Abstract
SARS-CoV-2 may activate both innate and adaptive immune responses ultimately leading to a dysregulated immune response prompting the use of immunomodulatory therapy. Although viral pneumonia increases the risk of invasive fungal infections, it remains unclear whether SARS-CoV-2 infection, immunomodulatory therapy, or a combination of both are responsible for the increased recognition of opportunistic infections in COVID-19 patients. Cases of cryptococcosis have previously been reported following treatment with corticosteroids, interleukin (IL)-6 inhibitors, and Janus kinase (JAK) inhibitors, for patients with autoimmune diseases, but their effect on the immunologic response in patients with COVID-19 remains unknown. Herein, we present the case of a patient with COVID-19 who received high-dose corticosteroids and was later found to have cryptococcosis despite no traditional risk factors. As our case and previous cases of cryptococcosis in patients with COVID-19 demonstrate, clinicians must be suspicious of cryptococcosis in COVID-19 patients who clinically deteriorate following treatment with immunomodulatory therapies.
Introduction
SARS-CoV-2 can activate both innate and adaptive immune responses in patients ultimately leading to a dysregulated immune response. 1 Due to the widespread use of immunomodulatory therapy, including high-dose corticosteroids, interleukin (IL)-1 and IL-6 inhibitors, as well as Janus kinase (JAK) inhibitors, as part of the medical management of COVID-19,2,3 an increasing number of patients may experience an impaired immune response. 4 Consequently, compromising host immunity in COVID-19 patients could provide the perfect opportunity for secondary infections with or reactivation of previously latent diseases, such as latent tuberculosis infections, strongyloidiasis, aspergillosis, mucormycosis, or cryptococcosis.
Specifically, Cryptococcus neoformans is frequently associated with advanced HIV disease manifesting as meningoencephalitis, but rates have been decreasing among persons living with HIV (PLH) due to increased uptake of antiretroviral therapy (ART). 5 However, an increasing number of cases of cryptococcosis have been identified among new populations of patients with cell-mediated immunodeficiencies, including solid organ transplant (SOT) recipients and non-HIV-infected, nontransplant patients with malignancies, autoimmune diseases, diabetes mellitus, cirrhosis, as well as those receiving immunosuppressive medications.6–10 Herein, we present the case of a patient with COVID-19 who received immunomodulatory therapy with high-dose corticosteroids and was later found to have cryptococcosis.
Patient case
A man in his early 70s, who was a nursing home resident, was sent to an outside hospital (OSH) in the Southeastern United States in late November 2020, due to hypoxia following a recent diagnosis of SARS-CoV-2 infection. His past medical history included a cerebrovascular accident resulting in left hemiplegia and hemiparesis, idiopathic peripheral autonomic neuropathy, spinal stenosis with bilateral lower extremity radiculopathy, atrial fibrillation, hypertension, coronary artery disease, dyslipidemia, chronic obstructive pulmonary disease, gastroesophageal reflux disease, stage 3 chronic kidney disease (CKD), benign prostatic hypertrophy, obesity, major depressive disorder, and vascular dementia for which he took amlodipine, aspirin, atorvastatin, dabigatran, donepezil, famotidine, memantine, metoprolol tartrate, omeprazole, pregabalin, tamsulosin, and valproic acid. On admission, he complained of fevers, shortness of breath, nonproductive cough, loose stools, and anorexia, but denied headache, visual disturbance, hemoptysis, arthralgia, myalgia, nausea, vomiting, or rashes. His O2 saturation was 91% on room air and computed tomography (CT) of the chest revealed multifocal infiltrates with a ground-glass appearance consistent with COVID-19. Baseline chemistry and hematology laboratory values revealed leukopenia and lymphopenia but were otherwise unremarkable and reflected his underlying CKD (Figure 1). He was provided supplemental oxygen via nasal cannula and treated with ceftriaxone and azithromycin for 7 days. Although he was deemed not to be a candidate for remdesivir at that time due to CKD, he received dexamethasone for 10 days in combination with convalescent plasma. Blood cultures and sputum cultures were collected on admission but remained sterile.

Timeline of hospitalization.
