Abstract
Intravenous push (IVP) antimicrobial administration refers to rapid bolus infusion of medication. This drug delivery method offers improved patient convenience, superior patient and nursing satisfaction, and cost savings when used in outpatient parenteral antimicrobial therapy (OPAT). Antimicrobial agents must demonstrate optimal physiochemical and pharmacologic characteristics, as well as sufficient syringe stability, to be administered in this manner. Additionally, impacts on medication tolerability, patient safety, and effectiveness must be considered. This narrative review summarizes the available data and practical implications of IVP administration of antimicrobials in the OPAT setting.
Introduction
Outpatient parenteral antimicrobial therapy (OPAT) is the administration of intravenous (IV) antimicrobials in a non-acute care setting for at least two consecutive days. OPAT shortens hospital length of stay, prevents hospital-associated complications, and yields significant cost savings for health systems. 1 OPAT can be provided in a skilled nursing facility (SNF), long-term care or transitional care facility (LTCF), infusion therapy center (ITC), ‘hospital in the home’, or at home by the patient, a caregiver, or a home health nurse. 2
Standard OPAT infusion times typically range from 30 minutes to several hours. Long infusion times can be disruptive to patients’ activities of daily living and decrease scheduling availability at ITCs. However, certain antimicrobials may be administered via intravenous push (IVP) bolus dosing as a rapid infusion over 1–5 minutes. 3 In addition to shortening infusion times, IVP requires less time for patient training, improves patient and healthcare-provider satisfaction, allows smaller fluid volumes to be used, and decreases costs. 3 This article reviews the available pharmacologic, clinical, and pharmacoeconomic implications of IVP use in OPAT.
Favorable antimicrobial characteristics for IVP
Many factors influence whether an IV medication is appropriate to administer rapidly in a small volume of fluid. The relationship between the physiochemical properties of the compound, the pharmacology of the active ingredient, and the technique required to prepare or administer the dose determine whether IVP is a viable option for a given medication. Characteristics of antimicrobials typically given via IVP in OPAT are summarized in Tables 1 and 2.
Ideal drug characteristics for IVP administration.

IVP, intravenous push.
Select IV OPAT antimicrobials.
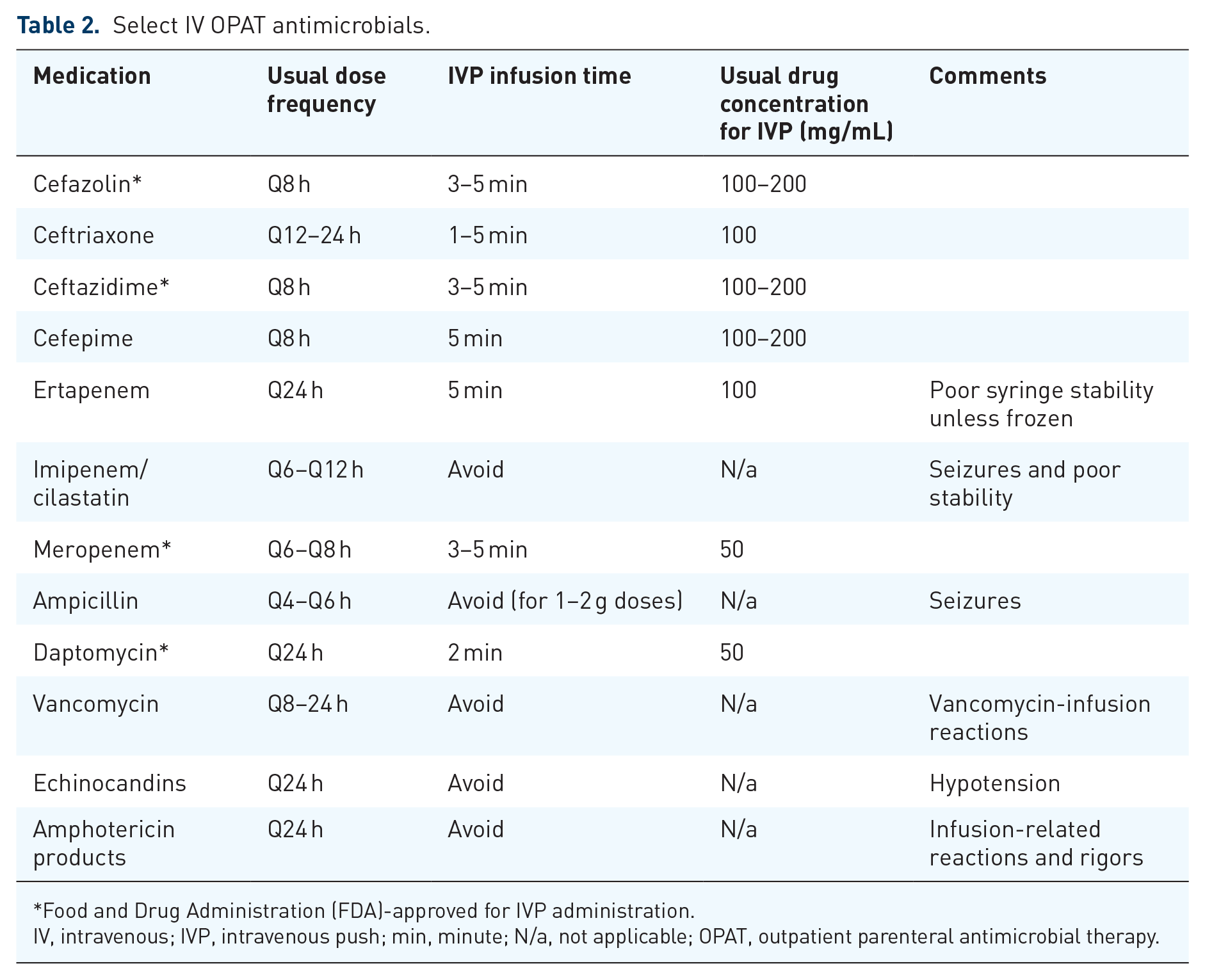
Food and Drug Administration (FDA)-approved for IVP administration.
IV, intravenous; IVP, intravenous push; min, minute; N/a, not applicable; OPAT, outpatient parenteral antimicrobial therapy.
Physiochemical characteristics
