Abstract
Objective:
Emerging infectious diseases challenge healthcare systems to implement new models of care. We aim to evaluate the rapid implementation of a new care model for monkeypox in our health system.
Design:
This is a retrospective case series evaluation under the Reach, Effectiveness, Adoption, Implementation, and Maintenance (RE-AIM) framework of implementation of a testing and care model for monkeypox in a large, integrated health system.
Methods:
Atrium Health implemented education of providers, testing protocols, and management of potential monkeypox cases using electronic health record (EHR) data capabilities, telehealth, and collaboration between multiple disciplines. The first 4 weeks of care model implementation were evaluated under the RE-AIM framework.
Results:
One hundred fifty-three patients were tested for monkeypox by 117 unique providers at urgent care, emergency departments, and infectious disease clinics in our healthcare system between 18 July 2022 and 14 August 2022. Fifty-eight monkeypox cases were identified, compared with 198 cases in the state during the time period, a disproportionate number compared with the health system service area, and 52 patients were assessed for need for tecovirimat treatment. The number of tests performed and providers sending tests increased during the study period.
Conclusion:
Implementation of a dedicated care model leveraging EHR data support, telehealth, and cross-disciplinary collaboration led to more effective identification and management of emerging infectious diseases and is important for public health.
Plain Language Summary
New infectious diseases challenge health systems to implement new care practices. Our health system responded to this challenge by implementing a care model for education, testing, and clinical care of monkeypox patients. We analyzed results from implementing the model. We were able to identify a disproportionate number of monkeypox cases compared with the rest of our state by using our model to educate medical providers, encourage testing, and ensure patients had access to best disease care. Implementation of care models for testing and management of new diseases will improve patient care and public health.
Background
Healthcare systems have been challenged by recent emerging infectious diseases to develop and implement new models of care. 1 With the coronavirus disease 2019 (COVID-19) pandemic, our healthcare system and others had to rapidly scale up diagnostic testing, vaccination, and treatment capabilities which included increased utilization of telehealth.2,3 Our health system and others used telehealth during the COVID-19 pandemic to screen all patients testing positive for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) for potential treatment with monoclonal antibody therapy. 4 We implemented a protocol similar to published studies where electronic health record (EHR) information was used to identify those needing treatment among all patients tested over a wide geographic area at multiple locations within the healthcare system. 4 Telehealth visits were then used to assess and consent patients for treatment prior to referral to an infusion treatment center. Recently, we translated those lessons learned to implement a similar testing, assessment, and treatment strategy for monkeypox within our healthcare system.
Monkeypox is a zoonotic infectious disease with a disease outbreak starting in Europe in May 2022.5,6 Currently, more than 30,000 cases in 89 nations worldwide have been reported since 1 January 2022.7,8 Described cases to date have primarily been in men who have sex with men (MSM) (98%) with 41% occurring in people living with HIV (PLwH).9,10 North Carolina, where our healthcare system is primarily located, identified its first case on 23 June 2022. 11 As an emerging infectious disease, most clinicians are unfamiliar with monkeypox’s clinical presentation, which at times has been atypical from previously published literature and may mimic other diseases such as herpes viruses.9,12 We identified a need to educate providers on appropriate testing and scale up testing capabilities across our healthcare system, particularly as delays in recognition and testing of cases could lead to further spread without appropriate case isolation. 10 We also identified a need to assess all patients tested for treatment with tecovirimat which is available for patients with severe disease. To meet this challenge, our healthcare system implemented an innovative care model to encourage testing and assess patients for treatment over a geographically dispersed area of an integrated healthcare system using healthcare information technology and telehealth.
