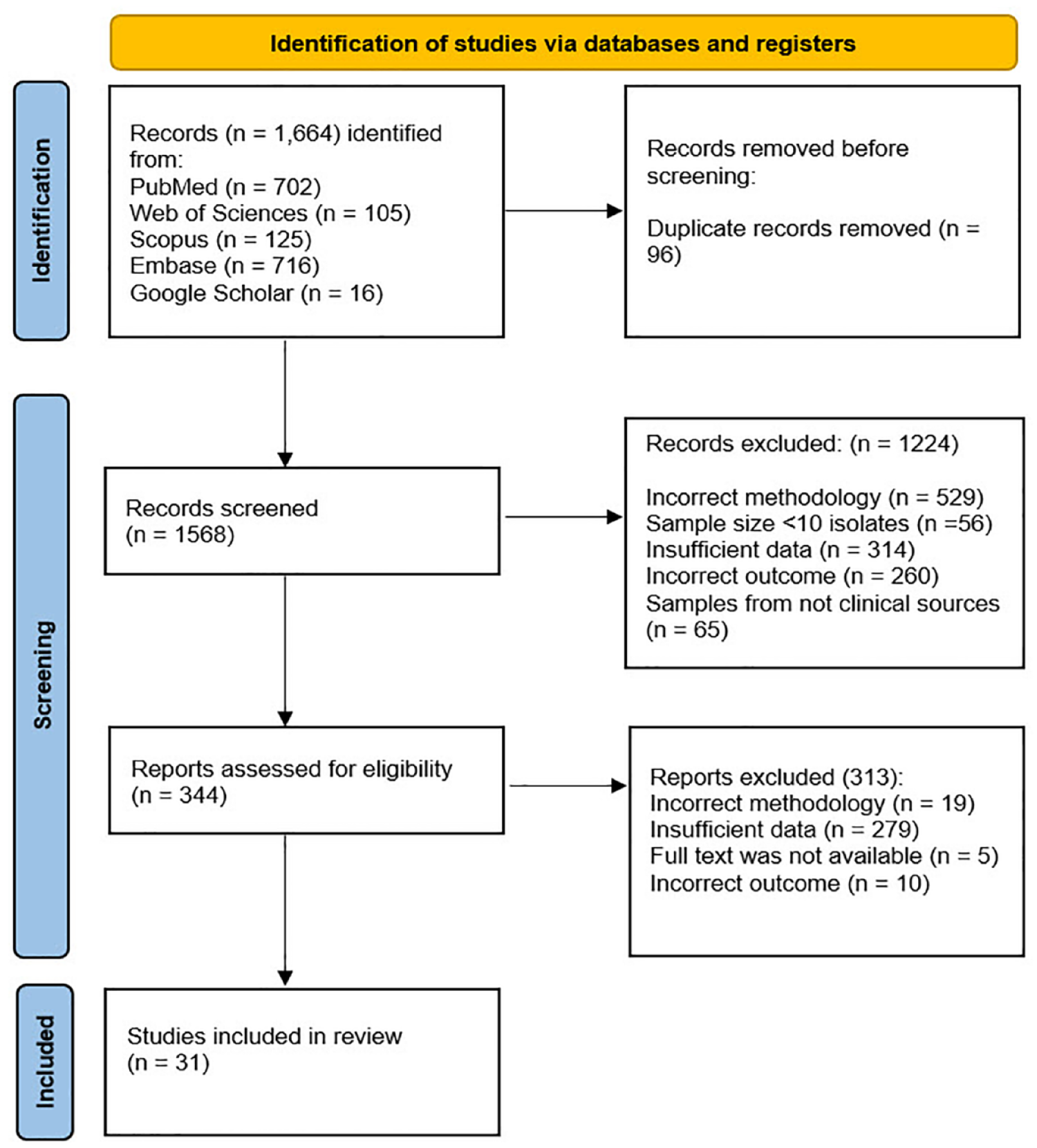
Flowchart of the literature search strategy and study selection.
The main characteristics of studies included in the meta-analysis for ESBLs-EC isolates.

Patients >65 years old.
Intensive care unit.
Immunocompromised patients.
Intra-abdominal infections
Urinary tract infections
ESBL, extended-spectrum β-lactamase; ESBLs-EC, ESBLs-producing
The main characteristics of studies included in the meta-analysis for ESBLs-KP isolates.

Patients >65 years old.
Intensive care unit.
Immunocompromised patients.