One benefit of IVP is the minimal fluid volume required for each dose and the rapidity at which the dose can be delivered. Therefore, it is critical to consider whether the chemical stability of the antimicrobial compound is compromised at high concentrations and if the resulting solution is locally and systemically tolerable for the patient. Ideal medication solutions for OPAT are isotonic and pH neutral at therapeutic doses as well as chemically stable in 10–20 mL of diluent for at least 1 week.1,4,5
Solubility and stability are highly dependent on the saturation point at a given temperature and pressure. 6 Supersaturation can occur as the concentration of a solution increases. Decreasing the diluent volume of a compound for IVP may pose a safety risk due to drug precipitation and/or loss of medication potency. Therefore, all medications considered for IVP should have published evidence supporting stability at the given concentration in a specific diluent. Ampicillin and ceftaroline are examples of medications that are not ideal to be delivered via IVP. The stability of ampicillin in solution decreases as concentration of the compound increases, 7 while ceftaroline lacks evidence to support stability in a diluent volume less than 50 mL or a concentration greater than 12 mg/mL. 8 Stability guidance for medications may be listed in the package insert, available through American Society of Health-System Pharmacists (ASHP) publications, or through other studies. For OPAT, stability for at least 1 week after preparation is ideal for pharmacy operational efficiency and patient convenience. 1 Shorter stability may be acceptable in an institutional or facility-based setting.
Chemically stable medications in small volumes must be evaluated for patient tolerability and safety. Tolerability depends largely on osmolality, tonicity, and pH which are affected by medication concentration. 5 Osmolality describes the osmotic pressure exerted by a solvent across a semi-permeable membrane, while tonicity describes the osmotic pressure exerted by a solvent across a cell membrane (endothelial vasculature) compared to blood. pH is a measure of a solution’s hydrogen ion concentration and its relative degree of acidity or basicity. Ideal IVP solutions are isoosmotic (isotonic), do not induce fluid shifts or result in endothelial cell damage, and are near physiologic pH. 1 Hypertonic and drastically acidic or basic solutions induce vascular damage, usually manifesting as local pain and phlebitis, though systemic reactions involving cerebral blood flow and blood–brain permeability changes are also possible. 9 However, hypertonic solutions or those at extremes of pH may also be better tolerated as IVP due to the reduced contact time with the endothelium compared to larger volume infusions administered in equivalent concentrations.