Over the next 10 days, he remained afebrile, but his respiratory function continued to deteriorate on nasal cannula, and he was transitioned to nonrebreather (NRB) mask. The previously identified multifocal infiltrates persisted on follow-up chest radiography (CXR). Due to decreased O2 saturation to 70% and bluish-gray appearance, the patient required intubation, mechanical ventilation, and initiation of vasopressors, which prompted a transfer to our institution.
Upon arrival, he was admitted to the intensive care unit (ICU), where he remained sedated, mechanically ventilated, and on vasopressors. He was subsequently started on remdesivir for 5 days, in addition to IV methylprednisolone 60 mg IV every 8 h for 2 days followed by a gradual taper throughout his hospitalization. His initial blood and urine cultures were sterile, but multiple tracheal aspirate cultures revealed methicillin-resistant Staphylococcus aureus. Despite an unchanged CXR and successful extubation, he was started on IV vancomycin on day 3, but was transitioned to linezolid after experiencing an acute kidney injury for a combined total of 16 days.
The patient continued to improve and was transferred out of the ICU on a high-flow nasal cannula over the next 4 days, but became febrile and experienced hypoxia, lethargy, as well as hypotension requiring intubation and mechanical ventilation with a subsequent transfer back to ICU. Blood cultures revealed yeast on Gram stain, and he was started on micafungin. Unfortunately, the patient’s blood pressure and respiratory function continued to deteriorate over the next 24 h, and he expired. Post-mortem, the previously detected yeasts grew on Sabouraud Dextrose Agar (Emmons modification) which were identified via Thermo Scientific™ RapID™ Yeast Plus System (RapID; Thermo Fisher Scientific, Lenexa, Kansas) as C. neoformans. Molecular identification and antifungal susceptibility testing were not performed due to limited capabilities in the local microbiology laboratory. Serum cryptococcal antigen (CrAg) lateral flow assay (LFA) was also not performed due to low suspicion of cryptococcosis in this patient and limited data describing cryptococcosis in patients with COVID-19 at the time of this patient’s care.
Discussion
There is increasing recognition of opportunistic infections (OIs) in COVID-19 patients with underlying medical conditions such as uncontrolled diabetes mellitus.11,12 Although the precise mechanism remains unknown, the most likely pathophysiology is multifactorial involving cytokine dysregulation and impaired cell-mediated immunity due to SARS-CoV-2 combined with receipt of immunomodulatory therapies as part of the therapeutic armamentarium for COVID-19. The most frequently identified OIs include invasive pulmonary aspergillosis and mucormycosis. Clinical presentation varies depending on organ involvement and overlap with COVID-19, but often develops 1 to 2 weeks after hospitalization.13,14 Advanced age, chronic pulmonary disease, and treatment with antimicrobial therapy were identified as risk factors for coronavirus disease-associated pulmonary aspergillosis (CAPA),13,15 whereas diabetes mellitus was the most common risk factor among patients with COVID-19-associated mucormycosis (CAM). 14 In addition, many of these patients received immunomodulatory therapies for COVID-19, specifically corticosteroids and IL-6 inhibitors, which likely contributes to the increasing incidence of CAPA and CAM.13–15 As our case demonstrates, cryptococcosis is yet another important opportunistic pathogen that should be considered by clinicians caring for patients with COVID-19 who received immunomodulatory agents.