Here, we evaluate implementation of our healthcare system’s monkeypox care model based on the Reach, Effectiveness, Adoption, Implementation, and Maintenance (RE-AIM) framework. The RE-AIM framework is an established method for evaluation of multilevel interventions with public health implications such as our protocol. 13 By examining implementation of our intervention, we hope to further refine and develop a sustainable model which can be implemented on a broader scale to address monkeypox and other new infectious disease outbreaks which may occur. 14
Methods
Setting
Atrium Health is a large, integrated healthcare system with 12 hospitals, 5 free-standing emergency departments, and 44 urgent cares across a metropolitan area. Across our health system, approximately 57% of patients identify as female, 19% African American, and 70% non-Hispanic White. The payor mix is 16% Medicare, 15% Medicaid, 49% commercial, and 20% self-pay. Our healthcare system also has two clinics to care for PLwH, providing HIV care to more than 3800 patients and receives Ryan White funding. In addition, our health system provides HIV Pre-exposure prophylaxis (PrEP) services and has dedicated programs for diagnosis and early linkage to care of individuals newly diagnosed with HIV in its emergency departments.
Development of monkeypox care model
Participants
Two distinct institutional task forces were established to respond to the monkeypox epidemic. One consisted of infectious disease physicians with expertise in epidemiology, antimicrobial stewardship, and HIV care. The second larger task force included collaborators from urgent care and emergency department leadership, healthcare system administration, pharmacy, laboratory services, employee health, ambulatory clinic leadership, information services, and corporate communications. Both groups met at least weekly to develop and implement the care model. Barriers to implementing the care model were identified and key stakeholders engaged to move the project forward (see Figure 1).
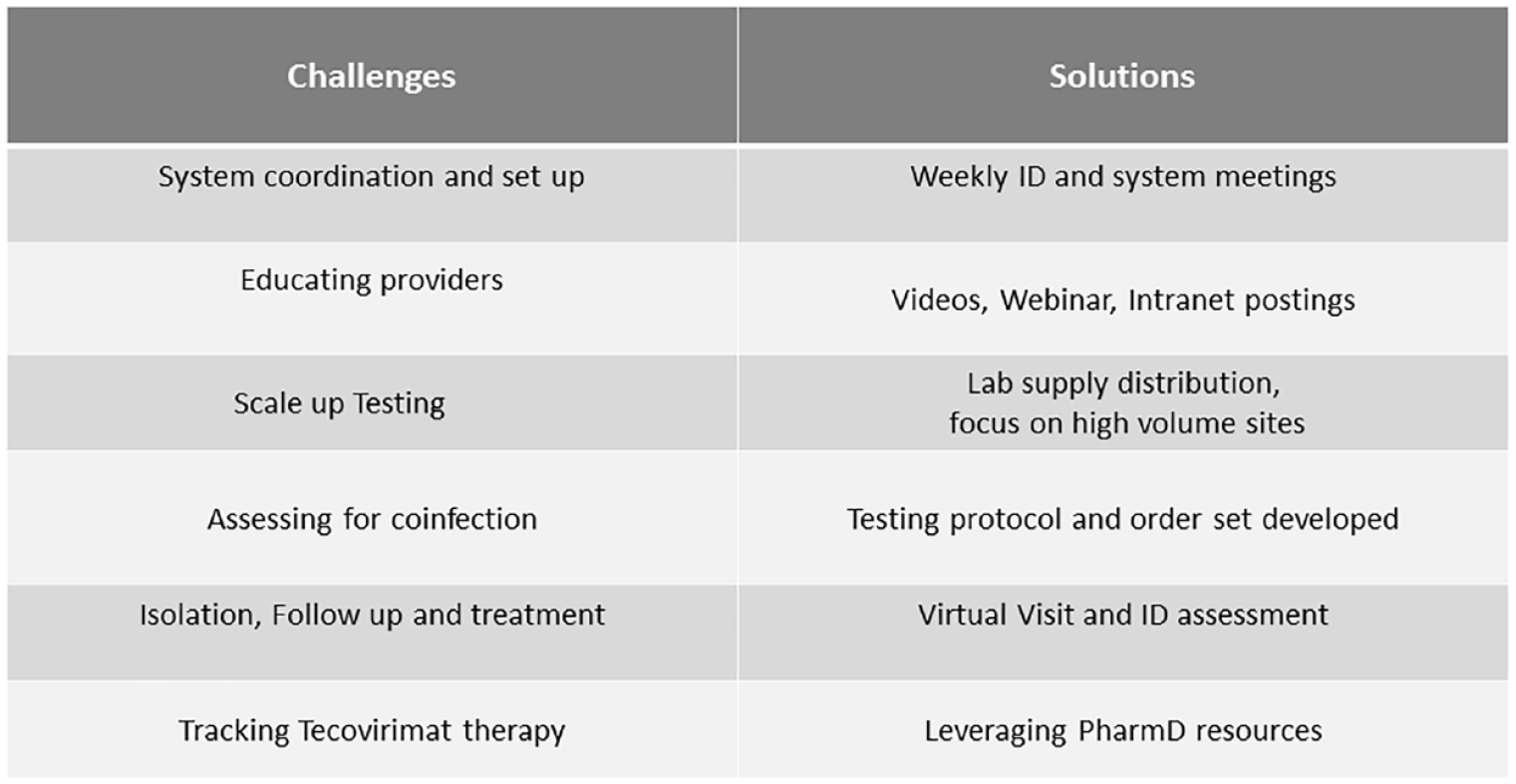
Challenges and solutions in developing a health system response to monkeypox outbreak.