Multiple professional organizations have published guidance for the ideal osmolality and pH of IV solutions. The Infusion Nurses Society (INS) 2021 Infusion Therapy Standards of Practice suggest solutions with osmolality >900 mOsm/kg be administered through a central line. 10 This is consistent with the American Society for Parenteral and Enteral Nutrition guidelines suggesting a maximum upper limit of 900 mOsm/L for peripheral lines. 11 However, both note that higher limits have been tolerated, and there is no official consensus on tolerability limits. Additional literature suggests an ideal range of 300 ± 30 mOsm/kg and recommends <1000 and <500 mOsm/kg for small- and large-volume injections, respectively. 5 Previous iterations of the INS guidelines have stated ideal infusates possess a pH range of 5–9, particularly when selecting type of vascular access, although more recent reexamination suggests that pH alone may not be an evidence-based predictor of tolerability. 9
Osmolality and pH thresholds may be difficult to apply to IVP antimicrobials due to the lack of standardized practice for IVP preparation and administration. 12 The theoretical osmolarity of a solution is the sum of osmolality of the solvent and diluent. This may differ from the measured osmolarity of a compound in practice due to changes in pH, temperature, and concentration. Therefore, to appropriately assess osmolality of IVP formulations, it is best to measure directly with all variables held constant. 13 Research has shown several medications routinely given via IVP may exceed suggested osmolality maximums when diluted in 0.9% sodium chloride (NS). For example, one study found that cefepime 1 g in 10 mL NS resulted in osmolality of 1040 mOsm/L, which exceeds the recommended 1000 mOsm/L threshold, however, this practice is generally accepted as safe and well tolerated. 14 Furthermore, a number of antimicrobial medications correspond to the recommended pH range of 5–9 and are associated with increased phlebitis risk.15,16 As such, there are no well-defined limits for pH or osmolality; rather all physiochemical drug characteristics should be considered in unison.
Overall, it is difficult to generalize conclusions impacting IVP medication practices due to the lack of standardization surrounding diluents [NS versus sterile water for injection (SWFI)] and administration rates. These differences across practices complicate interpretation of current research on patient tolerability; more well-designed, controlled studies are needed.
Pharmacologic characteristics
In addition to physiochemical properties, pharmacologic characteristics are essential for determining a compound’s suitability for IVP. An ideal formulation has low risk of infusion rate-related reactions. 1 Despite possessing proper stability, osmolality, and pH characteristics, some medications have a propensity to induce infusion reactions when administered rapidly. 3 For example, vancomycin-infusion reaction (VIR) is due to a non-immunoglobulin E-mediated, concentration or infusion rate-dependent release of histamine in patients receiving rapid doses of vancomycin. Histamine-mediated reactions, such as VIR, can induce vasodilation and marked hypotension. IVP may also increase the incidence and/or severity of dose-related adverse effects, such as corrected QT interval (QTc) prolongation with moxifloxacin or seizure risk with ampicillin.17,18 Such cases may be due to increased peak plasma drug levels from rapid administration.
Pharmacokinetic and pharmacodynamic (PK/PD) activity of the antimicrobial must also be considered. Drugs with concentration-dependent (peak or Cmax) associated antimicrobial activity are expected to be equally, if not more, effective when given IVP. Antimicrobials with time above minimum inhibitory concentration (MIC) (T > MIC) and total drug exposure (area under the curve/MIC)-related efficacy could hypothetically have decreased PD indices when given IVP, of particular concern for drugs with short half-lives. The clinical significance of this is examined in later sections of this review.