While cryptococcosis primarily affects immunodeficient patients, an increasing number of cases have been reported in immunocompetent patients, in which some of these patients do not have identifiable risk factors. 9 In most situations, immunocompetent hosts can eradicate C. neoformans after inhalation, but C. neoformans may survive by evading the host immune response and forming a cryptococcal granuloma or residing in phagocytic cells, thereby establishing a latent infection.16,17 Following the development of disease state- (e.g. diabetes mellitus, cirrhosis, sarcoidosis, etc.)6–8,10,18 or medication- (e.g. corticosteroids, chemotherapy, etc.)19,20 induced immune defects in these previously immunocompetent patients, the latent C. neoformans infection can reactivate resulting in detectable cryptococcosis. 21
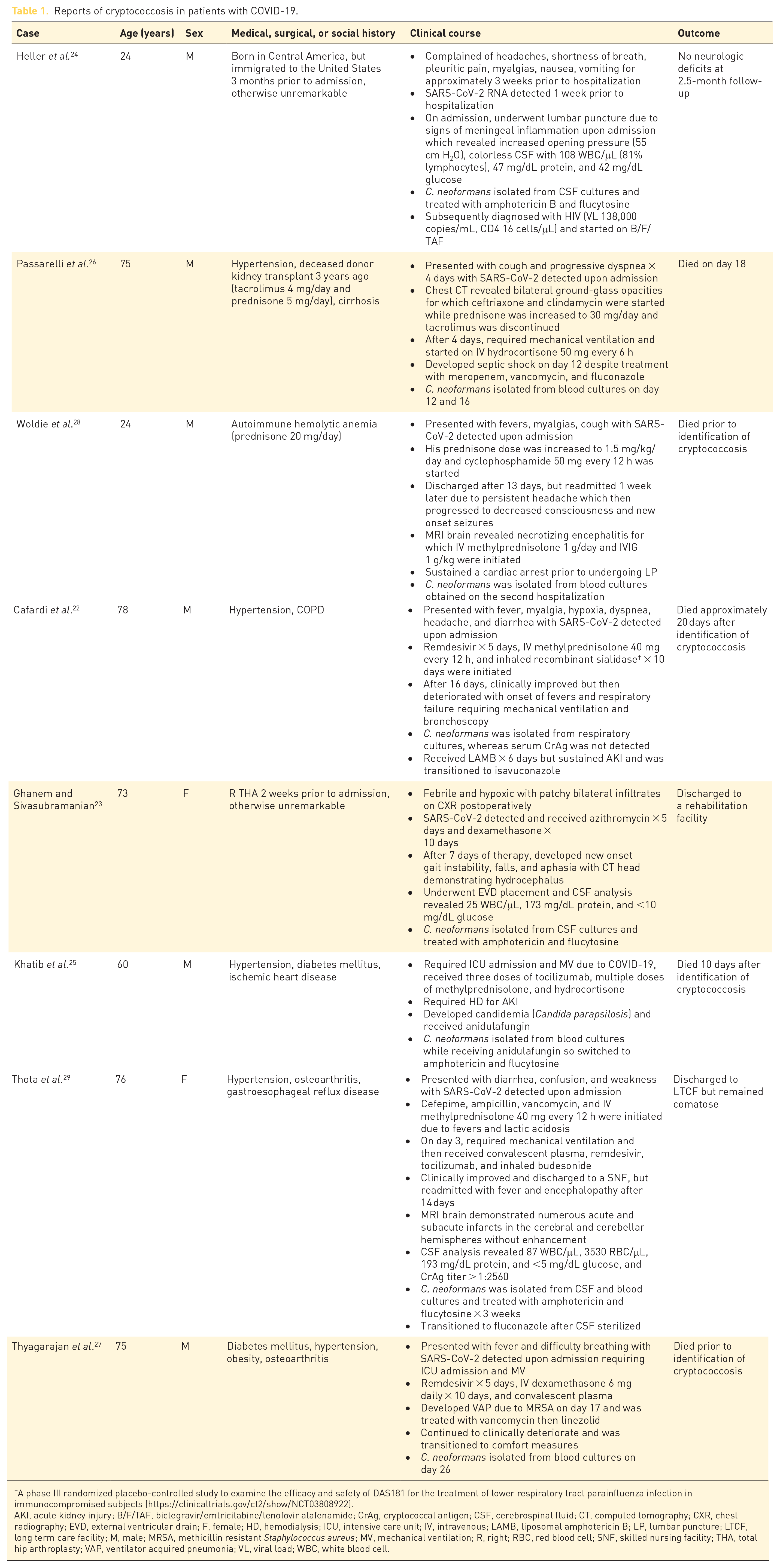
Indeed, eight previous cases of cryptococcosis in patients with COVID-19 have been reported (Table 1)22–29 based on a PubMed literature search on 14 September 2021, using ‘Cryptococcus’ and ‘COVID-19’. Of those reports, the median age was 74 (range: 24–78) years and 80% were men. Seventy-five percent had chronic comorbidities,22,25–29 of which hypertension (83%)22,25–27,29 and diabetes mellitus (33%)25,27 were most common. In addition, two patients were receiving immunosuppressive medications: prednisone for autoimmune hemolytic anemia 28 and tacrolimus in combination with prednisone due to renal transplantation. 30 Characterization of symptoms caused by cryptococcosis was challenging due to overlap with COVID-19. However, due to low suspicion for cryptococcosis many patients clinically deteriorated, some of which died prior to detection of cryptococcosis.