Pre-implementation planning
A testing protocol was developed by the infectious disease team to guide decisions on whom to test and appropriate additional diagnostic workup including assessment for sexually transmitted coinfections. Initially, monkeypox testing required approval of hospital epidemiology in consultation with the state health department, but this requirement ceased on 22 July 2022 with transition of testing to a commercial laboratory. Patients tested had swabs sent for orthopox polymerase chain reaction (PCR) from two separate unroofed skin lesion sites in accordance with state and Centers for Disease Control and Prevention (CDC) guidance at the time of protocol initiation, except in cases where in consultation with an infectious disease physician, a swab was sent from an alternative site. An EHR order set was built to standardize ordering of orthopox and sexually transmitted infection (STI) testing. The order set was available regardless of testing location (e.g. emergency department, urgent care, or ambulatory clinic). Laboratory testing supplies were deployed to locations dispersed over the geographic reach of the healthcare system where at-risk patients were considered most likely to seek care: HIV clinics, emergency departments, and urgent care sites. Significant focus was placed on meeting the challenge of educating more than 120 emergency department and urgent care providers on both disease recognition and test collection method.
Education was provided to providers at these sites on clinical presentation, testing criteria, order sets, and methods of testing with videos, webinars, and information posted on a dedicated institutional monkeypox intranet site.
Patients sent for testing were tracked in a healthcare system-wide database populated from EHR data. Those identified with positive orthopox virus testing were automatically flagged in an EHR report for phone contact by an infectious disease Advanced Practice Provider (APP). Patients were assessed and offered a virtual or in-person visit with a physician within 24 h of test results to further assess disease severity and need for treatment. Patients were also counseled on appropriate isolation and follow-up requirements. Patients with severe disease or immunosuppression qualifying for treatment were offered a subsequent clinic visit to initiate tecovirimat antiviral therapy (see Figure 2).

Process map for testing and treatment of monkeypox across a healthcare system.
Program evaluation
Data collection
Data including demographics, care locations, coinfection including HIV status, epidemiologic history, clinical course, and outcomes were collected from the EHR to describe patients who received care through our health system. Treatment information was also tracked. All healthcare system patients tested for orthopox were included. Patients were followed up through their last healthcare system contact relevant to monkeypox evaluation including receipt of a negative test result, reassessment of symptoms for consideration of treatment, or 21 days following tecovirimat therapy. Wake Forest University School of Medicine institutional review board approval was obtained to evaluate and analyze data from the monkeypox care model based on the RE-AIM framework with waiver of informed consent. For the analysis, all patients starting from when our care model implementation was initiated on 18 July 2022 and tested through 14 August 2022 were included.
Reach
Reach was defined as the number of patients tested within the healthcare system.