Preparation and administration
An ideal medication for the OPAT setting should be available in a ready-to-use IVP formulation. 4 Bedside manipulation requirements introduce opportunities for medication errors and contamination, therefore, use of commercially prepared or sterile compounded syringes that can be administered directly into the injection site is the best practice. If the medication must be reconstituted or compounded at the bedside, vial sizes containing exact doses available from the manufacturer are preferable. 19 In some cases, formulations provided by the manufacturer may limit the feasibility of IVP administration. For example, IV formulations of linezolid and metronidazole are only available in premixed bags of a fixed concentration and thus are always given via intermittent infusions.20,21
Clinical data
Effectiveness of IVP administration
Antimicrobial activity is based on the PK/PD characteristics of the agent. Generally, concentration-dependent antimicrobials maximize the rapidity of bacterial killing with high concentrations of drug per unit of time in relation to the bacterial MIC, whereas time-dependent antimicrobials maximize bacterial killing with longer durations where the drug concentration remains above the MIC (T > MIC).
Pharmacokinetic modeling and clinical outcomes data for antimicrobials given via IVP are limited, and nearly all available data pertain to beta-lactams. This is unsurprising, as other commonly used IV antimicrobials either have concentration (rather than time)-based activity and would be predicted to be equally or more efficacious when given via rapid push (i.e., daptomycin), 22 or are associated with infusion syndromes that preclude rapid administration (i.e., vancomycin and VIR). For time-dependent agents, such as beta-lactams, IVP may theoretically result in decreased T > MIC, potentially reducing efficacy. Several PK and clinical studies have evaluated whether this method of administration affects antimicrobial effectiveness.
One PK modeling study predicted that cefepime probability of target attainment (PTA) would be 2–5% lower when given via 2-min IVP versus 30-min piggyback infusion (IVPB), with greater reductions in PTA predicted for higher-MIC isolates (e.g., MICs of 1–4 mg/L versus 0.25–0.5 mg/L). 23 Notably, contemporary every 6-h dosing strategies for higher-MIC organisms were not modeled in this study, and creatinine clearance appeared to have a relatively insignificant effect on PTA between methods. Additional Monte Carlo simulation studies with ertapenem, meropenem, cefepime, aztreonam, and cefazolin have demonstrated similar T > MIC profiles with IVP compared to 30-min infusions.24 –26
Clinical data comparing outcomes of IV beta-lactams administered via IVP or IVPB are available from two retrospective pre/post studies. In September 2017, the Category 5 Hurricane Maria destroyed much of the power and economic infrastructure of Puerto Rico, a major manufacturer of US medical devices, leading to a months-long nationwide shortage of the small-volume sterile IV bags used to infuse antibiotics. As a result, two medical centers were compelled to switch to IVP beta-lactams and later reported comparative outcomes with standard IVPB. The first study, examining 213 patients with gram-negative bacteremia (primarily Escherichia coli of urinary origin), found IVP and IVPB of either cefepime or meropenem led to comparable times to defervescence, WBC normalization, weaning of vasopressors, and similar rates of in-hospital mortality. 27 The second, examining 200 patients receiving OPAT for either osteomyelitis or infective endocarditis, found that IVP and IVPB administration of cefazolin, ceftriaxone, cefepime, and daptomycin led to similar rates of mortality, ED visits, and hospital readmission at 30 days and 1 year, with no increased rates of adverse events in the IVP arm. 28 This study also found that patients receiving OPAT via IVP learned how to administer their antibiotics more quickly (92% versus 71% receiving IVPB were able to ‘teach back’ antimicrobial administration to their nurse in three or fewer attempts) and, perhaps as a consequence, that hospital length of stay was a median 1 day shorter with IVP (11 versus 12 days; p = 0.03). Similar clinical findings have been replicated in inpatient and emergency department (ED) settings.29,30 It is important to note that extended or continuous infusion beta-lactam administration has been associated with improved T > MIC and clinical outcomes in certain patient populations or organisms with elevated MICs, and IVP would not be an appropriate alternative in such cases. Otherwise, the available data, while scant, generally support the equivalent efficacy of IV beta-lactams when administered via IVP instead of traditional intermittent infusion.