Reports of cryptococcosis in patients with COVID-19.
A phase III randomized placebo-controlled study to examine the efficacy and safety of DAS181 for the treatment of lower respiratory tract parainfluenza infection in immunocompromised subjects (https://clinicaltrials.gov/ct2/show/NCT03808922).
AKI, acute kidney injury; B/F/TAF, bictegravir/emtricitabine/tenofovir alafenamide; CrAg, cryptococcal antigen; CSF, cerebrospinal fluid; CT, computed tomography; CXR, chest radiography; EVD, external ventricular drain; F, female; HD, hemodialysis; ICU, intensive care unit; IV, intravenous; LAMB, liposomal amphotericin B; LP, lumbar puncture; LTCF, long term care facility; M, male; MRSA, methicillin resistant Staphylococcus aureus; MV, mechanical ventilation; R, right; RBC, red blood cell; SNF, skilled nursing facility; THA, total hip arthroplasty; VAP, ventilator acquired pneumonia; VL, viral load; WBC, white blood cell.
During hospitalization, 25% of patients received tocilizumab, and 88% received corticosteroids. In the only case where corticosteroids were not administered for COVID-19, diagnosis of cryptococcosis subsequently led to a new diagnosis of HIV, 24 whereas C. neoformans was most commonly identified after receiving immunomodulatory therapy in all other cases; C. neoformans was isolated from blood cultures in 63% (n = 5) of patients. Of the 38% (n = 3) of patients who underwent cerebrospinal fluid (CSF) evaluation, C. neoformans was detected in all CSF cultures. Overall mortality was 63%, of which two patients died before identification of cryptococcosis.27,28
In four cases,22,23,29 including our patient, no traditional risk factors for cryptococcosis were identified. 9 Notably, our patient had a glycated hemoglobin (A1C) of 5.6% (A1C ⩾ 6.5% diagnostic for diabetes), 31 but did not undergo HIV testing. While viral pneumonia increases the risk of invasive fungal infections, it remains unclear whether SARS-CoV-2 infection, immunomodulatory therapy, or a combination of both were responsible for increased susceptibility to acute infection with or reactivation of C. neoformans in these patients with COVID-19. Although much of the pathogenesis of cryptococcosis in COVID-19 patients is poorly defined due to limited details in other case reports, we suspect our patient had latent cryptococcosis which reactivated because of SARS-CoV-2 infection–induced lymphopenia and corticosteroid treatment. Blood cultures obtained on admission at the OSH were sterile. Based on a rat model of pulmonary cryptococcosis which mirrors human infection, C. neoformans persists as a granuloma within the lung despite initial containment and no clinical symptoms.17,21 Following receipt of corticosteroid therapy, increased fungal burden and extrapulmonary dissemination were observed to a greater extent when corticosteroid therapy was administered within 4 weeks of initial infection with C. neoformans. However, similar findings were not observed with late stages of latent cryptococcosis suggesting multiple mechanisms might be necessary for reactivation, which in this case include SARS-CoV-2 infection–induced lymphopenia combined with prolonged high-dose corticosteroid treatment. Mechanisms to predict and prevent reactivation of latent cryptococcosis are needed given the increasing incidence of immunosuppressive diseases and widespread use of immunosuppressive medications.