Effectiveness
Effectiveness was defined as positive cases identified as percentage of the estimated at-risk population in the geographic service area of our healthcare system as compared with North Carolina state data. Many cases of monkeypox to date have been described with mild or atypical presentations, such that the disease may be underrecognized in the community with secondary spread.11,14 This could result in a lower case ascertainment rate, where the reported number of cases is an underestimate of the true number of infections in the community. 14 Effectively implementing a care model to encourage recognition and testing of cases should improve case recognition. Currently, the monkeypox outbreak is concentrated in MSM populations with 98% of published cases in 2022 in MSM or bisexual males.7,9 Drawing on experience using social determinants of health to focus healthcare testing, previous work estimated 15,700 sexually active MSM in North Carolina, 3632 in the 11 counties served by our health system (see Figure 3), who would be at significant risk of monkeypox acquisition.15,16 We postulated that through implementation of our testing protocol, we will identify a higher percentage of positive cases from the estimated at-risk population in our geographic service area compared with other areas of North Carolina without this intervention.
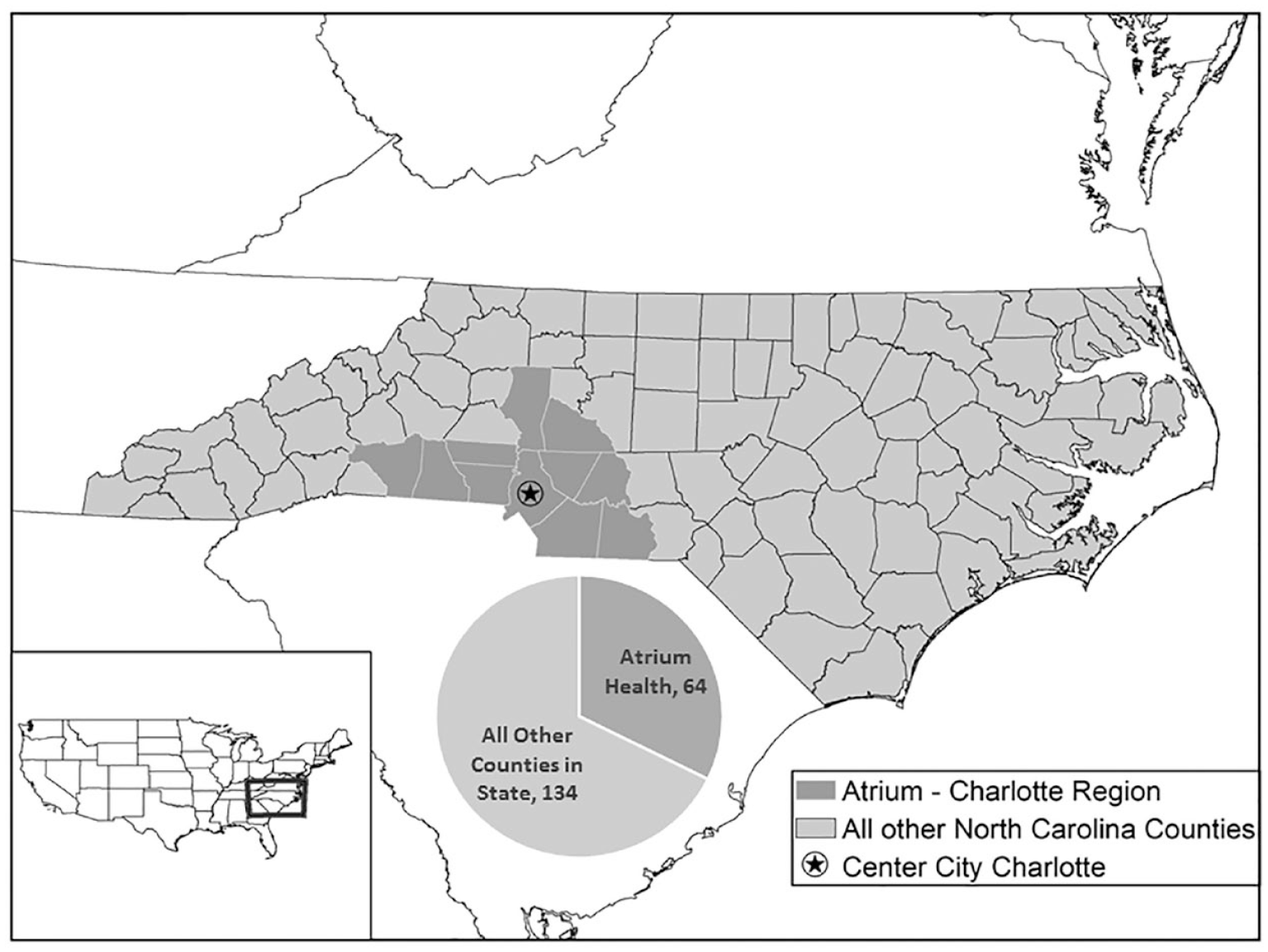
Healthcare system monkeypox case rates compared with remainder of state.