Safety of IVP antimicrobials
IVP requires administration of a high concentration of antimicrobial over a faster rate compared to other drug delivery methods, which raises unique safety considerations. 31 The Institute for Safe Medication Practices (ISMP) has developed guidelines that identify risks, examine the evidence, and make recommendations for safe practices related to adult IVP medication preparation and administration. 4 Additionally, the INS has published a review of best practices for IVP medication administration to minimize patient safety risks associated with this method. 19
The safety of IVP antibiotics has been explored in several studies and is best supported for daptomycin and selected beta-lactams.32 –37 A study including 1116 cases of IVP antimicrobials given in the home found no significant difference in complication rates in patients who received IVP versus intermittent infusion methods. The rate of phlebitis with IVP was found to be low, with an incidence of 0.6/1000 catheter days, and did not differ significantly from phlebitis rates with intermittent infusion. 38
A prospective observational study within an OPAT program was conducted to determine the safety and efficacy of IVP ceftriaxone over 4 min, daptomycin over 2 min, and meropenem and ertapenem over 5 min. Over a median treatment duration of 21 days and a combined 4326 doses given to 184 patients, the study found no rapid infusion-related adverse reactions and concluded that IVP for OPAT is clinically safe and well tolerated. 39
Another retrospective cohort study conducted within a health system evaluated the incidence of adverse events with IVP beta-lactams during the aforementioned 2017 national shortage of small-volume parenteral solutions. In adult patients who received a 5-min slow IVP of aztreonam, ceftriaxone, cefepime, or meropenem, a total of 10 adverse events were identified among 1000 patients (1%). Of these, five were determined to be allergic reactions unrelated to the IVP method. The remaining five adverse events included one infiltration of a peripheral venous line with grade two phlebitis in a patient receiving IVP ceftriaxone, and four instances of neurotoxicity with IVP cefepime. Four of these cases were graded as being ‘possibly’ related to IVP administration, with one case graded as ‘probably’ being related to IVP administration. The study concluded that IVP beta-lactams were safe and a feasible alternative to IVPB infusion. The median duration of beta-lactams in this study was 3–5 days. 31
Other studies have reported adverse effects related to prolonged use of IVP beta-lactams. A prospective observational study of 163 outpatients receiving IVP beta-lactams over 3–5 min, found that 11 patients (7%) developed a late-onset adverse reaction. Symptoms appeared at the time of infusion and included infusion-related abdominal pain (n = 9), cytopenias (n = 7), elevated liver enzymes (n = 6), rash (n = 5), and abnormal urine microscopy (n = 2). The median treatment duration prior to the reactions was 25 days. IVP beta-lactams received in patients experiencing these adverse events included benzyl penicillin (n = 6), flucloxacillin (n = 2), ceftriaxone (n = 1), cefuroxime (n = 1), and piperacillin/tazobactam (n = 1). These adverse events were not observed with non-beta-lactams or continuous infusion beta-lactams. 40 Recently, prolonged courses of IVP cefepime in OPAT have been linked to neutropenia. A retrospective cohort study of 326 adult OPAT patients found 11 cases of cefepime-induced neutropenia (CIN). The study reported that the cases with CIN were three times more likely to have received IVP administration compared to non-CIN cases. Each patient with CIN received cefepime for 2 weeks or greater. 41 Due to the small number of cases, additional studies are needed to further validate these findings.
In summary, IVP antimicrobials appear to be safe for OPAT patients. Although the optimal frequency has not been established, the Infectious Diseases Society of America (IDSA) OPAT guidelines highlight the importance of laboratory monitoring in OPAT patients, regardless of infusion method, to lower the risk of readmissions and mitigate medication adverse effects. 1
Patient comprehension
Patients prefer to receive OPAT at home compared to a healthcare facility after hospital discharge. 42 Risk factors for readmission on OPAT include age, history of drug-resistant organisms, prior hospitalizations, and aminoglycoside use. 43 However, there are no randomized studies to inform selection of patients or caregivers who will be successful at home antimicrobial administration. Instead, a healthcare team must consider the complexity of a patient’s clinical course and social situation to create a feasible OPAT plan. 44 For home infusion, this requires that patients and caregivers are taught how to administer IV antimicrobials and demonstrate these skills accurately prior to discharge. 45
Standard infusion via gravity drip requires multiple steps for successful administration. This includes piercing a bag of IV antimicrobial with additional IV tubing, which is then primed and attached to the patient’s IV catheter. An infusion rate is calculated by counting drops in the drip chamber over a defined period. These steps must be done while maintaining sterile technique. Syringe pumps offer some simplification of this method by removing the rate monitoring step; the patient, however, must still be able to load the syringe, program the pump, and connect additional kink-resistant tubing. Both methods require more training and time of administration compared to IVP. 10
Fewer steps are needed for administering IVP antimicrobials. IVP antimicrobials are delivered to the patient in a ready-to-administer form. 4 No additional tubing is required with the syringe connected directly to the patient’s IV catheter. The patient injects the medication over a few minutes based on the time of infusion printed on the syringe’s label. During administration, the syringe’s label is easily seen and includes other important identifiers including the patient’s name, antimicrobial, and antimicrobial dose. 40 Easier infusion technique, fewer supplies, and less administration time all make IVP an attractive alternative to standard infusions for reducing the burden of OPAT to the patient and their caregivers.