Intra-abdominal infections
Urinary tract infections
ESBL, extended-spectrum β-lactamase; ESBLs-KP, ESBLs-producing
This study reviewed 31 articles from various countries across different regions, including North and South America, Asia, the Middle East/Africa, Asia/Pacific, and Latin America. We investigated several collections of ESBLs-producing
According to this evaluation, in 28 studies,
In these studies, the pooled prevalence of Ceftolozane/Tazobactam susceptibility for ESBLs-EC isolates was assessed at 91.3% (95% CI: 90.1–92.5%) [Figure 2(a)]. There was significant heterogeneity among the 31 studies (χ2 = 91.621;

Forest plot of the pooled prevalence of Ceftolozane–Tazobactam susceptibility. (a) The prevalence of Ceftolozane–Tazobactam susceptibility in ESBLs-EC isolates and (b) The prevalence of Ceftolozane–Tazobactam susceptibility in ESBLs-KP isolates.
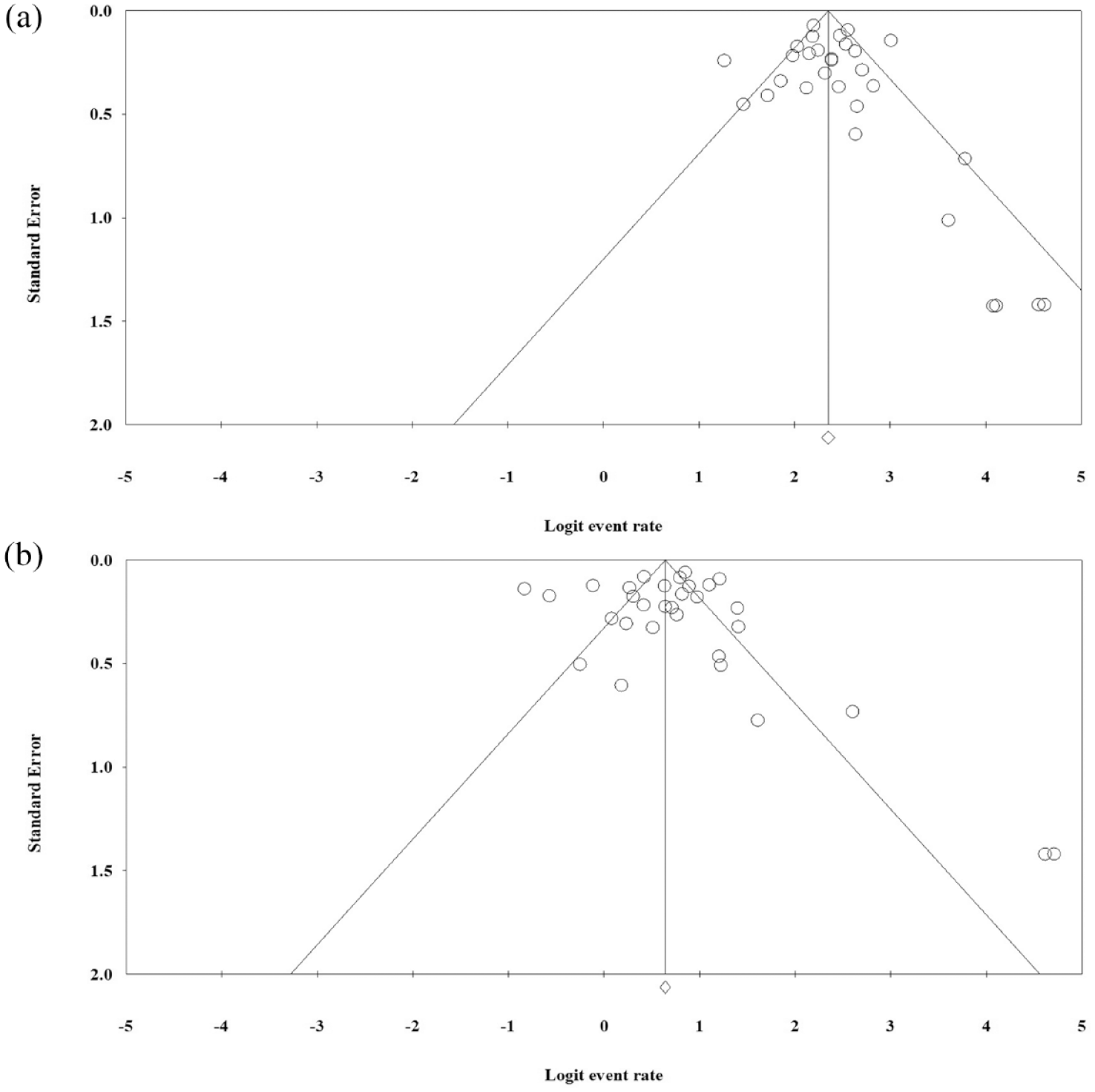
Funnel plot of meta-analysis on the pooled prevalence of Ceftolozane–Tazobactam susceptibility for evaluation of publication bias. (a) The Funnel plot of Ceftolozane–Tazobactam susceptibility in ESBLs-EC isolates and (b) The Funnel plot of Ceftolozane–Tazobactam susceptibility in ESBLs-KP isolates.