Adoption
Adoption was measured as the number of unique providers at different sites within the healthcare system which tested patients.
Implementation
Implementation was evaluated as the fidelity to completing all key components of our care protocol. Specifically, this was defined as (1) completion of an assessment visit by an infectious disease provider after receiving a positive orthopox test result and (2) assessment for other STIs including HIV; herpes simplex virus (HSV); syphilis; and urine, rectal, and pharyngeal chlamydia and gonorrhea. Assessment for other STIs was deemed important because of the high prevalence of coinfections in published cohorts with monkeypox. 9
Maintenance
Maintenance was defined as increasing capacity of the program as case numbers escalated. The number of orthopox tests sent and provider visits completed weekly for testing, treatment assessment, and dispensing of treatment was tracked following protocol implementation.
Statistical analysis
For analysis of data variables, means and frequencies were calculated. The Mann–Whitney U test was used to compare median values between groups and Chi-square tests were used to compare categorical values for cases and controls. A value of p ⩽ 0.05 was defined as statistically significant. A z test with Yates continuity correction was used for comparisons of proportions.
Results
Reach
Our care model began implementation on 18 July 2022. Over the four weeks of study between 18 July and 14 August 2022, there were 153 individuals tested for monkeypox within the health system with 58 positive cases. Prior to implementation of the care protocol on 18 July, only eight patients were tested within the healthcare system. Tests were sent from two skin lesions in each patient per protocol except in cases when sent from conjunctiva (n = 1), throat (n = 1), and rectum (n = 1).
Cases with monkeypox were all male or transgender women and more likely to be African American, MSM, and PLwH (p < 0.00001, see Table 1). Reports of exposure to an individual with monkeypox were not predictive of testing positive [p = not significant (NS), see Table 1]. Clinical presentation with prodrome of fever or lymphadenopathy or proctitis was more common in cases compared with those testing negative (p < 0.01), and most patients presented with fewer than 10 lesions (see Table 1).
Characteristics of individuals tested for and cases of monkeypox.

MSM, men who have sex with men; NS, not significant; PLwH, people living with HIV.
Three individuals had inconclusive tests but quickly resolved symptoms prior to retesting and retrospectively were not thought to be positive so included as negative.
Effectiveness
Including 6 cases identified prior to protocol implementation, our health system identified 64 individuals testing positive with monkeypox. These 64 cases were from an estimated at-risk population of 3632 sexually active MSMs in the health system geographic service area or 18 cases per 1000 at-risk individuals. 14 Comparatively, there were a total of 198 individuals identified in North Carolina with monkeypox by the end of the study from an estimated at-risk population of 15,700 sexually active MSMs or 13 cases per 1000 individuals (see Figure 3).11,14 This would indicate we detected an average of 6.5 more cases per 1000 individuals from the at-risk population compared with cohorts outside of our health system in our state [95% confidence interval (CI), 1.7–11, p = 0.003].
Adoption
Testing was sent by 117 unique providers during the study of 237 providers who work in our healthcare system’s emergency departments, urgent cares, and HIV clinics as testing sites. The largest volume of testing was from emergency departments and urgent cares with 75 (49%) and 71 (46%) tests sent, respectively; 6 tests (4%) were sent from the infectious disease clinic, and 1 patient tested during hospital admission.