Patient satisfaction
OPAT given via IVP has been linked to high rates of patient satisfaction,28,38 which may be attributed to several factors. First, IVP antibiotics have a simpler administration method which is easier for patients to learn and requires less time for patient teaching. 28 Additionally, IVP allows for shorter administration times and less equipment requirements leading to reduced costs, increased patient mobility, less daily restrictions, and a better quality of life.
A study assessing outcomes for patients receiving IV antimicrobials by various drug delivery methods in the home found high rates of client satisfaction across all delivery methods, including IVP. 38 In one study of OPAT patients surveyed to assess their satisfaction with the IVP method compared to IV drip infusion (n = 22), 96% of participants preferred the IVP method. Reduced administration times, convenience, and clear instructions were reasons cited for preference of IVP compared to intermittent infusion.
Another study surveyed 27 patients to compare satisfaction between adults receiving vancomycin versus daptomycin for OPAT. Vancomycin is commonly infused over a range of 60–120 min, whereas daptomycin can be administered IVP over 2 min. Patients reported more interference in daily routine with vancomycin compared to daptomycin. The investigators emphasized that patient satisfaction ties significantly into the ease of antimicrobial administration, of which, infusion time is a major factor. 46
Healthcare-provider satisfaction
The evidence surrounding healthcare-provider satisfaction of IVP antimicrobials is scarce. The few studies published assessing the healthcare team’s perspective are focused on nursing staff within an emergency setting. One report suggests that nurses overwhelmingly favor IVP administration of first-dose cephalosporins in the ED compared to traditional infusions. 47 This conclusion was drawn by a survey prior to the intervention in which 87% of nurses preferred IVP administration when possible. There was no post-intervention survey to compare results, however, the authors reported anecdotal post-intervention feedback which revealed satisfaction related to improved efficiency with nursing workflows. It is important to note that the institution supplied kits for reconstitution of each medication while also standardizing the diluent (SWFI) and administration rate (2 min). Therefore, results may not be generalizable to settings where nurses must gather supplies or when the diluent and/or rate is not standardized. Ready-to-use formulations recommended by ISMP and IDSA for OPAT are likely more preferred by nurses.1,4,48 Data highlighting physician satisfaction with IVP are even more lacking. To our knowledge, there is no research specifically addressing this topic, however, there are publications authored by physicians both supporting and discouraging the use of IVP antimicrobials in the emergency setting. Those in favor seem to support the improvements in workflow efficiency and time to first dose for sepsis. 49 Skeptics discuss the potential safety risks and insist that the significance of such benefits have not been shown to improve clinical outcomes. 50 Information on pharmacist satisfaction with IVP antibiotics is unavailable. More research is needed to assess the impact of IVP administration on the healthcare team, particularly in settings outside of the ED.
Pharmacoeconomic considerations
Home-based administration
The basic direct and indirect costs of IV antimicrobials have been grouped into four main categories: (i) drug acquisition costs, (ii) costs of additional materials used, (iii) costs related to the use of medical and nursing staff, and (iv) indirect costs such as overheads, storage space, and electricity used. 51 Potential basic OPAT costs with IVP and IV intermittent infusions are outlined in Table 3. Notable possible cost advantages to IVP antimicrobials when given in the home are decreased materials used, less drug preparation time for pharmacy and nurse/patient/caregiver, and lower shipping costs. Conversely, if there is a higher rate of adverse drug reactions with an IVP drug, such as phlebitis or pruritus, then a greater indirect management cost could be incurred.