Asymptomatic treatment-naïve PLH with CD4 cell counts ⩽100 to 200 cells/µL undergo screening for cryptococcosis using serum cryptococcal antigen (CrAg) testing.32,33 Among those with detectable CrAg, CSF should be evaluated to determine whether the cryptococci have disseminated to the central nervous system (CNS). Early identification and treatment of cryptococcus in PLH with low CD4 cell counts has led to decreased mortality attributable to cryptococcal meningoencephalitis.34–36 Unfortunately, similar recommendations to detect cryptococcal antigenemia and ultimately identify localized pulmonary cryptococcosis or disseminated cryptococcosis do not exist in HIV-uninfected immunodeficient patients nor immunocompetent patients. Diagnosis of cryptococcosis in non-HIV nontransplant patients is often missed or significantly delayed compared to PLH or organ transplant recipients resulting in lower survival rates,37,38 as the sensitivity of CrAg LFA to detect CrAg in serum, while still relatively high, is lower than that in PLH. 39 Unfortunately, the sensitivity of serum CrAg is unknown in COVID-19 patients with an impaired immune response. As such, the risk of dissemination is increased compared to immunocompetent patients, therefore necessitating an evaluation for meningoencephalitis in COVID-19 patients with serologic or microbiologic evidence of cryptococcosis.
An optimal treatment regimen for cryptococcosis has not been identified in non-HIV nontransplant patients due to limited data.40,41 As a result, most treatment regimens are based on efficacy data from PLH and SOT recipients, but depend on the extent of disease. A combination of lipid-associated formulations of amphotericin B (LFAB), such as liposomal amphotericin B (LAmB) or amphotericin B lipid complex (ABLC), plus flucytosine is most effective due to rapid fungicidal activity and is considered first line for induction therapy among patients with meningoencephalitis, disseminated (e.g. involvement of two or more noncontiguous sites), or severe pulmonary disease (e.g. diffuse pulmonary infiltrates). In cases of meningoencephalitis, antifungal therapy should be combined with daily therapeutic lumbar punctures or placement of a lumbar drain or ventriculostomy to reduce intracranial pressure (ICP). Indeed, most patients who were still alive after identification of cryptococcosis received an LFAB in combination with flucytosine,23–25,29 though few patients underwent CSF examination.23,24,29
While LFABs are preferred due to lower risk of nephrotoxicity, amphotericin b deoxycholate (AmBd) can be used if LAmB or ABLC are unavailable. 42 Many resource-limited countries are unable to access flucytosine due to limited availability and prohibitive costs,43,44 requiring use of less efficacious regimens such as high dose fluconazole with or without AmBd.40,41 A prolonged duration of induction therapy of 4 weeks, or longer in the presence of neurologic complications, is required in non-HIV nontransplant patients. After clinical improvement with induction therapy, fluconazole 800 mg per day should be initiated as consolidation therapy for at least 8 weeks followed by 200 to 400 mg per day as maintenance therapy for at least 1 year. However, the duration of antifungal therapy is somewhat dependent on resolution of the underlying immunodeficiency allowing for gradual restoration of immune function.
Conclusion
Cases of cryptococcosis have previously been reported following short- or long-term initiation of corticosteroids, IL-6 inhibitors, and JAK inhibitors for patients with autoimmune diseases, but it remains unknown how the use of one or a combination of these therapies will directly or indirectly alter immunologic response in patients with COVID-19. Clinicians must be suspicious of cryptococcosis in COVID-19 patients who clinically deteriorate following treatment with immunomodulatory therapies as signs and symptoms of cryptococcosis may overlap with COVID-19. Given the utility of CrAg LFA to detect CrAg in serum coupled with the high rate of missed opportunities to identify cryptococcosis and dire outcomes in non-HIV nontransplant patients, early recognition, perhaps through the use of a CrAg screen-and-treat strategy, prior to administration of immunomodulatory therapies in COVID-19 patients will result in prompt administration of antifungal therapy, most often a combination of LFAB plus flucytosine, thereby halting or eliminating further dissemination and improve overall mortality.
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our report did not require ethical board approval as it described the treatment of a single patient which does not meet the federal definition of human subjects research. This case was documented in the context of routine care and the information presented was anonymized in accordance with the Declaration of Helsinki.
Patient consent
Consent was unable to be obtained as the patient is deceased and their relatives were not contactable. As such, details have been removed from the case description to ensure anonymity.