Implementation
Fifty-two (90%) of 58 patients who tested positive completed an assessment visit with an infectious disease provider to assess severity of disease and need for tecovirimat therapy. Patients not completing a visit were unable to be reached in follow-up despite multiple attempts. The majority of, but not all, patients were also tested for some other STIs, but compliance with order set recommendations for pharyngeal and rectal gonorrhea and chlamydia testing was low (see Table 2). Coinfection with syphilis was identified in 22% (n = 13) of cases, with chlamydia and gonorrhea in one case each, and three patients of all those tested were newly diagnosed with HIV infection (see Table 2).
Testing and case of STI coinfection.

HSV, herpes simplex virus; PLwH, people living with HIV; STI, sexually transmitted infection.
Forty-eight known PLwH were tested, with 39 PLwH positive for monkeypox.
Positive for chlamydia not gonorrhea.
Positive for both chlamydia and gonorrhea.
Positive for chlamydia not gonorrhea.
Maintenance
Case rates, the number of tests performed, and provider visits completed increased after 1 week of protocol implementation. During week 1, 14 tests were performed and 9 cases were identified. In the subsequent 3 weeks, 30, 56, and 53 tests were performed and 18, 14, and 17 monkeypox cases were identified. All patients had subsequent infectious disease follow-up with a telephone encounter, video, or in-person visit except the six patients who were unable to be contacted. Ten patients were identified as requiring and treated with tecovirimat over the course of the study (see Table 3).
Testing and provider visits by week.

Discussion
We were able to use the strengths of an integrated health system to rapidly implement a diagnosis and care model for an emerging infection in context of a global outbreak. We were able to reach patients over a broad geographic area by providing system-wide education and focusing on implementing testing in emergency departments and urgent cares as the highest value sites where a large volume of STI testing was already performed. The success of our care model is demonstrated in the higher percentage of at-risk individuals identified with monkeypox in our health care system’s geographic service compared with North Carolina state data (18 versus 13 per 1000 at-risk individuals). Additional cases were identified by other health care entities within our service area as well, so our case rate detection was proportionally higher than expected. While it is certainly possible regional variances in disease prevalence may have contributed to this difference, we think failures at disease recognition and missed testing opportunities explain a large portion of the case rate variance. This is particularly likely given emerging data on atypical presentation of monkeypox during the 2022 global outbreak compared with prior documented outbreaks and our data revealing most patients had less than 10 lesions at presentation.17,18 Also notably, reports of exposure to an individual with monkeypox were not predictive of testing positive suggesting circulation of unrecognized cases in the community. Notably incidence of monkeypox case peaked nationally during the time of our implementation study but did not peak in North Carolina until 1–2 weeks following our study. 7 Whether delays in familiarity with case finding affected these peaks is uncertain. This demonstrates the importance of a systems approach to implementing testing for emerging infections where clinicians may be less familiar with the clinical presentation. By taking early action to engage key stakeholders across multiple disciplines and creation of a care model, we were able to drive change and implement best practice care more quickly, and importantly for identification of cases for public health purposes, although admittedly it is impossible to determine how many cases were missed even with our dedicated protocol and higher case detection rate.
Our higher case ascertainment rate might also help explain some of the race disparities we observed in our cohort compared with other published monkeypox cohorts. Individuals we identified with monkeypox were disproportionately African American compared with those testing negative, and African Americans were represented at higher rates than in our health system overall patient mix (19%) and compared with North Carolina state data where 68% (n = 134) of monkeypox cases were African American. 11 African Americans are less likely to present for health care and are known to be disproportionally affected by emerging infectious diseases.19,20 African Americans are also disproportionally affected by HIV, especially in the US South, where our healthcare system is located in one of the CDC’s 48 highest priority counties for addressing the HIV epidemic.21–23 PLwH made up a higher percentage (74%) of our monkeypox case compared with published cohorts (41%) which overlaid on the racial disparity.7,9 Extending testing and care into communities with historic health disparities will be critical for case detection and control of the current monkeypox outbreak, given the need to identify and isolate cases to prevent spread. While a limitation of our work is our inability to specifically develop outreach interventions to African American or minority communities as part of this implementation project, focused work on improving testing and care in specifically vulnerable communities represents an opportunity for further work.