Basic costs.
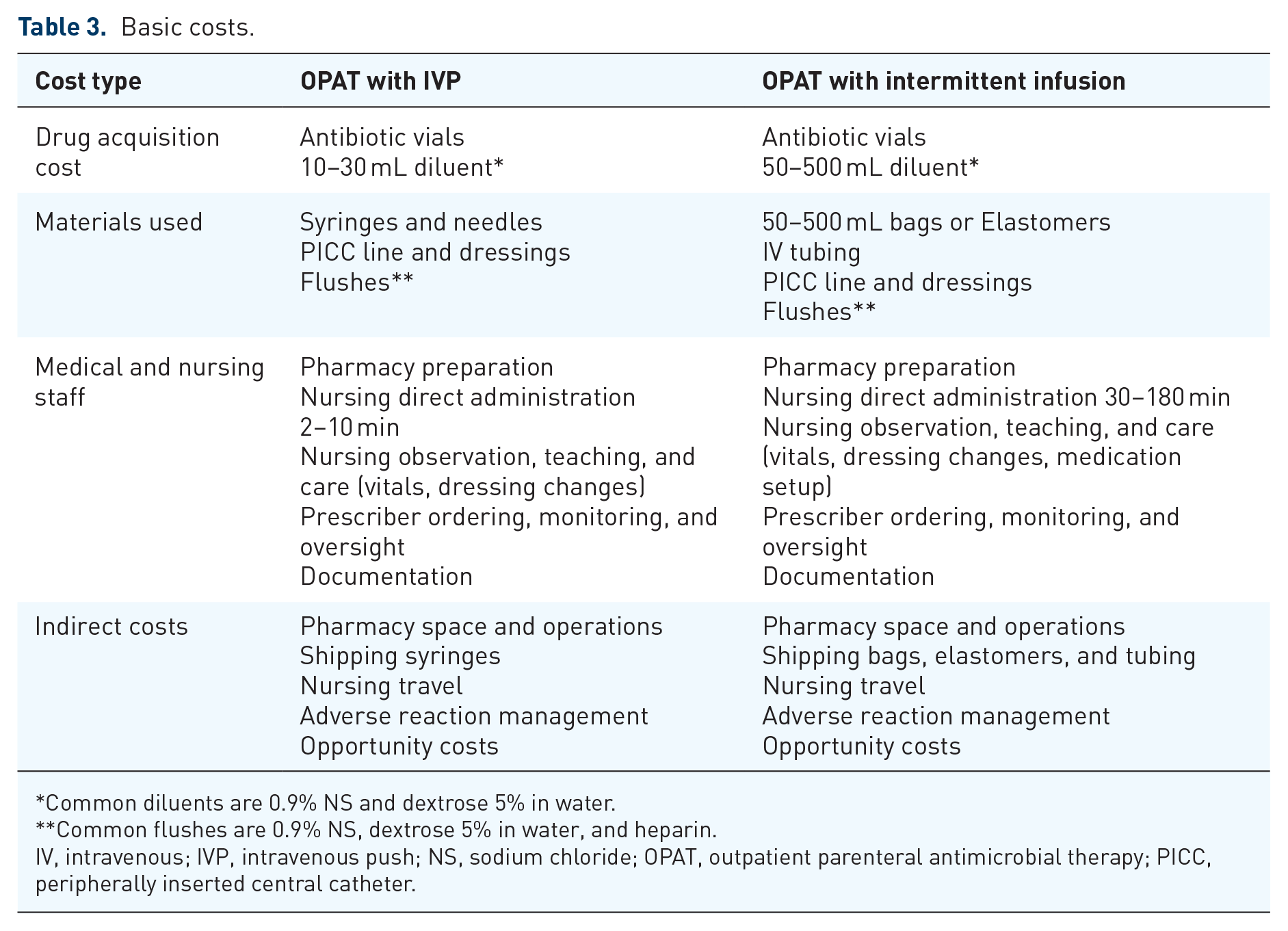
Common diluents are 0.9% NS and dextrose 5% in water.
Common flushes are 0.9% NS, dextrose 5% in water, and heparin.