Also, most clinical isolates of ESBLs-KP had MIC50 at a concentration of 1 µg/mL which 50% of the isolates were inhibited. In contrast, MIC90 was at 32 µg/mL which 90% of the isolates were inhibited.
The pooled prevalence of Ceftolozane/Tazobactam susceptibility for ESBLs-KP isolates was assessed at 65.6% (95% CI: 60.8–70.2%) [Figure 2(b)]. There was high significant heterogeneity among the 31 studies (χ2 = 348.72;
The sensitivity analysis was performed by excluding one study at a time to evaluate the impact of each study on the summary results and between-study heterogeneity. None of the studies alone in the sensitivity analysis showed any significant effect on estimated prevalence (Supplemental Figure S1A and B).
Meta-regression results for susceptibility rate of ESBLs-EC (coefficient: −0.033, 95% CI: −0.111–0.043,
Discussion
Faced with a growing global health threat posed by increasing resistance to main therapeutic drugs against ESBLs-producing Enterobacterales, which has left clinicians with few viable alternatives, there is now an even greater need for introducing new effective antibiotics that demonstrate activity against ESBLs-producing gram-negative bacteria, especially ESBLs-EC and ESBLs-KP strains. 31 To spare carbapenems as the first choice for treating infections caused by ESBLs-producing Enterobacterales and to prevent the increase of carbapenem-resistant strains, the use of alternative drugs is essential.5,41,42 This surveillance study evaluated the antimicrobial susceptibility profile of Ceftolozane/Tazobactam, an approved cephalosporin-beta-lactamase inhibitor combination, for treating infections caused by ESBLs-EC and ESBLs-KP strains on a global scale. To achieve this, we utilized the CLSI MIC interpretive criteria, which are predominant in the United States, Canada, and many regions outside Europe (such as Asia, the Middle East/Africa, Asia/Pacific, Latin America, etc.), instead of the European Committee on Antimicrobial Susceptibility Testing (EUCAST) 13 to determine susceptibility rates.43,44
Pfaller
Based on previous studies, the results indicated a lower activity of Ceftolozane/Tazobactam against clinical isolates of ESBLs-KP due to a high rate of non-susceptible isolates (34.6%). The evaluated MIC values of Ceftolozane/Tazobactam against ESBLs-KP were 1 µg/mL for MIC50 and 32 µg/mL for MIC90, which are much higher than the MIC values of carbapenems for ESBLs-KP reported in the literature.2,3,5,24,29–37,39,40,45 Ceftolozane/Tazobactam exhibits moderate activity against certain ESBLs-KP strains. This may be attributed to the coproduction of Carbapenemases in ESBLs-KP strains, which is less prevalent in
Considering the results of meta-regression (Supplemental Figures S2A), the susceptibility rate of Ceftolozane/Tazobactam against ESBLs-EC isolates has shown a slight decrease during recent years, leading to an increase in the prevalence of resistant phenotypes. This finding suggests that the usage of Ceftolozane/Tazobactam and the pressure of natural selection may contribute to an increase in the prevalence of antibiotic-resistant ESBLs-EC strains in the future. On the other hand, the meta-regression analysis related to the susceptibility rate of Ceftolozane/Tazobactam against ESBLs-KP isolates (Supplemental Figure S2B) did not reveal a significant increasing trend in the estimated pooled prevalence of susceptibility rate over time.
The main limitations of our study were the lack of a standard MIC breakpoint for defining Ceftolozane/Tazobactam susceptibility rates in some of the studies, and the absence of sufficient data about the molecular enzymes of the studied ESBLs strains.
In summary, the meta-analysis results demonstrate that ESBLs-EC isolates from different clinical sources show a higher susceptibility to Ceftolozane/Tazobactam compared to ESBLs-KP isolates. Although Ceftolozane/Tazobactam exhibits limited activity against ESBLs-KP strains when compared to carbapenems, it effectively inhibits the majority of clinical isolates of ESBLs-positive Enterobacteriaceae. Therefore, it has the potential to serve as a valuable empirical therapeutic agent, offering an alternative to carbapenems for treating patients with infections caused by ESBLs-producing Enterobacteriaceae.
Supplemental Material
sj-docx-1-tai-10.1177_20499361231212074 – Supplemental material for Global evaluation of the antibacterial activity of Ceftolozane/Tazobactam against ESBLs-producing Escherichia coli and Klebsiella pneumoniae: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tai-10.1177_20499361231212074 for Global evaluation of the antibacterial activity of Ceftolozane/Tazobactam against ESBLs-producing Escherichia coli and Klebsiella pneumoniae: a systematic review and meta-analysis by Marzieh Rahim Khorasani, Soodabeh Rostami, Arash Bakhshi and Raheleh Sheikhi in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