Through implementation of our care model, we were also able to ensure patients with severe disease or those at risk due to immunosuppression received therapy and could access treatment. During the study, 10 of the 58 (17%) patients were prescribed tecovirimat for severe disease, almost all (n = 9) were PLwH. Anecdotally, access to tecovirimat was severely limited in our region during the time frame of the study, with no other local providers providing treatment during the first weeks of our study. Access to treatment was particularly important in our population, given our high percentage (71%) who were PLwH at high risk of severe disease outcomes.
Our care model was built on lessons learned from the COVID-19 pandemic with centralized easily accessible education tools for clinicians, system messaging on best practice protocols, and deployment of EHR order sets to efficiently manage appropriate care. Leveraging the EHR, we were able to quickly identify and perform targeted outreach to affected patients and provide comprehensive care including using telehealth as an increasingly utilized care modality. Use of telehealth visits was well suited for care in this context as an efficient use of resources limiting contact time and reducing isolation needs for an infectious disease. All these tools will likely continue to be important in developing care models for future emerging infections and managing public health needs. Based on the success of our care model using a multidisciplinary team approach, a similar protocol has now been implemented for testing and education within our healthcare systems’ locations in other states.
Multiple barriers were encountered during implementation of our care model, one of which was limited access to testing which initially was conducted solely at the state microbiology lab and required a time-consuming approval process. We were able to quickly move to a commercial lab to improve capacity. We continue to be challenged by long turn-around time for testing results even from the commercial lab. Education of clinicians on how to perform testing was also required and managed with instructions and a video posted to the health system intranet site as well as availability of assistance from an on-call epidemiologist, which will need to continue to be updated as the science evolves with new testing and treatment guidance. The visit for initiation of treatment with tecovirimat required a significant amount of time, given current CDC requirements for prescription. We leveraged an infectious disease clinical pharmacist from our antimicrobial stewardship program to manage this process and who could ensure documentation compliance across multiple clinic providers.
Limitations in our care model include fidelity to providers sending associated STI testing and scalability. Despite building testing for associated STIs into an order set, few patients received the full recommended panel of tests. This may be partially explained by delayed roll out of the full EHR order set until 5 August, but compliance with testing remained suboptimal even after this date and likely reflects poor baseline rates of completion of appropriate STI testing. For patients who were later seen in person at the infectious disease clinics, STI testing could be completed on-site, but this was the minority (n = 18) of those with missing tests so diagnosis of other STIs could have been missed. Improved STI testing is clearly an opportunity for improving public health through our health system in the future. Testing for STIs and HIV should be a priority at time of monkeypox testing, given demonstrated rate of coinfection observed in studied cohorts. We were able to leverage our health system resources to maintain care under the model even as case rates increased, but other health systems or smaller community practice sites may not have resources to implement our care model. Due to limited supply of available vaccines, we were unable to integrate the vaccination approach in our current care model. However, we anticipate that this will become an important role in controlling the ongoing outbreak by vaccination of contacts and high-risk individuals.
As new infectious diseases continue to emerge, rapidly developing care models to facilitate diagnosis, management, and treatment will be important. Here, we demonstrate the feasibility and efficacy of implementing a care model for a new infectious disease. Given the disproportionate number of monkeypox cases diagnosed within our healthcare system compared with our state likely indicating a higher case ascertainment rate, our success in implementing multidisciplinary care demonstrates the need to develop and implement care models to quickly scale up best practice care. This may also be important in addressing disparities in health care and improving public health.
Footnotes
Acknowledgements
The authors wish to thank Christopher Hoang in helping implement the care protocol in urgent care locations and developing documentation and data tracking, Jessica Kearney-Bryan for invaluable assistance in regulatory work, Kaylee Smith for implementing laboratory testing, and Jennifer Terry for leadership in system implementation.