IV, intravenous; IVP, intravenous push; NS, sodium chloride; OPAT, outpatient parenteral antimicrobial therapy; PICC, peripherally inserted central catheter.
One United Kingdom-based study compared costs of different OPAT drug administration models to an equivalent duration inpatient stay stratified by the infectious syndromes treated. 52 An outpatient clinic model, nurse home visits, self (or caregiver)-administration by IVP, self-administration by a commercially prefilled elastomeric device, continuous IV infusion of piperacillin-tazobactam or flucloxacillin with elastomeric device once daily and oral antibiotics were all found to be less costly than an inpatient hospital stay. Staff time and antimicrobial medications were the primary OPAT costs. Savings extent differed by OPAT healthcare delivery models and infusion methods. Overall, IVP OPAT costs were consistently the lowest among the IV antibiotic strategies across infectious syndromes, both relative to an inpatient stay and to other OPAT administration strategies.
In a previously cited study of self-administered OPAT for endocarditis and osteoarticular infections, the shift from IV infusion by gravity to IVP resulted in savings of US$43,652 over 6 months from a reduction in infusion supplies and direct drug costs, while the related reduced length of stay avoided an additional US$550,000 in costs over the same period. 28
Thus, while limited, the available evidence supports lowest costs with IVP for common antimicrobials and infectious syndromes treated with OPAT in the home.
Facility-based administration
The most common subacute care settings where OPAT medications are given by a healthcare professional include ITCs, EDs, SNFs, and LTCFs. In these care-delivery models, antimicrobials are prepared in IV bags or IV syringes by an on-site or contracted pharmacy, delivered to facility staff, then administratered to the patient. A facility-provided drug model tends to align with a hospital drug model and, in settings such as the ED or certain ITCs, an acute care hospital pharmacy supplies the OPAT medication. Thus, for facility-based OPAT, the discussed pharmacoeconomic studies involve hospital dispensing of antimicrobials.
Pharmacoeconomic exploration of IVP-administered drugs dates back to the 1980s, with a sentinel publication examining the material and labor costs associated with preparing admixtures in mini-bags compared to polypropylene syringes in Canadian hospitals. 53 Medication syringes were diluted and administered as IV admixtures with standard volume-control sets. There was a major cost advantage and nursing preference for the syringe-based method. 53 In 1992, the safety, timing, and cost-effectiveness of IVP antimicrobials was compared to IVPB for surgical prophylaxis. 54 Drug preparation and nursing administration times were less with the IVP method, resulting in a cost avoidance of US$0.60 per dose. Material cost of the mini-bag and IV tubing was avoided at US$3.25 per dose. The authors concluded antimicrobials given via IVP were both safe and cost-effective, holding potential institutional cost savings of US$184,000 per year. 54
Two contemporary reports set in the ED have further supported cost savings using IVP antimicrobials. A ‘first-dose IVP’ protocol implemented in a 73-bed ED eliminated the need for an infusion pump, setup, and infusion documentation. The cost to administer IVP antibiotics was US$0.83 compared with US$9.53 for traditional IV infusion. 47 In a single-center pre-(IV infusion)-post (IVP)-study with a total of 696 administrations of ampicillin/sulbactam, piperacillin/tazobactam, and ertapenem given in an ED from 2015 to 2018, the supply acquisition cost savings totaled more than US$5,000 with IVP antimicrobials. 29 These results have been replicated in a multihospital health system, pediatric setting, and with IVP administration of non-antimicrobial medications as well.55 –57
Robust pharmacoeconomic analyses specific to OPAT in the facility-administered setting are much needed. This available basic expenditure-related data indicate IVP antimicrobials are associated with less cost, particularly from supplies, and less pharmacy and nursing personnel time compared to intermittent IV infusions.
Conclusion
When compared to other OPAT administration methods, IVP offers a simpler infusion technique that uses fewer supplies and takes less administration time. Consequently, IVP requires less time for patient training, improves patient and nursing satisfaction, and decreases overall costs to the healthcare system. Future research is needed to better understand the optimal physiochemical characteristics for IVP solutions, as well as multidisciplinary team satisfaction with this method of administration. For antimicrobials with suitable stability, safety, and effectiveness data, IVP should be the preferred administration method in the OPAT setting.
